Abstract
Recent advances in tissue engineering technologies have highlighted the ability to create functional liver systems using isolated hepatocytes in vivo. Considering the serious shortage of donor livers that can be used for hepatocyte isolation, it has remained imperative to establish a hepatocyte propagation protocol to provide highly efficient cell recovery allowing for subsequent tissue engineering procedures. Donor primary hepatocytes were isolated from human α-1 antitrypsin (hA1AT) transgenic mice and were transplanted into the recipient liver of urokinase-type plasminogen activator-severe combined immunodeficiency (uPA/SCID) mice. Transplanted donor hepatocytes actively proliferated within the recipient liver of the uPA/SCID mice. At week 8 or later, full repopulation of the uPA/SCID livers with the transplanted hA1AT hepatocytes were confirmed by blood examination and histological assessment. Proliferated hA1AT hepatocytes were recovered from the recipient uPA/SCID mice, and we generated hepatocyte sheets using these recovered hepatocytes for subsequent transplantation into the subcutaneous space of mice. Stable persistency of the subcutaneously engineered liver tissues was confirmed for up to 90 days, which was the length of our present study. These new data demonstrate the feasibility in propagating murine hepatocytes prior to the development of hepatic cells and bioengineered liver systems. The ability to regenerate and expand hepatocytes has potential clinical value whereby procurement of small amounts of tissue could be expanded to sufficient quantities prior to their use in hepatocyte transplantation or other hepatocyte-based therapies.
Keywords
Introduction
Liver tissue engineering using primary hepatocytes presents the opportunity to create a de novo liver system, which can lead to a new therapeutic modality towards the treatment of various liver diseases (4,10, 15,18,28) and may have the potential to obviate the need for organ liver transplantation. In fact, hepatocyte transplantation into the liver of a recipient patient has proven to be effective in some experimental and clinical settings (3,10,14,20,21,24,26).
We have recently developed a novel approach for engineering a functional ectopic liver system within the subcutaneous space in mice (17). The engineered liver systems have been shown to persist for at least 200 days, and the persistency of the de novo engineered tissue has been largely predicated on two key factors. First, the creation of a vascularized platform using a growth factor-releasing device at the target subcutaneous site prior to the transplantation of the donor hepatocytes (30). The prevascularization process plays a pivotal role in the survival of the transplanted hepatocytes by providing an adequate blood supply to these transplanted cells, due to their high metabolic activities that require an active nutrient supply and waste exchanges (13,16). The second factor is the creation of uniform hepatocyte sheets using the isolated individual donor hepatocytes in culture (17). The in vitro created hepatocyte sheets maintain their intercellular communications (e.g., desmosomes and bile canaliculi) and exhibit other essential cellular microstructures to document their continued hepatocyte phenotype and functionality. Recent studies by our group has shown that combining these two factors in which the in vitro-generated hepatocyte sheets were transplanted within a prevascularized subcutaneous site potentiated the formation of a fully functional, long-lasting two-dimensional liver systems (17). Moreover, a more spatially complex, three-dimensional liver systems could be successfully achieved by stratifying additional hepatocyte sheets in the subcutaneous space (17). The confirmed functionality of the engineered liver system includes liver-specific protein productions, chemical uptake and subsequent metabolizing enzyme expressions, and regenerative growth (17).
Although this approach has shown considerable promise in recent years, there are several hurdles that need to be overcome for continual advancement to occur using hepatocyte-based technologies. Most notably, the potential lack of donor hepatocytes could become an issue depending on the abundance of the liver mass obtained for this procedure, since isolated hepatocytes lose their ability to actively proliferate following transfer to culture conditions (9,14). For this reason, an efficient method to propagate donor hepatocytes needs to be established. However, we and others have shown that primary donor hepatocytes can be successfully proliferated in vivo following transplantation in the livers of several mouse models, including the urokinase-type plasminogen activator-severe combined immunodeficiency (uPA/SCID) transgenic mice (1,6,7,19,22,23).
Towards this end, we have designed the present study to determine whether primary donor mouse hepatocytes transplanted into the livers of uPA/SCID recipient mice can be efficiently propagated in vivo for subsequent downstream utilization as a valuable source for the creation of hepatocyte cell sheets. The efficacy in the generation of hepatocyte cell sheets in the prevascularized subcutaneous space of mice and their persistent biological function(s) will be investigated.
Materials and Methods
Animals
Transgenic mice expressing human α-1 antitrypsin (hA1AT) under the hepatocyte-specific promoter (hA1AT-FVB/N, H-2q; kindly provided by Dr. Bumgardner, Ohio State University, Columbus, OH) (2) at 13 weeks of age were used as donors for hepatocyte isolation. Wild-type female FVB/N mice (11–12 weeks of age), which were syngenic to the hA1AT-FVB/N, were used as the recipient animals. hA1AT transgenic (hA1AT-TG) and FVB/N mice were maintained in the Animal Center at Tokyo Women's Medical University. uPA/SCID mice used in the hepatocyte propagation experiments were generated at Hiroshima Prefectural Institute of Industrial Science and Technology (HPIIST) and PhoenixBio, Co. Ltd. as described previously (22,23,31). Presence of the uPA transgene in each SCID mouse was genotypically confirmed by PCR as described previously (23). Experimental protocols were developed in accordance with the guidelines of the local animal committees at HPIIST, PhoenixBio, Co., Ltd., and Tokyo Women's Medical University. Mice were placed in cages within a temperature-controlled room with a 12-h light/12-h dark cycle as well as ad libitum access to food and water.
Hepatocyte Isolation and Purification
Hepatocytes were isolated from hA1AT-TG mice or hA1AT hepatocyte-repopulated uPA/SCID mice using a modified two-step collagenase perfusion method as previously described (5,11,12,16,17,30). Briefly, the livers were primarily perfused with Hank's balanced salt solution (HBSS) (Sigma, St. Louis, MO) containing 0.09% EGTA followed by a second perfusion using HBSS containing 0.03% collagenase (Sigma) and 5 mM CaCl2. Isolated cells were filtered through a nylon mesh membrane and hepatocytes were then purified by slow speed centrifugation at 50 ×
Transplantation of hA1AT Hepatocytes for Propagation in the uPA/SCID Livers
Isolated hA1AT hepatocytes were resuspended with serum-free Dulbecco's modified Eagle medium (DMEM) (Sigma) to a final ratio of 1.5 × 107 hepatocytes/ml. A total of 5 × 105 viable hepatocytes were transplanted into the liver of uPA/SCID mice using an infusion technique into the inferior splenic pole as described previously (22,23,31).
Creation of Two-Dimensional Hepatic Tissue Sheets
The creation of hepatocyte sheets was performed as described previously (17). Briefly, hA1AT hepatocytes were isolated from hA1AT mouse livers and hA1AT hepatocyte-repopulated uPA/SCID livers were plated on the temperature-responsive polymer [poly(
Hepatocyte Sheet-Based Liver Tissue Engineering Procedure
Prior to the liver tissue engineering procedure, the basic fibroblast growth factor (bFGF)-releasing device was inserted into the subcutaneous space on the back of FVB/N mice as described previously (17,30). Ten days after the device insertion, a highly vascularized subcutaneous platform was developed. Right before the hepatocyte sheet transplantation, an L-shaped skin incision was made to open the vascularized platform followed by removal of the device. The harvested hepatocyte sheet was transplanted onto the vascularized platform with the support membrane. Approximately 5 min later, the support membrane was removed while leaving the hepatocyte sheet in vivo and procedure was finalized by closing the skin wound.
Enzyme-Linked Immunosorbent Assay (ELISA)
Propagation status of the hA1AT hepatocytes in the uPA/SCID livers and functional volume of the liver tissues engineered in the FVB/N mice were assessed by measuring the recipient serum hA1AT concentrations by ELISA. The ELISA used an antibody against hA1AT (DiaSorin, Stillwater, MN) coupled with a secondary goat IgG antibody conjugated with horseradish peroxidase (Resewarch Diagnostics Inc., Flander, NJ) as previously described (10).
Histological Analyses
In some recipient uPA/SCID mice, livers were harvested at 8 weeks after hA1AT hepatocyte transplantation. Portions of subcutaneous tissues of FVB/N mice containing engineered liver tissues were harvested 90 days after hepatocyte sheet transplantation. Liver specimens and subcutaneous tissues were fixed in 10% buffered formalin and embedded in paraffin. The specimens were sliced into 5-μm-thick sections, which were processed for hematoxylin & eosin (H&E) staining or immunohistochemical staining. Immunohistochemical staining was performed using the avidin-biotin complex method with the rabbit polyclonal hA1AT-specific antibody (1:200, YLEM, Roma, Italy). For the cellular glycogen detection, Periodic Acid Schiff (PAS) histochemical staining was performed as described previously (16). To confirm the staining specificity of the cellular glycogen, serial sections were pretreated with salivary amylase for 60 min followed by the same PAS staining procedures.
Statistical Analysis
All of the values calculated in the present study were provided as mean ± SD. Statistical differences in the values were determined by a Student's
Results
Proliferation of hA1AT Allogenic Hepatocytes in uPA/SCID Livers
Hepatocytes isolated from 13-week-old hA1AT-TG mouse were transplanted into uPA/SCID mice (

Proliferation and propagation status of the transplanted hA1AT hepatocytes in uPA/SCID mouse livers. (A) Proliferation status of the human α-1 antitrypsin (hA1AT) hepatocytes. At day 0, hA1AT hepatocytes were transplanted into the liver of urokinase-type plasminogen actiovator-severe combined immunodeficient (uPA/SCID) mice (
At random, we selected some of the recipient uPA/SCID mice to assess the state of hA1AT hepatocytes repopulation in the liver samples obtained from the uPA/SCID mice at week 8 after transplantation. hA1AT staining of native liver samples of nontransplanted uPA/SCID mouse (for control) did not show any positively stained cells, confirming the staining specificity for hA1AT (Fig. 1B). In contrast, week 8 liver samples of the recipient uPA/SCID mice showed that hA1AT hepatocytes had invaded the liver throughout the organ (Fig. 1C), indicating that the uPA/SCID livers had been fully repopulated with the transplanted hA1AT hepatocytes during the 8-week period after transplantation.
Isolation of Repopulated hA1AT Hepatocytes From uPA/SCID Livers and Creation of Hepatocyte Sheet
Our confirmation that there was full repopulation of uPA/SCID livers with hA1AT hepatocytes after week 8 allowed us to continue to determine whether these donor repopulated hepatocytes could be a viable option for isolation and recovery to develop them in the hepatocyte sheet protocol. To achieve this goal, we isolated and recovered the repopulated hA1AT hepatocytes form one uPA/SCID recipient at week 12. After the Percoll isodensity centrifugation step, 14.5 × 106 viable hepatocytes were obtained. These recovered hA1AT hepatocytes (recovered hepatocytes) were then plated on the PIPAAm culture dishes at a density of 0.75 × 106 per 35-mm dish as previously described (17) (Fig. 2). We also isolated hA1AT hepatocytes (fresh hepatocytes) from hA1AT mouse and plated the hepatocytes on the PIPAAm culture dishes at the same cell density. Plating efficiency (percent of attached hepatocytes per plated hepatocytes) assessed 24 h after the cell plating were 73.8 ± 8.8% and 75.5 ± 5.3% in the recovered hepatocytes and fresh hepatocytes, respectively (four different wells in each group, no statistically significant differences between groups). At day 3 of the culturing protocol, the recovered hepatocytes were found to reach confluence. Lowering the culture temperature from 37°C to 20°C for 15 min resulted in the spontaneous detachment of the cultured hepatocytes from the culture plates as a uniform cell sheet.
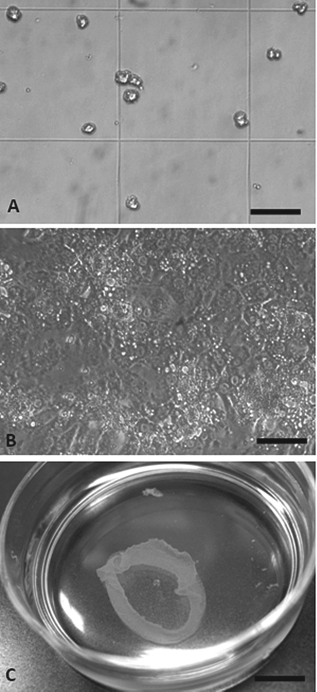
Morphological validation of the recovered hA1AT hepatocytes from the uPA/SCID livers that had been repopulated with the hA1AT hepatocytes. (A) Morphology of the isolated hepatocytes from uPA/SCID recipient. Photo was taken following the trypan blue exclusion test. (B) Recovered hA1AT-hepatocytes cultured on the poly(
Liver Tissue Engineering Using Hepatocyte Sheet Made of Recovered Hepatocytes
The recovered hepatocyte sheet was then used for liver tissue engineering with the FVB/N mice using a similar procedure as previously described (17). Hepatocyte sheet was transplanted to the subcutaneous site that had been prevascularized following the insertion of a bFGF-releasing device. Functional activity of the engineered liver tissues was assessed by measuring serum hA1AT levels in the recipient FVB/N mice. As shown in Figure 3A, recipient mice showed stable and persistent serum hA1AT levels (ranged between 2,000 and 5,000 ng/ml), suggesting that the engineered liver tissues could be viable and stably maintained throughout the 90-day experimental period.
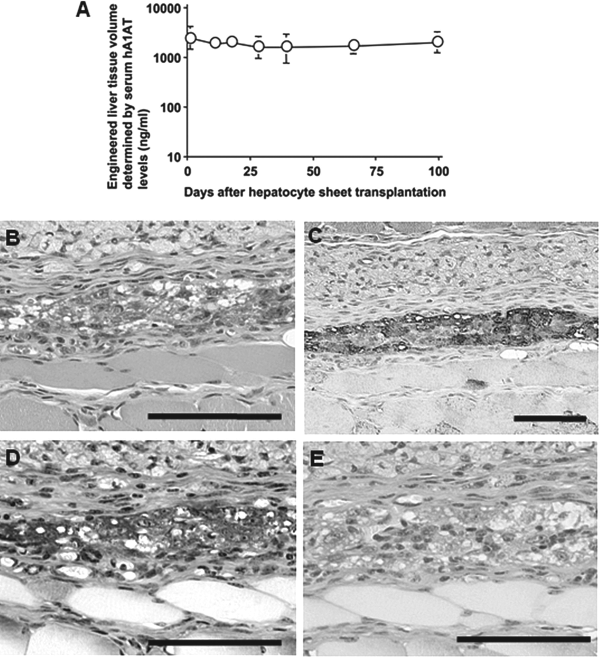
Functional maintenance of the engineered liver tissue within the subcutaneous space. (A) Functional volume of the liver tissues engineered in the subcutaneous space was determined by measuring recipient serum hA1AT levels by ELISA. Hepatocyte sheet composed of the hA1AT hepatocytes as shown in Figure 2 was generated and this harvested hepatocyte sheet was then transplanted into the subcutaneous space where an active vascular network had been induced using a basic fibroblast growth factor (bFGF)-releasing device. Histological findings by (B) H&E staining, (C) immunohistochemical staining for hA1AT, and (D, E) Periodic Acid Schiff (PAS) staining of the engineered liver tissue from samples obtained at day 90 after the hepatocytes sheet transplantation. Functions for cellular glycogen synthesis and storage were confirmed by PAS staining in sections that were either nontreated (D) or pretreated with salivary amylase (E). Scale bars: 100 μm (B–E).
Subcutaneous tissue samples taken 90 days after the tissue engineering procedure were assessed for histological examination. H&E staining and hA1AT immunostaining revealed that liver tissues were engineered in the subcutaneous space with the hepatocyte-specific phenotypes (Fig. 3B, C). PAS staining revealed that the engineered liver tissues were positive for the function of glycogen synthesis and storage (Fig. 3D). Negative signals in the PAS staining on salivary amylase-pretreated samples confirmed staining specificity for cellular glycogen (Fig. 3E). It is important to note that there was no evidence of tumor formation observed in any of the engineered tissues.
Discussion
The present study describes an in-mouse hepatocyte propagation system by which uPA/SCID mouse livers were found to fully reconstitute transplanted allogeneic mouse primary hepatocytes. This full reconstitution event was conducted by active and continuous hepatocyte proliferation as evidenced by the progressive increase in the hA1AT serum levels of the recipient uPA/SCID mice and massive occupation of hA1AT staining-positive hepatocytes in the recipient livers. The present study also showed that these propagated donor hepatocytes were able to be recovered, cultured, and subsequently used in the creation of hepatocyte sheets. The de novo generated hepatocytes sheets were able to be transplanted into a prevascularized subcutaneous space resulting in the successful engineering of an ectopic functional liver tissue.
We have established several innovative experimental approaches to create functional liver system in vivo (5,11–13,16,17,30). One of the key technologies in our series of developments is the creation of a uniform hepatocyte sheet using the PIPAAm culture dishes (17,27). The present study demonstrated that the hepatocytes recovered from the propagated livers of uPA/SCID mice were adhesive to the PIPAAm dish surfaces, and appeared to be at a similar efficiency of newly isolated primary hepatocytes that have not been previously isolated and propagated. The attached hepatocytes in the present study showed natural cell extension and became confluent by day 3 of culture. These cells showed the same morphological alterations associated with normal primary hepatocytes. This morphological similarity suggests that the microtubules and microfilaments were likely intact in the recovered hepatocytes.
This study as well as previous studies by our group have shown that hepatocyte sheets can be obtained by natural detachment of the primary cultured hepatocytes from the PIPAAm culture surfaces through a simple change in the ambient temperature for a brief period of time (17) without the need for any proteolytic enzyme treatment (28,29). This simple cell harvesting approach does not damage the cell-to-cell contact allowing for the retention of the intricate cell–cell intercommunication systems (i.e., bile canaliculi, desmosomes, gap junctions) (17). Although the ultrastructural examination was not performed in the present study, it is reasonable to postulate that the hepatocyte sheet made of the recovered hepatocytes possessed intercellular communications previously observed in our system using newly isolated hepatocytes considering that these functional intercellular communications are important in maintaining the structure of monolayered hepatocyte sheet. The integrity of the hepatocyte sheet using the recovered donor hepatocytes is similar to that of de novo isolated cells, and is readily transferable to a support membrane for the transplantation process.
We have previously reported that the uPA/SCID mice have the innate property to actively proliferate donor primary hepatocytes of xenogenic origin, including human hepatocytes, following transplantation into the liver (22, 23,31). The functional preservation of the human hepatocytes during their active proliferation phase in the uPA/SCID livers had been previously established as determined by the production of a hepatocyte-specific coagulation factor IX (FIX) (23). As an extension of the FIX findings, we have recently found that human hepatocytes in the uPA/SCID livers can maintain their cellular machinery to produce other hepatocyte-specific coagulation factors and anticoagulation factors (unpublished data). The availability of the propagated human hepatocytes in the uPA/SCID system for in vitro analyses was established in an earlier study by Yoshitsugu et al. (31). Since the recovered human hepatocytes can be cultured and express various drug-metabolizing enzymes, it was demonstrated that these recovered hepatocytes could be a valuable cell source for the study of a variety of liver functions (31). From these previous studies as well as the findings in our current study, we suggest that the propagated hepatocytes in the uPA/SCID livers could be as effective as the original primary hepatocytes with respect to their cellular functionalities. Although the present study clearly demonstrated that the hepatocytes obtained by in-mouse propagation system are useful for the creation of hepatocyte sheets, their availability for other hepatocyte-based approaches have not been investigated. Future studies will need to be conducted if the recovered hepatocytes can be transplanted and/or engraftable to other sites, such as the liver parenchyma itself or other ectopic sites within the body other than the subcutaneous space.
For potential clinical application for this type of hepatocyte-based therapeutic approach, the potential risk of oncogenesis deriving from the transplanted hepatocytes needs to be addressed, particularly due to the active level of proliferation that is being undergone following the transplantation procedure (8). In the present study, the gross morphology of all six of the recipient uPA/SCID livers at the time of sacrifice and/or hepatocyte isolation was normal. Histological investigation of the liver specimens did not show any cancerous or precancerous lesions. In parallel to these findings, no cancer development was observed in our previous study in which human hepatocytes were transplanted into the same strain of mice following the active proliferative phase of the donor hepatocytes (23). The best to our knowledge and as recently reviewed by Marongiu et al. (8), cancer cell development from the transplanted normal primary hepatocytes has not been reported in the uPA/SCID system. Other types of small animal models that are able to coordinate the process of proliferation of transplanted hepatocytes toward the replacement of recipient livers have been established, including the fumaryl-acetoacetatehydroxylase-null Rag2- mouse (1), the retrorsine-treated (6–8), or monocrotaline-treated rat models (25). In none of the models has there been any evidence of cancer cell development being documented. As far as we are aware from the previously documented and the present findings, it could be summarized that the risk of the cancer cell development from the normal hepatocytes during in-animal hepatocyte proliferation process appears minimal.
In all, we have shown that the use of the uPA/SCID mice as a viable recipient model to actively proliferate allogenic primary hepatocytes in their livers for subsequent use in the development of newly propagated hepatocytes or as a cell sheet system. These results taken together provide strong evidence that isolated hepatocytes can be efficiently propagated using an in-mouse liver procedure, and can be a potential valuable method to generate a new source of hepatocytes for hepatocyte-based therapies.
Footnotes
Acknowledgments
