Abstract
We recently developed a clinical grade ex vivo cord blood expansion procedure enabling a massive amplification of hematopoietic progenitors without any loss of stem cell potential. This procedure, based on day 14 liquid cultures of cord blood CD34+ cells, in medium Macopharma HP01 and in the presence of stem cell factor (SCF; 100 ng/ml), fms-related tyrosine kinase 3-ligand (Flt-3L; 100 ng/ml), megakaryocyte growth and developmental factor (MGDF; 100 ng/ml), and granulocyte colony-stimulating factor (G-CSF; 10 ng/ml) had to be modified due to the commercially unavailability of clinical grade MGDF molecule. So MGDF was replaced by thrombopoietin (TPO) in fivefold lower dose (20 ng/ml), and culture time was reduced to 12 days. That way, a mean expansion fold of 400, 80, and 150 was obtained for total cells, CD34+ cells, and colony-forming cells (CFCs), respectively. This amplification was associated with a slight enhancing effect on stem cells [Scid repopulating cells (SRCs)]. These are the ultimate preclinical modifications of a clinical grade expansion protocol, which is already employed in an ongoing clinical trial.
Introduction
We recently developed clinical scale expansion cultures of CD34+ cord blood (CB) cells enabling simultaneously to amplify committed progenitors and maintain stem cell activity (15). These cultures produce an important expansion of total cells (about 350-fold) and clonogenic progenitors (130-fold) and no decline in the activity of stem cells capable to reconstitute the human hematopoiesis in immunodeficient mice bone marrow [severe combined immunodeficient (Scid) repopulating cells (SRCs)] was detected. Thus, this technology would allow for engraftment to humans the whole CD34+ cell content of one cord blood unit after expansion and maintain the important fractions without additional manipulation being required.
However, a clinical grade molecule of megakaryocyte-derived growth factor (MGDF), a component of the expansion media, is no longer commercially available. We encountered a similar problem during our clinical trial concerning expansion and subsequent autologous transplantation starting from CD34+ cells mobilized in peripheral blood of myeloma patients (3,24). This problem was solved by replacing MGDF with thrombopoietin (TPO) (7). At a clinical level, the results with TPO were comparable to those obtained with the cells ex vivo expanded with MGDF. Initially, the cocktail comprised granulocyte colony-stimulating factor (G-CSF; 100 ng/ml), stem cell factor (SCF; 100 ng/ml), and MGDF (100 ng/ml). MGDF was then replaced by TPO, but at a lower dose (10 ng/ml) (7). Bearing in mind these results as well as the proliferative specificities of cord blood stem and progenitor cells (19), we modified the existing ex vivo expansion protocol by replacing MGDF (100 ng/ml) with TPO (20 ng/ml) in the cytokine cocktail comprising SCF (100 ng/ml), fms-related tyrosine kinase 3-ligand (Flt-3L; 100 ng/ml), and G-CSF (10 ng/ ml). In addition, the culture was shortened from 14 to 12 days in order to avoid the culture saturation noticed for some CB units of previous day 14 series. This way, in clinically compatible ex vivo cultures, we obtained with TPO an expansion product similar from a functional viewpoint to one with MGDF.
Materials and Methods
CD34+ CB Cell Isolation
CB units, previously banked in the CB bank of Bordeaux Transfusion Center (Etablissement Français du Sang Aquitaine-Limousin) as volume-reduced (5), were thawed in the presence of DNase and CD34+ selection made by Variomacs and Clinimacs (mean purity, 94.85 ± 5%) (Miltenyi Biotech, Paris, France) as described previously (8). Only CB samples rejected for technical reasons (lower volume or CD34+ cell number with respect to the minimal banking standard) were used. The informative consent to use CB for experimental purposes (in case it was not suitable for banking) was systematically obtained from mothers before sampling. The experiments were performed on discarded CB units in compliance with French regulation (article R1243-49 of Code de Santé Publique) and with a related authorization granted to the Laboratory.
Total Cells Counting
The cells were counted by automatic cell counter (Cell Dyn 3000, Abbott, Rungis, France).
Expansion Cultures
The cultures were initiated with 20 × 103 CD34+ cells per milliliter in HP01 medium (Macopharma, Tourcoing, France) (16) supplemented with SCF (100 ng/ml; Peprotech, Rocky Hill, NJ), TPO (20 ng/ml; Peprotech, Rocky Hill, NJ), Flt-3L (100 ng/ml; Peprotech, Rocky Hill, NJ), and G-CSF (10 ng/ml; Neupogen, Amgen SAS Neuilly sur Seine). The volume of culture varied, depending on the number of CD34+ cells obtained from each CB sample. This cell suspension was injected into culture cell bags (VueLife 25 or 118, AFC, Gaithersburg, MD) and cultured at 5% CO2 in water-saturated air (Incubators Stericult 200, Thermo Scientific, or IGO 150 Cell Life Jouan) for 6 days; the cultures were then fed with the fresh cytokine-supplemented medium (4 volumes of fresh medium to 1 volume of culture) and continued until day 12. The analyses of cellular content and phenotype were performed at day 12 of culture.
The serum-free medium (16), all cytokines (SCF, TPO, G-CSF, and Flt-3L), bags, CD34+ selection devices, etc., used in this study were of clinical grade, and the volume-reduced cord blood units (5) were previously banked and thawed before expansion (8).
CD34+ Cell Detection and Immunophenotypical Analysis
The CD34+ cell concentrations/purities in CB samples before freezing and after selection as well as the CD34+ cell number in cultures were determined following the recommendation of the International Society of Hematotherapy and Graft Engineering (26) as previously described (18). Briefly, three-color fluorescence was used to detect viable CD45+/CD34+ cells that were counted directly using tricount microbeads (Beckton Dickinson, San Jose, CA) by means of a flow cytometer (FACS Calibur, Becton Dickinson, San Jose, CA). For phenotypic characterization of cultured cells, the following fluorescein-coupled monoclonal antibodies were used: anti-CD13 (phycoerythrin; PE), anti-CD14 (fluorescein isothiocyanate; FITC), anti-CD33 (PE), anti-CD61 (FITC) (all from Beckton Dickinson, San Jose, CA), and anti-CD41 (PE) (Pharmingen, San Diego, CA).
Detection of Committed Progenitors (CFCs)
The committed progenitors CFCs = [colony forming unit-granulocyte-macrophage (CFU-GM) + CFU-Mix + burst forming unit-erythroid (BFU-E)] were detected using the cytokine-supplemented methylcellulose ready-to-use kit ID (Stem Alpha, Saint Clement les Places, France) (9). CD34+ cells were seeded in methylcellulose cultures in concentration of 500 cells/ml in 35-mm Petri dishes (NUNC, Roskilde, Denmark). The cultures were incubated for 14 days, and the colonies (>50 cells) were enumerated on an inverse microscope (Leica, Wetzlar, Germany).
Detection of Stem Cells by Their In Vivo Repopulating Capacity (SRC)
The animal experiments were performed in compliance with French regulation (License No: 3306002). One thousand day 0 CD34+ cells as well as their whole day 12 progeny (13,20) were injected to nonobese diabetic/severe combined immunodeficient/interleukin 2 receptor δ-chain null mice (NSG; central animal-keeping facility of Bordeaux Segalen University) conditioned by two injections of busulfan (-2 and -1 day) at the dose of 25 mg/kg (25). After 7 weeks, the animals were sacrificed, and their femoral mononuclear bone marrow cells were isolated and analyzed by flow cytometry (FACSCalibur; Becton Dickinson, San Jose, CA) for the presence of human CD45, CD19, and CD33 [phycoerythrin-cyanine 5.1 (PC5)-coupled anti-human antibody, Immunotech, Marseille, France]. Femora were isolated, and the bone marrow was flushed with 1 ml of RPMI 1640 complemented with 20% fetal calf serum (FCS). Cells were incubated with anti-human antibodies at 4°C and washed with PBS. Cells were then analyzed on the flow cytometer. To avoid false-positive results due to control isotype, we used the noninjected mice as controls (17).
Results
Expansion Efficiency and Phenotype of Expanded Cells
Mean day 12 expansion fold was approximately 400, 80, and 150 for total cells, CD34+ cells, and CFCs, respectively (Fig. 1A). This differentiation profile is comparable to the one obtained with MGDF (17). A mean absolute number of 98 × 106 CD34+ cells (i.e., 1.4 × 106/kg for a 70-kg patient) was obtained (Fig. 1B) per CB unit, which should be sufficient to obtain a posttransplantation agranulocytosis-shortening effect. Furthermore, since we used the CB units rejected from banking due to their lower cell content and/or volume, the “regular” CB units are expected to yield even more CD34+ cells. More than 80% of expanded cells expressed CD13 and CD33 and a much lower percentage expressed CD41, CD61, and CD14 (Fig. 1C).
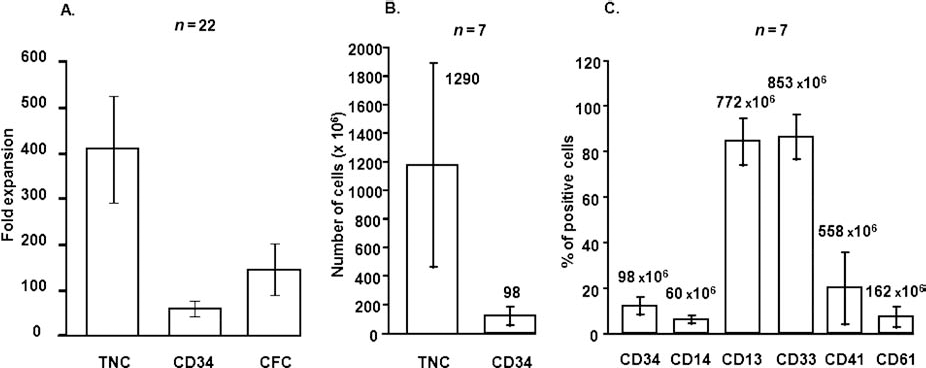
(A) Fold expansion of total nucleated cells (TNCs), CD34+ cells, and colony-forming cells (CFCs) and of the culture (day 12). (B) Absolute number of total cells and CFCs at day 12. (C) Immunophenotype of expanded cells. The absolute number of cells per culture is presented at the top of the bars.
SRC Activity
When determined on the basis of human CD45-expressing cells in murine bone marrow 7 weeks after transplantation, the dose of 1,000 CD34+ cells represents a limiting dilution dose (84% of mice were engrafted). After ex vivo expansion, the progeny of this same number of cells transplanted to NSG mice resulted in 100% of positive mice (Fig. 2A). The analysis of human CD33 and CD19 expression revealed a similar phenomenon: if transplanted with day 0 cells, 12.5% and 47% mice were “positive” for human CD33 and CD19, respectively, while after 12 days of expansion, the proportion raised to 50% (Fig. 2B) and 60% (Fig. 2C). Altogether, these results suggest some enhancing of SRC activity after ex vivo expansion of cord blood CD34+ cells with TPO (20 ng/ml) instead of MGDF (100 ng/ml) in the cytokine cocktail.
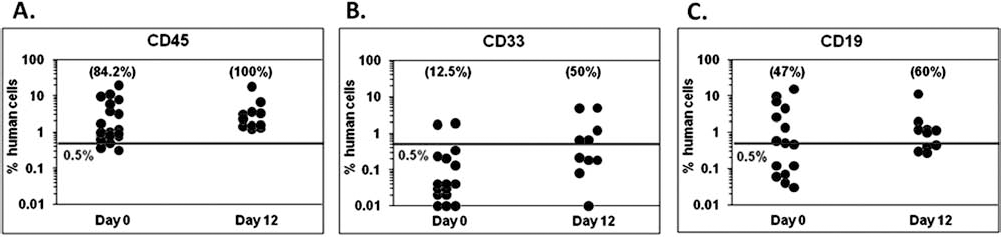
Maintenance of stem cells at the end of expansion cultures with respect to day 0: evaluation on the basis of engraftment of NSG mice. The day 0 CD34+ cells (1,000) or their whole day 12 progeny (expansion product) were injected (IV) to the mice preconditioned as described in Materials and Methods. The animals were sacrificed after 7 weeks, and the presence of human cells endowed by immunophenotypical markers [(A) CD45, (B) CD33, (C) CD19] in femoral bone marrow was analyzed. Each point represents one mouse. The line represents a background of fluorescence found in noninjected mice.
Discussion
MGDF is a truncated protein exhibiting homology with the erythropoietin (EPO)-like amino terminus of human TPO (6,10). This EPO-like domain is crucial for activation of c-MPL (myeloproliferative leukemia virus oncogene/CD110), the TPO receptor. The coupling to polyethyleneglycol (“PEGylation”) markedly increased the in vivo half-life of MGDF molecules without altering their ability to bind and activate c-MPL [reviewed in Abraham and Basser (1)]. Thus, PEG-MGDF, a clinical grade product, was used to obtain a positive effect on stem cell maintenance in course of ex vivo expansion, initially evidenced when TPO was associated with Flt-3L (23).
In this article, we are showing that PEG-MGDF could be replaced by TPO during ex vivo expansion in our cultures, clearly demonstrating that the pegylation is irrelevant for a use in ex vivo cultures.
TPO/mpl signaling is considered to be an important microenvironmental factor of stem cell maintenance (2,28). The other important microenvironmental factor, both for stem cell maintenance (4,12,14) and megakaryocyte production (21), is a low O2 concentration [reviewed by Ivanovic (11)]. These two factors could be related due to the fact that TPO regulates HIF-1α (hypoxia-inducing factor-1α) levels through generation of mitochondrial reactive spaces, leading to the stabilization of HIF-1α (27). That way, TPO exhibits some kind of “hypoxia mimicking effect.” The results presented here are in line with the “low O2 concept” [reviewed by Ivanovic et al. (13)] since the association of a powerful antioxidant system-equipped medium (16) with TPO seems to be very effective in our cultures both in terms of progenitors amplification and stem cells maintenance.
We can figure out that the future developments will concern the replacement of TPO by a “small-molecule agonist of c-MPL,” NR-101, which seems to exhibit an even more pronounced effect on stem and progenitor cell expansion than TPO itself (22).
Since the present results obtained with TPO replacing MGDF and 12 days of expansion are similar to those obtained in MGDF-supplemented cultures expanded for 14 days, it could be concluded that these ultimate modifications of our clinical grade expansion protocol did not modify its main result: a massive expansion of hematopoietic progenitors without exhaustion of stem cell potential, as determined by SRC assay.
The protocol is already employed in an ongoing clinical trial (http://clinicaltrials.gov/show/NCT01034449). Eight patients were transplanted so far with expanded cells, and the results are very promising.
Footnotes
Acknowledgment
This work was supported by R&D funds of Aquitaine-Limousin Branch of French Blood Institute (Bordeaux). The authors declare no conflicts of interest.
