Abstract
Endothelial progenitor cells (EPCs) promote revascularization and tissue repair mainly by paracrine actions. In the present study, we investigated whether EPC-secreted factors in the form of conditioned medium (EPC-CM) can protect cultured brain microvascular endothelial cells against an ischemic insult. Furthermore, we addressed the type of factors that are involved in the EPC-CM-mediated functions. For that purpose, rat brain-derived endothelial cells (rBCEC4 cell line) were exposed to EPC-CM pretreated with proteolytic digestion, heat inactivation, and lipid extraction. Moreover, the involvement of VEGF and IL-8, as canonical angiogenic factors, was investigated by means of neutralizing antibodies. We demonstrated that EPC-CM significantly protected the rBCEC4 cells against an ischemic insult mimicked by induced oxygen–glucose deprivation followed by reoxygenation. The cytoprotective effect was displayed by higher viable cell numbers and reduced caspase 3/7 activity. Heat inactivation, proteolytic digestion, and lipid extraction resulted in a significantly reduced EPC-CM-dependent increase in rBCEC4 viability, tube formation, and survival following the ischemic challenge. Notably, VEGF and IL-8 neutralization did not affect the actions of EPC-CM on rBCEC4 under both standard and ischemic conditions. In summary, our findings show that paracrine factors released by EPCs activate an angiogenic and cytoprotective response on brain microvascular cells and that the activity of EPC-CM relies on the concerted action of nonproteinaceous and proteinaceous factors but do not directly involve VEGF and IL-8.
Introduction
Neuronal tissue integrity is strongly dependent on appropriate blood perfusion. In addition to the importance of a functional vascular system under normal physiological conditions, the activation of angiogenesis plays a crucial role in tissue regenerative processes (31,50). In the context of therapeutic interventions aimed at promoting tissue recovery, cell transplantation represents a promising approach. Evidence accumulated over the last several years indicates that the therapeutic effect of transplanted stem/progenitor cells in injured tissues primarily relies on the secretion of paracrine factors (4,45,59). In fact, improved tissue functionality is typically associated with low levels of cell engraftment and transdifferentiation (1,15). Therefore, there is a general consensus that stem/progenitor cells support tissue regeneration mainly by trophic and immunomodulatory actions (18,38). There are a variety of factors secreted by stem/progenitor cells, including growth factors, cytokines, and chemokines, a group of factors generally referred to as a secretome (43). The composition of the secretome varies depending on the type of stem/progenitor cells being studied (19).
Encouraging results have been obtained in preclinical studies and clinical trials that used endothelial progenitor cells (EPCs) to promote revascularization and repair of the ischemic heart and limbs (6,20,23,4). In line with this notion, successful neuronal regeneration after ischemic stroke by administration of EPCs has also been reported recently (39,46).
Exploiting tissue repair by means of paracrine factors in the form of cell-free therapy is a growing area of interest in regenerative medicine (2,5,7,4,32). We have previously shown that soluble factors secreted by EPCs promote angiogenic functions in rat brain microvascular endothelial cells through AKT and ERK (13). Despite the progress made in deciphering the secretome (24,35), a clear molecular signature of the effectors of EPC-conditioned medium (EPC-CM)-mediated angiogenesis and cytoprotection is still missing. A considerable number of studies reported on the identification of specific growth factors and cytokines as key mediators of paracrine-induced activities by different stem/progenitor cells (30,36,42). Among these factors, the angiogenic factors interleukin-8 (IL-8) and vascular endothelial growth factor (VEGF) were identified to be crucial in attenuating injury and promoting recovery of damaged tissue (9,10,29,4). Moreover, recent studies have shown evidence that lipids play a key role as mediators of the paracrine actions of mesenchymal stem cells (MSCs) (48,54).
In the present work, we explored the cytoprotective capacity of EPC-CM on rat brain microvascular endothelial cells using an in vitro model of ischemia. Moreover, we sought to further characterize the factors involved in the activity of EPC-CM. Thus, in the present study, we investigated whether the properties of EPC-CM rely on proteinaceous factors with special emphasis on the involvement of VEGF and IL-8. We also addressed whether lipids in EPC-CM might function as mediators of the paracrine actions of EPCs.
Materials and Methods
Endothelial Cell Culture
The rat brain microvascular cell line rBCEC4 was kindly provided by Dr. I. Blasig (Forschungsinstitut für Molekulare Pharmakologie, Berlin, Germany). Cells were grown on 1% gelatin (Bio-Rad, Cressier, Switzerland)-coated plates in Dulbecco's modified Eagle medium (DMEM; Thermo Fisher Scientific, Reinach, Switzerland) containing 5% fetal bovine serum (FBS; Gibco, Reinach, Switzerland), 1% antibiotics/antimycotics (Sigma-Aldrich, St. Louis, MO, USA), 1.2 mM glutamine (Invitrogen, Reinach, Switzerland), 100 μg/ml heparin (Sigma-Aldrich), and 10 μg/ml endothelial cell growth factor (ECGF; Millipore, Temecula, CA, USA), at pH 7 in a humidified incubator at 37°C and 5% CO2 as previously described (13).
EPC Culture and Conditioned Medium Preparation
EPCs were isolated from peripheral blood mononuclear cells from healthy human anonymous donors as previously described (20). The buffy coats employed for mononuclear cell isolation were obtained from a commercial source (Interregional Transfusion Centre of the Swiss Red Cross, Bern, Switzerland) approved by the Swiss Accreditation Service (SPTS 003); therefore, no experimental approval or informed consent was further needed. Moreover, all samples were handled according to the regulations and guidelines of the University Hospital Bern. In total, 21 buffy coats were processed for EPC cultures. While the identity of the donors of the buffy coats was unknown (the samples were anonymized), all 21 buffy coats used were from different lot numbers. Therefore, we assume that some variability between outcomes of the experiments was due to this circumstance. Mononuclear cells were isolated by gradient centrifugation using Lymphoprep (Axis-Shield, Dundee, UK) and cultured on six-well plastic dishes (BD Falcon, Allschwil, Switzerland) coated with human fibronectin (Sigma-Aldrich) at a density of 1 million cells/cm2 using endothelial cell basal medium-2 (EBM-2; Lonza, Basel, Switzerland) supplemented with endothelial growth medium SingleQuots (Lonza) and 5% FBS (Lonza). After 4 days in culture, adherent cells were passaged at the density of 0.1 million cells/cm2 and cultured through day 7 to obtain early outgrowth EPCs. To produce EPC-CM, EPCs were exposed to hypoxia, since we have previously observed that these culture conditions enhance soluble factor secretion by EPCs (14). Notably, with the aim of limiting the effect of endogenous factors present in the serum, in this step a medium with reduced serum content was used (1% FBS). Accordingly, EPCs were washed three times with phosphate-buffered saline (PBS; Inselspital Hospital Pharmacy, Bern, Switzerland) and incubated in 2.5 ml growth factor-free EBM-2 containing 1% FBS for 48 h under hypoxic conditions (1.5% O2, 5% CO2, 93.5% N2) using a humidified gas-sorted anoxic incubator-gloved box (InVivo2 400; Ruskinn Technologies, Bridgend, UK). After incubation, EPC-CM was collected and centrifuged at 200 ° g at 4°C; the supernatant was filtered under sterile conditions and snap-frozen until use (14). Overall, the experiments were conducted using individual EPC-CM preparations. The EBM-2 containing 1% FBS was treated in parallel to the cell cultures and served as control medium. Throughout all of the experiments, EPC-CM and control medium were exposed simultaneously to the same treatments.
Conditioned Medium Dilution
For the dose–response experiment, immediately before use, EPC-CM was diluted in control medium to the final concentrations of 10%, 20%, and 50% (1:10, 1:5, and 1:2, respectively).
Multiplex Assay
The concentrations of IL-8 (in eight different samples) and VEGF (in four different samples) were assessed in supernatants of EPC cultures grown for 48 h (in two independent experiments) using a multiplex assay (Bioplex; Bio-Rad, Cressier, Switzerland) as previously described (14).
Conditioned Medium Treatments
Heat treatment of EPC-CM was performed in order to denature the proteinaceous components of EPC-CM. Briefly, 15-ml polypropylene test tubes (BD Falcon, Allschwil, Switzerland) containing 6 ml of medium were immersed in boiling distilled water for 10 min, and then the tubes were transferred to ice-cold water for 5 min.
For the protease treatment, the medium was incubated with proteinase K immobilized on agarose beads (Sigma-Aldrich) as described by others with minor modifications (48). Briefly, 3 ml of the medium was incubated with agarose-proteinase K (5 U) in 15-ml polypropylene test tubes (BD Falcon, Allschwil, Switzerland) for 4 h at 37°C in a rotating roller drum. Thereafter, the tubes were centrifuged for 5 min at 780 ° g to pellet the agarose-proteinase K, and the proteinase K-free supernatant was carefully collected and used for subsequent experiments.
Removal of lipid components from EPC-CM was performed by chloroform extraction (56). Briefly, equal volumes (6 ml) of EPC-CM and chloroform were mixed and centrifuged for 5 min at 1,750 ° g, the aqueous phase was collected, and the procedure was repeated. Finally, the medium was transferred into an open 1-cm Petri dish, and the traces of chloroform were left to evaporate in the sterile hood for 10 min. Thereafter, the water lost during evaporation was reconstituted with distilled sterile water.
Viable Cell Number Assay
A PrestoBlue assay (Invitrogen) was used as previously described (13) to assess the viable cell numbers. Briefly, 5,000 rBCEC4 were seeded into gelatin-coated 48-well plates and cultured as described above. After 24 h of starvation in EBM-2 containing 1% FBS, the cells were then incubated with the processed EPC-CM for 24 h. After this period, the experimental media were replaced with EBM-2 containing 1% FBS and 10% PrestoBlue. Fluorescence readouts were acquired with a microplate reader (Tecan Infinite 1000, Ex. 560 nm, Em. 590 nm; Tecan, Männedorf, Switzerland), and the values were corrected for the background.
In Vitro Tubular Structure Formation
The morphogenic capacity of rBCEC4 in vitro was investigated as previously described (57). In brief, melted growth factor-reduced Matrigel (Becton Dickinson AG, Allschwil, Switzerland) was layered in 24-well plates and allowed to become gelatinous. Forty thousand cells were resuspended in control medium, as well as the other experimental culture media and seeded on the Matrigel layer. After 16 h of incubation, the formation of endothelial cell tubular structures was assessed. Digital microphotographs were taken at low magnification with a light microscope from three randomly selected fields for data analysis. The angiogenic response of rBCEC4 was assessed measuring the total length of the tubular structures with the aid of ImageJ software (NIH, Bethesda, MD, USA). Digitalized photomicrographs were slightly modified for better visualization of the tubular structures.
In Vitro Ischemia–Reoxygenation
Ischemic injury (RI-I) was mimicked in vitro by exposing rBCEC4 to oxygen–glucose deprivation (OGD) followed by reoxygenation as described by others with minor modifications (16,21,51). Endothelial cells were incubated in a glucose-free buffer (BSS0, 116 mmol/L NaCl, 1.8 mmol/L CaCl2, 0.8 mmol/L MgSO4, 5.4 mmol/L KCl, 1 mmol/L NaH2PO4, 14.7 mmol/L NaHCO3, and 10 mmol/L HEPES, pH 7.4; all from Merck KGaA, Darmstadt, Germany) for 6 h in an atmosphere composed of 95% N2 and 5% CO2 at 37°C by using a hypoxia chamber (Billups-Rothenberg Inc., San Diego, CA, USA). For reoxygenation, BSS0 was replaced with unconditioned medium or EPC-CM (either native or treated EPC-CM), and the cells were returned to standard incubation conditions (20% O2, 5% CO2, 37°C). As a control (Nor-Ctr), cells were incubated in a buffer supplemented with 5 mM of glucose (BSS5) in standard incubation conditions for 6 h and replaced with unconditioned medium. Cultures were kept in reoxygenation conditions for 3 h to measure caspase 3/7 activity or for 18 h to assess the cell viability using PrestoBlue reagent (Invitrogen) as described above.
Neutralization Experiments
The blocking of IL-8 and VEGF in EPC-CM was performed using specific neutralizing antibodies. In these experiments, EPC-CM was pretreated for 30 min at 37°C with anti-VEGF (goat anti-VEGF; AF 293-NA, 0.2 μg/ml; R&D Systems Inc., Abingdon, UK) or anti-IL-8 (goat anti-IL-8; MAB208-NA, 2 μg/ml; R&D Systems Inc.). Subsequently, rBCEC4 were incubated with EPC-CM or the angiogenic factors in the presence or absence of the neutralizing antibodies.
Caspase 3/7 Activity Assay
Caspase 3 and 7 activity was measured using the Apo-ONE homogenous caspase 3/7 kit (Promega AG, Dübendorf, Switzerland). Fluorescence intensity was acquired with a microplate reader (Ex. 499 nm, Em. 521 nm; Tecan Infinite 1000) as previously reported (58). The relative caspase 3 activity of each sample was then normalized to the number of viable cells in the same sample measured with PrestoBlue and expressed as percentage relative to the not-ischemic control group.
Statistical Analysis
For statistical analysis, a commercially available software package was used (Prism; GraphPad Software, La Jolla, CA, USA). One-way ANOVA followed by Bonferroni's multiple comparison test was used to compare group means. Comparisons between two groups were done with Welch's t-test. Statistical significance was inferred at a two-sided p ≤ 0.05. Data are presented in percentage relative to the controls, and values are given as mean ± standard error of the mean (SEM).
Results
Dose–Response Activity of EPC-CM
To assess the response of rBCEC4 to different doses of EPC-CM, we tested the effect of different dilutions of EPC-CM on viability and tube-like formation in vitro (Fig. 1A and B, respectively). In line with our previous observations (13), viable cell numbers and tube length were substantially augmented by incubation with undiluted EPC-CM (CM100%) compared to controls (1.4-fold p = 0.002 and 2.4-fold p = 0.017, respectively) and steadily declined with increasing dilutions of EPC-CM (Fig. 1A). At a 20% dilution of EPC-CM (CM20%), the capacity to enhance cellular viability was not significantly different from untreated controls (p = 0.48). In contrast, in the presence of CM20%, the formation of longer tubular-like structures dropped to 43%, but it was still significantly different when compared to full EPC-CM (p = 0.0.034) (Fig. 1B). EPC-CM-dependent tube formation was lost at 10% dilution (p = 0.323).

Effects of dilution of EPC-CM on viable cell number and tube length. Effects of different dilutions of the EPC-CM on viable cell numbers (A, n = 4) and total tubule length (B, n = 3) in rBCEC4 cultures. Note that a twofold dilution (CM50%) significantly elevated cell numbers (A), while up to a dilution of five times (CM20%) significantly increased tube length was detected (B). Data are presented as a percentage of controls, and values are given as mean + SEM. *p < 0.05 versus corresponding controls.
EPC-CM-Induced Activities Involve Proteinaceous and Heat-Labile Factors
We and others have asserted that EPCs secrete a wide variety of growth factors and cytokines (8,14). With the aim of gaining insight into the nature of the EPC paracrine factors, we assessed the thermostability of EPC-CM. The cell viability induced by native EPC-CM was completely abrogated by the heat treatment (p = 0.008) (Fig. 2A), whereas the total tube length was reduced by 44% (p = 0.0004) (Figs. 2B and 3). Accordingly, proteinase K treatment of EPC-CM (CM prot K) resulted in a significant reduction in cell viability (18%, p = 0.049) (Fig. 2C) and tubular structure length (42%, p = 0.032) (Figs. 2D and 3) compared with untreated EPC-CM.
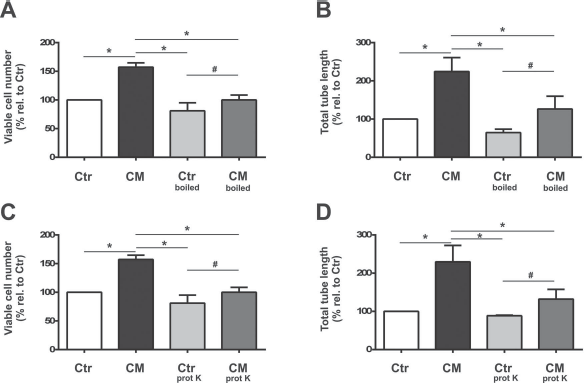
Heat and proteinase K treatments reduce viable cell number and tube length. Quantitative analyses of heating (A, n = 5; B, n = 4; CM-boiled) and treating the media with proteinase K treatment (C, n = 3; D, n = 4; CM-prot K) on viable cell numbers (A, C) and tube length (B, D) in cultures of microvascular endothelial cells. Both treatments significantly reduced the EPC-CM (CM)-mediated effects on number of viable cells (A, C) and total tube length (B, D). Data are presented as a percentage of controls, and values are given as mean + SEM. *p < 0.05 versus EPC-CM-treated cultures; #p < 0.05 versus corresponding controls.

Heat and proteinase K treatments reduce EPC-CM-induced tubular network formation. Representative microphotographs of tubular network formation by microvascular endothelial cells on growth factor-reduced Matrigel. EPC-CM incubation (CM) strikingly promoted the tube-like structure formation compared to control medium (Ctr). Supplementation of EPC-CM, which underwent heat treatment (CM-boiled), substantially attenuated EPC-CM-induced tube formation. Similarly, proteinase K treatment (CM-prot K) resulted in a decrease of tube formation capacity. Scale bar: 100 μm.
EPC-CM Exerts Cytoprotection Against In Vitro Ischemia Reoxygenation
In view of a potential use of EPC-CM for tissue regeneration, we assessed whether EPC-CM promotes the viability of brain microvascular endothelial cells challenged by the detrimental effects of ischemia–reoxygenation (Fig. 4). Exposure of rBCEC4 to RI-I significantly reduced the cell viability to 58% compared to controls treated with unconditioned medium during the reoxygenation phase (p = 0.003; RI-I-Ctr) (Fig. 4A, C). This decline was significantly attenuated by 71% when the reoxygenation was carried out in the presence of EPC-CM (p = 0.008; RI-I-CM) (Fig. 4A, C). RI-I also induced an increase in caspase 3/7 activity in the cultures (1.9-fold relative to controls, p = 0.0016), which was significantly attenuated in the presence of EPC-CM (1.2-fold relative to controls, p = 0.0002) (Fig. 4B, D). Similar to the outcome observed under standard conditions, both heat and proteinase K treatment caused significant reduction of the cytoprotective properties of EPC-CM with cell viability reaching almost the level of untreated ischemic cultures (p = 0.008 and p = 0.003) (Fig. 4A and C, respectively). Correspondingly, the activity of caspase 3/7 increased by 40% and 38% in the cultures incubated with heat- and proteinase K-treated EPC-CM (RI-I-CM-boiled and RI-I-CM-prot K; p = 0.0002 and p = 0.024) (Fig. 4B and D, respectively) compared to native EPC-CM.

Heat and proteinase K treatments reduce EPC-CM-mediated cytoprotection against an ischemia–reoxygenation insult. Quantitative analyses of heat (A, n = 4–5; B, n = 3; boiled) and proteinase K treatment (C, n = 4; D, n = 4; prot K) of the media on viable cell numbers (A, C) and caspase 3/7 activity (B, D) in cultures of microvascular endothelial cells exposed to a RI-I. While treatment with EPC-CM (CM) exerted cytoprotective effects on cell viability (A, C) and significantly reduced the augmented caspase 3/7 activity induced by the RI-I, both heat (CM-boiled) and proteinase K treatment (CM-prot K) significantly reduced the effects of EPC-CM (CM). Data are presented as a percentage of controls, and values are given as mean + SEM. *p < 0.05 versus corresponding EPC-CM-treated cultures.
EPC-CM-Induced rBCEC4 Angiogenic Response Is Not Directly IL-8 and VEGF Dependent
The results described above support the view that growth factors and/or cytokines are involved in the EPC-CM-induced effects. In agreement with previous observations (13,14), the quantitative analysis of EPC-CM disclosed the presence of high levels of IL-8 (7,089 ± 2,637 pg/ml) and lower levels of VEGF (49.8 ± 21.7 pg/ml). Therefore, we investigated, in greater detail, the contribution of IL-8 and VEGF to the protective effects of EPC-CM.
We first performed experiments to assess the validity of the experimental setup based on neutralizing antibodies. As expected, VEGF and IL-8 supplementation promoted rBCEC4 viability in a dose-dependent manner, reaching a maximal effect at the concentration of 50 ng/ml for VEGF and 100 ng/ml for IL-8 (1.9- and 1.6-fold increase relative to untreated controls, respectively, p = 0.0001 and p = 0.009, respectively) (Fig. 5A, B). The tube formation capacity was likewise enhanced by 100 ng/ml VEGF and by 50 ng/ml IL-8 (1.6- and 1.8-fold increase relative to untreated controls, respectively, p = 0.005 and p = 0.0001, respectively) (Fig. 6A, B). These effects were abrogated by administration of the respective neutralizing antibodies (p = 0.006 for VEGF and p = 0.0005 for IL-8) (Fig. 6). Importantly, however, both anti-VEGF and anti-IL-8 antibody treatment did not significantly affect the increase in rBCEC4 viability (p = 0.73 and p = 0.92) (Fig. 7A, C) nor tube formation (p = 0.68 and p = 0.18) (Fig. 7B, D) induced by EPC-CM. Similar observations were made when cultures were exposed to RI-I (Fig. 8). Thus, neutralization of VEGF and IL-8 in EPC-CM did not affect the EPC-CM-induced cytoprotection with regard to cell viability (RI-I-CM-anti-VEGF and RI-I-CM-anti-IL-8, 74% and 79%, respectively, compared to control ischemic cultures, p = 0.85 and p = 0.51) (Fig. 8A) and nor the levels of caspase 3/7 activity (1.4-fold increase for both compared to control ischemic cultures, p = 0.99 for RI-I-CM vs. RI-I-CM-anti-VEGF and p = 0.91 for RI-I-CM vs. RI-I-CM-anti-IL-8) (Fig. 8B).

VEGF and IL-8 promote viable cell number. Dose–response analysis of vascular endothelial growth factor (VEGF; A, n = 3–5) and interleukin-8 (IL-8; B, n = 3–5) on viable cell numbers in cultures of microvascular endothelial cells in the absence (open bars) or presence of neutralizing antibodies (gray bars) against VEGF (anti-VEGF; A) and IL-8 (anti-IL-8; B). Data are presented as a percentage of corresponding controls, and values are given as mean + SEM. *p < 0.05 versus corresponding controls.

VEGF and IL-8 promote tubular structure. Effects of vascular endothelial growth factor (VEGF; A, n = 4–6) and interleukin-8 (IL-8; B, n = 5) administration on total tube length in cultures of microvascular endothelial cells in the absence (gray bars) or presence of neutralizing antibodies (dark gray bars) against VEGF (anti-VEGF; A) and IL-8 (anti-IL-8; B). The significant increase in tube length induced by the growth factor treatment was abolished in the presence of the neutralizing antibodies. Data are presented as a percentage of corresponding controls, and values are given as mean + SEM. *p < 0.05 versus corresponding growth factor-treated cultures.
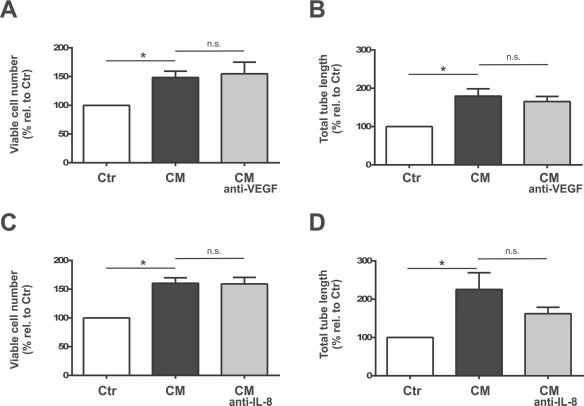
Neutralizing antibodies do not alter EPC-CM-induced effects. Effects of neutralizing antibodies against vascular endothelial growth factor (CM-anti-VEGF; A, n = 5; B, n = 4) and interleukin-8 (CM-anti-IL-8; C, n = 5; D, n = 3) on viable cell numbers (A, C) and total tube length (B, D) in cultures of microvascular endothelial cells exposed to EPC-CM (CM). Untreated cultures served as controls (open bars). The significant increase in viable cell numbers (A, C) and total tube length (B, D) induced by EPC-CM (filled bars) was not altered by the presence of neutralizing antibodies (gray bars). Data are presented as a percentage of corresponding controls, and values are given as mean + SEM. *p < 0.05 versus corresponding controls.

VEGF and IL-8 neutralization does not alter cytoprotection induced by EPC-CM against an ischemia–reoxygenation insult. Quantitative analyses of neutralizing antibodies against vascular endothelial growth factor (anti-VEGF) and interleukin-8 (anti-IL-8) on viable cell numbers (A, n = 6) and caspase 3/7 activity (B, n = 4) in cultures of microvascular endothelial cells exposed to an RI-I. While treatment with EPC-CM (CM, filled bars) exerted cytoprotective effects on cell viability (A) and significantly reduced the augmented caspase 3/7 activity induced by the RI-I, the presence of neutralizing antibodies (gray bars) did not alter the EPC-CM-mediated effects. Data are presented as a percentage of untreated controls, and values are given as mean + SEM. *p < 0.05 versus corresponding controls.
Lipid Extraction From EPC-CM
Similar to the outcome seen for heat and proteinase K treatment, lipid extraction of EPC-CM diminished EPC-CM-mediated effects (Figs. 9–11). Lipid extraction (CM-chlorof) caused a slight, but significant, reduction of cell viability (from 1.5- to 1.2-fold increase relative to untreated controls, p = 0.0002) (Fig. 9A). Chloroform treatment triggered a reduction of 26% in the total tube length compared to untreated EPC-CM (p = 0.03) (Figs. 9B and 10).

Lipid extraction attenuates the effects of EPC-CM. Quantitative analyses of the lipid content in EPC-CM-mediated effects on viable cell numbers (A, n = 6) and total tube length (B, n = 4) in cultures of microvascular endothelial cells. While chloroform extraction (Ctr-chlorof) had no effects on control media-treated cultures (gray bars), EPC-CM (CM; filled bars) induced an increase in viable cell numbers, and total tube length was significantly decreased (CM-chlorof; dark gray bars). Data are presented as a percentage of controls, and values are given as mean + SEM. *p < 0.05 versus corresponding EPC-CM-treated cultures; #p < 0.05 versus corresponding controls.

Lipid extraction reduces EPC-CM-induced tubular network formation. Representative digital microphotographs of tubular network formation by microvascular endothelial cells on growth factor-reduced Matrigel. EPC-CM incubation (CM) robustly promoted the tube-like structure formation compared to control medium (Ctr). Lipid extraction of the EPC-CM (CM-chlorof) significantly attenuated EPC-CM-induced tube formation. Scale bar: 100 μm.
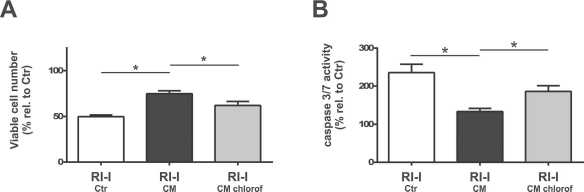
Lipid extraction reduces EPC-CM-mediated cytoprotection against an RI-I. Quantitative analyses of the lipid content in EPC-CM-mediated effects on viable cell numbers (A, n = 4) and caspase 3/7 activity (B, n = 5) in cultures of microvascular endothelial cells exposed to an RI-I. Treatment with EPC-CM (CM; filled bars) exerted cytoprotective effects on cell viability and significantly reduced the augmented caspase 3/7 activity induced by the RI-I. Chloroform extraction (CM-chlorof; gray bars) was found to abolish the EPC-CM-induced effects on cell viability and caspase 3/7 activity. Data are presented as a percentage of untreated controls, and values are given as mean + SEM. *p < 0.05 versus corresponding EPC-CM-treated cultures.
Moreover, lipid extraction by chloroform also significantly impaired the cytoprotective capacity of EPC-CM displayed by a reduced viability (18% decline compared to native EPC-CM, p = 0.004) (Fig. 11A), paralleled by an increase in caspase 3/7 activity (38% rise compared to native EPC-CM, p = 0.02) (Fig. 11B).
Discussion
In the present study, we describe that the soluble factors secreted by EPCs enhanced brain microvascular endothelial cell growth, tube formation capacity, and cell survival in an oxygen–glucose deprivation (RI-I) culture model that mimics the acute ischemic stroke situation in humans. These findings further support the cytoprotective properties of EPCs against ischemic insults in various organs and tissues including the brain vascular cells (28,46,52). We provide evidence that the paracrine actions of EPC-CM are mediated by thermo-labile, protease-sensitive, as well as chloroform-extractable factors. Moreover, our data suggest that VEGF and IL-8 might not directly contribute to the effects of EPC-CM.
It is now a widely accepted opinion that paracrine actions of stem/progenitor cells are the major player in tissue protection/repair (53). Despite the critical importance of this aspect of regenerative medicine, the molecular effectors and the mechanisms of action of EPC-paracrine factors are still elusive. Consequently, great emphasis has been given to the need for dissection of the entire spectrum of growth factors and cytokines secreted by EPCs. In this line, in the present work, we initially aimed at verifying whether the effects of EPC-CM rely solely on proteins. Indeed, the observed susceptibility of EPC-CM to heat and to protease treatment implies that proteinaceous factors contribute, at least in part, to the cytoprotective and proangiogenic properties of EPC-CM.
In order identify the potential factors involved in the enhanced endothelial survival, tubular structure formation, and protection against an ischemic insult, we focused our attention on IL-8 and VEGF. These two factors were selected out of several growth factors and cytokines found to be secreted by EPCs, since they have been described as master regulators of endothelial cell survival and vascular growth (33,40). In the present work, quantitative measurements of IL-8 and VEGF as well as the dose–response analysis using the recombinant peptides indicate that the concentration of VEGF and IL-8 found in EPC-CM is at subthreshold level. This observation combined with our previous findings that IL-8 is among the angiogenic factors found in EPC-CM in highest concentration (13) suggests that the effects of EPC-CM rely on mechanisms different from the canonical response to single factor treatment. It should also be considered that the cultures exposed to EPC-CM might display an altered sensitivity to growth factors compared to untreated controls. In fact, effects of potentiation of growth factor activity might occur possibly as a result of synergic actions of multiple factors contained in EPC-CM (57).
In control experiments, the activity of the recombinant IL-8 and VEGF could be effectively blocked by neutralizing antibodies, whereas these failed to attenuate the EPC-CM-induced effects. In light of these in vitro results, we concluded that, taken individually, IL-8 and VEGF appear to not be major players of the EPC-CM-enhanced viability, tubulogenesis, and protection from the ischemia–reperfusion damage. However, it cannot be excluded that the lack of blockade of IL-8 and VEGF could result from the presence of splice variants or redundant growth factors structurally and functionally related to these two. Importantly, these findings replicate our previous observations showing that the antiapoptotic response promoted by EPC-CM was not diminished by individual or collective neutralization of VEGF, hepatocyte growth factor (HGF), matrix metalloproteinase-9 (MMP-9), and IL-8 (58). In contrast, other reports have shown significant reduction of the angiogenic properties of myeloid angiogenic cells and late (outgrowth) EPCs by blocking IL-8 (17,33). The reasons underlying these discrepancies remains to be investigated and could be, in part, due to the different experimental systems employed, such as the use of a different type of endothelial cells to test the angiogenic functions of EPC-CM or a different method of EPC-CM production. In the present study, we employed EPC-CM produced in hypoxic cultures, since it has been demonstrated that low oxygen promotes the angiogenic properties of EPCs (22). This is consistent with enhanced release of proangiogenic factors in EPCs exposed to low oxygen tension (14,27). Additionally, hypoxia as well as tissue ischemia have been shown to promote EPC mobilization and tissue revascularization thereby augmenting the therapeutic capacity of transplanted cells (34,55).
The observations that heat and proteinase K treatment of EPC-CM significantly lowered, but not completely abolished, the effects EPC-CM in brain endothelial cells imply that the EPC-CM-dependent endothelial cell viability and tubulogenesis might be mediated by proteinaceous as well as nonproteinaceous factors. Indeed, the inhibitory effect of chloroform extraction on EPC-CM activity suggests that lipids are one group of bioactive factors contained in EPC-CM, which might act in concert with growth factors and cytokines to promote the functions of microvascular endothelial cells.
Despite the growing attention given to the lipids released by stem cells as paracrine mediators (25,48), the involvement of lipids released by EPCs has not been thoroughly investigated so far. In fact, the findings that EPCs secrete signaling lipids such as prostanoids and endocannabinoids (11,37) further underscore the potential of EPCs as vascular and neuronal therapeutics. Also, shingosine-1-phosphate seems to play a pivotal role in endothelial autocrine/paracrine cross talk and is a potent regulator of different vascular functions (41,47). In the present work, the specific identity of the lipids that might be involved in the enhanced tube formation and increased cell survival under ischemic conditions in vitro was not addressed. In particular, whether the chloroform-extractable factor(s) in EPC-CM are freely soluble lipids or consist of exosomes or as moieties of lipid-modified proteins, such members of the Wnt family [e.g., Wnt 5a (3)] remains to be investigated. Our findings showing that both proteinaceous factors and lipids play a role in the EPC-CM-mediated actions may hint at the idea that these effects involved released exosomes or microvesicles. It has been reported that exosomes and microvesicles are involved in many types of cell–cell communication, being able to shuttle virtually all kinds of signaling molecules, including proteins, lipids, and RNA (12,44). Nevertheless, we did not perform experiments that would prove such an assumption, and follow up studies are needed to investigate the role of exosomes in EPC-CM-mediated protection and the possible interplay between proteinaceous factors and lipids, all of which represent important aspects of the paracrine signaling that indeed deserve deeper analysis (49).
In conclusion, we provide the evidence that EPC-CM promotes endothelial cell survival, tube-formation capacity, and survival in an in vitro model of ischemia, likely through a combination of factors of a proteinaceous and lipidic nature. Overall, these results complement the known cytoprotective and angiogenic functions of EPC-derived soluble factors (13,22). It is important to note, while stressing again the vast complexity of composition of the factors released by EPCs, the findings described here encourage verification through in vivo studies. Insights into the in vivo translation of mechanisms of action of EPC-CM are hence mandatory and fundamental to enable the development of therapeutic interventions based on paracrine factors.
Footnotes
Acknowledgment
This study was supported by SNF NRP63 (406340_128124). We thank Prof. Ingolf Blasig (Leibniz-Institute of Molecular Pharmacology, Berlin) for providing the rBCEC4 cell line. This work has in part been presented at the 21st and 22nd Annual Meetings of the American Society for Neural Therapy and Repair, Clearwater, FL, USA, Cell Transplant. 23(6):780, 2014, and Cell Transplant. 24(4):756, 2015. The authors declare no conflicts of interest.
