Abstract
Natural killer cells not only play important roles in protecting against viral infection and cancer but also involved in the pathogenesis of Graves’ disease. Killer Ig-like receptor (KIR) genes encode receptors which are mostly expressed on and regulate the activation of natural killer cells. Our previous research found that the KIR2DS4 gene frequency was lower in patients with Graves’ disease than in controls. Nevertheless, the specific mechanisms by which natural killer cell act is obscure in Graves’ disease. In total, 178 participants including newly diagnosed Graves’ disease patients (n = 95) and healthy individuals (n = 83) were recruited in this study. TSH (thyrotropin), FT3 (free triiodothyronine), and FT4 (free thyroxine) were assayed using electro chemiluminescent immunoassays. The counts of natural killer cell (CD3−CD56+ natural killer cell), activated natural killer cell (CD3−CD56+CD69+ natural killer cell), and KIR2DS4-expressing natural killer cell (CD3−CD56+CD158i+ natural killer cell) in peripheral blood were analyzed using flow cytometry. The proportions of natural killer cells and activated natural killer cells were lower in the newly diagnosed Graves’ disease patients than in the controls; the difference was statistically significant (
Introduction
Graves’ disease (GD) is an autoimmune thyroid disease (AITD) that involves the breakdown of self-tolerance. It is the most common cause of hyperthyroidism. 1 Studies have shown that cellular and humoral immunity are all involved in the process. GD patients develop auto-antibodies that activate the thyroid stimulating hormone receptor on thyrocytes, inducing the production of excess thyroid hormone and goiter.2,3 Natural killer (NK) cells are defined by the expression of CD56 and lack of CD3(CD56+CD3−). In the peripheral blood, NK cells represent only 10% of lymphocytes. 4
Decreased NK cell proportion and cytotoxicity have been linked to several autoimmune disorders 5 including Crohn’s disease, 6 type 1 diabetes, 7 multiple sclerosis, and systemic lupus erythematosus. 8 Previous studies have indicated a possible link between autoimmune thyroid disease and NK cell proportion as well as NK cell immunocompetence. Some studies showed that the activity of NK cell was decreased in GD and Hashimoto’s thyroiditis (HT).9–11 However, another study found an increased activity of NK cell in GD patients, and NK cell activity may be independent of the level of thyroxine. 12 A similar result was found in patients with Hashimoto’s thyroiditis; the activity of NK cell has nothing to do with thyroid status, as well as thyroid goiter size and antithyroid antibody titers. However, another study indicates that the treatment of Graves’ disease, including radioiodine, thyroidectomy, and drug, affects the function of NK cell. NK cell function was low in untreated GD patients but normal in hypothyroid patients after thyroidectomy or radioiodine treatment. 9 Additional study observed loss of the NK cell population upon methimazole therapy. 13 Therefore, there is no unified view on changes in the population and activity of NK cell in GD so far, and more research on this topic is needed.
Killer cell immunoglobulin-like receptors (KIRs) are a class of regulatory molecules that mainly expressed on NK cell. KIRs can impact the function of NK cell. Our previous research found that the KIR2DS4 (CD158i) gene frequency was significantly reduced in the GD patients compared with control subjects, so we speculated that KIR2DS4 might be a protective gene in GD.14,15
The primary focus in this study was to identify abnormalities in the proportions of NK cells, activated NK cells, and KIR2DS4-expressing NK cells in untreated patients with GD, explore the potential role of NK cell in the pathological mechanism of GD, as well as seek out genetic basis of NK cell dysfunction in GD.
Materials and methods
Study design and study subjects
The study was a cross-sectional in nature. A sample size calculation was done for the primary outcome only using a Software program. 16 A total of 95 new-onset Graves’ disease patients were included from the Department of Endocrinology and Metabolism at Shandong Provincial Hospital between 2012 and 2013. The diagnosis of GD is supported biochemically when serum thyrotropin (TSH) levels are reduced (<0.3 µIU/mL) in the presence of elevated free thyroxine (FT4 > 22.7 pmol/L), free triiodothyronine (FT3 > 6.5 pmol/L), and TRAb (>1.22 IU/L) concentrations. A total of 83 healthy individuals were recruited. Patients with other autoimmune diseases, infection diseases, and cancer were excluded in this study. All the participants signed informed consent before the experiment. This study was approved by the ethics committee of Shandong Provincial Hospital affiliated with Shandong University (ethics approval number NO.2009-021). Demographic data were collected from participants by endocrinologists. Peripheral blood samples were obtained and used for serum or plasma preparations. Laboratory procedures were performed according to standard operating procedures.
Flow cytometry cell sorting
About 2 mL fasting venous blood samples were collected in the morning from GD patients and healthy volunteers. The collection tubes contained 0.2 mL of sodium heparin, stored at 4°C and examined within 12 h. About 200 µL of each sample was transferred to 24 well plates, and 200 µL RPMI-1640 was added to each well. After fully mixing, 2 µL PMA was added to the well. Then, the 24-well plate was incubated at 5% CO2 and 37°C for 3 h. There were two samples from each well and 100 µL per sample were removed and placed into polypropylene tubes; one tube was incubated in a mixture of anti-CD3 (2.5 µL), anti-CD56 (2.5 µL), anti-CD69 (2.5 µL) all from ebioscience (San Diego, USA), and anti-CD158i (5 µL) from BD Bioscience(San Diego, USA) monoclonal antibodies for 20 min at 20–25°C. As negative control, the other tube did not add antibodies. Next, add 2 mL lysing solution (FACSlyse; Becton Dickinson) to each tube; after vortex mixing, the liquid mixture was incubated for 10 min at room temperature. The cells were washed twice with phosphate-buffered saline (PBS) to remove cell debris and unbound antibodies. Flow cytometry was performed on XB-08 (BD Bioscience), and the results were analyzed by CytExpert software (v2.3.0.84). For scatter diagram analysis, the NK cell surface makers were CD3−CD56+, and R1 was set by the light scattering property (the forward-angle light scattering (FSC) and lateral-angle light scattering (SSC) settings were adjusted), namely, the lymphocyte region. Through the CD3 and CD56 molecular gate R2, namely, CD3−CD56+NK cells, CD69 was further used as activation markers for NK cells, and cyclor-r3, namely, CD3−CD56+CD69+-activated NK cells. CD158i was used as marker of KIR2DS4 receptor for NK cells, that is, CD3−CD56+CD158i+NK cells expressing KIR2DS4. Fl4h was the CD56-apc channel.
Biochemical measurements
Serum thyroid hormones and TRAb levels were measured by chemiluminescence methods (Cobas E601; Roche, Basel, Switzerland). Reference ranges were obtained from the manufacturer. The examinations of these variables were all carried out by the Shandong Provincial Hospital laboratory department.
Data analysis and statistics
All data were analyzed with SPSS software (Version 18.0). The significance of the difference in positive KIR2DS4 expression in NK cells between the GD group and healthy controls was analyzed by chi-square test of four-fold table. Significant differences in immunocyte typing, NK cell proportions, and NK cell activities were analyzed using t test for two independent samples. The relation between numerical variables was examined with Spearman’s correlation analysis. For all analyses, a final
Results
Summary of the 95 case and 83 control samples included in the study
A total of 95 patients with new-onset GD including 29 males and 66 females, as well as 83 healthy controls including 25 males and 58 females were collected in this study.
Table 1 shows the baseline characteristics of all participants. There was no obvious difference in age or gender between the two groups, and were considered to be comparable. The level of blood TSH in the GD patients was significantly less than that in the controls. The level of blood FT3, FT4, and TRAb in the GD patients were significantly greater than those in the controls.
Comparison between GD patients and Healthy controls of general clinical data.
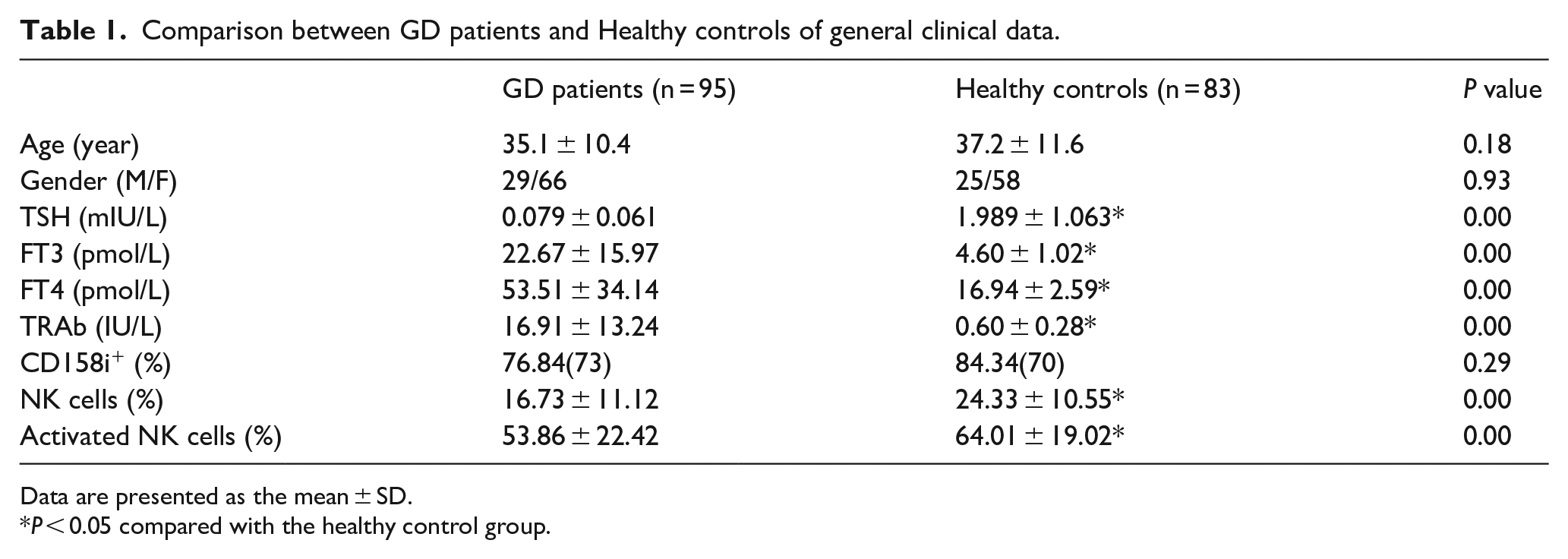
Data are presented as the mean ± SD.
The proportion of CD3−CD56+NK cells and CD3−CD56+CD69+NK cells in GD patients
To compare the proportion of NK cells (CD3−CD56+NK cells) and activated NK cells (CD3−CD56+CD69+NK cells) in peripheral circulation from study participants, we used fluorescence cytometry to analyze the percentages of NK cells (CD3−CD56+NK cells) and activated NK cells (CD3−CD56+CD69+NK cells) in peripheral circulation from all the participants (Table 1). GD patients showed a significant reduction in the proportion of NK cells compared with healthy subjects.
The proportion of NK cells in the GD patients decreased significantly compared with the healthy controls (16.73%± 11.12% vs 24.33%± 10.28%, respectively;

Flow cytometry analysis of NK cells. After treatment with red blood cell-lysing buffer, remaining cells were stained induplicate with Percp-anti-CD3, APC-anti-CD56, FITC-anti-CD69, and PE-anti-CD158i, respectively. The percentages of CD3−CD56+, in total mononuclear cells in individual subjects were determined by flow cytometry. The cells were first gated on lymphocytes and then gated on CD3−CD56+ cells. A total of at least 10,000 lymphocytes from individual samples were analyzed. Data shown are representative charts from different groups of subjects and expressed as individual percentage values which can represent the cohort of different groups of subjects. (a) Flowcy tometry and quantitative analysis of CD3−CD56+ cells. (b) Flowcy tometry and quantitative analysis of CD3−CD56+CD69+ cells. (c) Flow cytometry and quantitative analysis of CD3−CD56+CD158i+ NK cells.
The proportion of CD3−CD56+CD158i+NK cells in GD patients
To evaluate the differences between the circulat-ing CD3−CD56+CD158i+NK cells (KIR2DS4-expressing NK cells) populations in GD patients and healthy controls, we also used fluorescence cytometry to analyze the percentage of CD3−CD56+CD158i+NK cells in peripheral blood from all the study participants (Table 1). Our findings show that a lower percentage of CD3−CD56+CD158i+NK cells in the GD group than in the healthy controls (76.84% versus 84.34%, respectively;
The relationship between CD3−CD56+CD69+NK cells and CD3−CD56+CD158i+NK cells in GD patients
There is no obvious relationship between activated NK cells (CD3−CD56+CD69+NK cells) and KIR2DS4-expressing NK cells (CD3−CD56+CD158i+NK cells) (r = 0.081,

The relationship between CD3-CD56+CD69+NK cells and CD3−CD56+CD158i+NK cells in GD patients. No significant correlation was found between activated NK cells and KIR2DS4-expressing NK cells.
The relationship between CD3−CD56+CD69+NK cells and FT3, FT4, and TSH levels
There was negative correlation between the proportion of activated NK cells (CD3−CD56+CD69+NK cells) and the blood FT3 level (r = −0.358,
The relationship between CD3−CD56+NK cells and FT3, FT4, and TSH levels
We found no relationship between the proportion of NK cells (CD3−CD56+NK cells) and the blood FT3 level (r =−0.201,
Discussion
NK cells are not only major effector cells of the innate immune system, but also immune-modulatory cells regulating the adaptive immune system through produce cytokines. 17 GD is an autoimmune disease that involves abnormal B and T lymphocyte responses. Because NK cell has Fc receptors that bind to Ig, the activity of NK cell maybe modulated by plasma antibodies. NK cell activation also regulates antibody responses against autoantigens. These findings suggested that NK cell is involved in B cell-mediated autoimmunity.
There are two main functions of NK cells: cellular cytotoxicity and cytokine secretion. 18 The dysfunction of NK cytotoxicity and cytokine production was found in Hashimoto’s thyroiditis patients.11,19,20 Many studies have also found abnormalities in NK cell activity and phenotype of GD, but the result is still controversial. Tezuka found that peripheral NK cells functioned normally in GD patients. 10 However, a subsequent study demonstrated that GD patients exhibited abnormalities in NK cell counts and activity and cytokine secretion.9,11,13,17
NK cells can inhibit the activities of adaptive immune responses by affecting the activity of T cells 21 and dendritic cells (DCs) 22 through well-identified surface receptors. A previous study showed that activated T lymphocytes from patients with newly diagnosed and untreated GD are increased in number. 23 Therefore, NK cell could be either directly involved in autoimmune response, or interact with other immune cells (B cell, T cell and dendritic cell) regulating the immune inflammatory response. We infer that decreased count and activity of NK cells more likely enhance T-cell activation and promote the occurrence of GD.
CD69 is an early activation marker expressed on the surface of NK cell. In human NK cells, increased interferon (IFN)-γ and tumor necrosis factor (TNF)-α production was associated with expression of CD69. CD69 also has been shown to correlate with NK cell cytotoxicity.24,25 We have shown that the percentage of NK cell (CD3−CD56+NK cell) and activated NK cell (CD3−CD56+CD69+NK cell) in patients with GD are significantly decreased compared with a normal population. This is consistent with previous research.9,10,26,27
KIRs, which can activate or inhibit the function of NK cell by binding to human leukocyte antigen (HLA) class I molecules, are essential in the regulation of NK cell function. 28 Receptor and ligand binding is necessary to induce functional activity. 29 Many studies have shown the possible role of KIRs in the development of autoimmune disease, including atopic dermatitis, systemic lupus erythematosus, and multiple sclerosis.30–32 Our previous research found that the KIR2DS4 (CD158i) gene frequency in GD patients was significantly lower than that of the healthy controls.14,15 This result also supports the insufficiency of activated KIR genes in GD patients. Since KIR2DS4, one of the activating KIRs, can activate the killing activity of NK cells, the above findings suggest a genetic basis for NK cell dysfunction in GD patients. Further research on the KIR2DS4 present at the surface of NK cells suggested that the surface expression of KIR2DS4 receptors have been significantly lower in GD patients than the healthy controls; however, the difference did not reach statistical significance. Thus, the decrease in the frequency of KIR2DS4 gene might not be the main cause for the impaired immunocompetence of NK cells in GD patients.
In Zhang’s 27 study, there was a negative relationship between the blood FT4 levels and the proportion of NK cell (CD3−CD56+NK cell) and the proportion of NK cell subgroups expressing the activating receptor NKG2D (NKG2D-expressing NK cell), while the blood FT4 levels was positively correlated with the proportion of NK cell subgroups expressing the inhibitory receptor KIR3LD1 (KIR3LD1-expressing NK cell) in GD patients. In GD patients, after radioactive iodine therapy or thyroidectomy, circulating thyroxine levels decrease and NK cell activity recovers.9,33 This result suggests that the abnormal activity of NK cell may be linked to the initiation and progression of Graves’ disease. In the current study, we did not find any correlations between thyroid function and the proportion of NK cell (CD3−CD56+NK cell) in GD patients. Our study revealed that the proportion of activated NK cell (CD3−CD56+CD69+NK cell) was negatively related to the blood FT3 and FT4 levels, whereas the proportion of activated NK cell (CD3−CD56+CD69+NK cell) was positively related to the blood TSH levels in GD patients.
Conclusion
The pathogenesis of GD is complex, in which the reduction of the number or the functional defects of NK cell may participate in the process of immune regulation; however, the results have been controversial and inconclusive.9,10,11,13,16 Our results suggest that GD occurrence corresponded to decreased NK cell counts and activity. But several limitations exist in our study. For instance, many kinds of immune cells play a role in GD, the current research has only examined the proportion and activity of NK cell. Lack of data regarding other cells of the immune system such as neutrophils, T lymphocytes, and macrophages. It also remains to be determined whether other immune cells are involved in GD. Second, a single measurement of proportion and activity of NK cell in patients may not represent long-term proportion and activity of NK cell. Moreover, our study Lacks data on the treatment of GD. Treatment-induced alterations in the level of FT3, FT4 and TSH may affect NK function in GD patients. More comprehensive studies are required to elucidate how decreased NK cell count and activity may be causative for the pathogenesis of GD.
Footnotes
Acknowledgements
We are grateful to all investigators and subjects, for without their effort and dedication, this study could not have been accomplished.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the National Natural Science Foundation of China (81670721) for financing the research.
