Abstract
Bone marrow-derived endothelial progenitor cells (EPCs) have been shown to contribute to not only angiogenesis in ischemic tissue but also neovascularization in uterine endometrium formation. Reduced neovascularization and elevation of serum soluble Flt1, a functional blockage of VEGF, in the development of placenta is thought to be one of the major causes of repeated miscarriages in gestation. We then examined whether transfusion of VEGF-expressing extrinsic EPCs prevented frequent miscarriage via its promotional effect on neovascularization with a VEGF–eNOS signaling pathway in a mouse miscarriage model. The results showed that systemic EPC transfusion significantly reduced the rate of miscarriage, and EPCs were frequently observed in the miscarriage placenta. In contrast, only a few EPCs were detected in the placenta of normal gestation. The vascular pattern was irregular, and vessel size was small in the miscarriage placenta compared with that of normal gestation. The placental vascular pattern in miscarriage tended to be normalized with increased vessel size up to a similar level as normal gestation by EPC recruitment. For the mechanistic insight, since soluble Flt1 inhibits EPC functions, it was suggested that the increased soluble Flt1 could suppress the recruited EPC functional activity in the miscarriage placenta. In vitro experiments by soluble Flt1 treatment in cultured EPCs suggested that the vascular abnormality could be partly due to the inhibition of eNOS expression by the increased amounts of soluble Flt1. These findings from animal experiments indicated that autologous EPC therapy may be a novel therapy to prevent miscarriage in high-risk pregnancies, such as preeclampsia.
Keywords
Introduction
Miscarriage is one of the most common complications of pregnancy and is defined as the spontaneous loss of a pregnancy before the fetus reaches viability, including all pregnancy losses from the time of conception until 24 weeks of gestation. Although approximately 15% of clinically recognized pregnancies result in miscarriage, total reproductive losses are over 40% (4,33). Specifically, recurrent miscarriage affects about 1% of couples (31), and at least 25% of women experience one or more sporadic miscarriages generally due to unspecified fetal genetic abnormalities (30,31). The risk of miscarriage has been reported to rise along with increased maternal age (23). Angiogenesis is the development of new blood vessels from preexisting vasculature and is a crucial event for fetal growth and placental development (36). A balance of angiogenic and antiangiogenic factors is essential for a successful pregnancy (15,28). Recent emerging evidence suggests that an imbalance of angiogenic/antiangiogenic factors is involved in the pathophysiology of preeclampsia (PE) (3,9), a pregnancy complication often associated with small-for-gestational-age (SGA) neonates (10,25), spontaneous preterm labor and delivery (29), and stillbirth (24). The elevated serum level of circulating soluble fms-like tyrosine kinase 1 (sFlt1), a receptor of angiogenic vascular endothelial growth factor (VEGF), has been reported to play a pathogenic role in patients with preeclampsia (13,16).
Bone marrow (BM)-derived vascular precursor cells have the potential to differentiate into endothelial cells and are collectively referred to as endothelial progenitor cells (EPCs) (1). Emerging evidence indicates that EPC-participating vasculogenesis contributes significantly not only to postnatal neovascularization but also to endometrial vascular development along with the menstrual cycle (19). These novel insights that contribute to the formation of new blood vessels suggest a potential strategy for the treatment of ischemic diseases. Thus, the therapeutic transplantation of EPCs was developed to induce neoangiogenesis, which also plays a critical role for tissue regeneration including functional recovery of organs, at least in part, by activation of the VEGF–PI3K/Akt–endothelial nitric oxide synthase (eNOS) pathway (6). Furthermore, chemoattractants for EPCs, such as stromal cell-derived factor-1 (SDF-1) (5), insulin-like growth factor 2 (IGF-2) (18), VEGF, and placental growth factor (PlGF) (14) have also been reported to be produced in the placenta (18,32,35). We therefore hypothesized that EPCs played a role in placental vascular development or normalization, specifically in the case of miscarriage.
The objective of this study was to determine whether frequency of miscarriage is reduced by EPC transfusion via placental neovascularization and to explore a certain mechanism for the effect of sFlt1 on EPC functions involving the VEGF–eNOS pathway in miscarriage using a mouse recurrent miscarriage model.
Materials and Methods
Animal Experiments
All animal procedures were performed according to the guidelines of the Osaka Medical College Animal Care and Use Committee with the approval protocol number 24010. Twenty male DBA2 mice (8–14 weeks old; CLEA, Tokyo, Japan) were mated with 20 female CBA/J mice (10–14 weeks old; CLEA) to generate pregnant mice with frequent miscarriages (miscarriage group, MisCar). Twenty male BALB/c mice (8–14 weeks old; CLEA) were mated with 20 female CBA/J mice (10–14 weeks old; CLEA) to generate pregnant mice with normal gestation (normal gestation group, N-Gest).
We used CM-DiI dye (CellTracker™; Invitrogen, Tokyo, Japan) as a cell tracker because it does not affect viability and is retained in living cells through several generations, maintaining similar intensity. The dye is transferred to daughter cells, but not adjacent cells in a population, and will not remain in situ by sweeping away with macrophages after the labeled cells die. The red fluorescent (CM-DiI)-labeled cultured EPCs [5 × 10 5 in 100 μl of phosphate-buffered saline (PBS)/mouse] isolated from CBA/J bone marrow or PBS alone (control; Invitrogen) were intravenously transfused via a tail vein in the MisCar group (MisCar EPC group) and the N-Gest group (N-Gest EPC group) on days 7 and 14 after checking vaginal plugs in female mice. On day 18, the miscarriage rate was calculated, and the placentas were examined by fluorescence immunohistochemistry (IHC) and quantitative real-time RT-PCR.
Quantitative Real-Time RT-PCR Analyses
Total RNA was extracted from the placenta using an RNeasy Mini Kit (Qiagen, Hilden, Germany), and reverse transcription was performed using a PrimeScript™ II 1st strand cDNA Synthesis Kit (Takara Biochemicals, Kyoto, Japan) according to the manufacturer's instructions. For quantitative RT-PCR, the converted cDNA samples (2 μl) were amplified in triplicate by a real-time PCR machine (CFX Connect; Bio-Rad, Hercules, CA, USA) in a final volume of 10 μl using SsoFast EvaGreen Supermix reagent (Bio-Rad) with gene-specific primers (Table 1). Melting curve analysis was performed with the CFX Manager software (Bio-Rad), and the mean quantification cycle (Cq) values were used to calculate gene expression with normalization to mouse glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Supplementary Fig. 1; supplementary material is available at https://www.dropbox.com/s/njvl5otgdouwtrk/Supplmentary%20Figures.pdf?dl=0). Primers were designed with a basic local alignment tool (BLAST; http://www.ncbi.nlm.nih.gov/BLAST/) to avoid regions of similarity between biological sequences, and the amplification efficacy was calculated by standard curve (Supplementary Fig. 2; https://www.dropbox.com/s/njvl5otgdouwtrk/Supplmentary%20Figures.pdf?dl=0). We first confirmed that GAPDH is a suitable housekeeping gene for normalization of each gene expression in EPCs (Supplementary Fig. 3; https://www.dropbox.com/s/njvl5otgdouwtrk/Supplmentary%20Figures.pdf?dl=0).

Gene profiling of the placenta in normal gestation versus miscarriage and circulating EPC levels. (A) A littermate in one pregnant mouse with miscarriage on day 18 in gestation (arrow, miscarried fetus; arrowhead, placenta of miscarried fetus; ∗normal fetus with placenta). (B) The mRNA expressions of IGF-2, PIGF, SDF-1, sFlt1, and VEGF were measured by a quantitative real-time RT-PCR, and each expression level of the MisCar group (n = 8) versus that of the N-Gest group (n = 10) was analyzed statistically (ns, not significant; ∗p < 0.05; ∗∗∗p < 0.001; ∗∗∗p < 0.0001). (C) The percentage of CD34+ circulating EPCs was measured in nonpregnant mice (N-Preg group, n = 7), N-Gest group, and MisCar group (ns, not significant).

Effect of sFlt1 on cultured EPC migration, proliferation, and growth factor/chemokine gene expression. (A) The migration and (B) proliferation activities of cultured EPCs with [sFlt1 (+)] or without [sFlt1 (-)] recombinant mouse sFlt1 protein (300 ng/ml) were assessed (∗p < 0.05). (C) The mRNA expressions of VEGF-A, eNOS, and SDF-1 of cultured EPCs (n = 11/each) with [sFlt1 (+)] or without [sFlt1 (-)] recombinant mouse sFlt1 protein (300 ng/ml) were measured by a quantitative real-time RT-PCR (ns, not significant; ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001).

Effect of sFlt1 on cultured EPC differentiation into functional endothelial lineage cells. Double fluorescence immunocytochemistry for ILB4 (green) and eNOS (red) (A) and ILB4 (green) and CD31 (red) (B) was performed in cultured EPCs with [sFlt1 (+)] or without [sFlt1 (-)] recombinant mouse sFlt1 protein (300 ng/ml). The merged images of ILB4 and eNOS or CD31 are shown in the right, and negative staining controls with PBS without antibodies are also shown as PBS for ILB4 and PBS for CD31.
Specific Primers for Quantitative Real-Time RT-PCR Amplifications

CD34+ Cell Analysis
Freshly isolated mononuclear cells (MNCs) from mouse peripheral blood were incubated with 0.5% bovine serum albumin (BSA; Sigma-Aldrich, St. Louis, MO, USA) in PBS (Invitrogen) containing fluorescein isothiocynanate (FITC)-conjugated monoclonal antibody against CD34 (1:100; BD Pharmingen, San Diego, CA, USA) for 30 min on ice. The cells stained with FITC-conjugated rat anti-mouse IgG2a (BioLegend, San Diego, CA, USA) were used as negative controls. The cells were washed with PBS and fixed with 0.5% paraformaldehyde (PFA) (Wako, Osaka, Japan)/PBS for 15 min at room temperature (RT) to avoid fading the fluorescent signal. Samples were analyzed using an image-based cytometer (Tali™; Invitrogen) according to the manufacturer's instructions. Briefly, nine different high-power fields per sample were analyzed for calculating the CD34+ percentage. Three independent assays were performed with three different MNC samples, and the representative result was demonstrated.
EPC Isolation and Culture
EPC isolation and culture were performed as described previously (11,27). Briefly, MNCs were harvested from female CBA/J mice (10–14 weeks old; CLEA) BM and seeded onto culture dishes. After 24 h in culture with 10% fetal bovine serum (FBS; HyClone, Logan, UT, USA)/Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich), a subpopulation of floating cells were further seeded onto rat vitronectin (Sigma-Aldrich)-coated dish. After 3 days in culture with EPC differentiation medium, cells differentiated into endothelial lineage cells exhibiting adherent phenotypes and endothelial characteristics of acetyl low-density lipoprotein (LDL) uptake/isolectin B4 (ILB4) binding and were used as an EPC-rich population for experiments. We previously confirmed that the cells isolated with this method had an endothelial phenotype by immunocytochemistry, fluorescence-activated cell sorting (FACS) analysis, and real-time RT-PCR analysis for gene profiling (27).
Migration Assay
Migration activity of cultured EPCs was evaluated with a chemotaxis chamber assay as described previously (12). Briefly, the polycarbonate filter (5-μm pore size) (Transwell; Corning, Corning, NY, USA) was placed between the upper and lower chambers. The cultured EPCs were coincubated with recombinant mouse sFlt1 protein (300 ng/ml; R&D Systems, Minneapolis, MN, USA) for 24 h in 0.5% FBS/endothelial basal medium 2 (EBM2) (Lonza, Tokyo, Japan) before migration assay. The sFlt1-pretreated EPC suspensions (5 × 10 4 cells/well) were placed in the upper chamber, and the lower chamber was filled with 0.5% FBS/EBM2 (Lonza) with growth factors alone or that with a chemoattractant SDF-1α (100 ng/ml) (R&D Systems). The cells were incubated for 16 h at 37°C in 5% CO2. The total number of migrated cells on the lower chamber side of polycarbonate filter was evaluated in each well and expressed as the migration activity. The polycarbonate filter with migrated cells was fixed with 2% PFA/PBS and stained with 4,6-diamidinophenylindole (DAPI) (Sigma-Aldrich) to visualize nuclei. The DAPI-positive nuclei were counted in three polycarbonate filters and averaged for comparison. The experiment was repeated for over three times, and the representative result was demonstrated.
Proliferation/Viability Assay
Proliferation activity and viability of the cultured EPCs were examined with the use of Cell Counting Kit-8 (Dojindo, Tokyo, Japan) according to the manufacturer's instructions (12). Briefly, cultured EPCs (10,000 cells/well) were seeded on 96-well flat-bottomed plates with 100 μl of growth medium (EGM2-MV BulletKit; Lonza) Then cells were incubated in the presence (100 ng/ml) or absence of recombinant mouse sFlt1 protein (R&D Systems) for 48 h at 37°C in 5% CO2. The absorbance at 570-nm wavelength was recorded in eight wells in each group with the use of a 96-well enzyme-linked immunosorbent assay (ELISA) plate reader (SPECTRA MAX 190; Molecular Devices, Tokyo, Japan), and the data were averaged for comparison. The experiment was repeated for over three times, and the representative result was demonstrated.
Histological Assessment of the Placenta
For fluorescence IHC, tissues were embedded in optimum cutting temperature (OCT compound; Sakura, Tokyo, Japan) and snap frozen in liquid nitrogen. Frozen cross sections of 6-μm thickness were mounted on silane-coated glass slides and stained with FITC-isolectin B4 (B-1205; 1:200; Vector Laboratories, Burlingame, CA, USA) for assessment of vascularity in green, and the recruited DiI-labeled EPCs were visualized in red under a fluorescence microscope (BZ-X700; Keyence, Osaka, Japan), and the acquired images were analyzed by stereology-based software (BZ-3XD, BZ-H3D, and BZ-H3M; Keyence) (8,22). All histological sections were scanned in multiple layers with a 1-μm interval through a full-focus image-processing module (BZ-H3XD; Keyence). The recruited DiI-positive (red) EPCs were counted, and vessel area (green)/vessel perimeter was measured using a measurement module (BZ-H3M; Keyence) and a hybrid cell counting module (BZ-H3C; Keyence) in five selected high-power fields (HPF)/section (magnification: 100×) and averaged for quantitative analysis, respectively.
Statistical Analysis
Statistical analyses were performed with commercially available software (GraphPad Prism 6™; MDF Co. Ltd., Tokyo, Japan). Statistical comparisons between two groups were performed by Mann–Whitney U tests, and among multiple groups by Kruskal–Wallis tests followed by Dunn's post hoc procedure to determine statistical significance. All values are expressed as median and interquartile range. All in vitro experiments were repeated at least in triplicate and analyzed. The authors had full access to the data and take full responsibility for its integrity.
Results
Placental Gene Expression Profile Is Altered in Miscarriage
As shown in Figure 1A, two out of seven placentas exhibited undergrown fetus in a littermate of the MisCar group. Although there was no significant difference in PlGF and SDF-1 mRNA expressions between the MisCar group and the N-Gest group, the mRNA expressions of IGF-2, VEGF, and sFlt1 were significantly upregulated in the MisCar group compared with those in the N-Gest group (Fig. 1B). sFlt1 is a spliced variant of Flt1, and we cannot distinguish the mRNA of sFlt1 from that of Flt1. However, since the placenta is a major source of sFlt1 production in gestation (7), the mRNA expression level of Flt1 reflects sFlt1 production activity in the placenta. On the other hand, the percent of circulating EPCs, which were detected as CD34+ cells (2,19), exhibited no significant difference among nonpregnant mice (Normal group), N-Gest group, and MisCar group (Fig. 1C), suggesting that the number of circulating EPCs was not influenced regardless of whether a miscarriage occurred. We then focused on the upregulation of sFlt1 mRNA among the upregulated genes because only sFlt1 might inhibit functions of EPCs that need VEGF for their activity, including its contribution to placental vascular development even if circulating EPCs recruit to the placenta by IGF-2 and VEGF, and therefore examined the effect of sFlt1 on EPC functions in the next series of experiments. Interestingly, the gene expression of VEGF-A165b, which has been reported to be an antiangiogenic form of VEGF-A, tends to be upregulated along with that of VEGF-A in the placenta with miscarriage (Supplementary Fig. 4 https://www.dropbox.com/s/njvl5otgdouwtrk/Supplmentary%20Figures.pdf?dl=0), suggesting that VEGF-A165b production might also negatively contribute to placental vascular development in miscarriage.

Effect of exogenous EPC administration on miscarriage rate in pregnant mice. (A) Fluorescence immunohistochemistry for ILB4 was performed with placentas in the N-Gest and MisCar groups. The tissue cross sections were stained with FITC-isolectin B4 for assessment of vascularity in green and the recruited DiI-labeled EPCs were visualized in red under a fluorescence microscope. Arrows, DiI-labeled EPCs. (B) The recruited DiI-positive (red) EPCs were counted in five selected high-power fields (HPF) and averaged for quantitative analysis. n = 10; ∗∗∗p < 0.0001. (C) The miscarriage rates were calculated by checking the littermates in the N-Gest (n = 11), MisCar (n = 8), and MisCar EPC groups (n = 11) on day 18 of gestation (ns, not significant; ∗p < 0.05; ∗∗p < 0.01).
sFlt1 Inhibits EPC Functions
To examine the effect of sFlt1 on EPC functions, we evaluated the migration activity, proliferation activity, and growth factor/cytokine production with or without sFlt1 treatment in cultured EPCs. The sFlt1 significantly inhibited migration activity and proliferation activity of EPCs (Fig. 2A and B). Although the mRNA expression of VEGF was not affected by sFlt1, sFlt1 significantly reduced eNOS and SDF-1 mRNA expressions in EPCs (Fig. 2C). The protein expression level of eNOS merged with isolectin B4, an endothelial cell marker, was also decreased by the treatment of sFlt1 (Fig. 3A), whereas the expression level of CD31 was not changed (Fig. 3B), suggesting that sFlt1 does not affect endothelial differentiation of EPCs at all, but inhibits eNOS production. Since eNOS plays a role in the microcirculation production of NO in the development of neovessels, the lack of eNOS production in EPCs might result in the reduction of EPC's favorable paracrine effect on neovascularization by placenta-derived sFlt1.
Transfused EPCs Home to the Abnormal Placenta and Prevent Miscarriage
We next examined whether exogenously transfused cultured EPCs reduce the frequency of miscarriage in gestation. The rate of miscarriage was over 20% in the MisCar group, while it was limited to around 5% in the N-Gest group. The cultured EPCs labeled with DiI-red fluorescent dye were systemically infused into pregnant mice via a tail vein, and the placenta/fetus was examined. The EPC transfusion significantly reduced the rate of miscarriage (Fig. 4C). We then evaluated EPC recruitment to the placenta with or without miscarriage. The transfused EPCs were frequently recruited to the placenta with miscarriage, but to a lesser extent to the placenta with normal gestation (Fig. 4A). The quantitative analysis also showed that the number of recruited EPCs was significantly higher in the MisCar group than in the N-Gest group (Fig. 4B). Vascularity was also assessed by FITC-isolectin B4 staining. Interestingly, the vascular pattern was irregular, and vessel size was significantly smaller in the MisCar group compared to the N-Gest group. On the other hand, the placental vascular pattern tends to be normalized in the MisCar EPC group (Fig. 5A). Indeed, both vessel area and perimeter were significantly reduced in the MisCar group compared with the N-Gest group, and EPC transfusion significantly increased the parameters (MisCar EPC group) up to similar levels as that of the N-Gest group (Fig. 5B). These findings suggest that the exogenously transfused EPCs recruited to the placenta with miscarriage normalized the vascular pattern, leading to the reduced rate of miscarriage.
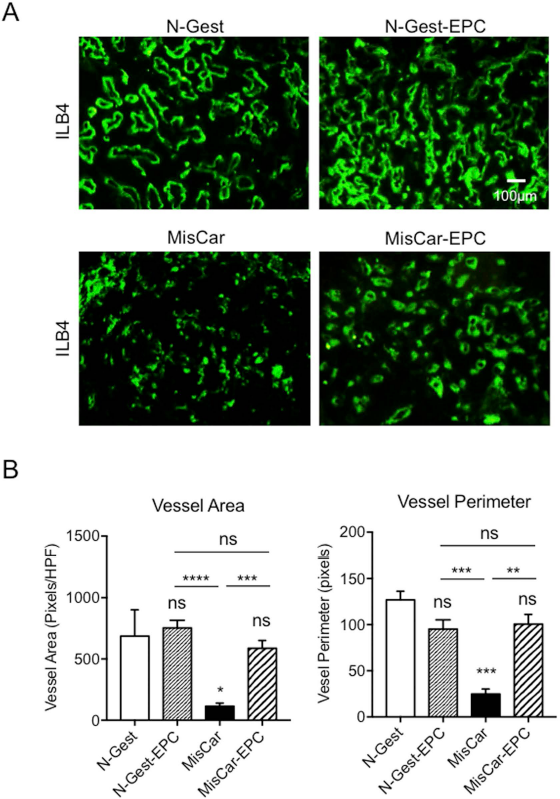
Effect of exogenous EPC administration on placental vasculature in pregnant mice. (A) Fluorescence immunohistochemistry for ILB4 was performed with placentas in N-Gest, N-Gest with exogenous EPC administration (N-Gest EPC), MisCar, MisCar with exogenous EPC administration (MisCar EPC). Frozen cross sections were stained with ILB4 in green. The normal placentas in pregnant mice (N-Gest) were used as a control. (B) To examine the effect of exogenous EPCs on the vascular development of placentas, the vessel area (green fluorescent area, left) and the vessel perimeter (right) were measured and analyzed quantitatively (ns, not significant; ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001; ∗∗∗p < 0.0001 versus N-Gest, n = 10 in each group.
Discussion
In the present study, we have investigated the role of bone marrow-derived circulating EPCs in gestation, specifically focusing on EPC contribution to placental vascular development, and obtained the following evidence: 1) vascular development was impaired, and gene expressions of sFlt1, IGF-2, and VEGF were upregulated in the placenta with miscarriage; 2) EPC functions were reduced by sFlt1; and 3) exogenously transfused EPCs homed to the placenta and normalized vascular structure, resulting in the reduced rate of miscarriage in gestation.
sFlt1, a soluble receptor for VEGF, has been reported to be one of the key molecules for recurrent miscarriage in clinical settings (25,29). Since sFlt1 is able to neutralize biological activity of VEGF, even though the VEGF gene is upregulated in the placenta (Fig. 1B), the effect on neovascularization could be inhibited by the production of sFlt1 in the placenta with miscarriage. Circulating EPCs are known to contribute to neovascularization in the development of the placenta (19). Our in vitro data suggest that increased serum levels of sFlt1 and upregulation of sFlt1 in the placenta (21) could inhibit EPC functional activities before and after the homing to the placenta, resulting in impaired vascular development. Since the upregulated genes IGF-2 (18) and VEGF in the placenta with miscarriage are chemoattractants for circulating EPCs, a number of exogenously infused cultured EPCs, and perhaps endogenous circulating EPCs as well, homed to the placenta with miscarriage compared with that without miscarriage (Fig. 4A and B), toward the secreting IGF-2 from the placenta with miscarriage. Since RNA was isolated from whole placentas for quantitative RT-PCR analysis, it remains unclear whether the IGF gene was upregulated at either the maternal site or the fetal site of the placenta. These results suggest that patients with frequent miscarriages have lower numbers of EPCs in their blood (26,34).
Since the exogenously infused cultured EPCs are not impaired functionally and produce proangiogenic factors, they could home to sites of ischemia in the dysfunctional placenta, exhibiting the promotional effect on angiogenesis and vascular development (Fig. 5). Why did exogenously infused cultured EPCs reduce the rate of miscarriage in gestation? It is likely that the external EPCs were recruited to the placenta probably due to the release of IGF-2 in the placenta. The endogeneous EPCs are thought to be inactivated by high concentrations of sFlt1, which might inhibit the VEGF-induced activation of EPCs in an autocrine manner. The exogenously infused EPCs might act as promoters of placental vascular development before the inhibition of their activities by sFlt1.
Endothelium-derived NO might play an important role in the maintenance of basal vascular tone in the placenta (17,31). As shown in Figure 3A, sFlt1 significantly inhibited the expressions of eNOS mRNA and protein in cultured EPCs. Since placental sFlt1 and fetal liver kinase 1 (Flk1) have been reported to be upregulated and downregulated, respectively (20), reduced production of NO may cause vascular abnormality/miscarriage, perhaps by both neutralization of VEGF activity with sFlt1 and reduced vascular Flk1 expression resulting from the inhibition of the VEGF–PI3K/Akt–eNOS signaling pathway. Although the mechanism by which sFlt1 is upregulated in the miscarriage placenta remains unclear, the increased VEGF might be the trigger. The placenta rapidly grows and stops growing when it reaches the appropriate size for fetal development during gestation. In the meantime, neovascularization occurs very rapidly and is mediated by VEGF in the beginning, and it terminates when the fetus acquires sufficient blood supply. For this reason, the placenta is thought to produce sFlt1 to inhibit VEGF. Although the abnormal regulation of sFlt1 production should exist in the placenta with miscarriage, we have shown that exogenously infused EPCs ameliorated miscarriage, indicating that the freshly isolated EPCs could be a therapeutic tool for miscarriage. Nevertheless, further investigation will be required to reveal the precise mechanism for the favorable effects of EPCs on miscarriage.
In conclusion, autologous EPC transfusion is considered to be a novel therapy to prevent miscarriage in high-risk pregnancies, such as those with preeclampsia. To establish a safe and feasible EPC therapy for miscarriage, further optimization would be required to explore the role of EPCs in the placenta.
Footnotes
Acknowledgement:
We thank Ms. Eiko Kohbayashi for her technical assistance in the real-time RT-PCR analysis, immunohistochemistry, and in vitro experiments in this study. The authors declare no conflicts of interest.
