Abstract
Natural killer (NK) cells play important roles in adoptive cellular immunotherapy against certain human cancers. This study aims to establish a new human NK cell line and to study its role for adoptive cancer immunotherapy. Peripheral blood samples were collected from 54 patients to establish the NK cell line. A new human NK cell line, termed as NKG, was established from a Chinese male patient with rapidly progressive non-Hodgkin's lymphoma. NKG cells showed LGL morphology and were phenotypically identified as CD56bright NK cell with CD16-, CD27-, CD3-, αβTCR-, γδTCR-, CD4-, CD8-, CD19-, CD161-, CD45+, CXCR4+, CCR7+, CXCR1-, and CX3CR1-. NKG cells showed high expression of adhesive molecules (CD2, CD58, CD11a, CD54, CD11b, CD11c), an array of activating receptors (NKp30, NKp44, NKp46, NKG2D, NKG2C), and cytolysis-related receptors and molecules (TRAIL, FasL, granzyme B, perforin, IFN-γ). The cytotoxicity of NKG cells against tumor cells was higher than that of the established NK cell lines NK-92, NKL, and YT. NKG cell cytotoxicity depended on the presence of NKG2D and NKp30. When irradiated with 8 Gy, NKG cells were still with high cytotoxicity and activity in vitro and with safety in vivo, but without proliferation. Further, the irradiated NKG cells exhibited strong cytotoxicity against human primary ovarian cancer cells in vitro, and against human ovarian cancer in a mouse xenograft model. The adoptive transfer of NKG cells significantly inhibited the ovarian tumor growth, decreased the mortality rate and prolonged the survival, even in cases of advanced diseases. A number of NKG cells were detected in the ovarian tumor tissues during cell therapy. In use of the new human NK cell line, NKG would a promising cellular candidate for adoptive immunotherapy of human cancer.
Keywords
Introduction
Natural killer (NK) cells as part of the innate immune system are important players in the first-line defense against malignancies. NK cells play an important role in adoptive cellular immunotherapy against certain human cancers such as renal-cell carcinoma and malignant melanoma (29,30), and are involved in the eradication of experimentally induced and spontaneously developing tumors in mice (17,44). The use of NK cells in human cancer immunotherapy has been proposed and such treatment has recently entered clinical trials (29). NK cell-based cellular immunotherapy can be implemented by using any of the following approaches: administrating cytokines or immunomodulatory drugs to activate endogenous NK cells; transplantation of alloreactive NK cells with allogeneic stem cells; adoptive transfer of ex vivo expanded autologous NK cells, donor-derived NK cells, autologous lymphokine-activated killer (LAK) cells, and cytokine-induced killer (CIK) cells; and adoptive transfer of an NK cell line (6,29,31,33,34). By comparison, there are some limitations in the efficacy of the reinfusion of ex vivo expanded autologous or allogeneic NK cells (25). Autologous NK cells are inhibited by self-major histocompatibility complex (MHC)-I molecules, and NK cells isolated from patients with malignant diseases often show impaired function (41). In addition, endogenous NK and LAK cells may be insufficiently cytotoxic in advanced disease (35). The isolation and large-scale ex vivo expansion of donor-derived NK cells is technically difficult as a result of contamination by other lymphocytes. T-cell contamination poses a risk for graft versus host disease (GVHD). The expansion potential of activated NK cells is poorly standardized between different clinical trials, with different phenotypes affecting the therapeutic potential and the risk for adverse reactions (41). Furthermore, expanded NK or LAK have not been well characterized and their antitumor activities have not been well defined due to complicated populations. Clinical use of human permanent NK cell lines would help us to better define their antitumor activities and overcome some of these limitations. Most importantly, their antitumor activities can be further enhanced, which are more cytotoxic and can be easily expanded and maintained in vitro (41).
Six malignant NK cell lines, including NK-92, YT, NKL, HANK-1, KHYG-1, and NK-YS, have been established. Except for YT cells, which were derived from an undefined acute lymphoblastic lymphoma, the other cell lines were established from patients with various NK cell malignancies (12). Among them, YT and NK-YS cells were established in Japan, and the other cell lines were established in Western countries (13,16,32,42,46,47). Three cell lines, including YT, NK-YS, and HANK-1, are Epstein-Barr virus (EBV) positive (type II latency). These cells are useful for studying the biological characteristics of EBV associated lymphoma/leukemia, but not for studying their antitumor activity (20, 42,47). NK-92 and KHYG-1 cell lines do not carry the EBV genome, and their antitumor activities have been well documented (45). NK-92 is the only NK cell line that has entered clinical trials. It is safe and has generated antitumor effects in advanced renal-cell carcinoma and malignant melanoma (23,29,30). It has been proposed that NK cell lines established in Western countries, such as NK-92, might not be suitable for Chinese patients. The lack of killer immunoglobulin like receptor (KIR) ligand(s) (human lymphocyte antigens; HLA) in the recipient induces NK cell function, and this may be a potential beneficial effect of NK cell line immunotherapy (43). However, there are significant differences in HLA molecules between races. Therefore, HLA antibodies in the recipient may inhibit the immune effects.
In this study, a newly established NK cell line, named NKG, was generated from a Chinese male patient with rapidly progressive non-Hodgkin's lymphoma. With the characteristics of activated NK cells, NKG cells were highly cytotoxic against various tumor cells. Successful immunotherapy against human ovarian cancer with irradiated NKG cells was firstly demonstrated in a xenograft mouse model. Thus, in use of the new human NK cell line, NKG would be a promising candidate in cellular immunotherapy for the clinical control of human cancer.
Materials and Methods
Establishment of the NKG Cell Line
Ten milliliters of peripheral blood was collected from a male patient with rapidly progressive non-Hodgkin's lymphoma. The peripheral blood was obtained under informed consent and with ethical approval. Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Hypaque gradient centrifugation, and the primary human NK cells were obtained by magnetic activated cell sorting (MACS; Miltenyi Biotec) using anti-CD3 microbeads (negative selection) and anti-CD56 microbeads (positive selection) (Miltenyi Biotec, Auburn, CA) as previously described (48). These enriched human NK cells were cultured in α-MEM (Gibco BRL, USA) with 10% heat-inactivated fetal bovine serum (FBS) (ExCell Biology, Shanghai, China), 10% horse serum (Hyclone, USA), and 100 IU/ml human recombinant interleukin-2 (rhIL-2) (Changsheng, Changchun, China) in humidified 5% CO2 at 37°C. Purified NK cells were cloned by limited dilution, and the most stable clone was selected, cultured, and named NKG.
Cell Lines and Cell Culture
NK-92 cells (human natural killer cell line) were obtained from American Type Culture Collection (ATCC, USA) and maintained in α-MEM with 12.5% FBS, 12.5% horse serum, and 100 IU/ml rhIL-2. YT cells (NK-like cell line) were obtained from NCI and maintained in RPMI-1640 medium (Gibco BRL) with 10% FBS. NKL cells (human natural killer cell line) were kindly provided by Professor BQ Jin (Department of Immunology, Fourth Military Medical University, Xi'an, PR China). The NKL cells were cultured in RPMI-1640 medium with 10% FBS and 100 IU/ml rhIL-2.
Ho8910 (human ovarian carcinoma) and SGC7901 (human gastric adenocarcinoma) cells were purchased from the Shanghai Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and maintained in RPMI-1640 medium with 10% FBS. K562 (erythroleukemia), A549 (human lung carcinoma), HCT-116 (epithelial colon colorectal carcinoma), HepG2 (hepatocellular carcinoma), Hep2 (human larynx cancer), and Daudi (human Burkitt's lymphoma) cells were obtained from ATCC and maintained in RPMI-1640 medium with 10% FBS. SKBR3 (human breast ademocarcinoma) cells were obtained from ATCC and maintained in DMEM medium (Gibco BRL) with 10% FBS. LoVo (human colorectal adenocarcinoma) cells were obtained from ATCC and maintained in F-12K medium (Gibco BRL) with 10% FBS.
Primary Ovarian Tumor Cell Isolation
Ovarian tumor tissues were obtained under informed consent and with ethical approval from female patients with epithelial ovarian cancer. The case information is shown in Table 1. The tumor tissue was washed, cut into pieces (<1 mm3) and placed in RPMI-1640 medium. Fresh ovarian tumor cells were isolated by the human tumor cell isolation and purification kit (Labkit, Shenzhen, China) according to the manufacturer's instructions. The primary ovarian tumor cells were cultured in RPMI-1640 medium containing 100 U/ml penicillin, 100 μg/ml streptomycin, and 10% FBS for 7–14 days. These cells were used as target cells for 51Cr release assays as previously described (6).
Clinical Characteristics of the 10 Patients With Epithelial Ovarian Cancer

The pathologic stage was determined as I, II, III, or IV according to the International Federation of Gynecology and Obstetrics (FIGO) staging of ovarian neoplasm.
Animals
Four- to 5-week-old female nude mice (BALB/c background) were obtained from the Model Animal Research Center of Nanjing University (Nanjing, China). All mice were maintained under specific pathogen-free (SPF) and controlled conditions (22°C, 55% humidity, and 12-h day/night rhythm). The animal experiments were performed in compliance with the US NIH guidelines.
Immunofluorescence Assay
NKG cells (1 × 105) were adhered to glass slides that were precoated with poly-L-lysine for 15 min at 37°C. Cells were rinsed twice with PBS, fixed in 2% paraformaldehyde, and stained with 5 μl of Alexa-488 conjugated anti-CD56 antibody (BD Pharmingen, CA, USA) for 30 min at room temperature in the dark. The stained cells were washed twice with PBS and imaged using a Zeiss LSM 510 laser-scanning confocal microscope.
Electron Microscopy
NKG cells (1 × 107) were washed with PBS and fixed in 2.5% glutaraldehyde overnight at 4°C. Electron microscopy were performed as previously described (15). Ultrathin sections (70 nm) were examined using a JEM-1230 transmission electron microscope.
Flow Cytometry Analysis
The following monoclonal antibodies, purchased from BD Pharmingen (San Diego, CA, USA), were used in this study: fluorescein isothiocyanate (FITC)-conjugated anti-CD2 (clone RPA-2.10), anti-CD4 (clone RPA-T4), anti-CD8 (clone RPA-T8), anti-CD16 (clone 3G8), anti-CD19 (clone HIB19), anti-CD25 (clone M-A251), anti-CD27 (clone M-T271), anti-CD28 (clone CD28.2), anti-CD48 (clone TU145), anti-CD62L (clone DREG-56), anti-CD85j (clone GHI/75), anti-CD94 (clone HP-3D9), anti-CD158a (clone HP-3E4), anti-γδTCR (T-cell receptor; clone B1), anti-IFN-γ (interferon; clone B27) and anti-granzyme B (clone GB11); Alexa-488-conjugated anti-CD11b (clone ICRF44) and anti-TNF-α (tumor necrosis factor clone MAb11); phycoerythrin (PE)-conjugated anti-CD11a (clone HI111), anti-CD11c (clone B-ly6), anti-CD45 (clone HI30), anti-CD54 (clone HA58), anti-CD56 (clone B159), anti-CD58 (clone 1C3), anti-CD69 (clone FN50), anti-CD112 (clone R2.525), anti-CD122 (clone Mik-β3), anti-CD127 (clone HIL-7R-M21), anti-CD132 (clone AG184), anti-CD158b (clone CH-L), anti-CD161 (clone DX12), anti-CD226 (clone DX11), anti-CD244 (clone 2-69), anti-NKp30 (clone P30-15), anti-NKp44 (clone P44-8.1), anti-NKp46 (clone 9E2/NKP46), anti-NKG2D (clone 1D11), anti-αβTCR (clone T10B9.1A-31), anti-TRAIL (TNF-related apoptosis-in-ducing ligand; clone RIK-2), anti-perforin (clone δG9), anti-IL-4 (clone 8D4-8), and anti-IL-10 (clone JES3-9D7); PE-CY5-conjugated anti-CD3 (clone UCHT1) and anti-CD56 (clone B159) and PE-CY7-conjugated anti-CCR7 (clone 3D12). FITC-conjugated anti-CXCR1 (clone 8F1-1-4), PE-conjugated-anti-CD95L (clone NOK-1), and anti-CXCR4 (clone 12G5) were obtained from eBioscience (San Diego, CA, USA). PE-conjugated anti-NKG2A (clone 131411), anit-NKG2C (clone 134591), and anti-CX3CR1 (clone 528728) were obtained from R&D Systems (Minneapolis, MN, USA). For surface phenotype assays, NKG cells (5 × 105) were blocked with purified mouse IgG (BIODESIGN, Saco, ME, USA) for 30 min at 4°C and then stained with the indicated antibody for 30 min at 4°C in the dark. For the intracellular assay, after the surface markers were labeled, NKG cells (1 × 106) were fixed, permeabilized, and then labeled with the indicated intracellular antibody for 30 min at 4°C in the dark. All data were acquired by a FACSCalibur (fluorescence-activated cell sorter; Becton Dickinson, Franklin Lakes, NJ, USA) and analyzed with WinMDI 2.8 software.
Gamma Irradiation
A gamma irradiation instrument (Biobeam 2000, STS, Braunschweig, Germany) was used to irradiate NKG cells. For the cytotoxicity assay, NKG cells (2 × 106/ml) were collected and resuspended in RPMI-1640 medium with 10% FBS and exposed to 4, 8, or 12 Gy of radiation. After irradiation, cells were examined for cytotoxicity or cultured for an additional 3, 18, 24, 48, or 72 h. NKG cells (1 × 108/ml or 6.67 × 107/ml) were resuspended in α-MEM and irradiated with 8 Gy for adoptive transfer in vivo.
Cytotoxicity Assay (4-h 51Cr Release)
The cytotoxicity assay was performed as previously described (7). Target cells (tumor cells, 1 × 106) were labeled by incubation with 200 μCi Na51CrO4 (Perkin Elmer, Billerica, MA, USA) in a final volume of 100 μl for 1 h at 37°C in 5% CO 2. Cells were washed three times with culture medium before use. NKG, NK-92, NKL, or YT cells were used as effector cells. Labeled target cells (T; 1 × 104/well) were incubated with effector cells (E) in a total volume of 200 μl RPMI-1640 medium with 10% FBS in round-bottom 96-well plates. Cells were plated at various cell densities to achieve different E/T ratios (20:1, 10:1, 5:1, 2:1, 1:1). Following the standard 4-h incubation, 100 μl supernatants were harvested from each well and monitored with a γ-counter (GC-911, ZONKIA, Hefei, China). The specific cytotoxicity was determined as follows: % specific release = (CPMexp – CPMspontaneous)/(CPMmaximum - CPMspontaneous) x 100%. Spontaneous release of 51Cr, as determined by incubating the target cells in RPMI-1640 medium with 10% FBS alone, was always less than 10%. Maximal release was determined by adding Triton X-100 (2.5% final concentration) to the target cells.
Cytokine Neutralization and Receptor Blockade
Anti-IFN-γ mAb (R&D Systems, clone 25718) was used for cytokine neutralization, and anti-NKG2D (R&D Systems, clone 149810), anti-FasL (R&D Systems, clone 100419), anti-NKp30 (BioLegend, clone p30-15), and anti-TRAIL (R&D Systems, catalog number AF375) were used to block the receptor function at 10 μg/ml in a total volume of 200 μl in the 4-h 51Cr release cytotoxicity assay.
Proliferation Assay ([3H]Thymidine Riboside Incorporation)
NKG cells (4 × 105/ml) were resuspended in α-MEM containing 10% FBS and 10% horse serum and exposed to 1, 2, 4, 6, 8, 10, 12, or 16 Gy of radiation (1 Gy/min). Irradiated NKG cells (4 × 104 cells/200 μl per well) were plated in 96-well flat-bottom plates with α-MEM containing 10% FBS, 10% horse serum, and 100 IU/ml rhIL-2 for 48 h. [3H]Thymidine riboside (1.5 μCi, GE HealthCare, USA) was added to each well 16 h prior to cell harvest. Tritium uptake was measured in a liquid scintillation counter (FJ-2107A 262 factory, Xian, China). Thymidine incorporation was determined: % [3H]thymidine incorporation = (CPMexp – CPMspontaneous)/(CPMcon – CPMspontaneous) x 100%.
ELISA
NKG cells (5 × 105 cells/ml) were incubated without IL-2 overnight (12 h) and then irradiated with 8 Gy (100 cGy/min) or not. NKG cells were stimulated by individual interleukins or combinations of interleukins for 24 h. The supernatant was collected, and the ability of NKG cells to produce the cytokines IFN-γ, TNF-α, IL-10, and IL-6 was analyzed. Human IFN-γ ELISA kit (catalog: BMS228) was obtained from Bender Medsystems (Vienna, Austria); human TNF-α ELISA kit (catalog: BMS2034), human IL-10 ELISA kit (catalog: BMS215/2), and human IL-6 ELISA kit (catalog: BMS213/2) were obtained from eBioscience. The interleukins used included recombinant human IL-2 (3000IU/ml, Changsheng, Changchun, China), recombinant human IL-12 (10 ng/ml; 100 U/ml, catalog: 95/544, NIBSC, the National Institute for Biological Standards and Control), recombinant human IL-15 (50 ng/ml, catalog: 200-15, Peprotech), recombinant human IL-18 (50 ng/ml, catalog: 4179-25, Biovision), and recombinant human interferon-α2 (2000 IU/ml, product number: 11105-1, PBL Biomedical Laboratories).
Histological Examination
The tissue samples (liver, spleen, and tumor) from sacrificed nude mice were fixed in 10% neutral-buffered formalin and embedded in paraffin. Sections 4 μm thick were stained with hematoxylin and eosin using routine methods. For cell smear detection, 300 μl of malignant ascitic fluid harvested from the nude mice with ovarian tumors was air dried at room temperature, fixed in 95% ethanol for 2 h, washed, and stained with hematoxylin and eosin.
Safety of NKG Cells In Vivo
NKG cells (6.67 × 107/ml), with or without 8 Gy irradiation, were resuspended in α-MEM and injected IP (10 μl/g body weight) into nude mice three times per week for 3 consecutive weeks. The same volume of α-MEM alone was used as the control. There were five mice in each group, and body weight and survival were observed. After 6 weeks, mice were sacrificed to determine NKG cell level in peripheral blood by flow cytometry analysis. NKG cells in liver, spleen, kidney, lymphoid nodes, and bowel were examined by tissue pathology using hematoxylin and eosin staining.
Human Ovarian Tumor Model in Nude Mice
Ho8910 cells (2 × 106) were resuspended in 200 μl RPMI-1640 medium and injected IP into nude mice. The same volume of RPMI-1640 medium alone was used as the control. The body weight and circumference of the abdomen were determined two times each week to evaluate the establishment of the tumor model. Mice that developed ascites or lost 10% of body weight were sacrificed to confirm tumor growth.
Adoptive Transfer of NKG Cells
Ho8910 cells (2 × 106) alone or mixed with NKG cells (1 × 107) irradiated with 8 Gy (E/T = 5:1) were injected IP into nude mice in a final volume of 200 μl. The mice were examined twice each week for the body weight, abdomen circumference, palpable tumor, and the survival. To determine the therapeutic efficacy of NKG cells on established ovarian tumors, nude mice were injected IP with Ho8910 cells (2 × 106) in a volume of 200 μl RPMI-1640 medium. At 21 or 35 days after tumor cell implantation, NKG cells (6.67 × 107/ml in α-MEM) irradiated with 8 Gy were injected IP (10 μl/g body weight) into mice three times per week for 3 consecutive weeks. Body weight and circumference of abdomen were examined twice each week to determine tumor progression.
Immunohistochemistry
NKG cells (6.67 × 107/ml in α-MEM) irradiated with 8 Gy were injected IP (10 μl/g body weight) into mice with palpable tumor three times per week for 1 week. At 12 h after NKG cell transplantation, tumor tissues were collected to measure the prevalence of CD56+ NKG cells. Sections 4 μm thick were stained according to the manufacturer's procedures. The reagents used included neural cell adhesion molecule (NCAM; 123C3; anti-human CD56, Catalog: sc-7326, Santa Cruz Biotechnology, Inc.), Histostain™-Plus Kits (SP-9002, Zhongshan Goldenbridge Biotechnology Co., Ltd.), and 3,3′-diaminobenzidine tetrahydrochloride (ZLI-9033, Zhongshan Goldenbridge Biotechnology Co., Ltd.). The images were observed using a microscope (Axioskop 2 Plus, ZEISS).
Statistical Analysis
All data are shown as mean ± SEM. The differences between individual data were analyzed by Student's t-test or analysis of variance where appropriate. Least significant difference test (LSD, 0<α<1) was used post hoc. A value of p < 0.05 was considered statistically significant. Mouse survival was analyzed using the Kaplan-Meier method.
Results
Establishment of the NKG Cell Line
Peripheral blood samples were collected for many years in our lab from a total of 54 patients in attempts to establish the NK cell line. Ultimately, the NK cell line was successfully established in 2003 from a male patient with rapidly progressive non-Hodgkin's lymphoma. CD56+ cells (NK cells) made up 64.82% of his peripheral blood mononuclear cells. NK cells were purified and cloned by limited dilution. A suitable culture was selected in α-MEM with 10% FBS, 10% horse serum, and 100 IU/ml rhIL-2. The selected NK cell clone was continuously cultured in vitro for more than 352 days. The cells were then frozen, thawed, and cultured for more than 6 months (180 days). The morphologic features remained stable during this time. As shown in Figure 1A, the cell line grew with aggregate-forming potential, and the cell doubling time was 2–3 days. The cells were identified as NK cells using the specific phenotypic marker CD56 (Fig. 1B). Further, the cells were confirmed to be malignant cells that originated from NK cells with the ultrastructure of large granular lymphocytes (LGL). The cells showed no signs of pathogen infection or contamination (Fig. 1C). These results indicate that a novel human NK cell line, which we termed NKG cells, had been established, which is the first NK cell line generated from a Chinese patient.
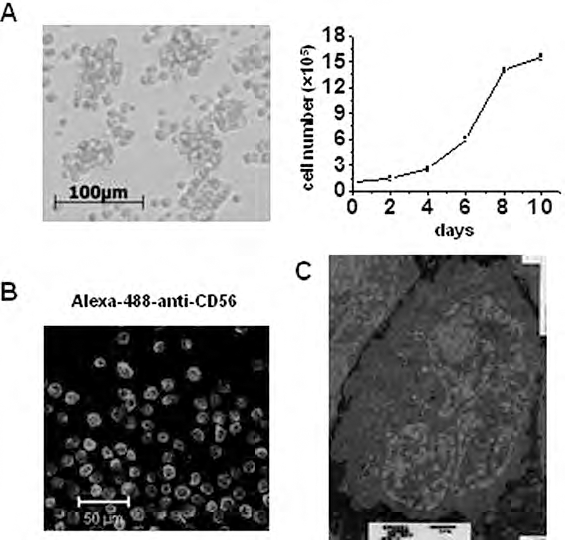
Establishment of the natural killer cell line, NKG. The morphology, growth, and internal structure of NKG cells were observed. (A) Microscopic picture of cell growth morphology. NKG cells (1 × 105) were seeded, the medium was changed by centrifugation, and the cell number was determined at the indicated time points (2, 4, 6, 8, 10 days). Date are shown as mean ± SEM from three samples. (B) NKG cells were stained by Alexa-488-conjugated anti-CD56 mAb for the immunofluorescence assay. (C) NKG cells were inspected by electron microscopy.
Phenotypic and Functional Characteristics of NKG Cells
As shown in Figure 2, NKG cells were characterized by flow cytometry as CD56+, CD16, CD27-, CD3-, αβ-TCR-, γδTCR-, CD4-, CD8-, CD19-, CD161-, CD45+, CXCR4+, CCR7+, CXCR1-, and CX3CR1-. NKG cells had characteristics of CD56bright NK cells, as evidenced by high expression levels of CXCR4 and CCR7 and no expression of CXCR1 and CX3CR1. IL-2R α/β/γ chains were expressed on NKG cells and may explain why the cells needed to be cultured in rhIL-2. They exhibited high expression of adhesion molecules, including CD2, CD58, CD11a, CD54, CD11b and CD11c, but CD62L expression was low. Among the costimulatory receptors expressed by NK cells, NKG cells only showed high level of CD48, and not CD226, CD112, CD244, or CD28. Although the inhibitory receptors CD94/NKG2A, CD158b, and CD85j were detected, NKG cells expressed high levels of activating receptors NKp30, NKp44, NKp46, NKG2D, and NKG2C. The cells also expressed cytolysis-related receptors and molecules TRAIL, FasL, granzyme B, perforin, and IFN-γ. NKG cells displayed characteristics of activated NK cells, making them a valuable tool to study activated NK cell function.

Phenotypic and functional characteristics of NKG cells. Cultured NKG cells were harvested and analyzed by flow cytometry using the indicated antibodies. The gray histogram represents the isotype-control antibody, and the open histogram represents the observed molecule. These were from a single experiment representative of three independent experiments. TNF, tumor necrosis factor; TRAIL, TNF-related apoptosis-inducing ligand; IFN, interferon; IL, interleukin; TCR, T-cell receptor.
Further, the ability of NKG cells to express cytokines in response to interleukin stimulation was examined. As shown in Figure 3A, NKG cells were highly responsive to IL-2, IL-12, and IL-15, and their combinations (IL-2 + IL-12 and IL-12 + IL-15), as evidenced by the high levels of cytokines they produced such as IFN-γ, TNF-α, IL-10, and IL-6. However, NKG cells were unable to produce IL-22 in response to these interleukin stimulations (data not shown). The natural killing of tumor cells is the most important role of activated NK cells. NKG cells showed high cytotoxicity to several kinds of tumor cells including Ho8910, K562, SGC7901, A549, Hep2, HepG2, HCT116, SBKR3, LoVo, and Daudi cells. In particular, NKG cell cytotoxicity against the Ho8910 and SGC7901 cells was more than 80% at the E:T ratio of 20:1 (Fig. 3B). Compared with NK-92, NKL, and YT cells, NKG cells were the most cytotoxic against the MHC-I+ cell lines (Ho8910 and K562) and the MHC-I cell lines (LoVo and Daudi) (Fig. 3C).

Cytokine production and cytotoxicity of NKG cells. (A) The ability of NKG cells to produce cytokines in response to exogenous stimulation. IFN-γ, TNF-α, IL-10, and IL-6 were measured by ELISA. (B) Four-hour 51Cr release assay was used for NKG cell cytotoxicity against a series of tumor cells. (C) Ho8910 and K562 cell lines [major histocompatibility complex (MHC)-I+], and LoVo and Daudi cell lines (MHC-I-) were selected as the target cells. The cytotoxicity of NKG cells was compared with NK-92, NKL, and YT cells. The percent cytotoxicity was calculated as shown in Materials and Methods. Data are shown as mean ± SEM of triplicates from one of three experiments. Similar results were obtained in all three experiments. ∗p < 0.05 versus the corresponding control groups.
According to the results in Figure 2, the molecular mechanisms underlying the cytotoxicity of NKG cells against Ho8910 cells were investigated. NKG cells cytotoxicity against Ho8910 cells was not affected when IFN-γ was neutralized and TRAIL or FasL was blocked (Fig. 4A). However, blockade of NKG2D or NKp30 in NKG cells significantly inhibited the cytotoxicity, especially with NKp30. Importantly, the cytotoxicity was completely abolished with concurrent blockade of NKG2D and NKp30 (Fig. 4A). These results demonstrate that the high cytotoxicity of NKG cells against Ho8910 cells depends on NKG2D and NKp30 recognition. Furthermore, the expression levels of NKG2D and NKp30 on the NKG cells were correlated with their cytotoxicity to Ho8910 cells (Fig. 4B).

The cytotoxicity of NKG cells against tumors depends on NKG2D and NKp30. Four-hour 51Cr release assay was performed to test the cytotoxicity of 8 Gy irradiated NKG cells against Ho8910 ovarian cancer cells at the Effector/Target (E:T) ratios of 10:1 and 5:1. (A) A dose of 10 μg/ml anti-IFN-γ mAb, anti-FasL mAb, anti-TRAIL mAb, anti-NKG2D mAb, or anti-NKp30 mAb was used for cytokine neutralization or receptor function blockade in the 4-h 51Cr release cytotoxicity assay. Data are shown as mean ± SEM of triplicates from one of three experiments. Similar results were obtained in the three experiments. ∗p < 0.05 versus the corresponding control groups. (B) NKG cells were stained with phycoerythrin (PE)-Cy5-conjugated anti-CD56 mAb and PE-conjugated anti-NKG2D mAb or PE- conjugated anti-NKp30 mAb at the indicated time point (0, 0.5, 1, 2, or 4 h) for flow cytometry analysis during cytotoxicity against Ho8910 at the E:T ratio of 5:1. Median fluorescent intensity at each time point is shown in parentheses.
Immunotherapy of Human Ovarian Cancer with Irradiated NKG cells in a Xenograft Mouse Model
To explore their ability against solid cancers, adaptive transfer of nonproliferative NKG cells was performed. To limit their proliferation capacity in vivo after transfer, we explored various doses of radiation. NKG cells irradiated with up to 8 Gy still possessed high cytotoxicity against tumor cells within 48 h, and no proliferation was observed (Fig. 5A–C). Adoptive transfer of 8 Gy irradiated NKG cells to nude mice demonstrated that NKG cells could not be detected in peripheral blood, liver, spleen, lymph node, kidney, and bowel (Fig. 5D, E). As the control, nonirradiated NKG cells also could not be detected in peripheral blood, liver, spleen, lymph nodes, kidney, and bowel after cell transfer, demonstrating that nonirradiated NKG cells could not grow in the mice, which further confirms the safety of NKG cells in vivo. Irradiated NKG cells were still able to produce cytokines in response to interleukin stimulation (Fig. 5F), although a small decrease was observed when compared with nonirradiated NKG cells (Fig. 3A). These results suggest 8 Gy should be an effective dose for NKG irradiation, suitable for immunotherapy, and would not produce oncogenicity or acute toxicity. High cytotoxicity (from 45% to 75%) of the irradiated NKG cells was observed against the primary ovarian tumor cells from patients with epithelial ovarian cancer at the E:T ratio of 20:1 (Fig. 6A), and the mean cytotoxicity was more than 60% against primary cancer cells obtained from 10 patients with ovarian tumors (Fig. 6B). The data suggest irradiated NKG cells show high antitumor activity against ovarian cancer.

Cell activity and safety of irradiated NKG cells. (A) The proliferation of irradiated NKG cells with different doses was determined by [3H]thymidine riboside (TdR) incorporation. (B) The cytotoxicity of irradiated NKG cells against tumor cells (Ho8910, K562, and SGC-7901) was determined by 4-h 51Cr release assay as described in Materials and Methods. (C) 8 Gy irradiated NKG cells were cultured for 0, 3, 18, 24, 48, or 72 h and then the cytotoxicity against Ho8910 was determined by 4-h 51Cr release assay. (D) NKG cells in peripheral blood were detected by flow cytometry to assess the safety in vivo described in materials and methods. (E) Tissue pathology was performed to determine the safety of NKG cells in vivo as described in materials and methods. (F) The ability of 8 Gy irradiated NKG cells to produce cytokines in response to exogenous stimulation was determined by ELISA. Data are shown as mean ± SEM of triplicates from one of three experiments. Similar results were obtained in the three experiments. ∗p < 0.05 versus the corresponding control group.
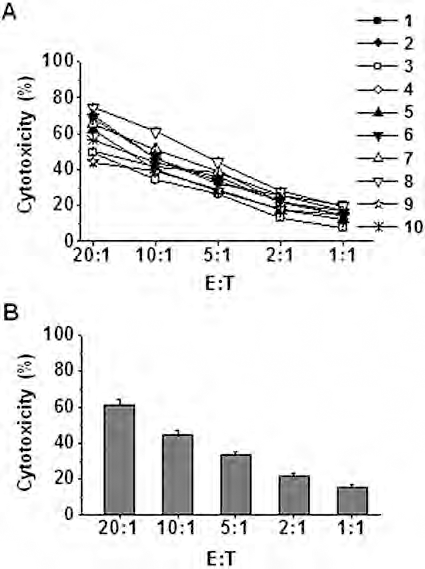
Irradiated NKG cells are cytotoxic against primary ovarian tumor cells from patients. Primary ovarian tumor cells were isolated from the female patients with epithelial ovarian cancer and cultured for 7–14 days. Cells were then used as target cells. Four-hour 51Cr release assay was used to determine the cytotoxicity of 8 Gy irradiated NKG cells. (A) The cytotoxicity of irradiated NKG cells against the primary ovarian tumor cells from each patient is shown. Data are shown as mean ± SEM of triplicates from one experiment. There were 10 patient samples for the study. (B) The mean cytotoxicity of irradiated NKG cells against primary ovarian tumor cells is shown. Data are shown as mean ± SEM of the 10 patient samples.
Further, Ho8910 cells (2 × 106) were injected IP into nude mice to establish a human ovarian tumor model (Fig. 7A–D). When the irradiated NKG cells were adoptively transferred into mice together with Ho8910 cells at a ratio of 5:1, tumorgenesis was significantly decreased from 100% to 50%, and the time to palpable tumor detection was delayed from 28–44 days to 90–135 days (Fig. 8A). The abdomen circumference (AC) was significantly decreased with NKG cell treatment, indicating attenuation of the severity of ovarian cancer (Fig. 8B). Impressively, the mean survival time in NKG-treated group was obviously prolonged from 61.5 to 210.2 days (Fig. 8C). These results demonstrated that the irradiated NKG cells owned the high cytotoxicity against the ovarian tumor cells in vivo.

Establishment of an ovarian tumor model with Ho8910 cells in nude mice. Ho8910 cells (2 × 106) were injected IP into nude mice in 200 μl RPMI-1640 medium. The same volume of RPMI-1640 medium alone was used as the control. (A) Mice with palpable tumor were calculated and the tumorgenesis (%) is shown. (B) The survival rates were measured. There were 10 nude mice in each group. ∗p < 0.05 versus the corresponding control group. (C) The liver, spleen, and celiac tumor tissue are shown at 42 days after Ho8910 cell implantation. Two representative samples from each group are shown. (D) Liver, spleen, celiac tumor, and ascitic fluid samples were collected from nude mice at 42 days after Ho8910 cell implantation and stained with hematoxylin and eosin.
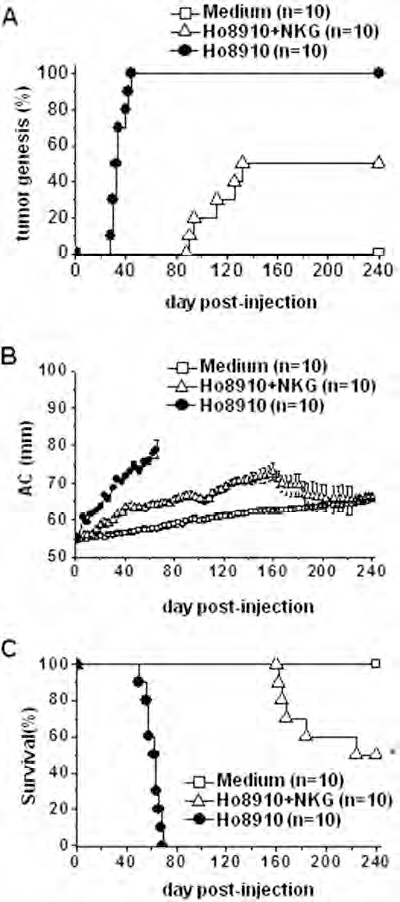
Irradiated NKG cells inhibit the growth of ovarian tumors in xenograft mice. Ho8910 cells (2 × 106) alone or mixed with 8 Gy irradiated NKG cells (1 × 107) (E:T = 5:1) were injected IP into nude mice in a total volume of 200 μl. The medium was used as the control. (A) Mice with palpable tumor were calculated and the tumorgenesis (%) is shown. (B) The abdomen circumference (AC) of each mouse was measured. Data are shown as mean ± SEM in each group. (C) The survival rates were measured. There were 10 nude mice in each group. ∗p < 0.05 versus the corresponding control group.
Clinically, adoptive cell transfer could be performed on tumor patients. Therefore, NKG cell treatment was performed on mice injected IP with Ho8910 cells (2 × 106) for 21 days. As shown in Figure 9A, NKG cell treatment significantly inhibited the tumorgenesis (from 100% to 62.5%) and delayed the time it took to develop a palpable tumor (from 28–38 days to 56–66 days). The severity of ovarian cancer was attenuated, as indicated by the significantly decreased AC (Fig. 9B). At 138 days after tumor cell implantation, 37.5% of the mice in the NKG cell treatment group showed no palpable tumor. The mean survival time was prolonged from 64.6 to 119.6 days (Fig. 9C). Further, the therapeutic efficacy of NKG cells was also examined in advanced ovarian cancer. Thirty-five days following Ho8910 cell (2 × 106) injection, the mice with palpable tumors were selected. NKG cell treatment attenuated the severity of ovarian cancer, shown by the decreased AC in the early stage of treatment (Fig. 9D). The mean survival time was prolonged from 64.2 to 93.6 days in NKG transferred group (Fig. 9E). A number of NKG cells could be detected in the ovarian tumor tissue of NKG-treated mice, demonstrating that irradiated NKG cells could migrate into the ovarian tumor tissue in vivo during adoptive cell therapy (Fig. 10). Together, these results suggest use of the new human NK cell line, NKG, represents a novel immunotherapy for the control of human ovarian cancer.

Therapeutic effects of adoptive transfer of irradiated NKG cells on the ovarian tumors in a mouse xenograft model. The nude mice were IP challenged with Ho8910 cells (2 × 106) to establish the tumor model. (A–C) Adoptive transfer of NKG cells was performed 21 days after tumor cell implantation. Eight Gy irradiated NKG cells (6.67 × 107/ml) were injected IP (10 μl/g body weight) into the mice three times per week for 3 weeks. Medium was used as the control. The mice were observed for ovarian tumor progression. (A) Mice with palpable tumor were calculated and the tumorgenesis (%) is shown. (B) The abdomen circumference (AC) of each mouse was measured. Data are shown as mean ± SEM in each group. (C) The survival rates were measured. There were 8 nude mice in each group. ∗p < 0.05 versus the corresponding control group. (D, E) Ten nude mice with ascitic fluid (palpable tumor) were selected 35 days after tumor cell implantation. Adoptive transfer of NKG cells was performed as described above. (D) The abdomen circumference (AC) of each mouse was measured. Data are shown as mean ± SEM in each group. (E) The survival rates were measured. There were five mice in each group. ∗p < 0.05 versus the corresponding control group.

Migration of irradiated NKG cells into the ovarian tumor tissues in a mouse xenograft model. The nude mice were IP challenged with Ho8910 cells (2 × 106) to establish the tumor model. Eight Gy irradiated NKG cells (6.67 × 107/ml) were injected IP (10 μl/g body weight) into the mice with palpable tumors three times per week for 1 week. Twelve hours after cell transplantation, mice were sacrificed, and CD56+ cells (NKG cells) were detected in the tumor tissue. Medium was used as the control. There were five mice in each group.
Discussion
Generation of well-characterized NK cell line will allow us to better understand NK cell immunobiology and its potential in enhanced immunity against cancers. In this study, a novel human NK cell line, named NKG, was established from a Chinese male patient with rapidly progressive non-Hodgkin's lymphoma. This NK cell line is the first to be established in China. With the characteristics of activated CD56bright NK cells, NKG cells were highly responsive to exogenous stimulation by producing high levels of cytokines. They were also highly cytotoxic against a series of tumor cell lines, most notably the ovarian cancer cells. Successful immunotherapy of human ovarian cancer with irradiated NKG cells was firstly demonstrated in a xenograft mouse model.
To date, NK-92 cells are the most potent and suitable NK cell line for potential clinical use. They are characterized by an almost complete lack of KIRs and conserved ability to induce cytotoxicity via the perforin and granzyme B-mediated pathway. NK-92 cells were established from a 50-year-old male patient with rapidly progressive non-Hodgkin's lymphoma (24,39,41). The growth characteristics of NKG cells were similar to NK-92 cells in an IL-2-dependent manner (100 IU/ml). However, the doubling time of NKG cells (2–3 days) was longer than that of NK-92 cells (24–36 h) (41). NKG cells were confirmed free of pathogen infection or contamination with virus, bacteria, fungi, or mycoplasm (Fig. 1C). EBV infection was excluded with genomic DNA analysis much like that performed in NK-92 cells. The morphology of NKG cells was identical to that of NK cells, as a population of LGL (Fig. 1B, C). The NKG cells were characterized as CD56+, CD16-, CD27-, CD3-, αβTCR-, γδTCR-, CD4-, CD8-, CD19-, CD161-, and CD45+ (Fig. 2), which is similar to NK-92 cells (12). Compared with NK-92 cells (data not shown), the expression levels of NKp30, NKp44, NKG2D, NKG2C, granzyme B, perforin, and IFN-γ were significantly higher in NKG cells. The expression of CD11b and CD11c was also higher in NKG cells. The inhibitory receptors including CD94/NKG2A and KIRs showed similarly expression in both NKG and NK-92 cells. The balance of the activating and inhibitory signals transmitted by cell surface receptors is critical for the natural killing of NK cells against target cells (26). NKG cell cytotoxicity against tumor cells, including MHC-I cell lines and MHC-I + cell lines, was significantly higher than that of NK-92, NKL, and YT cells (Fig. 3C). The higher expression levels of NKG2D and NKp30 in NKG cells may explain the differences in the cytotoxicity against tumor cells. The Fc-receptor CD16, which usually mediates antibody-dependent cytotoxicity (ADCC), was absent on NKG cells (Fig. 2), which was also absent on NK-92 cells (41). KHYG-1, another NK cell line, induces apoptosis of tumor cells by the granzyme M/perforin pathway in addition to the activation of NKp44. Greater cytotoxicity of KHYG-1 versus NK-92 cells was observed (37). This study did not compare the cytotoxicity of NKG cells to that of KHYG-1 cells.
The safety of NK cells must be considered due to their potential for adoptive transfer and immunotherapy for tumors. Irradiation of NK-92 cells with 5 Gy prevents further cell division, whereas the substantial cytotoxicity can be maintained with up to 10 Gy irradiation for 48 h in vitro (29,38). Irradiation (at least 10 Gy) of KHYG-1 cells inhibits their proliferation but does not diminish their enhanced cytolytic activity against several tumor targets, which suggests that KHYG-1 cells may be a feasible immunotherapeutic agent in the treatment of cancers (36). In our study, irradiation of NKG cells with 8 Gy inhibited their proliferation and maintained their cytotoxicity against tumor cells within 48 h (Fig. 5A–C), indicating that 8 Gy would be an effective dose for NKG cell irradiation. As with NK-92 cells, the safety aspects of NKG cells were examined in vivo (Fig. 5D, E).
NK-92 cells have achieved FDA approval to be tested in patients with advanced malignant melanoma and renal cell carcinoma in Europe and US (41). Immunotherapy of malignant melanoma with NK-92 cells was firstly demonstrated in a severe combined immunodeficient (SCID) mouse model (40). In that study, mice challenged with MEWO melanoma cells were administered IV with NK-92 cells, which resulted in a 1.5–2.5-fold increase in average length of survival, and reduced primary tumor size by 30–75% (40). Our study used a nude mouse xenograft model implanted with Ho8910 ovarian cancer cells. Adoptive NKG cell transfer, in combination with ovarian cancer cell implantation (IP), significantly inhibited the tumor growth, decreased the mortality rate, and prolonged survival (Fig. 8). The mean survival time increased from 61.5 to 210.2 days (greater than threefold). As for the xenografted mice without palpable tumor in early stage, adoptive NKG cell transfer therapy was much effective, significantly inhibiting the tumor growth (from 100% to 62.5%), prolonging the survival time (about twofold) and decreasing the mortality rate. At 138 days after tumor cell implantation, 37.5% of the mice were still alive and without palpable tumor (Fig. 9A–C). When the xenografted mice were in advanced stage with palpable ovarian tumors, adoptive NKG cell transfer also significantly inhibited the tumor growth and prolonged the survival time (about 1.5-fold) (Fig. 9D, E). In the field of ovarian cancer immunotherapy, it is difficult to assess tumor load in the peritoneal cavity. The effectiveness of NKG cell immunotherapy was demonstrated in our mouse model, indicating its potential for clinical use.
Ovarian cancer is responsible for the highest mortality rate in patients with gynecologic malignancies (19). The development of novel immunotherapies is impending to complement the traditional treatments with surgery and chemotherapy for the control of ovarian cancer. Adoptive cell transfer, including LAK, CIK, and tumor infiltrating lymphocytes (TILs), has been performed for ovarian cancer therapy (1,4,6,8,14,18,22,27). Inhibition of the tumor cell growth in the early stages of CIK cell therapy was observed, but the limitations of the reinfusion of ex vivo expanded cells would hamper their clinical use (6,22,27). Clinical trials involving the adoptive transfer of TILs have been performed, and such treatment may be an effective adoptive immunotherapy approach for the control of ovarian cancer (1,14,18). However, the difficulty in the achievement of TILs from the patients is a barrier to this type of treatment. Chimeric NKG2D expressing T cells could eliminate immunosuppression and activate immunity within the ovarian tumor microenvironment (2,3). Unfortunately, a phase I study on adoptive immunotherapy using gene-modified T cells for ovarian cancer demonstrated that these cells do not persist in large numbers in the long term, and no reduction in tumor burden was observed in the patients (21). Therefore, the clinical use of an NK cell line, such as NKG, deserves further clinical testing. High expression of NKG2D ligands, such as MHC class I chain-related gene A/B (MICA/B) and UL16 binding proteins (ULBPs), has been detected in ovarian cancer cells, and strong expression of ULBP2 is an indicator of poor prognosis (3,28). Human ovarian carcinoma also expresses lymphocyte effector cell toxicity-activating ligand (Letal), a novel NKG2D ligand (9,10). Chronic ligand exposure might induce alterations in NK cell receptor expression, which may hamper immune surveillance and promote tumor progression (5). It has been speculated that ovarian tumor cells expressed high levels of NKG2D ligands and NKp30 ligand (not known until now); however, the NK cell-mediated rejection of ovarian cancer was limited by perturbed NKG2D and NKp30 expression on tumor-associated NK cells in patients. Thus, adoptive transfer of NKG cells with high levels of NKG2D and NKp30 would be highly cytotoxic against ovarian tumor cells in vivo. In addition, ovarian tumor cell growth, even in advanced stages, is almost always restricted to the peritoneal cavity (11). Therefore, repeated IP injections of NKG cells would be practical. In the peritoneal cavity, NKG cells were cytotoxic to ovarian tumor cells, and it was observed that NKG cells migrated into the tumor tissues (Fig. 10). Intraperitoneal injection is more convenient and safe than cell transfer via IV injection.
The NKG cell line, which was established in China, would be more appropriate for use in Chinese patients. The HLA genotype of NKG cells has been assessed and the common HLA compatibility to Han population in China demonstrated (data not shown). Furthermore, NKG cell line can be genetically modified, which provides more opportunities for future therapeutic trials. Of course, crucial questions such as conditioning of patients prior to therapy, the clinical context of therapy, the criteria for patient selection, and strategies for the cell transfer will require consideration when developing successful NKG cell immunotherapy for human cancer.
Footnotes
Acknowledgments
Financial support from National 863 High-Tech Grant (2007AA021109), Important National Science & Technology Specific Projects (2008ZX10002-023), Knowledge Innovation Project of the Chinese Academy of Sciences (KSCX1-YW-22), Ministry of Public Health Special Fund for Healthy Industry (200902002-2), and the Science and Technology Key Project of AnHui province (08010302101). The authors declare no conflicts of interest.
