Abstract
Tissue necrosis resulting from critical limb ischemia (CLI) leads to amputation in a significant number of patients. Autologous cell therapy using angiogenic cells such as endothelial progenitor cells (EPCs) holds promise as a treatment for CLI but a limitation of this treatment is that the underlying disease etiology that resulted in CLI may also contribute to dysfunction of the therapeutic EPCs. This study aimed to elucidate the mechanism of EPC dysfunction using diabetes mellitus as a model and to determine whether correction of this defect in dysfunctional EPCs ex vivo would improve the outcome after cell transplantation in the murine hind limb ischemia model. EPC dysfunction was confirmed in a homogenous population of patients with type 1 diabetes mellitus and a microarray study was preformed to identify dysregulated genes. Notably, the secreted proangiogenic protein osteopontin (OPN) was significantly downregulated in diabetic EPCs. Furthermore, OPN-deficient mice showed impaired recovery following hind limb ischemia, suggesting a critical role for OPN in postnatal neovascularization. EPCs isolated from OPN KO mice showed decreased ability to adhere to endothelial cells as well as impaired angiogenic potential. However, this dysfunction was reversed upon exposure to recombinant OPN, suggesting that OPN may act in an autocrine manner on EPCs. Indeed, exposure of OPN knockout (KO) EPCs to OPN was sufficient to induce the secretion of angiogenic proteins (IL-6, TGF-α, and FGF-α). We also demonstrated that vascular regeneration following hind limb ischemia in OPN KO mice was significantly improved upon injection of EPCs preexposed to OPN. We concluded that OPN acts in an autocrine manner on EPCs to induce the secretion of angiogenic proteins, thereby playing a critical role in EPC-mediated neovascularization. Modification of cells by exposure to OPN may improve the efficacy of autologous EPC transplantation via the enhanced secretion of angiogenic proteins.
Keywords
Introduction
Critical limb ischemia (CLI) is the end stage of peripheral vascular disease (PAD) and leads to ischemia, resulting in pain at rest and tissue loss. Improvement of blood flow often requires surgical or endovascular revascularization. However, in over one third of patients revascularization either fails or is not possible due to comorbid complications (1). In these patients major amputation is often necessary and results in a 5-year survival rate of less than 30%. Thus, the need to develop new therapies is imperative to improving both the quality of life and survival rate of patients. Currently, bone marrow-derived stem and progenitor cells show some promise as a therapeutic for CLI [reviewed in (21)]. Endothelial progenitor cells (EPCs) show particular promise as a therapeutic agent as numerous studies support their role in augmenting angiogenesis (14). However, a potential limitation of autologous EPC therapy is that the disease state that results in CLI may also adversely affect EPC function and thus limit therapeutic efficacy. Indeed, in diabetes mellitus, where CLI is a common complication, patients have fewer EPCs than healthy volunteers (9,23,31), while patients with type 2 diabetes complicated by peripheral vascular disease have an even further reduction in the number of EPCs and in these patients EPC number inversely correlates with glycemic control (9).
If a simple reduction in EPC number is the primary issue then autologous cell therapy, where an infusion of cells is delivered directly to the diseased area, should be sufficient to restore normal function. However, the issue is more complex as studies have revealed that EPCs from diabetic patients and animals are not only decreased in number but are also dysfunctional. EPCs from patients with type 2 diabetes mellitus (T2DM) exhibit decreased adhesion to activated endothelial cells and to matrix molecules including collagen and fibronectin, and EPCs derived from patients with both T1DM and T2DM are impaired in their ability to form tubules in angiogenesis assays (23,31). Similarly, bone marrow-derived mononuclear cells derived from streptozotocin-induced diabetic mice yield fewer EPCs and they exhibit dysfunction, as they are less likely to incorporate into newly forming vascular structures in a Matrigel plug assay than those from healthy mice (29). Beyond effects mediated directly by the cells, it has also been noted that conditioned media from T1DM EPCs has a reduced capacity to mediate angiogenesis and may actually inhibit angiogenesis in vitro (23). Taken together, these data suggest that the use of autologous cells may be limited by the inherent dysfunction of the transplanted cells. Therefore, we used EPCs from type 1 diabetic patients as a model for EPC dysfunction with the goal of elucidating techniques to manipulate the EPCs to increase their therapeutic potential in a model of cell transplantation.
We used microarray analysis to identify genes that are dysregulated in diabetic EPCs and determine if these genes contribute to EPC-mediated angiogenesis. The expression of osteopontin (OPN) was found to be decreased in diabetic EPCs. OPN is a secreted glycoprotein that is involved in cell migration, cell survival, regulation of immune cell function, inhibition of calcification, and control of tumor cell phenotype (5,10,11,32). Interestingly, OPN has been implicated in the process of neovascularization in various cancer models and peripheral ischemia (10,11). OPN overexpression is associated with the increased invasiveness of cancers while inhibition of OPN suppresses tumorigenicity (12,25,27,30,35). Studies in noncancerous animal models also support a role for OPN in neovascularization (7,8).
Despite these studies, the precise role of OPN in vessel formation is poorly understood. Further, the relationship between OPN and EPC function is currently unknown. Therefore, we sought to elucidate the underlying cause of EPC dysfunction in patients with type 1 diabetes mellitus, a model for EPC dysfunction, and assess the putative role of OPN in ischemia and neovascularization. We found that recovery from hind limb ischemia is impaired in OPN knockout (KO) mice and we went on to demonstrate that EPCs from OPN knockout mice are dysfunctional. We found that OPN may act on EPCs in an autocrine manner, resulting in the secretion of additional angiogenic factors. Furthermore, in a murine model of hind limb ischemia in the OPN knockout mouse, a model of EPC dysfunction, injection of OPN KO EPCs preincubated with OPN resulted in improved blood flow to the injured limb. These data indicate that OPN is a potential target for the regulation of the EPC-mediated angiogenic response to ischemic vascular injury and suggest a novel method to enhance therapeutic efficacy of autologous EPC transplantation in peripheral vascular disease.
Materials and Methods
Subject Recruitment
Patients with poorly controlled type 1 diabetes (as defined by HbA1/c > 10%) who were on insulin for more than 1 year and not on any other medications were recruited from the Diabetes Day Centre, University College Hospital Galway, Ireland. Patients with micro- or macrovascular complications were excluded from the study. Microvascular complications were defined as the presence of microalbuminuria, diabetic retinopathy, and neuropathy. Macrovascular complications were defined as the presence of any previous history of acute coronary syndrome, peripheral vascular disease, and cerebrovascular disease. All subjects gave informed consent and were recruited with ethical approval from University College Hospital Galway Clinical Research Ethics Committee and the investigation conforms to the principles outlined in the Declaration of Helsinki.
Isolation of EPCs
EPCs were isolated according to previously described techniques with some modifications (16). Briefly, peripheral blood mononuclear cells (PBMNCs) were isolated by Ficollpaque (GE Healthcare) density centrifugation. After purification with three washing steps, 10 × 106 or 2 × 106 cells (for cell quantification) were plated on fibronectin-coated six-well plates or four-well glass slides, respectively. Cells were incubated for 4 days at which point the supernatant was discarded, cells were washed with PBS, and fresh endothelial basal media (EBM2) was added and changed daily until day 7, when EPCs were used.
Adhesion to Endothelial Cells
Adhesion to endothelial cells was determined according to previously described techniques (16). In brief, a monolayer of human umbilical vein endothelial cells (HUVECs; Lonza) was prepared 48 h before the assay by plating 2 × 105 cells (passage 5 to 10) in each well of a four-well glass slide. Twelve hours before the assay the HUVECs in some experiments were treated with tumor necrosis factor-α (TNF-α; BD Biosciences) 1 ng/ml or media. After the 12 h, 1 × 105 EPCs labeled with 1,1′- dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine iodide (DiI) were added to the HUVECs and incubated for 3 h. Nonattached cells were then gently removed with PBS and adherent cells were fixed with 4% paraformaldehyde (PFA) and counted by a blinded observer.
Matrigel Tubule Assay
The assay was performed according to previously described techniques (16). Matrigel (BD Biosciences) was thawed on ice and placed in four-well glass slides and incubated at room temperature for 30 min to allow for solidification. EPCs were incubated at 37°C for 24 h with or without 5 μg/ml OPN (Sigma) and labeled with DiI. Labeled EPCs (2 × 104) were coplated with 4 × 104 HUVECs and tubule formation was determined at 12 h for human EPCs and 24 h for murine EPCs and conditioned media. Tubule formation was defined as a structure exhibiting a length four times its width. The number of tubules formed was assessed by a blinded observer.
Alloxan-Induced Diabetic Rabbit Model
Diabetes was induced in male New Zealand White rabbits using intravenous injection of alloxan (150 mg/kg). Rabbits with plasma glucose of >22 mmol/L were included for this study. Phlebotomy was performed via the marginal artery under anesthesia. This study was approved by the National University Ireland, Galway (NUIG) Animal Care and Use Committee.
Total RNA Extraction
Total RNA was isolated from day 4 EPCs using RNA Mini Kit (Qiagen) as described by the manufacturer. The concentration of isolated total RNA was analyzed using NanoDrop counter. QuantIt DNA, High Sensitivity kit (Invitrogen) was used to detect the presence of any genomic DNA in the total RNA samples.
Microarray Analysis
The GeneChip, HG-U133 Plus 2.0 (Homo sapiens) in situ oligonucleotide array (Affymetrix. Inc., Santa Clara, CA) was used. The GeneChip covers the transcribed human genome, comprised of 54,675 probe sets and ESTs. The CEL file, which stores the results of the intensity calculations on the pixel values of probes, was preprocessed as the first-order data analysis using Microarray Suite (MAS) algorithm, Affymetrix GeneChip® Operating Software (GCOS). The data sets were normalized by per-chip, per-gene normalization method using GeneSpring bioinformatics software (GeneSpring GX 7.3.1, Silicon Genetics, Redwood City, CA). Gene filtering on normalized intensity followed by one-way ANOVA, multiple testing correction, Benjamini and Hochberg false discovery rate, and Tukey's post hoc testing based on values of p < 0.05 and fold changes >2.0-fold was performed to generate list of gene expression profile.
Primer Sequences
Primers for OPN (CAGAGCACAGCATCGTCGG; GGCAAAAGCAAATCACTGCAA) and the housekeeping gene, cyclophilin A (TGCTGGACCCAACA CAAATG; CATGCCTTCTTTCACTTTGCC) (Sigma Genosys) were designed using PrimerExpress software.
Real-Time PCR
The ABI Prism 7000 Sequence Detection System (Applied Biosystems) with One Step QuantiTect SYBR Green PCR kit (Qiagen) was used for all PCR experiments. The reactions were performed according to the manufacturer's instructions with minor modifications. A sample volume of 25 μl was incubated for 30 min at 50°C; 95°C for 15 min; followed by 40 cycles of 15 s at 95°C and 30 s at 60°C. The dissociation curves were generated using a temperature range between 60°C and 95°C. All reactions were further analyzed by electrophoresis on 2% agarose gels stained with SyBrGreen dye to confirm the PCR results. Each sample was analyzed in triplicate.
Isolation and Identification of Murine Endothelial Progenitor Cells
The hind limbs were separated from the body and the epiphyses were removed from both ends. The marrow was flushed from the bone in 5 ml of endothelial growth medium 2 (EGM-2) and the media with bone marrow was collected and centrifuged at 1,700 rpm for 25 min. The supernatant was removed and cells were resuspended in 1 ml of red cell lysis buffer (Sigma). Cells were collected at 1,700 rpm for 5 min and washed twice in PBS. Cells were resuspended in 4 ml of EBM2 and plated on a fibronectin-coated plate. EPCs were identified by direct fluorescent staining to detect fluorescein isothiocyanate (FITC)-Ulex europaeus agglutinin-1 lectin (Sigma) and DiI-acetylated low density lipoprotein (acLDL) (Invitrogen) as previously described (13).
Murine Hind Limb Ischemia Model
C57BL/6 (strain code 027) (wild-type; WT) and OPN-/OPN-[B6.12956(Cg)-spp1tm1Blh/J] (OPN-KO) [B6.12956(Cg)-spp1tm1Blh/J] mice were purchased from Charles River Lab and Jackson Laboratory, respectively. OPN KO and WT mice aged between 8 and 10 weeks of age were used. The mice were housed at the animal facility in the Regenerative Medicine Institute (REMEDI), NCBES, NUIG. All procedures were approved by the Minister of Health and Children under the Cruelty to Animals Act, 1876. Unilateral hind limb ischemia was created in WT and OPN KO mice as previously described (19). In brief, the left femoral artery and its associated branches were isolated, ligated, and excised. The contralateral hind limb served as an internal control within each mouse. Animals were anesthetized with ketamine and xylazine and maintained with isoflurane. In the second set of experiments, injection of 1 × 106 EPCs resuspended in 200 μl of EBM2 (with no supplements added) was performed. There were six injections into the thigh muscle at the time of surgery.
Laser Doppler Blood Flow Assessment (LDBF)
Animals were anaesthetized and placed on a heating table at 37°C before scanning to minimize temperature variation. The blood flow in the foot pads of both limbs was measured using a laser Doppler blood flow analyzer (PeriScan PIMII, Perimed Inc.) immediately before and after surgery as well as on postoperative days 7, 14, and 28. Microvasulature blood flow was displayed in a color-coded image. Analysis was performed by comparing blood flow in the ischemic limb (left) to the nonischemic limb (right) to further minimize variations due to ambient light and temperature.
Collection of Conditioned Media
EPCs were grown as described until day 6. On the 6th day of culture the media was removed and cells were washed three times in PBS. One milliliter of fresh EBM2 was then added to each well of a six-well plate and the plates were then incubated for 24 h at which point the media was collected for use in the Matrigel tubule assay. Conditioned media for the angiogenesis array was obtained by incubating day 6 EPCs in growth factor-free EBM2 for 48 h.
Angiogenesis Array
Conditioned media (100 μl) was hybridized to each membrane of an antibody-sandwich angiogenesis array (Panomics) according to manufacturer's guidelines. Expression intensities were determined with Image J and background values were subtracted. All data were normalized to the positive control spots to control for differences between arrays.
Statistical Analysis
Data are presented as mean ± SEM. Statistical analysis was performed by unpaired Student's t-test for comparisons between two groups and by one-way analysis of variance followed by Bonferroni correction for comparison between more than two groups. A value of p < 0.05 was considered significant.
Results
EPC Dysfunction in Type 1 Diabetes Mellitus
We first sought to determine if EPC number and function were impaired in a homogenous population of patients with T1DM that did not have micro- or macrovascular complications. EPCs were isolated from four patients with T1DM with a mean HbA1/c of 12.5 ± 4.3% and a mean duration of diabetes of 7 ± 2.3 years, and four gender- and age-matched (22.3 ± 4.3 vs. 22.8 ± 2.1 years old) healthy volunteers were recruited (Fig. 1). EPCs from patients with T1DM were reduced in both number and function when compared to EPCs from healthy volunteers (Fig. 2).

Human endothelial progenitor cell (EPC) characterization. EPCs were characterized by their ability to uptake acetylated low-density lipoprotein (LDL) and Ulex europaeus agglutinin-1 (UEA-1) lectin. (A) Brightfield picture of EPCs. (B) Uptake of acetylated LDL and (C) UEA-1 lectin were observed. (D) Colocalization shows that greater than 90% of the cells were dual stained for both acLDL and UEA-1 lectin. Scale bar: 100 μm.
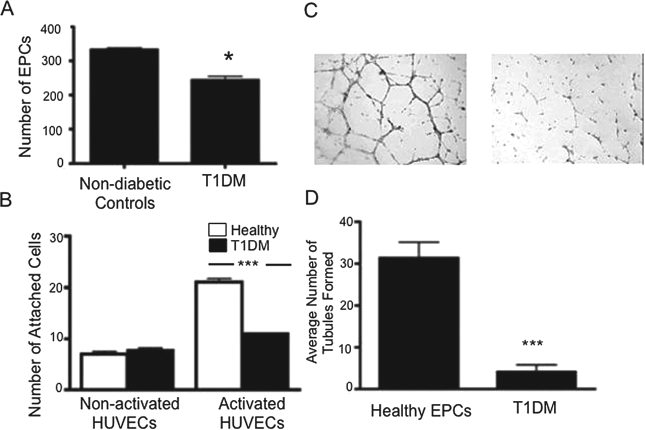
EPCs from patients with type 1 diabetes mellitus (T1DM) are reduced in number and dysfunctional. (A) EPCs were isolated from the peripheral blood of patients with T1DM, cultured, and counted 4 days after plating on fibronectin-coated dishes. Patients with T1DM had fewer EPCs than nondiabetic controls (n = 4), *p < 0.05. (B) Adhesion to tumor necrosis factor-α (TNF-α) activated but not nonactivated endothelial cells was impaired in diabetes mellitus (n = 3), ***p < 0.001. (C) Representative images from a Matrigel tubule assay showing EPCs from patients with T1DM had a reduced ability to support tubule formation. Scale bar: 20 μm. (D) Quantification of images shown in (C). T1DM cells have a reduced ability to induce angiogenesis in vitro (n = 4), ***p < 0.001. HUVEC, human umbilical cord vein endothelial cell.
OPN in EPC Dysfunction in DM
Having identified diabetes as a model for EPC dysfunction, we sought to elucidate the underlying cause of this dysfunction. Therefore, a microarray was performed on EPCs obtained from diabetic and control subjects and the top 10 genes up- and downregulated are shown (Fig. 3A, Tables 1 and 2). Three hundred and sixty genes were found to be upregulated in T1DM EPCs and 15 genes downregulated twofold or greater. OPN was found to be markedly reduced in diabetic EPCs and this was confirmed by qRT PCR where OPN was downregulated 26.89-fold in diabetic EPCs.
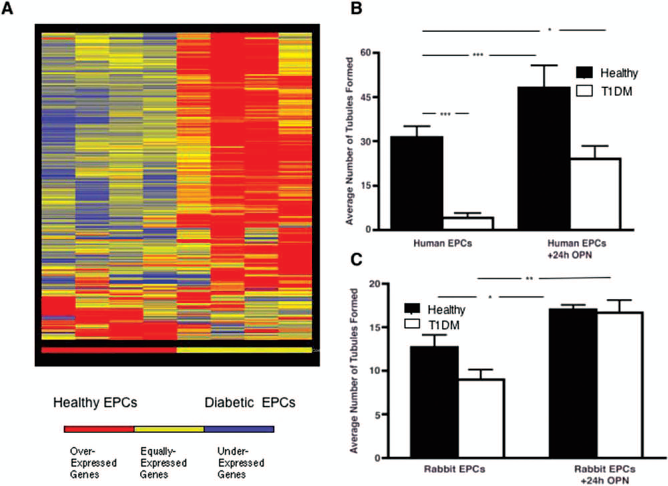
Decreased osteopontin (OPN) expression is observed in diabetic EPCs. (A) A heat map showing the gene expression pattern of genes with a greater than twofold difference in EPCs from healthy controls (n = 4) and EPCs from type 1 diabetes mellitus (T1DM). Three hundred and sixty genes were upregulated in T1DM and 15 were downregulated. OPN was identified as having a 9.6-fold decrease in T1DM EPCs. (B) To test if OPN will restore function to diabetic EPCs, EPCs from both healthy and diabetic subjects were exposed to recombinant OPN. Exposure to OPN was able to significantly restore tubule formation mediated by EPCs in both human and in New Zealand white rabbits (C) (n = 3). *p < 0.05; **p < 0.01; ***p < 0.001.
Top 10 Genes Underexpressed in Type 1 Diabetes Mellitus Endothelial Progenitor Cells (EPCs) Compared to Healthy Control EPCs

SPP1/OPN, secreted phosphoprotein 1/osteopontin; CD109, cluster of differentiation 109; KIAA0555, Janus kinase and microtubule-interacting protein 2; NDN, necdin; CLDN23, claudin 23; B4GALT1, β-1,4-galactosyltransferase 1; COL23A1, collagen, type XXIII, α1; KIAA1706, endonuclease/exonuclease/phosphatase family domain containing 1; LTBP2, latent transforming growth factor-β binding protein 2.
Top Genes Overexpressed in Type 1 Diabetes Mellitus EPCs Compared to Healthy Control EPCs
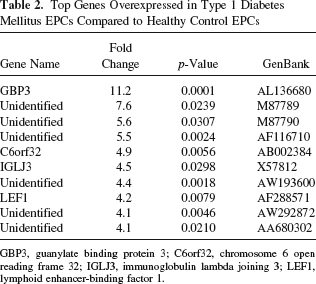
GBP3, guanylate binding protein 3; C6orf32, chromosome 6 open reading frame 32; IGLJ3, immunoglobulin lambda joining 3; LEF1, lymphoid enhancer-binding factor 1.
To further address the potential importance of OPN in EPC-mediated angiogenesis, diabetic and healthy EPCs were incubated with OPN and a Matrigel tubule assay was performed. Impairment in tubule formation from diabetic EPCs (compared to nondiabetic EPCs) was reversed upon exposure to recombinant OPN. Furthermore, this finding was also confirmed with EPCs from an alloxan-induced diabetic rabbit model: tubule formation was restored to nondiabetic levels after exposure to recombinant OPN protein (Fig. 3C).
Role of OPN in Angiogenesis
Having observed that EPCs from patients with T1DM were dysfunctional and expressing decreased levels of OPN, we went on to explore the role of OPN in angiogenesis and vascular wound healing. Hind limb ischemia was induced in OPN KO mice and laser Doppler blood flow analysis was performed. Postoperative results in the ischemic limb demonstrate that blood flow was restricted to about 15% of the flow in the control limb in both WT and KO animals. By day 28 blood flow in the ischemic footpad of WT mice was restored to levels found in the control limb; however, blood flow was restored to only about 23% of WT levels in the KO animals (Fig. 4A, B). In addition to decreased blood flow, we also found that microvascular density was decreased in the ischemic limbs of OPN KO mice when compared to the ischemic limb of WT mice (Fig. 4).

Absence of osteopontin (OPN) impairs neovascularization. Hind limb ischemia was induced in wild-type (WT) and OPN knockout (KO) mice. Laser Doppler blood flow (LDBF) analysis was performed at days 7, 14, and 28 and animals were sacrificed on day 28. (A) Representative Doppler images before and after induction of ischemia. Images at day 28 show an improvement in blood flow in WT animals but not in OPN KO mice. (B) Quantitative analysis of the laser Doppler images displayed a significant decrease in recovery in OPN-deficient mice. Results are shown as the ratio of the ischemic limb to the control limb. (C) Microvascular density was decreased in the ischemic limb of OPN KO mice when compared to the ischemic limb of WT mice. No difference was noted in microvascular density in the nonischemic limbs between OPN KO and WT animals (n = 5). *p < 0.05.
Role of OPN in EPC Function
We next sought to determine if there was a direct link between the impaired angiogenesis observed in KO mice and EPC function. We first determined the ability of WT and KO EPCs to adhere to activated endothelial cells. EPCs were obtained from the bone marrow of the mice and dual staining of DiI-acetylated LDL and FITC-lectin were confirmed (Fig. 5). EPCs from OPN KO mice displayed impaired adherence when compared to WT EPCs (Fig. 6A). Furthermore, when OPN KO EPCs were compared to WT EPCs in a Matrigel tubule assay we observed that WT EPCs induced significantly more tubule formation than KO EPCs (Fig. 6B). Taken together, these data suggest that OPN KO EPCs are impaired in their ability to adhere to the site of injury as well as their ability to induce angiogenesis. Interestingly, although the DiI-labeled EPCs did appear to associate with the tubules formed in the angiogenesis assay they did not appear to actually incorporate into the tubule structures. Thus, we next questioned if the EPCs need to be physically present to enhance angiogenesis or if they are secreting factors that act in a paracrine manner to enhance angiogenesis.

Murine endothelial progenitor cell (EPC) characterization. EPCs were characterized by their ability to uptake acetylated LDL and UEA-1 lectin. (A) Nuclei were stained with Hoescht. (B) Uptake of acetylated LDL and (C) UEA-1 lectin was observed. (D) Colocalization shows that greater than 90% of the cells were dual stained for both acLDL and UEA-1 lectin. Scale bar: 20 μm.
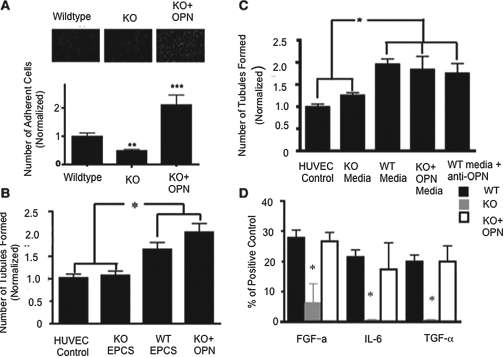
OPN exerts autocrine effects on EPCs inducing the secretion of angiogenic factors. (A) To assess the ability of EPCs to adhere to sites of injury, adhesion to TNF-α activated endothelial cells was measured in murine EPCs. EPCs from OPN knockout (KO) mice did not adhere as well as wild-type (WT) EPCs. Exposure to OPN increased the ability of KO EPCs to adhere, suggesting that OPN plays a role in adhesion (n = 3). **p < 0.01; ***p < 0.001. (B) Angiogenesis induced by murine EPCs was determined with a Matrigel assay. WT EPCs induced the formation of significantly more tubules than OPN KO EPCs. Exposure to OPN reversed this dysfunction in OPN KO EPCs. All tubule counts were normalized to the baseline levels of tubules formed with HUVECs alone (n = 3). *p < 0.05. (C) Angiogenesis induced by conditioned media from murine EPCs was measured with a Matrigel tubule assay. Conditioned media from WT EPCs induced tubule formation, suggesting angiogenesis may be mediated by the secretion of factors from the EPCs. Neutralization of OPN did not dampen tubule formation, indicating other factors may be increasing angiogenesis (n = 3). *p < 0.05. (D) Protein secretion was measured with an angiogenesis array. Three proteins that had decreased expression in OPN KO cells were identified and expression of all three protein expression levels was returned to WT levels upon exposure to OPN, suggesting OPN may induce the secretion of angiogenic factors (n = = 3). *p < 0.05. FGF, fibroblast growth factor; IL-6, interleukin-6; TGF-α, transforming growth factor-α.
EPCs Enhance Angiogenesis in a Paracrine Manner
To determine if EPCs function to enhance angiogenesis in a paracrine manner, conditioned media (CM) from WT and OPN KO EPCs was obtained and incubated with HUVECs in the Matrigel tubule assay. CM from WT but not OPN KO EPCs induced a significant angiogenic response (Fig. 6C). To determine if OPN was responsible for this paracrine effect CM was collected from WT cells and incubated with an OPN neutralizing antibody. If OPN induces tubule formation, its neutralization in the CM should abrogate the angiogenic response. Surprisingly, incubation with the OPN neutralizing antibody did not significantly decrease tubule formation (Fig. 6C), suggesting that OPN does not enhance angiogenesis directly. Thus, we went on to explore other possible roles OPN could play in mediating angiogenesis.
OPN Acts on EPCs in an Autocrine Manner to Enhance Angiogenesis
An alternate mechanism for OPN-mediated angiogenesis is via an autocrine effect on EPCs to secrete angiogenic factors. Thus, incubation with exogenous OPN should be sufficient to elicit the secretion of proteins that enhance the angiogenic response. Indeed, we found that incubating EPCs with recombinant OPN was sufficient to enhance tubule formation to levels obtained from WT cells (Fig. 6B). In fact, incubation with recombinant OPN-induced KO cell adhesion to activated endothelial cells to levels surpassing WT cells (Fig. 6A). Furthermore, if KO cells are incubated with OPN for 24 h, followed by removal of the OPN-containing media, and fresh media (without OPN) is added and collected after 24 h this media is still able to enhance angiogenesis, supporting the possibility that OPN is inducing the secretion of angiogenic proteins from EPCs (Fig. 6C). To further address this, a chemiarray was used to determine if there was differential secretion between WT EPCs, KO EPCs, and KO EPCs exposed to OPN. The secretion of three angiogenic proteins, fibroblast growth factor-α (FGF-α), interleukin 6 (IL-6), and transforming growth factor-α (TGF-α), were found to be downregulated from KO cells when compared to WT (Fig. 6D). Interestingly, exposure of KO cells to OPN restored the secretion of these proteins to WT levels. Taken together, these data suggest that OPN acts in an autocrine manner on EPCs, resulting in the secretion of angiogenic proteins, which then act to enhance both the ability of the EPCs to adhere to sites of injury and to induce angiogenesis.
Exposure to OPN Enhances the Angiogenic Potential of EPCs in OPN KO Mice
Although it appears that exposure to recombinant OPN induces the secretion of angiogenic proteins and enhances angiogenesis, it is unclear if this is sufficient to restore the angiogenic potential of EPCs in vivo. Therefore, we induced hind limb ischemia in OPN KO mice and intramuscularly injected untreated KO EPCs, OPN-pretreated KO EPCs, or diluent alone (control). EPCs from OPN KO animals that were pretreated for 24 h with recombinant OPN improved blood flow compared to injection of cells that had not been preconditioned or diluent alone (Fig. 7), suggesting that exposure to OPN is sufficient to increase the angiogenic potential of OPN-deficient EPCs.

Treatment with OPN-exposed EPCs enhances blood flow to ischemic limbs. Hind limb ischemia was induced in OPN KO mice and intramuscular (IM) injection of EPCs was preformed. (A) Representative Doppler images before and after the induction of ischemia demonstrate an increase in blood flow in the ischemic limbs of mice treated with OPN preconditioned EPCs. (B) Quantitative analysis of the laser Doppler blood flow data demonstrates an accelerated angiogenic response to ischemia. At day 7 mice injected with OPN-preconditioned EPCs showed significantly improved blood flow when compared to mice injected with diluent alone and this improvement is further enhanced at day 14. Results are shown as the ratio of the ischemic limb to the control limb (n = at least 5). *p < 0.05, **p < 0.01 compared to control.
Discussion
Recent clinical trials, such as Transplantation of Progenitor Cells and Regeneration Enhancement in Acute Myocardial Infarction (TOPCARE-AMI) and Therapeutic angiogenesis by cell transplantation (TACT), have suggested that autologous bone marrow-derived progenitor cell transplantation holds promise for the treatment of acute myocardial infarction (3,26) and peripheral vascular disease (24). While these studies demonstrate the safety of autologous cell transplantation and potential for therapeutic benefit, the outcome may be compromised by disease-induced cellular dysfunction. This highlights the need to understand the mechanism of dysfunction associated with each disease state and how this may limit the effectiveness of treatment. Understanding disease-associated EPC dysfunction may allow reversal prior to transplantation. In the current study we used diabetes mellitus as a model of EPC dysfunction and determined a mechanistic role for OPN. Furthermore, we showed that OPN plays a crucial role in EPC function in general and that the mechanism is via autocrine secretion of angiogenic factors from EPCs. We went on to show that preincubation of dysfunctional EPCs (from OPN KO mouse) with OPN enhanced the therapeutic efficacy after cell transplantation.
We used T1DM as a model of EPC dysfunction in order to identify factors involved in cell dysfunction. Others have established that there are reduced numbers of circulating EPCs in patients with both T1DM and T2DM (23,31). However, patients with microvascular complications were not excluded from these studies and it has been shown that EPC numbers are increased in patients with diabetic retinopathy (2). Thus, we studied a population of T1DM patients without diabetic retinopathy or other confounding factors and found, in accordance with the previous studies, that the number of circulating EPCs was significantly reduced. Furthermore, we demonstrated that these EPCs were impaired in both their ability to adhere to activated endothelial cells and their ability to induce tubule formation, which are two critical indicators of angiogenic potential in vivo.
Having confirmed that EPC dysfunction exists in a homogenous cohort of T1DM patients, we used microarray analysis to identify gene expression differences, which may explain this observation. OPN was one of a number of genes found to be dysregulated in diabetic EPCs following microarray analysis and was confirmed to be downregulated by qRT-PCR. We proceeded to assess the vascular response to hind limb ischemia in OPN KO mice. Our results were consistent with those of Duval et al., who found OPN KO mice demonstrate impaired angiogenesis in response to hind limb ischemia (8). Interestingly, many studies have shown that OPN is upregulated in response to ischemia (4,18,20). These studies suggest a critical role for OPN in the angiogenic response but the mechanism is poorly understood. As we demonstrated OPN expression is decreased in EPCs from diabetes mellitus, a condition with abnormal EPC function, we hypothesized that the mechanism of OPN-mediated angiogenesis was via an interaction with EPCs.
We next studied the potential role of OPN in EPC dysfunction using cells isolated from OPN KO mice. OPN KO EPCs have a reduced ability to adhere to activated endothelial cells and to induce angiogenesis. Furthermore, we found that conditioned media was able to induce angiogenesis in vitro, which suggests that EPCs are secreting angiogenic factors, and this is supported by recent studies where EPC-conditioned media was demonstrated to contain angiogenic proteins and was able to induce an angiogenic response in vivo (6,15,17, 33). Interestingly, neutralization of OPN was not sufficient to dampen the angiogenic response induced by CM. This implied that OPN was not directly responsible for the induction of angiogenesis as its presence was not required in the assay itself. However, exposure of EPCs to OPN during culture was critical to inducing angiogenesis, which suggested OPN may be acting in an autocrine manner, stimulating EPCs to turn on a “master switch”, thereby promoting angiogenesis via the secretion of a variety of angiogenic proteins. Indeed, we found that exposure to OPN was sufficient to restore the secretion of at least three proangiogenic proteins to levels observed in WT cells. All three angiogenic proteins identified, IL-6, FGF-α, and TGF-α, have been implicated in wound healing and angiogenesis (22,28,34). However, OPN has various cleavage and phosphorylation sites and additional studies need to be performed to determine how OPN activates EPCs to release these angiogenic factors.
While we and others have shown that a deficiency in OPN suppressed the angiogenic response in a murine hind limb ischemia model (8), this cannot be directly attributed to OPN expression in EPCs. Using the OPN KO mouse as a model for EPC dysfunction, we examined the effectiveness of transplantation of OPN KO EPCs preexposed to OPN in the response to hind limb ischemia. Injection of OPN-treated EPCs significantly enhanced and accelerated healing in the ischemic limbs, suggesting that preincubation of dysfunctional EPCs with OPN may enhance their therapeutic efficacy.
This study shows for the first time that OPN plays a crucial role in EPC function and that this is at least in part due to an autocrine effect whereby OPN increases the secretion of angiogenic proteins from EPCs. This observation followed on the recognition that OPN expression in EPCs was reduced in diabetes mellitus, a model of EPC dysfunction and impaired angiogenesis and vascular healing. The data provide an important insight into the mechanism of EPC-mediated angiogenesis and also provide a target for reversal of EPC dysfunction prior to autologous cell transplantation. In addition, the data provide a rationale for attempted modulation of OPN expression in EPCs as a therapy to restore endogenous EPC function in vivo.
