Abstract
The survival of engineered tissue requires the formation of its own capillary network, which can anastomose with the host vasculature after transplantation. Currently, while many strategies, such as modifying the scaffold material, adding endothelial cells, or angiogenic factors, have been researched, engineered tissue implanted
Introduction
Tissue engineering paves a new way to restore or replace defective organs or tissues 1,2 . However, the potential clinical implementation of engineered tissues remains limited, at least in part because of the lack of effective strategies to ensure that engineered tissues are timely and adequately vascularized after transplantation 3,4 . Somatic cells are never more than 100 to 200 µm from a capillary 5 , which provides them with oxygen, essential nutrients, and transports away carbon dioxide and metabolic waste, all of which are critical for cell survival. Therefore, the successful transplantation of engineered tissue requires the construction of its own vascular network, which can anastomose with the host vasculature as soon as possible 6,7 .
To overcome the problem of vascularization, strategies, such as modifying the scaffold material, embedding angiogenic factors, 3D bioprinting technologies to create constructs containing vascular-like networks, and co-culture of endothelial cells with stem or target cells, have been researched 8 –11 . However, vascularization of thicker or complex tissues remains an unmet need in terms of clinical application 12 –14 . To ensure the implanted grafts can timely access to sufficient blood supply, it is necessary not only to construct the microvascular network of the engineered tissue itself, but also to ensure that the microvascular network can immediately anastomose with the host vascular network and transport blood.
The capsule tissue formed between an implanted device and the body’s own soft tissues was found to develop its own unique blood supply 15,16 . By placing a tissue expander close to large vessels, vascularization could be controlled and guided so that the induced capsule tissue will be supplied by an arteriovenous bundle, which was observed in our pre-experiments. The superficial circumflex iliac vessels (SCIs), adjacent to the bladder, were chosen to develop an axial capsule bed, which was then used to construct pedicled tissues to repair the damaged bladder. Cell sheet engineering is a promising technique applied to tissue engineering. This technique is capable of keeping extracellular matrix (ECM) and cell–cell interactions intact, which shows more advantages than traditional biodegradable scaffolds or cell suspension injections 17 . In this study, bladder smooth muscle cell (SMC) sheets were harvested to fabricate engineered smooth muscle tissues, as the muscle layer is the main part of the bladder tissue. To construct vascularized engineered tissue with its own vascular pedicle, we developed a three-stage rabbit model combining expander capsule tissue with cell sheet engineering. First, we transplanted a skin expander next to the separated SCIs, followed by expansion for different times, to identify which time point is the most effective for constructing an axial vascular bed. In the second stage, we compared the outcome of SMC sheets transplanted onto two different vascular beds including the expander capsule and subcutaneous tissue, which was widely used in other studies for vascularization. Finally, we transplanted multilayered SMC sheets onto the axial capsule bed twice to verify whether multi-transplantation of cell sheets was feasible to fabricate thick pedicled engineered tissues.
The objective of this study was to explore the feasibility of the tissue expander capsule as a novel platform for vascularizing autologous cell sheets and fabricating vascularized engineered tissue with its own axial vessels, which can deliver nutrients and oxygen immediately after pedicled transfer to the target region.
Materials and Methods
Animals
For the three stages of the animal experiments, male New Zealand white rabbits (stage I:
Expander Capsule Induction
Rabbits were kept on clear fluid 12 h before surgery and received cefuroxime sodium (50 mg/kg, Esseti FarmaceuticiS.r.l, Napoli, Italy) immediately before surgery. Following general anesthesia with 2% pentobarbital sodium (30 mg/kg, Sigma-Aldrich, St Louis, MO, USA), skin incisions were cut in the bilateral inguinal region (Fig. 1A), and SCIs surrounded by the inguinal fat pad were carefully isolated (Fig. 1B). A sterile, spherical skin expander (10 ml) was placed close to the separated SCIs below the bilateral abdominal skin to induce a vascularized capsule (Fig. 1C). Three milliliters of sterile saline solution was injected into the expander before the skin incision was closed with a 4–0 nylon interrupted suture (Fig. 1D). The incisions were allowed to heal for approximately 8 days. Following this, the expanders were repeatedly inflated with 3 ml of saline solution with a 2-day interval, increasing the amount to 12 ml (Fig. 1E). To identify which time point is the most appropriate for constructing an axial vascular bed, the expander capsule was photographed and resected for histological analyses 1 d, 1 week, 2 weeks, 3 weeks, and 4 weeks, respectively, after the expander was fully expanded (each

The induction and evaluation of the expander capsule. (A–E) A skin expander was placed close to the separated SCIs below the abdominal skin, after which the skin incision was closed with a interrupted suture. Then the expanders were repeatedly inflated with saline solution to induce a vascularized capsule tissue. (F–J) The gross appearance of the expander capsules at five different time points: 1 d, 1 week, 2 weeks, 3 weeks, 4 weeks, respectively, after the expander was fully expanded. (K–O) Masson staining of the expander capsules at five different time points, respectively. (P–T) CD31 staining of the expander capsules at five different time points, respectively. (U) Quantitative comparisons of CD31 positive microvessels within the expander capsules at five different time points, which indicted the vessel density of the capsule tissue 1 week after full expansion was significantly greater than those at other time points. The data are expressed as the mean ± SD, and the error bars represent the SD. **P < 0.01, compared with other time points. (V) The macroscopic appearance of the expander capsules infused with India ink. (W, X) Cross-sections of ink-perfused capsule tissue showed that the vascular network within the capsule was filled with the black ink, indicating that the blood supply of capsule was derived from the axial artery. Scale bar = 200 μm (K–T, W); Scale bar = 50 μm (X).
Bladder Muscular Layer Biopsy
Rabbits were anesthetized as described above, and a 3-cm vertical midline incision was made in the lower abdomen to expose the bladder. Next, 5 ml of sterile saline solution was injected into the bottom of the fat paddle at the vesical vertex to form a water sac, which was resected with ophthalmic scissors against the surface of the underlying muscle layer. Afterward, a small opening was cut into the exposed muscle layer and bluntly separated from its underlying mucosa layer with a curved clamp. Finally, an approximately 10 × 5 mm strip was excised from the muscle layer without disruption of the urothelium. The muscular incision was then sutured with 6–0 polyglycolic acid sutures.
SMC Sheet Fabrication
Muscle biopsies were incised into segments of approximately 2 mm2 with dissection scissors and incubated in Dulbecco’s modified Eagle medium (DMEM; Gibco, Grand Island, NY, USA) containing 1.5 mg/ml of collagenase NB4 (Serva, Heidelberg, Germany) and 1.0 mg/ml of dispase II (Roche, Mannheim, Germany) at 37°C for 30 min to create single-cell suspensions. The digestion was stopped by adding 1 ml of DMEM supplemented with 10% fetal bovine serum (Gibco). The isolated cells were filtered through a 70 μm cell strainer (Falcon, Corning, NY, USA), followed by centrifugation at 300 ×
Cell Sheet Viability Assay
To assess the viability of the cell sheets before and after harvest, a Live/Dead Viability/Cytotoxicity Kit (Invitrogen) was used. Green-fluorescent calcein-AM was used to indicate viable cells, while red-fluorescent ethidium homodimer-1 was used to indicate dead cells. Cell sheets before and after harvest were washed with phosphate buffered saline (PBS; Gibco) twice to eliminate the serum esterase activity present in the growth medium and then incubated in 2 μM of calcein-AM and 4 μM of ethidium homodimer-1 in Dulbecco’s PBS (DPBS; Gibco) at room temperature for 40 min. The stained cells were imaged by fluorescence microscopy (DM2500, Leica, Wetzlar, Germany). Cells killed using 70% methanol for 30 min were also stained as control. Image analysis (
Transplantation of Autologous SMC Sheets
Cell sheets were transplanted onto both the expander capsule and dorsal subcutaneous tissue in each rabbit. For the transplantation onto the expander capsule, the rabbits were prepared as described for the expander capsule induction to fully expose the capsule tissue which was used as a vascular bed to adopt cell sheets to construct vascularized engineered tissues. SMC sheets (1–3 layer, each
Multi-Transplantation of SMC Sheets to Construct Pedicled Tissues
After the vascularized expander capsule was induced and SMC sheets were fabricated, male rabbits (
Morphological and Histological Analyses
Macroscopic images of induced expander capsules were photographed by a digital camera (ILCE-6000 L, SONY, Tokyo, Japan) at five different time points. Tissue expander capsules, cell sheets, parts of the subcutaneous tissue and expander capsule transplanted with SMC sheets were harvested at the specific times mentioned above. All samples were fixed with 4% paraformaldehyde, embedded in paraffin, and sagittally sectioned into 4 µm-thick sections. Hematoxylin and eosin (HE) or Masson’s trichrome staining were performed according to the standard staining protocols. For immunohistochemistry, 4 µm-thick sections were deparaffinized and blocked with donkey serum for 30 min. To accurately measure the thickness of the SMC sheets transplanted
In Vivo Apoptosis Assay
Two days after transplantation, the resected cell sheet grafts were detected with TUNEL staining using a cell death detection kit (TMR red, Roche) following the manufacturer’s instructions. Six microscopic fields (at 400×) of each slide were randomly selected and photographed. The number of apoptotic cells within grafts was counted separately with ImageJ software. Apoptosis assay was expressed as the percentage of TUNEL-positive cells in total cells (DAPI).
Statistical Analyses
Data were expressed as the mean ± standard deviation (SD). Statistical analyses using the Student’s
Results
Characterization of the Induced Expander Capsule
To determine which time point is the most effective for constructing an axial vascular bed, we set five different time points. A macroscopic vascularized capsule tissue was consistently found around the skin expander after induction at five time points (Fig. 1F–J). SCIs were located in the center of the capsule tissue as axial vessels. In addition, numerous small vessels were observed originating from the axial vessels. Expander capsules were resected and fixed for histological analyses at different time points. Masson staining showed that the capsule tissue consisted of three layers (Fig. 1K–O): cellular layer, central layer, and fibrous layer. The thin cellular layer, which is closest to the expander, contains fibroblasts, inflammatory cells, and a few collagen fibers. The central layer mainly consists of loose collagen fibers, fibroblasts, and well-developed arterioles, venules, and capillaries. The fibrous layer mainly consists of dense collagen fibers and axial vessels. As a vascular bed, vessel density is a pivotal parameter to be considered, and immunostaining of CD31 was conducted (Fig. 1P–T). Quantitative comparisons indicated that vessel density of the capsule tissue 1 week after full expansion (160.0 ± 20.9/mm2) was significantly greater than those at other time points (P < 0.01) (Fig. 1U). Furthermore, the results of Indian ink injection showed that the ink first flowed through the femoral artery and superficial circumflex iliac artery, and then diffused into the capsule tissue, which was confirmed by the macroscopic observation (Fig. 1V). Cross-sections of ink-perfused capsule tissue indicated that the vascular network within the capsule was filled with the black ink (Fig. 1W, X), confirming our hypothesis that axial SCIs sprouted new blood vessels into capsule tissue so that the blood supply of the capsule was indeed derived from the axial artery.
Characterization of SMC Sheets
Bladder SMCs were successfully isolated and exhibited a characteristic spindle morphology at passage 0 (Fig. 2A). The SMCs at passages 3 to 5 were trypsinized and plated onto temperature-responsive culture dishes at a high density. The SMCs became excessively confluent after 5-day culture (Fig. 2B). Afterward, SMC sheets were harvested by reducing the culture temperature from 37°C to 25°C for approximately 30 min. The diameter of those cell sheets shrunk horizontally to approximately 1 cm due to the contraction of the ECM (Fig. 2C). As a result, harvested SMC sheets consisted of 3–5 cell layers, and the thickness of the cell sheets was approximately 50 μm (Fig. 2D). Immunofluorescent staining demonstrated that the cell sheets expressed typical SMC markers, such as α-SMA (Fig. 2E) and desmin (Fig. 2F), and cell nuclei were counterstained with DAPI (Fig. 2G). The merged image (Fig. 2H) was in accordance with our previous study 18 . Cell viability assay showed that almost all SMCs before harvest were live (Fig. 2I–K), while the control SMCs treated with methanol showed extensively dead (Fig. 2L). The cell sheet harvest did not damage the cells (Fig. 2M–O). Quantitatively, the SMC sheets before harvest showed 99.0% (±0.4%) live cells, compared with 98.6% (±0.5%) for the cell sheets after harvest (P > 0.05) (Fig. 2P). Immunofluorescent staining of Caspase-3 also demonstrated that few cells presented apoptosis in the SMC sheets (Fig. 2Q). Furthermore, immunofluorescent staining of Ki-67 (Fig. 2R) showed that approximately 15.3% of cells remained proliferative, although the cells were very dense and exhibited proliferation inhibition. ECM is critical for high cell transplantation efficiency. Immunofluorescent staining demonstrated that collagen IV (Fig. 2S) and laminin (Fig. 2T) both existed extensively in the cell sheets.

Characterization of SMC sheets. (A) Phase-contrast micrographs of rabbit primary bladder SMCs. (B, C) Phase-contrast and macroscopic images of SMC sheets. (D) Cross-sectional views of SMC sheets colored by hematoxylin and eosin staining. (E–H) The SMC sheet was stained with α-SMA (red color) and anti-desmin (green color) antibodies, while the nuclei were stained with DAPI (blue color). (I–K) Viability assay of the SMCs after 5-day culture. Live cells (green color); dead cells (red color). (L) Viability assay of the SMCs killed by 70% methanol as control. (M–O) Viability assay of the harvested SMC sheet. Live cells (green color); dead cells (red color). (P) For the viability, there was no significant difference between the sheets before and after harvest. The data are expressed as the mean ± SD, and the error bars represent the SD. n.s. denotes not significant. (Q–T) Immunofluorescent staining of Caspase-3, Ki-67, collagen IV and laminin (red color), respectively; nuclei were counterstained with DAPI (blue color). Scale bar = 100 μm (A, B, I–O); Scale bar = 50 μm (D–H, Q–T).
Outcome of SMC Sheets Transplanted onto Two Vascular Beds
The fate of cell sheets transplanted
On day 2, fluorescence microscopy of the cell sheets showed that cell sheets were successfully transplanted onto both vascular beds (Fig. 3, first–third column). The width of red fluorescence increased with the number of cell sheets, while the fluorescence intensity in the expander capsule transplantation group was stronger than that in the subcutaneous transplantation group. HE staining showed engineered smooth muscle tissues located on the surface of capsule and subcutaneous tissue, and erythrocytes were found in the vessels of cell sheet grafts (Fig. 3, fourth column), indicating that the vasculature of the grafts had connected to the host circulation. The thickness of the transplanted grafts was measured at five points in the α-SMA staining images (Fig. 3, fifth column) and the mean was calculated. Triplicate samples were measured for each group. There was significant difference (P < 0.01) between the SMC sheets transplanted onto capsule tissue and those transplanted onto subcutaneous tissue (1-layer: 148.0 ± 12.0 μm vs. 87.7 ± 6.3 μm; 2-layer: 211.0 ± 7.4 μm vs. 133.2 ± 9.4 μm; 3-layer: 269.5 ± 7.0 μm vs. 165.2 ± 14.3 μm, each
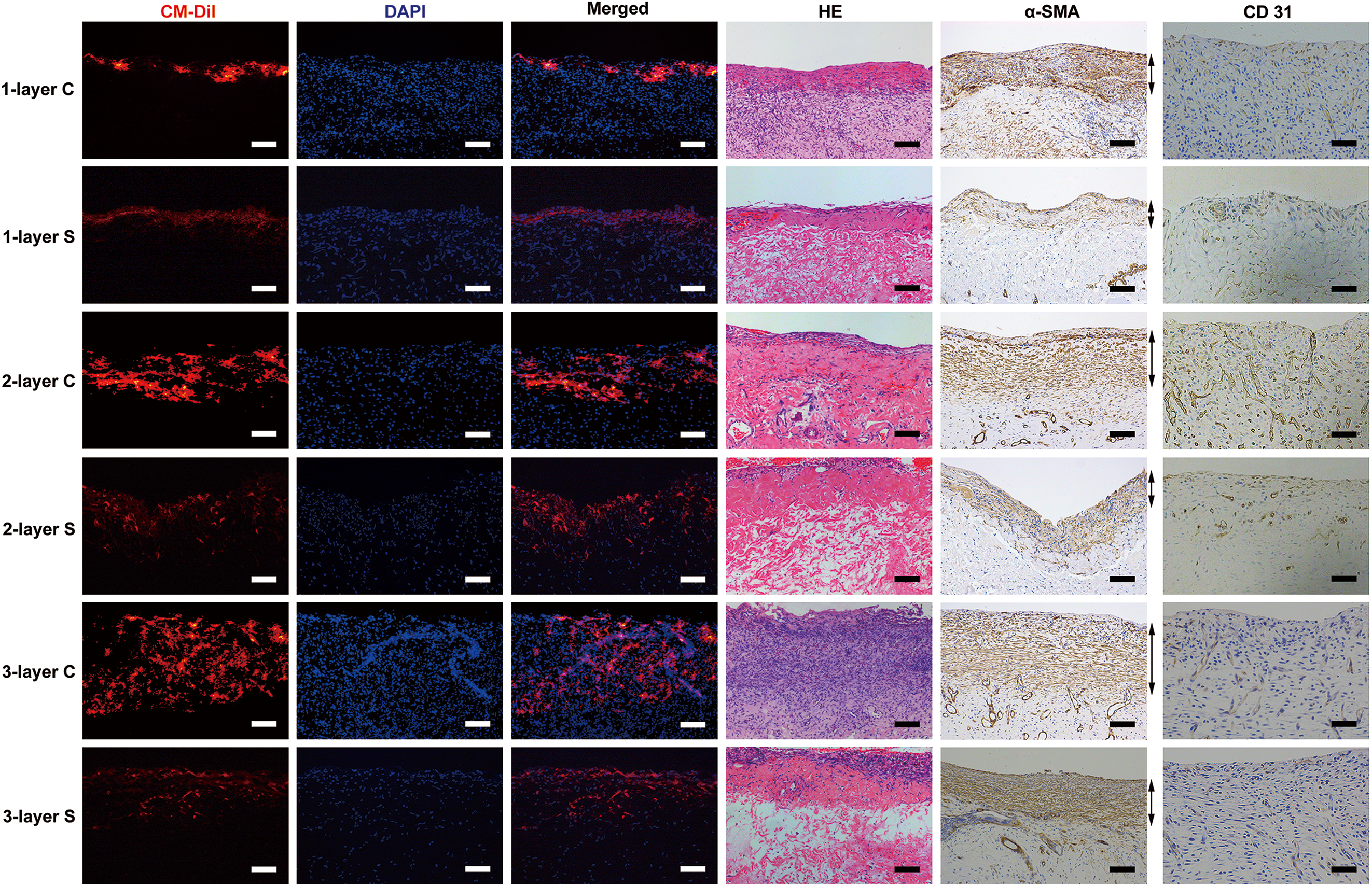
Outcome of SMC sheets transplanted onto two vascular beds on day 2. The transplanted cell sheets were labeled with CM-DiI (red color; first column). The width of red-fluorescent dye increased with the number of cell sheets, while the fluorescence intensity in the expander capsule transplantation group was stronger than that in the subcutaneous transplantation group. Nuclei were counterstained with DAPI (blue color; second column). The first and second column images were merged (third column). Hematoxylin and eosin staining of transplanted SMC sheets onto two vascular beds on day 2 (fourth column) showed engineered smooth muscle tissues located on the surface of two vascular beds and erythrocytes were found in the vessels of cell sheet grafts. Immunostaining of α-SMA was conducted to measure the thickness of cell sheet grafts (fifth column). Bidirectional arrows indicate viable cell sheet grafts. Immunostaining of CD31 was conducted to evaluate vessel density of cell sheet grafts (sixth column). “1-layer C” denotes 1-layer SMC sheets transplanted onto the capsule tissue. “1-layer S” denotes 1-layer SMC sheets transplanted onto the subcutaneous tissue. “2-layer C” denotes 2-layer SMC sheets transplanted onto the capsule tissue. “2-layer S” denotes 2-layer SMC sheets transplanted onto the subcutaneous tissue. “3-layer C” denotes 3-layer SMC sheets transplanted onto the capsule tissue. “3-layer S” denotes 3-layer SMC sheets transplanted onto the subcutaneous tissue. Scale bar = 100 μm (first–fifth column); Scale bar = 50 μm (sixth column).

TUNEL staining of cell sheet grafts 2 days after transplantation. For the one-layer and two-layer SMC sheets transplanted onto two vascular beds, almost all cells within cell sheet grafts were live. However, for the three-layer SMC sheets, apoptosis in the subcutaneous transplantation group showed an observed increase compared with that in the capsule transplantation group. TUNEL-positive nuclei (red color); nuclei were counterstained with DAPI (blue color). Scale bar = 50 μm.

Quantitative comparisons of thickness, vessel density and apoptosis of cell sheet grafts onto two vascular beds on day 2. (A) The thickness of SMC sheets transplanted onto capsule tissue was significantly larger than that of SMC sheets transplanted onto subcutaneous tissue for one-layer, two-layer, and three-layer, respectively. (B) The vessel density of SMC sheets transplanted onto capsule tissue was significantly greater than that of SMC sheets transplanted onto subcutaneous tissue for one-layer, two-layer, and three-layer, respectively. (C) TUNEL assay showed a remarkable reduction of apoptosis in the three-layer SMC sheets transplanted onto capsule tissue compared with those transplanted onto subcutaneous tissue. The data are expressed as the mean ± SD, and the error bars represent the SD. **P < 0.01.
On day 7, fluorescent images showed that the width of cell sheets traced with CM-DiI increased compared with that on day 2, indicating that the transplanted cells proliferated and formed thicker tissues. Meanwhile, some SMCs traced with red fluorescence migrated into host tissues (Fig. 6, first–third column). HE staining also showed that engineered smooth muscle tissues on two vascular beds became thicker over time and contained plenty of small vessels, which were filled with red blood cells (Fig. 6, fourth column). The thickness of transplanted SMC sheets was also measured by α-SMA staining (Fig. 6, fifth column). As with the results on day 2, there were significant difference (P < 0.01) between the SMC sheets transplanted onto capsule tissue and those transplanted onto subcutaneous tissue (1-layer: 245.2 ± 17.9 μm vs. 144.2 ± 13.1 μm; 2-layer: 355.0 ± 9.6 μm vs. 222.8 ± 26.4 μm; 3-layer: 499.5 ±22.3 μm vs. 244.3 ± 10.3 μm, each

Outcome of SMC sheets transplanted onto two vascular beds on day 7. The transplanted cell sheets were labeled with CM-DiI (red color; first column). The width of cell sheets traced with red fluorescence increased compared with those on day 2, indicating that the transplanted cells proliferated and formed thicker tissues. Nuclei were counterstained with DAPI (blue color; second column). The first and second column images were merged (third column). Hematoxylin and eosin staining of transplanted SMC sheets onto two vascular beds on day 7 showed engineered smooth muscle tissues on two vascular beds became thicker and contained plenty of small functional vessels (fourth column). Immunostaining of α-SMA was conducted to measure the thickness of cell sheet grafts (fifth column). Bidirectional arrows indicate viable cell sheet grafts. Immunostaining of CD31 was conducted to evaluate vessel density of cell sheet grafts (sixth column). The denotations are the same as those of Fig. 3. Scale bar = 100 μm (first–fifth column); Scale bar = 50 μm (sixth column).
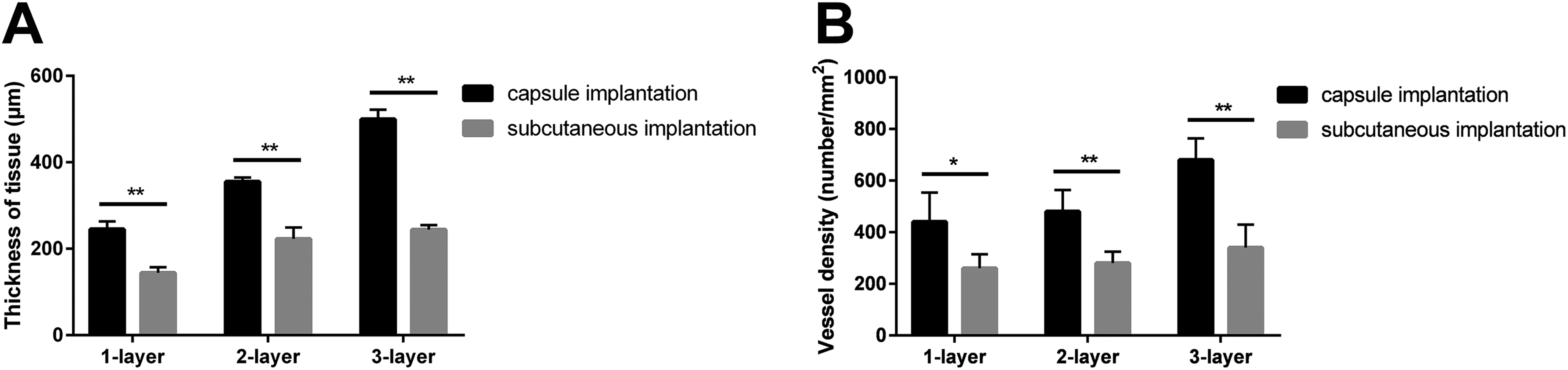
Quantitative comparisons of thickness and vessel density of cell sheet grafts onto two vascular beds on day 7. (A) The thickness of SMC sheets transplanted onto capsule tissue was significantly larger than that of SMC sheets transplanted onto subcutaneous tissue for one-layer, two-layer, and three-layer, respectively. (B) The vessel density of SMC sheets transplanted onto capsule tissue was significantly greater than that of SMC sheets transplanted onto subcutaneous tissue for one-layer, two-layer, and three-layer, respectively. The data are expressed as the mean ± SD, and the error bars represent the SD. *P < 0.05, **P < 0.01.
Thick Pedicled Smooth Muscle Tissues Constructed by Multi-Transplantation
Multi-transplantation of cell sheets after a certain time interval, which generates sufficient vascularization within the previously implanted cell sheets, could overcome the limit of cell sheet tissue thickness. The three-layer SMC sheets transplanted onto axial capsule tissue completely integrated with the vascular bed after 2 days, which was the basis for multi-transplantation. After 1 month, the transplanted six-layer SMC sheets formed macroscopically viable tissue, which could be easily distinguished from the surrounding capsule tissue (Fig. 8A). Macroscopically, the resected grafts contained axial vessels (Fig. 8B). Fluorescent images showed that the cells traced with CM-DiI still existed, and the fluorescence became sparse (Fig. 8C), indicting the transplanted cell sheets had proliferated since transplantation. Masson staining showed that the cell sheet graft was stratified (Fig. 8D): a thin layer of fibrous tissue located between two three-layer SMC sheet grafts. In addition, the graft was mainly composed of layered smooth muscle tissues, with less fibrous tissue distributed among the former. In regard to the thickness of the transplanted grafts, three points in the Masson staining images (

Thick pedicled smooth muscle tissues constructed by multi-transplantation of SMC sheets. (A) The macroscopic image of six-layer SMC sheets transplanted onto axial capsule tissue after 1 month. The dashed lines indicate the cell sheet graft. (B) The resected grafts contained axial vessels. (C) Fluorescent images showed the cells traced with CM-DiI (red color) still existed. Nuclei were counterstained with DAPI (blue color). (D) Masson staining showed the cell sheet graft was stratified and mainly composed of layered smooth muscle tissues. (E) Immunostaining of Ki-67 showed that plenty of proliferative SMCs existed in the grafts. Scale bar = 200 μm (C, D); Scale bar = 50 μm (E).
Discussion
In the current study, we have shown the capsule tissue can serve as an axial vascular bed for constructing vascularized engineered tissue with its own axial vessels using cell sheet engineering. When repairing damaged organs or tissues, we can choose large vessels, which are adjacent to the target organ or tissue and can be sacrificed without sequelae, as axial vessels to induce an axial capsule vascular bed. Then, cell sheets or cell-scaffold composites can be transplanted onto the vascular bed for vascularization and subsequent pedicled transfer without vascular suture. This study paves a new way for the vascularization of engineered tissues.
For successful vascularization of engineered tissues, it is necessary to develop strategies to accelerate the formation of the vascular system, inosculation with host vasculature, and succeeding blood perfusion into the engineered tissues. However, under the current technical conditions, the construction of an engineered tissue with its own fully functional vasculature
The capsule tissue has been shown to possess its own unique blood supply and contain extensive blood vessels 15,27 . In addition, the arteriovenous bundle technique has been proven to help to solve the problem of post-transplant vascularization 28,29 . Hence, the combination of capsule tissue and the arteriovenous bundle technique may be very promising to reach efficient vascularization within engineered tissues. The main purpose of the expander in clinical practice was to expand skin, and capsule tissue was confirmed to provide enough vascularity to support the skin graft 30,31 . However, the study of capsule tissue as a vascular bed in tissue engineering was rare 27 , and it was unknown at which time point the capsule tissue was the most vascularized. In this study, we set five time points after the skin expander was fully expanded. The macroscopic observation indicated that the capsule tissue was the most vascularized 1 week after full expansion. Masson’s trichrome staining indicated that the capsule tissue consisted of three layers: cellular layer, central layer, and fibrous layer, which was consistent with the experimental study performed by Kostakoğlu and colleagues 32 . The central layer consisting of abundant microvessels was relatively close to the silicone expander, which was convenient and minimally invasive for engineered-tissue implantation similar to the subcutaneous tissue. Vessel density is a key parameter to evaluate a vascular bed 33 , as the principal mechanism of vascularization by the vascular bed is sprouting angiogenesis, whereby endothelial cells branch out from existing blood vessels and expand into surrounding ischemic tissue to form a new vasculature 34,35 . Therefore, the efficiency of neovascularization increases with the vessel density of the vascular bed within limits. Quantitative comparisons of CD31-positive microvessels also identified that the capsule tissue was the most vascularized 1 week after full expansion, and could be used as an axial vascular bed for vascularization. Finally, we verified that the blood perfusion within the capsule tissue was derived from the SCI artery with Indian ink injection. This is the anatomic basis for constructing a pedicled flap and subsequent pedicled transfer.
Cell sheet engineering, which has advantages compared with other constructs, has been widely researched for the vascularization in tissue engineering
36
–38
. This technique allows for a noninvasive harvest of cultured cells as an intact sheet, including the ECM, without any enzymatic digestion. In this study, we successfully harvested bladder SMC sheets using thermo-responsive culture dishes. The harvested cell sheets consisted of multilayer cells because of cytoskeleton reassembly and contraction of the ECM
39
. In addition, the SMC sheets before and after harvest both showed high cell viability, which is a vital requirement for tissue engineering. Cell-generated ECM is probably more effective in regulating cell growth and maturation than artificial scaffolds
14
. ECM molecules are crucial for the attachment of transplanted cells to the target site without suture, and assure the implanted cells are supplied by diffusion until revascularization. Previous research has shown the ECM of intracorporal individual bladder SMCs mainly consists of collagen IV and laminin
40
. We also verified this point in bladder SMC sheets. Many previous studies have co-cultured endothelial cells or endothelial progenitor cells with other cell types, resulting in a capillary-like network formation
Vascularizing engineered tissues under the skin is regarded as the safest, simplest, and least invasive
The above results indicated that three-layer SMC sheets (approximately 150 μm) could be successfully transplanted onto the capsule vascular bed at one time, which was inconsistent with the previous opinion that cell sheet grafts more than 100 μm could not be successfully transplanted at single time 49,50 . The possible reasons include the superiority of the capsule vascular bed and different responses to hypoxia between bladder SMCs and cardiomyocytes researched in previous studies. However, for the thickness and vessel density on day 7, there was no significant difference (P > 0.05) between the two-layer and the three-layer SMC sheets transplanted onto subcutaneous tissue, indicating that the thickness of SMC sheets transplanted onto subcutaneous tissue at one time was also limited to approximately 100 μm, as with cardiomyocyte sheets. This indicated that the responses to hypoxia between bladder SMCs and cardiomyocytes were similar. In conclusion, the capsule vascular bed was more favorable to the growth and vascularization of engineered tissues than the subcutaneous tissue.
To overcome the problem of thickness limitation, multi-transplantation of cell sheets has been conducted to construct three-dimensional thicker tissues either
Although the results of the present study are encouraging, some limitations still exist. While we found the superiority of the capsule vascular bed over dorsal subcutaneous tissue for incubating and vascularizing engineered tissues, the underlying mechanisms are not clearly understood. Regulating and optimizing a vascular bed at cellular and molecular levels may promote the vascularization of engineered tissues. Second, we merely explored the possibility of three-layer SMC sheets, while the maximum layer of SMC sheets which could be successfully transplanted on the axial capsule vascular bed at single time was not investigated. Recent studies have attempted to increase the maximum layer of transplanted cardiomyocyte sheets in a single procedure through the addition of VEGF-releasing mats or insertion of gelatin hydrogel microspheres 53,54 . When the thickness limitation is overcome, cell sheet engineering will provide groundbreaking treatment options for injured organs or tissues. Finally, as the main objective of this study was to explore the feasibility of constructing pedicled engineered tissue by combining the capsule tissue and cell sheet engineering, pedicled transfer of the flap was not conducted. This is what we will continue to research to repair adjacent injured bladder or urethra.
Conclusion
The most vascularized axial capsule tissue can be successfully induced 1 week after full expansion when a tissue expander is placed close to the separated SCIs, which is more favorable to the growth and vascularization of transplants than classical subcutaneous tissue. Furthermore, thick pedicled smooth muscle tissues can be constructed by multi-transplantation of cell sheets on the capsule bed. The combination of the axial capsule tissue and cell sheet engineering may provide an innovative strategy to overcome the problem of vascularization in tissue engineering.
Footnotes
Authors’ Note
Zhiming Jia and Hailin Guo contributed equally to this work.
Ethical Approval
The experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Shanghai Jiao Tong University School of Medicine (B-2015-009).
Statement of Human and Animal Rights
All animals received care in accordance with the guidelines of IACUC and monitored by institutional veterinary staff.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by the National Natural Science Foundation of China (No. 81470911).
