Abstract
Tumor recurrence is the main limitation of liver transplantation (LT) in patients with hepatocellular carcinoma (HCC) and can be promoted by immunosuppressants. However, there is no prevention or treatment for HCC recurrence after LT. Here we describe a clinical-scale method for an adoptive immunotherapy approach that uses natural killer (NK) cells derived from deceased donor liver graft perfusate to prevent tumor recurrence after LT. Liver mononuclear cells (LMNCs) that were extracted from deceased donor liver graft perfusate contained a high percentage of NK cells (45.0 ± 4.0%) compared with peripheral blood mononuclear cells (PBMCs) (21.8 ± 5.2%) from the same donor. The CD69 activation marker and the natural cytotoxicity receptors, NKp44 and NKp46, were expressed at high levels in freshly isolated liver NK cells. Furthermore, interleukin-2 (IL-2)-stimulated NK cells showed greater upregulation of activation markers and the tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), which is critical for NK cell-mediated antitumor cell death and increased production of interferon. Moreover, IL-2 stimulation induced LMNCs to exhibit a strong cytotoxicity against NK-susceptible K562 target cells compared with PBMCs (p < 0.01). Finally, we also showed that the final product contained a very low T-cell contamination (0.02 ± 106 cells/kg−1), which reduces the risk of graft-versus-host disease (GVHD). Collectively, our results suggest that the adoptive transfer of IL-2-stimulated NK cells from deceased donor liver graft perfusate could be a promising treatment for LT patients with HCC.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is one of the most common reasons for liver transplantation (LT). In the past decade, the number of LT for patients with HCC has increased since the Milan criteria for HCC have been used for organ allocation in the US (16,23). However, the rate of recurrence of HCC after LT is 10–20% (21,32). This recurrence remains the most serious issue for LT in patients with HCC. The necessity of using postoperative immunosuppressants in the transplant recipient poses an additional risk for recurrence and hinders the use of cytotoxic chemotherapy drugs (14,23,41,46). However, there is no definitive treatment or prevention for the recurrence of HCC after LT (35,48). Hence, alternative therapies are needed for immunosuppressed HCC patients.
Natural killer (NK) cells are the major components of innate immunity and the first line of defense against invading infectious microbes and neoplastic cells (38). Functional impairment and decreased numbers of NK cells have been identified in HCC or cirrhotic patients (1,5,17). These functional defects in the NK cells might be responsible for the failure of antitumor immune responses after LT with HCC. Since the immunosuppressive regimen that is currently used after LT reduces the adaptive immune components but effectively maintains the innate components of cellular immunity (12,13,24), augmentation of the NK cell response may be a promising immunotherapeutic approach (28).
Recently, we characterized the phenotypical and functional properties of liver NK cells extracted from living donor liver graft perfusate (17). We have also proposed a novel strategy of adjuvant immunotherapy to prevent tumor recurrence after LT. This immunotherapy involves intravenously injecting LT recipients with activated donor liver allograft-derived NK cells. This immunotherapy has been successfully performed in 14 living donor LT recipients at Hiroshima University, Japan (27). Some research groups have shown that deceased donor liver graft contains a unique subset of NK cells (18,25,26). However, the function and characteristics of liver NK cells that are derived from deceased donors and processed for clinical immunotherapy are not well known. Here, we demonstrated for the first time the phenotypical and functional properties of liver NK cells that were extracted from deceased donor liver graft perfusate under current good manufacturing practice (cGMP) conditions.
Patients and Methods
Collection of Samples
Fourteen donors who underwent organ recovery for LT were involved in this study. The donors included 11 men and 3 women aged 20–71 years (mean age ± SD, 43.4 ± 17.6 years). Informed consent was obtained from each donor, and the study protocol was approved by the Ethics Committee at the University of Miami. Standard testing for infectious disease, including assays for the detection of hepatitis B and C and human immunodeficiency virus (anti-HCV, anti-HIV, anti-HBcore, and HBsAg), was performed. A donor who tested positive for any of the infectious disease markers listed above was excluded from this study. Peripheral blood (40 ml) was collected from the organ donors. Subsequently, peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Hypaque (GE Healthcare, Sweden) density-gradient centrifugation and resuspended in X-VIVO 15 medium (LONZA, Walkersville, MD) supplemented with 100 μg/ml of gentamycin (APP Pharmaceuticals, Schaumburg, IL), 10% human AB serum (Valley Biomedical, Winchester, VA), and 10 U/ml of sodium heparin (APP Pharmaceuticals, Schaumburg, IL) (culture medium). During organ recovery, the aorta was clamped and the liver flushed in situ with up to 4 L of University of Wisconsin (UW) solution to remove blood from the vasculature. After organ recovery, the liver was placed in a bag and perfused through the portal vein with an additional 2 L of UW solution at the back table. This perfusate was collected from the vena cava and used to study liver mononuclear cells (LMNCs). The perfusate was retrieved in our cGMP cell processing facility (4,9,37). Since the UW solution has a high viscosity (45), the perfusate was centrifuged at 2,800 × g for 30 min at 4°C in order to ensure adequate centrifugation. The cell pellet was then subjected to Ficoll-Hypaque density-gradient centrifugation. A cell viability of 90% was ensured by trypan blue exclusion prior to all assays.
Cell Culture
LMNCs and PBMCs were cultured with 1000 U/ml of human recombinant interleukin-2 (IL-2) (Proleukin, Novartis, Emeryville, CA) in culture medium at 37°C in an atmosphere supplemented with 5% CO2. Anti-CD3 monoclonal antibody (mAb) (Orthoclone OKT3, Ortho Biotech, Raritan, NJ) was added to the culture medium (1 μg/ml) 1 day prior to cell harvesting. After 4 days of culture, the cells were harvested for further analysis. Testing for lot release included cell counts, viability, Gram stain, and endotoxin. Cell counts and viability were performed using the trypan blue dye exclusion method. Test samples were stained with trypan blue and then microscopically examined with a hematocytometer. A minimum of 1 × 107 cells with a cell viability of >80% was required to release the NK cell product for infusion. The Gram staining was performed at the Clinical Microbiology Laboratory (Jackson Memorial Hospital, Miami, FL) by using standard methods, with the lot release criterion of “no organisms seen.” Endotoxin testing by the Limulus Amebocyte Lysate assay was performed on the final product by using the Endosafe-PTS (portable test system; Charles River, Wilmington, MA). An endotoxin value of not more than 5 EU/kg was used for lot release. Although not included as a lot release criterion, the final product was tested for sterility by collecting specimens for aerobic, anaerobic, and fungal cultures and inoculating them in vials filled with soybean-casein digest broth and fluid thioglycollate media (BD Bactec, Becton Dickinson, Sparks, MD). The specimens were cultured for 14 days at 37°C. Mycoplasma testing was performed using the VenorGeM Mycoplasma Detection Kit (Sigma-Aldrich, St. Louis, MD).
Flow Cytometry
All flow cytometry (FCM) analyses were performed on a FACSCalibur cytometer or LSR II Flow Cytometer (BD Biosciences, San Jose, CA). For phenotyping of the surface markers, the leukocytes were stained with the following monoclonal antibodies (mAbs): fluorescein isothiocyanate (FITC)-conjugated anti-CD3 and anti-CD15 (BD Biosciences), goat anti-mouse IgG and anti-CD56 (BioLegend, San Diego, CA); phycoerythrin (PE)-conjugated anti-CD16, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), NKp44, NKp46, CD69, CD94, CD25, CD14, CD19, and CD7; allophycocyanin (APC)-conjugated anti-CD56 (B159) and CD11b (BD Biosciences); APC-eFluor 780-conjugated anti-CD3; eFluor 625-conjugated anti-CD15; biotin-conjugated anti-CD4; peridinin chlorophyll protein complex (PerCP)-eFluor 710-conjugated anti-CD11c (eBioscience, San Diego, CA); Qdot565-conjugated anti-CD8; Qdot655-conjugated anti-CD19; Alexa Fluor 568-conjugated streptavidin; and Alexa Fluor 700-conjugated anti-CD14 (Invitrogen, Carlsbad, CA). Dead cells were excluded by light scatter and 7-aminoactinomycin D (7-AAD) or 4′,6-diamidino-2-phenylindole (DAPI) (Invitrogen) staining. Cytokine production of lymphocytes was measured by a combination of cell surface and cytoplasmic mAb staining according to the manufacturer's instructions. Briefly, 4 h after treatment with Leukocyte Activation Cocktail (BD GolgiPlug, BD Biosciences), the lymphocytes were stained with anti-CD3-FITC and anti-CD56-APC surface markers (BD Bioscience). After washing, the cells were fixed and permeabilized with Cytofix/Cytoperm solution (BD Biosciences) and washed with Perm/Wash Buffer (BD Biosciences). Subsequently, aliquots were stained with either a mAb against intracellular cytokines; anti-interferon-γ (IFN-γ)-PE, antitumor necrosis factor-α (TNF-α)-PE, or anti-IL-2-PE (BD Biosciences).
Cell Targets
K562, a human chronic myelogenous leukemia cell line (ATCC #CCl-243), was cultured in DMEM medium (Invitrogen) supplemented with 10% heat-inactivated fetal calf serum (Mediatech, Inc., Manassas, VA), 100 U/ml penicllin, and 100 μg/ml streptomycin (Invitrogen) (complete medium) at 37°C in 5% CO2. Target cells were harvested during the logarithmic phase of growth, washed in PBS, and counted using trypan blue staining prior to use.
Cytotoxicity Assay
The cell cytotoxicity assay was performed by FCM as described previously (20). Briefly, target cells were labeled with 0.1 μM carboxyfluorescein diacetate (CFDA) SE Cell Tracer Kit (Invitrogen) for 5 min at 37°C in 5% CO2. The labeled cells were washed twice in PBS, resuspended in complete medium, and counted using trypan blue staining. The effector cells were coincubated at various effector/target ratios of target cells for 1 h at 37°C in 5% CO2. As a control, either target cells or effector cells were incubated alone in a complete medium to measure spontaneous cell death; 7-AAD was added to every tube. The data were analyzed using the Flowjo software (Tree Star, Inc. Ashland, OR). The cytotoxic activity was calculated as a percentage by using the following formula: % cytotoxicity = [(% experimental 7-AAD+ dead targets) - (% spontaneous 7-AAD- dead targets)]/[(100 - (% spontaneous 7-AAD+ dead targets)] x 100.
Statistical Analysis
For comparison between two groups, the Student's t-test (two-tailed) was performed. For comparison of more than two groups, one-way ANOVA followed by the Student-Newman-Keuls post hoc analysis was performed. A value of p < 0.05 was considered statistically significant. Values are expressed as the mean ± SEM.
Results
Deceased Donor LMNCs Contain a Large Population of NK and NT Cells
As an initial step, we compared the characteristics between LMNCs and PBMCs derived from deceased donors to determine whether liver NK cells could be used for clinical immunotherapy. To characterize the donor liver and peripheral NK cells, we collected liver graft perfusate and peripheral blood during regular organ procurement. The liver graft perfusate contained a large number of mononuclear cells (1.2 ± 0.2 × 109 cells), with a viability of 90 ± 3%. The phenotype of these cells was markedly different from that of matched donor PBMCs (Table 1). The proportions of CD3-CD56+ NK and CD3+CD56+ natural killer-like T (NT) cells in the LMNCs were significantly higher than those in the PBMCs. In contrast, the LMNCs possessed a smaller number of T cells and B cells than did the PBMCs. There was no significant difference in the number of monocytes or granulocytes. Phenotypical flow cytometry analysis of other surface markers was then performed in a comparative analysis between liver and blood NK cells (Table 1). The CD69 early activation marker was expressed on the majority (75.0%) of liver NK cells, whereas the same subset in the PBMCs showed a significantly lower frequency of expression (9.6%). When the expression of the nonmajor histocompatibility complex class I-specific-activating NK cell receptors (natural cytotoxicity receptors; NKp44 and NKp46) was examined in both liver and peripheral blood, nearly all NK cells (>90%) expressed NKp46. In agreement with Vitale et al. (40), NKp44 was not detectable in peripheral blood NK cells, while a mean of 3.8% of liver NK cells expressed NKp44. These results indicate a physiological activation status for liver NK cells. The percentage of NK cells expressing CD16, an NK cell lysis receptor (22), was higher in PBMCs than in LMNCs. Both the liver and peripheral blood NK cells expressed the C-type lectin receptor CD94. This molecule binds human leukocyte antigen (HLA)-E loaded with leader peptides from major histocompatibility complex (MHC) class I molecules (10).
Immunophenotypical Comparison of NK Cells in Liver Perfusate and Peripheral Blood
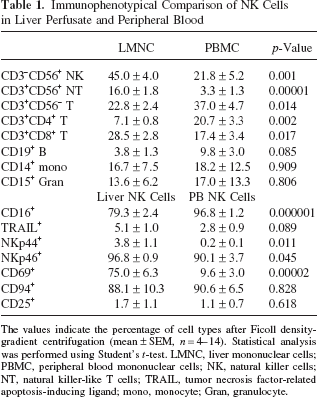
The values indicate the percentage of cell types after Ficoll density-gradient centrifugation (mean ± SEM, n = 4–14). Statistical analysis was performed using Student's t-test. LMNC, liver mononuclear cells; PBMC, peripheral blood mononuclear cells; NK, natural killer cells; NT, natural killer-like T cells; TRAIL, tumor necrosis factor-related apoptosis-inducing ligand; mono, monocyte; Gran, granulocyte.
Next, we analyzed the response of NK cells in LMNCs and PBMCs after IL-2 stimulation. TRAIL is a type II transmembrane protein that belongs to the TNF family, which preferentially induces apoptotic cell death in a wide variety of tumor cells but not in most normal cells (30,43,44). We previously reported that in vitro IL-2 stimulation upregulated the expression of TRAIL and induced strong cytotoxicity for liver NK cells extracted from living donor liver graft perfusate (17). As shown in Figure 1, freshly isolated liver NK cells and peripheral blood NK cells barely expressed TRAIL. Stimulation with IL-2 significantly upregulated the expression of TRAIL in liver NK cells, but this effect was barely observed for peripheral blood NK cells. IL-2 stimulation also resulted in an increased expression of the activation molecule NKp44 and maintained the expression of the inhibitory receptor CD94. These results indicate that cultivated NK cells have a compensatory mechanism to protect the self-MHC class I-expressing cells from NK cell-mediated cell death.

Comparison of surface marker expression patterns in response to interleukin-2 (IL-2) stimulation in liver perfusate and blood natural killer (NK) cells. Flow cytometry (FCM) analysis of freshly isolated or cultured with IL-2 (1,000 U/ml) liver mononuclear cells (LMNCs) and peripheral blood mononuclear cells (PBMCs) obtained from deceased donor after staining with anti-CD3 and anti-CD56 mAbs. The numbers indicate the mean percentage of each positive subset on electronically gated CD3-CD56- NK cells (mean ± SEM, n = 4–14). Statistical analyses were performed using Student's t-test (*p < 0.01 vs. fresh control).
Characteristics of the Liver NK Cell-Enriched Product
For determining whether NK cells from deceased donor liver graft perfusate could be processed using cGMP-compliant components, the LMNC cultivation was analyzed. At the start of the culture (preculture), the mean percentage of NK cells was 45.0% (range: 21.2–76.2%), whereas T cells constituted 22.8% (range: 6.6–35.2%). After processing, NK cells were enriched to 52.0 ± 5.0%. The viability of the enriched NK cells, as determined by trypan blue staining, remained >90% during the process. No microbial contamination was detected in the final product or in the culture medium. In addition, the cell processing resulted in a significant reduction of T cells in the final product. The percentage of CD3+CD56- T cells decreased to 0.6 ± 0.2% (0.18 × 105 cells/kg). Other CD56+ components of the final product included NT cells (0.2 ± 0.1%). Next, we further examined the phenotype of the CD56- fraction of the final product. After IL-2 stimulation, the phenotype of the final product was assessed using another T-cell marker, CD7, with or without the addition of OKT3. As shown in Table 2, the final product contained CD7+ T cells at 24.7%. Goat anti-mouse IgG antibody detection of OKT3 (isotype: mouse IgG) on T cells showed that 14.4% of the final product was bound with OKT3. After administration of the final product to the recipient, these T cells would be depleted by several mechanisms, including T-cell opsonization and clearance by mononuclear phagocytic cells, and complement-mediated cell lysis (6,7,39). The remainder of the T cells (10.3% of the final product) is involved in CD3 internalization or modulation, which induce T-cell dysfunction (6,36). Other components of the final product are shown in Table 2.
Phenotypical Characteristics of the CD56- Fraction of the Final Product From the Liver Perfusate

The values indicate the percentage of each marker (mean ± SEM, n = 5).
The percentage of the final product was calculated as follows: % of CD56- fraction x CD56- percentage (43.4)/100.
For phenotypically characterizing the NK cells in the final product relative to those in the starting material, a detailed flow cytometry analysis was undertaken. As shown in Figure 2, freshly isolated liver NK cells barely expressed TRAIL, NKp44, and CD25 (IL-2αR) and produced little cytokines. The cell processing significantly upregulated the expression of TRAIL and NKp44 in liver NK cells, but these changes were not seen in peripheral blood NK cells. The expression of CD69 and CD25 in liver NK cells also increased, but not significantly. In contrast, NKp46 expression significantly decreased after the cell processing. The activating receptors are defined by their ability to directly mediate the killing of the targets. Nevertheless, recent findings have demonstrated that the activation of some of the NK-triggering receptors requires the synergistic stimulation of more than one receptor (3). Our results are compatible with this theory. Intracellular staining flow cytometry showed that IL-2 stimulation induced significant cytokine production [IFN-γ and TNF-α (5.8–37.0% and 4.1–59.2%, respectively, n = 4, p < 0.01)] in liver NK cells (Fig. 2). These results are similar to those of studies of living donor liver graft perfusate (17,27). Next, NK cell cytotoxicity assays using LMNCs and PBMCs isolated from the deceased donor as effectors and K562 as targets were performed. Cytotoxicity against the standard NK cell target K562 was markedly elevated using effector cells from the final products relative to those from precultured LMNCs and PBMCs (Fig. 3). At a 20: 1 effector/target cell ratio, 56.3% of the K562 targets were killed on average by the final products, whereas precultured LMNCs and pre- and post-PBMCs killed only 11.8%, 2.5%, and 23.8% of K562 targets, respectively. We also tested the difference between with and without addition of OKT3 after IL-2 stimulation. The addition of OKT3 did not significantly enhance the NK cytotoxicity of either PBMCs or LMNCs.

Liver NK cells inductively express significant levels of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and cytokines after cell processing. FCM analysis of LMNCs obtained from deceased donor liver graft perfusate before the culture (upper panel) and after the culture (lower panel), after staining with mAbs against CD3 and CD56. Lymphocytes were gated by forward and side scatter. The FCM dot plot profiles are representative of 14 independent experiments. The percentages of CD3+CD56- (T), CD3-CD56+ (NK), and CD3+CD56+ (NT) cells are indicated at each quadrant. Histograms show the logarithmic fluorescence intensities obtained on staining for each surface marker or intracellular protein after gating on the CD3-CD56+ NK cells. Shaded regions indicate negative control staining with isotype-matched mAbs. The numbers indicate the mean percentages of positive cells in each group (n = 4–14). The histogram profiles are representative of 14 independent experiments.

LMNC final products show strong cytotoxicity against NK-susceptible target cells. The NK cytotoxic activities of freshly isolated (circle) and IL-2 stimulated (triangle) LMNCs (black) and PBMCs (white) with or without anti-CD3 monoclonal antibody (OKT3; square) against K562 target cells were analyzed by a FCM-based cytotoxic assay. All data are expressed as the mean ± SEM (n = 5). Statistical analysis was performed using one-way ANOVA followed by Student-Newman-Keuls post hoc analysis (*p < 0.01 vs. PBMC fresh and LMNC fresh, **p < 0.01 vs. PBMC IL-2 and PBMC IL-2 + OKT3).
Discussion
In this study, we demonstrated the phenotypical and functional properties of NK cells extracted from deceased donor liver graft perfusate under cGMP conditions. Methods for processing allogeneic NK cell products for human use on a clinical scale are limited to FDA-approved selection facilities and devices. The cGMP facility at University of Miami has published methods for processing different products (4,9). Lot release testing is described in the Patients and Methods section and is as dictated by the FDA guidelines for cellular products. First, LMNCs were shown to contain a large number of NK and NT cells, with both cell types possessing characteristics different from those of PBMCs. Second, in vitro stimulation with IL-2 induced liver NK cells to strongly upregulate activation markers, cytotoxicity, and cytokine production and to maintain the expression of inhibitory receptors. These results were compatible with those for living donor liver graft perfusate (17). Finally, we confirmed that the final product met the lot release criteria and contained low T cell numbers, thereby reducing the possibility of GVHD in a recipient.
This study demonstrated that deceased donor liver graft perfusate contained an average of 1.2 ± 0.2 × 109 mononuclear cells and 5.3 × 108 NK cells, whereas living donor liver graft perfusate, which contained 9.1 ± 0.8 × 108 mononuclear cells (Ohdan H et al., Hiroshima University, Japan, unpublished data). However, this number is several fold higher than the numbers calculated in previous studies on deceased donor liver perfusate (2,18,25). This discrepancy may be due to the fact that we collected the liver perfusate at the time of organ procurement while others did so just before liver transplantation.
NK cells can destroy many solid tissue-derived malignant cells through death receptor–ligand interactions (42). Previously, we found that normal hepatocytes express TRAIL-DR4 and TRAIL-DR5 together with TRAIL-DcR1 and TRAIL-DcR2, but that moderately or poorly differentiated HCCs highly express TRAIL-DR4 and TRAIL-DR5 but do not express TRAIL-DcR1 and TRAIL-DcR2, which indicates a susceptibility to TRAIL-expressing NK cell-mediated activity toward HCC (17,28). We have now shown that IL-2 stimulation significantly increases the expression of TRAIL in liver NK cells that are extracted from deceased donor liver graft perfusate (Fig. 1). Functionally, we also have shown that IL-2-activated liver NK cells were highly cytotoxic against tumors compared with PBMCs (Fig. 3). In addition to having an antineoplastic effect, NK cells are important components of the innate immune response due to their ability to lyse virus-infected cells and to recruit cells involved in adaptive immune responses. IFN-γ is a known host mediator that shapes the tumor phenotypes in a broader process known as “immunoediting” (19). Mice that lack either IFN-γ or its functional receptor are more susceptible to both viral and bacterial infections, indicating that IFN-γ plays an important role in antiviral and antibacterial responses (33,47). It is possible that these liver NK cells can prevent the replication of viruses including hepatitis C virus through an IFN-γ-dependent mechanism. Further studies are required to address this possibility.
The induction of GVHD is a major risk factor associated with the use of lymphocyte infusions from unrelated or haploidentical family donors (8,15). For clinical-scale experiments, OKT3 was added to the culture media 1 day prior to cell harvesting. The administration of OKT3-coated T cells in vivo has been shown to result in the opsonization and subsequent trapping or lympholysis of cells by the reticuloendothelial system (6,7,39). This method has been performed for clinical NK therapy in Japan, with no GVHD cases reported (27). Our final product from the cadaveric donor liver perfusate contained 0.02 × 106 CD3+CD56- T cells/kg. There are some clinical studies regarding T-cell contamination. For example, Miller et al. reported that the final T-cell dose was 0.18 × 106 cells/kg and that GVHD did not occur after haploidentical NK cell infusion (24). Schulze et al. reported that T-cell contamination was 0.01 × 106 cells/kg in allogeneic stem cell transplantation and that no GVHD occurred (34). Frohn et al. performed allogeneic NK cell infusion for renal cell carcinoma. The T cell contamination was 1.0% (0–7%) in their study (11). Passweg et al. defined the upper limit of acceptable T cell contamination as 0.1 × 106/kg BW for allo-NK cell infusion in stem cell transplantation. No patients developed clinical signs of GVHD after NK cell infusion (29). Compared with other clinical studies, our final product contains an acceptable level of T-cell contamination. NK cells exert alloreactivity after mismatched haploidentical transplantations due to an incompatibility between killer cell inhibitor receptors of donor NK cells and the recipient HLA type C (31). However, there is no known evidence of NK-mediated GVHD in humans.
In conclusion, liver NK cells derived from deceased donor liver graft perfusate inductively expressed TRAIL and secreted IFN-γ. IL-2-stimulated liver NK cells showed strong cytotoxicity against NK-susceptible K562 targets. Hence, these cells are potentially useful for the immunotherapy of LT recipients with HCC. This study is the first attempt to apply cadaveric donor liver NK cells to clinical cell transplantation. Our results will have a positive effect on adoptive immunotherapy using liver NK cells. However, further clinical studies are needed to elucidate the role played by donor liver NK cell infusions in the treatment of HCC patients after LT.
Footnotes
Acknowledgments
This work was supported by a grant from the Florida Department of Health and the Bankhead-Coley Cancer Research Program (1BG-08). The authors declare no conflicts of interest.
