Abstract
Transplantation of human endothelial progenitor cells (hEPCs) may improve vascularization and left ventricular function after myocardial infarction. The scope of this study was to explore, whether cross-linking of EPCs may enhance the deposition of cells in the rat heart after clinical-like, intracoronary transplantation. To this end, 111In-oxinate-labeled hEPCs were infused by a minimally invasive technique into the coronary arteries of immunosuppressed Wistar rats under control conditions and after ischemia/reperfusion. In a second set of experiments hEPCs were treated with phytohemagglutinin to create small cell clusters prior to transplantation. Continous three-dimensional HiSPECT images for 1 h and after 48 h revealed that cell deposition was significantly higher when hEPCs were cross-linked. Therefore, cross-linking of hEPCs may provide a promising approach to enhance the number of trapped cells also in a clinical setting.
Introduction
Endothelial progenitor cells (EPCs) are considered to be immature cells, which have the capacity to proliferate, migrate, and differentiate into endothelial cells. EPCs have been shown to contribute to neovascularization and left ventricular function after myocardial ischemia (2, 8).
Due to several promising results in animal models (9, 10, 13), clinical studies of EPC application for the treatment of ischemic heart diseases have been initiated in the past. After acute myocardial infarction the intracoronary infusion of an EPC-enriched population resulted in an improvement of left ventricular function and viability (2, 3), demonstrating feasibility, efficiency, and safety of EPC transplantation. A randomized, placebo-controlled, dose-ranging clinical trial of intramyocardial transplantation of GCSF-mobilized CD34+ cells has been started in patients with intractable angina pectoris (11). Notably, most of the cited studies have been performed on early EPCs or circulating angiogenic cells (14).
Critical for the outcome of EPC homing in the infarcted myocardium is the engraftment of a sufficient number of cells (12). In the present study we have cross-linked EPCs using phytohemagglutinin prior to intracoronary transplantation into Wistar rats to increase the number of engrafted cells. For cell tracking EPCs were labeled with 111In-oxinate and tracked using continuous three-dimensional SPECT imaging. Our results indicate that the overall number of trapped EPCs in the ischemic and nonischemic rat heart can be significantly increased after intracoronary infusion of cross-linked EPCs.
Materials and Methods
Cell Culture, Cellular Staining, and Cross-Linking
Human EPCs (hEPCs) were isolated from blood of human volunteers by density gradient centrifugation with Biocoll Separating Solution (Biochrom AG) and subsequent hemolysis. Cells were plated on culture dishes for 10 min and the supernatant containing hEPCs was replated and maintained in EBM MV2 (Promocell) supplemented with EGM MV2 SingleQuots. Cells were incubated for 8 days with medium changes every second day.
Fluorescent chemical detection was performed on attached hEPCs with 10 μg/ml DiI-Ac-LDL (Cell Systems) overnight and 20 μg/ml BS-1 (Vector Laboratories) for 1 h. For cross-linking experiments, detached hEPCs were incubated with 62.5 μg/ml phytohemagglutinin (Sigma) for 1 h under gentle shaking. Thereafter, cells were carefully detached, centrifuged, and resuspended in M199 (Gibco, Karlsruhe).
For SPECT measurements, hEPCs were incubated with 20 MBq 111In-oxinate (Tyco Healthcare) in serum-free M199 medium for 20 min at room temperature. Labeling efficiency was measured using a dose calibrator (MED Nuklear-Medizintechnik). Vitality of cells was tested by trypan blue exclusion.
hEPC Transplantation Animal Model
The investigation was approved by the Animal Research Committee at the University of Duesseldorf in accordance with federal law. Male Wistar rats (~350 g) were intubated and anesthetized with isoflurane (1.5% v/v; Abbott). Myocardial ischemia was induced by ligating the left anterior descending coronary artery for 2 h. After 4 days, cell transplantation was performed as previously described (6). Briefly, a homemade 3F balloon catheter was placed into the right atrium via the internal jugular vein to obstruct the venous return. A 2F through-lumen balloon catheter built according to our specifications (Edwards Life Science, USA) was placed into the aortic root via the right carotid artery. After the inflation of both catheters a transient cardiac arrest [1 mM esmolol (Brevibloc®, Gensia), 1 μM acetylcholine (Sigma)] was induced and 1 × 106 hEPCs (in 500 μl) were gently infused into the coronaries. After an incubation time of 1 min, spontaneous circulation was restored by an intravenous bolus injection of epinephrine (0.02 mg/kg; Hoechst, Germany) and sodium bicarbonate (0.5 mEq/kg; Braun, Germany), and cardiopulmonary resuscitation was performed at a compression rate of about 200 per minute. All animals received 30 mg/kg cyclosporine (Sandimmun®, Novartis) the day prior to transplantation and 15 mg/kg cyclosporine daily, thereafter. One animal of the +MI/+PHA group failed to reestablish stable circulation and died 1 h after transplantation.
SPECT Measurement and Radioactive Analysis
MPH tomography is an extension of conventional single-pinhole SPECT with more than one pinhole in a collimator. The main advantage of the MPH technique is the possibility of creating overlapping projections. The overlap increases the percentage of the used detector surface and leads to an improved system sensitivity. This HiSPECT technique is based on a commercial human SPECT scanner (PRISM 2000 S, Philips Medical, Eindhoven, The Netherlands), which employs a NaI(Tl) crystal of 8 mm thickness and has a rectangular FOV of 390 × 510 mm2. The MPH collimator is based on an original pinhole frame for the PRISM and consists of a 12-mm lead shielding and a MPH aperture, which is made of a 10-mm thick tungsten alloy (HPM1850). The used aperture for dynamic studies of a whole body has nine holes, which measure 4 mm in diameter, leading to a reconstructed resolution of 2.5 mm FWHM. The image quality of this setup benefits variable distance between object and aperture plate, which allows an individual adjustment to the object size. Continuous SPECT measurements were started 10 min after cell transplantation using a two-headed gamma camera (PRISM 2000 S). Tomographic data sets, each consisting of 14 projections, were acquired every 2.5 min for 50 min and after 48 h. For multipinhole geometry the MLEM reconstruction algorithm (HiSPECT, Scivis GmbH, Göttingen, Germany) and for regional analysis the imaging tool amide (version 0.7.14) were used. The three-dimensional region data of the heart (coronal slices of 1 mm thickness) were halftime corrected and normalized on the frame length and compared with the whole body value of the visible field of view (80 × 80 × 80 mm3).
Evaluation of Heart Samples
After final measurement hearts were excised and separated into infarcted area, border zone, and healthy region. Wet weight and inclosed radioactivity using a dose calibrator (MED Nuklear-Medizintechnik) was determined.
Statistics
Data are presented as mean ± SEM. Student's t-test was used for differences between two groups. A value of p < 0.05 was considered to be statistically significant.
Results and Discussion
In a typical experiment a total of 6.3 ± 2.3 × 106 (n = 12) circulating endothelial progenitor cells (hEPC)/ml human blood were isolated. After cultivation of 2 × 107 cells per six-well plate for 8 days typical spindle-shaped cells were visible (data not shown). In order to determine the phenotype, cells were stained with DiI-Ac-LDL and BS-1 and 93.80 ± 5.20% of the attached cells were positive for both markers. The phenotype was further characterized by measuring the expression of surface proteins such as von Willebrand factor (54.97 ± 29.61%), CD31 (40.45 ± 23.49%), CD144 (22.53 ± 7.95%), and CD34 (10.52 ± 4.29%) using flow cytometry (n = 3 each). Additionally, a substantial fraction of the cells were positive for CD3 (70.19 ± 9.69%), which is in agreement with the literature (7).
In order to enhance the deposition of cells within the heart, hEPCs were cross-linked with phytohemagglutinin (PHA) prior to radioactive labeling and transplantation. When using 62.5 μg/ml PHA, clusters of 2–4 cells were created. Vitality of cells was not affected by this method, as could be shown by trypan blue staining. Cells treated similarly, except that PHA was omitted, did not cluster (data not shown).
For in vivo cell tracking hEPCs were labeled with 111In-oxinate. hEPCs incorporated 3.34 ± 0.86 MBq/1 × 106 cells (n = 17). Cell viability did not decline under these conditions. Untreated and cross-linked hEPCs (1 × 106 cells in 500 μl) were transplanted into the coronary arteries of rats of either healthy animals (controls) or animals that were subjected to ischemia/reperfusion injury (3 each group) using a minimally invasive method (6). Immediately after transplantation, dynamic SPECT measurements were performed up to 48 h thereafter. Representative SPECT images of each group are shown in Figure 1A. The images show that deposition of hEPCs is mainly restricted to the heart and can be clearly visualized. Figure 1B summarizes the results from all experiments. It is evident that in the control/+PHA group more radioactivity is retained within the heart after 60 min compared to the other groups. But, overall, the amount of radioactivity declines within time (15 and 60 min postdelivery) in all groups.

Dynamic changes of radioactivity over the rat thorax followed for 60 min after transplantation of 1 × 106 hEPCs labeled with 111In-oxinate. (A) Representative coronal views of maximum intensity projections (MIPs) at indicated points of time after cell transplantation. Head is up. Scale bar: 3 cm. (B) Statistical evaluation of radioactivity in the heart (n = 3 each).
After 2 days overall radioactivity over the entire heart as measured by SPECT was below 1% and at the given sensitivity of the method the observed changes did not reach the level of significance between the groups: control, 0.19 ± 0.14%; control/+PHA, 0.21 ± 0.09%; +MI, 0.17 ± 0.13%; +MI/+PHA, 0.17 ± 0.06%. This low percentage of detected radioactivity is consistent with data from the literature (1, 4). In these studies, 111In-oxinate-labeled hEPCs or CD34+ cells were injected into the cavity of chronically infarcted left ventricles of rats and only ~3% or ~1% of the radioactivity was detected in the heart 96 h thereafter. For these measurements only planar views of the animals were used, which certainly overestimated the number of trapped cells in the heart. In contrast, we used 3D-SPECT images for calculation, which permitted a more precise estimation of the amount of radioactivity within the heart.
At the end of the experiment (2 days), lung, liver, and hearts were excised and the residual radioactivity in the MI-unaffected region, border zone, and infarcted region was measured in a bore hole with higher sensitivity. As summarized in Figure 2A, cross-linking hEPCs with PHA more than doubled the deposition of the cells in the infarcted heart of both ventricles. There was also a higher number of labeled cells in the left ventricular border zone (LV-BZ) and the center of the infarct (LV-MI) when compared with the unaffected myocardium (LV). From Figure 2B it is evident that most of the radioactivity was found within the liver. Interestingly, radioactivity tended to be higher within the lungs of animals that had received cross-linked cells compared to lungs of animals that had received single hEPCs.
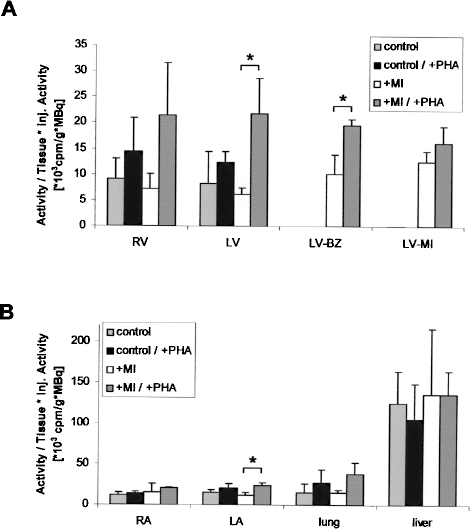
Distribution of radioactivity within different regions of the heart (A) and different organs (B) 2 days after coronary hEPC transplantation with and without myocardial infarct (MI) and with and without PHA treatment of cells. Hearts were dissected into right ventricle (RV), left ventricle (LV), left ventricular border zone (LV-BZ), and ischemic area of left ventricle (LV-MI) (A). Right atrium (RA) and left atrium (LA) are compared with values from lung and liver (B). Radioactivity of all samples was measured using a bore hole. Values given were calculated taking into account measured radioactivity, weight of the sample, and injected radioactivity.
Although we measured only radioactivity, we believe that these results can be related to cell numbers for two reasons. First, cell viability in vitro after labeling with 111In-oxinate and cross-linking with PHA did not significantly decline in hEPCs. Secondly, we have previously performed similar experiments using HUVECs (5) and were able to demonstrate that cell numbers calculated from radioactivity measurements and manually counting of fluorescent HUVECs showed no differences, thereby demonstrating the reliability of the method.
In summary, the present study demonstrates that the amount of hEPCs transplanted into the heart considerably decreases with time. However, cross-linking of hEPCs with PHA significantly enhances the deposition of hEPCs, particularly into the border zone of an infarct, and delays the clearance from the heart significantly. Thus, cross-linking of cells may provide a promising approach to enhance the number of transplanted cells in the infarcted heart.
