Abstract
The de novo engineering of a uniform hepatocyte sheet in vitro is considered as a novel approach for liver-directed therapeutics. Hepatocytes can be cultured on a temperature-responsive culture dishes coated with poly(N-isopropylacrylamide) (PIPAAm). Following multiple days of culturing, the hepatocytes can be easily harvested as a uniform sheet by decreasing temperature from 37°C to 20°C. By modifying the sheet harvesting protocol, we have noticed that two different forms of the hepatocyte sheets, “extended” and “shrinking,” were obtained. This study describes the methods for harvesting the two different forms of sheets, and their cellular structure and hepatocyte-specific functions. To obtain an “extended sheet” form, a cluster of hepatocytes covered with a support membrane was harvested by the temperature reduction. For the “shrinking sheet” form, the hepatocyte sheet was floated after reducing the culture temperature, and the floating process allowed the sheet to shrink spontaneously. Histological analysis revealed that the hepatocytes in the extended sheet form were predominantly flat, whereas the shrinking sheet contained cuboidal shaped hepatocytes. The preservation of hepatocyte-specific ultrastructures was confirmed in both types of sheets. To investigate hepatocyte-specific functionality, the harvested hepatocyte sheets were recultured on Matrigel-coated dishes. Assessment of protein production levels and chemical metabolizing activities showed the similar functionalities for each form. In contrast, the recalculation of these values per sheet versus per square centimeter of sheet surface demonstrated that the function of the shrinking sheet was significantly higher than that of the extended sheets. This study demonstrated that the hepatocyte sheets created on the PIPAAm dish could spontaneously shrink in size, but retain their hepatocyte functionality. This type of hepatocyte sheet could be utilized for the engineering of liver tissue in limited areas that are unable to give adequate transplant space.
Introduction
Hepatocytes play essential roles in detoxification, the production of enzymes and plasma proteins, and the metabolism of cholesterol, carbohydrate, and lipid. A hepatocyte culture system has been extensively used for the basic researches of pharmacology (13,32,34) and pathophysiology (22,28), and for the study of hepatic diseases (8). A culture system using primary hepatocytes was also applied to tissue engineering where hepatocytes are seeded on a biomaterial scaffold (14), extracellular matrix components (5), or thermoresponsive culture surfaces (31). Once hepatocytes favorably attached to each other, the cultured composites were then implanted into experimental animals for creating functional liver systems in vivo. This tissue engineering approach to create liver system in vivo has been highly expected as a next generation therapy toward liver diseases (12,24–31).
Under conventional culture conditions, once hepatocytes complete a cell attachment process on a culture surface, the hepatocytes start to spread and expand their pseudopods toward extending their shapes. As the progress of the primary culturing, the hepatocytes gradually deteriorate various hepatocyte-specific functions (35). Researchers reported that the changes of hepatocyte shape closely related with their cellular functions. The cuboidal cell shape of hepatocytes was reported to be maintained on micropatterned surfaces (35) and on Engelbreth-Holm-Swarm sarcoma-derived extracellular matrix (18,19,23). The culturing hepatocytes between two collagen layers like sandwich (18) allowed the hepatocytes to maintain their cuboidal shape and restored several differentiated functions such as albumin, transthyretin, and apolipoprotein A-1 productions. In contrast, Singhvi et al. (35) found that flatly extended hepatocytes showed high levels of DNA synthesis while losing some hepatocyte-specific functions. These reports suggested that hepatocyte shape was one of key regulators in hepatocyte-specific functions, dedifferentiation, cell proliferative growth, and cell survival, and thus prompted us to regulate hepatocyte shape morphology for using the cells for liver tissue engineering.
In the fields of tissue engineering and regenerative medicine, the creation of lamellar assembled tissues in vitro has been one of the major paradigms for recreating complex and functional tissues. Recently, our group created a temperature-responsive culture dish made of covalently grafted temperature-responsive polymer, poly (N-isopropylacrylamide) (PIPAAm) with nanometer-scale thickness (33,40). The lower critical solution temperature (LCST) of PIPAAm is 32°C (33). At regular culture temperature (37°C), PIPAAm is hydrophobic in water, but reversibly becomes hydrophilic at the temperature below the LCST. This hydrophobic nature of PIPAAm at 37°C allows various kinds of cells to adhere, spread, and proliferate on the culture surface, and to create numerous intercellular communications. After cultured cells reach a confluent state, a temporal temperature decrease to 20°C forces PIPAAm to be a hydrophilic form, resulting in the detachment of the cultured cells as an intact cell sheet while maintaining the intercellular matrix.
Using PIPAAm dishes, our laboratory recently established a procedure for fabricating hepatocyte sheets that were transplantable and engraftable in vivo for liver tissue engineering purposes (31). During the course in harvesting the hepatocyte sheets from PIPAAm dishes, two different kinds of hepatocyte sheets, which had the same number of cells and different surface area, could be obtained. When a hepatocyte sheet was prepared by the usual procedure without using the sheet-supporting membrane described above, a “shrinking sheet” form could be obtained. Once the hepatocyte sheets floated in the culture medium, the sheets naturally shrunk. When the support membrane was covered over the hepatocyte cluster in the culture dish before the temperature change to 20°C, an unshrinking hepatocyte sheet, “extended sheet” form, which had the same surface area as the hepatocyte cluster, was obtained. It would be essential to determine and understand the possible structural and functional disturbances of the cells of the hepatocyte sheet that appear by the two different harvesting procedures for preparing useful hepatocyte sheets.
This study aimed to elucidate the structural and functional differences between the two different shapes of hepatocyte sheet forms prepared by two different harvesting procedures. Histological analysis revealed that the sheet forms, which were harvested by the two different procedures, were composed of two different shapes of hepatocytes: cuboidal hepatocytes or flatly extended hepatocytes. This study was then designed to determine hepatocyte-specific functions by reculturing the two hepatocyte sheet forms in terms of protein expressions, drug metabolism and hepatocyte-specific mRNA expressions.
Materials and Methods
Animals
Male transgenic mice (hA1AT-TG; kindly provided by Dr. Bumgardner, Ohio State University, Columbus, OH, USA) [expressing human α1-antitrypsin (hA1AT) driven by the liver-specific A1AT promoter], 14–19 weeks old, were used for the isolation of hepatocytes (3). All animals were caged in an environmentally controlled room with alternating 12-h dark/light cycles and ad libitum access to food and water. All animal experiments were performed in accordance with the guidelines outlined by the institutional animal committee located at Tokyo Women's Medical University.
Isolation of Hepatocytes and Preparation of Hepatocyte Sheets
hA1AT-TG mouse hepatocytes were isolated by a two-step in situ collagenase perfusion as described previously (25,31,41). Isolated hepatocytes were filtered and then purified by low speed centrifugation at 50 × g, followed by 40% Percoll (GE Healthcare Bio-Sciences, Piscataway, NJ) gradient centrifugation. Hepatocyte viability was determined by trypan blue exclusion test. Hepatocytes with more than 90% viability were plated on a 35-mm temperature-responsive PIPAAm culture dish (UpCell; CellSeed, Tokyo, Japan) at a density of 8 × 105 cells/dish. Hepatocytes were cultured as described previously (28,31) at 37°C for the first 24 h, and then the medium was replaced daily with fresh culture medium with supplemental reagents containing 0.5 μg/ml insulin (Wako Pure Chemical Industries, Osaka, Japan) and 10 ng/ml epidermal growth factor (EGF) (Invitrogen, Carlsbad, CA) until cell sheet harvesting (9).
Harvesting of Hepatocyte Sheets
After 3 days, the temperature was reduced to 20°C for 20 min for inducing the detachment of hepatocyte cluster from the culture surfaces, resulting in a hepatocyte sheet. The following two different hepatocyte harvesting procedures were used for preparing hepatocyte sheets that were made from the same number of cells with two different surface areas. (i) For obtaining an extended sheet form (Ex-form), prior to reducing culture temperature, a support membrane (CellShifter; CellSeed) was covered over the hepatocyte cluster. Culture dishes were then placed in a 20°C incubator for 20 min, and a hepatocyte sheet with the support membrane was obtained. (ii) For obtaining a shrinking sheet form (Sh-form), the culture dish was placed in 20°C incubator for 20 min. By gentle pipetting the culture medium toward the edge of the hepatocyte clusters, the hepatocyte sheet floated in the culture medium. The floating process was found to spontaneously shrink in size while maintaining its 2D structures. After the aspiration of culture medium, the support membrane was covered onto Sh-form for 10 min for handling. Both types of hepatocyte sheet forms were processed for histological analyses, electron microscopic analyses, and reculturing functional analyses.
Histological Analysis of Harvested Hepatocyte Sheets
Hepatocyte sheets harvested as Ex-form and Sh-form were fixed with 4% paraformaldehyde for 48 h. After being embedded in paraffin and sliced into a 5-μm section, the hepatocyte sheet specimens were stained with hematoxylin and eosin (H&E) by conventional methods. The thickness of Ex- and Sh-forms was assessed by measuring the cell heights of six cells at randomly selected points per cell sheet. The representative cell height value was expressed by the mean of six data in each sheet. The length of the horizontal axis of hepatocytes was measuring at randomly selected 20 hepatocytes per sheet. The representative value of cell axis in each sheet was expressed by the mean of 20 data measured in each sheet.
Reculture of Hepatocyte Sheets
The harvested hepatocyte sheets were recultured with culture medium containing supplemental reagents in Matrigel-coated dishes (BD BioCoat Matrigel Cellware, 60 mm; BD Biosciences, Bedford, MA). Before the plating of hepatocyte sheets, the Matrigel-coated dishes were incubated with the culture medium for hydrating the dish surface. After the removal of the culture medium, hepatocyte sheets attached to the support membranes were placed on the dishes. Hepatocyte sheets were recultured for 12 h with 1 ml of culture medium, followed by the removal of the support membranes. Medium change (3 ml) was performed at 24 h of the reculture experiment, and the medium was collected at 48 h.
Fluorescence Staining of Recultured Hepatocyte Sheets
Recultured hepatocyte sheets were stained with phalloidin-rhodamine (Sigma-Aldrich, St. Louis, MO) and Hoechst 333258 (Wako Pure Chemical Industries) for F-actin and nuclei stainings, respectively (32). The thickness of the extended and the shrinking sheets was obtained by the measuring cell height at randomly selected 13 points per sheet. The mean of 13 obtained data became the representative value of the cell axis each sheet.
Transmission Electron Microscopy
For ultrastructural analyses, the harvested and 2-day cultured hepatocyte sheets were fixed with 2% paraformaldehyde and 2% glutaraldehyde in 0.1 M phosphate buffer. The specimens were dehydrated through a series of graded ethanol and embedded with Quetol-812. Ultrathin sections (70 nm) were stained with uranyl acetate and lead citrate and observed at 80 kV by a transmission electron microscope (JEM-1200EX; JEOL, Tokyo).
Enzyme-Linked Immunosorbent Assay (ELISA)
The amount of albumin and hA1AT secreted into the culture medium from recultured hepatocyte sheet for 24 h (between day 1 and 2 of reculture) was determined by ELISA. For albumin measurement, an Albumin ELISA Quantitation Kit (Bethyl Laboratories, Montgomery, TX) was used. For hA1AT measurement, anti-human α1-antitrypsin IgG (DiaSorin, Stillwater, MN) and goat anti-α1-antitrypsin horse radish peroxidase (HRP)-conjugate (Fitzgerald Industries International, Concord, MA) were used as described previously (25,30,31,41).
The Drug Metabolic Activity of the Hepatocyte Sheet
Twenty-four hours after the start of reculture experiment, 150 μg lidocaine (xylocaine) (AstraZeneca, London, UK) or 16.05 μg ammonium chloride (Wako Pure Chemical Industries) was inoculated into the culture medium for 6 h. The residual amount of lidocaine in the culture medium was determined by enzyme immunoassay at the Special Reference Laboratories (SRL; Tokyo). The residual amount of ammonia was determined by an Ammonia-Test-Wako kit (Wako Pure Chemical Industries). The amounts of metabolized lidocaine and ammonia were calculated by subtracting the values of experimental group from those of non-sheet control groups.
Gene Expression Analysis
The hepatocyte sheets were collected at 48 h after the start of reculture. Total RNA extraction and the synthesis of first-strand cDNA were performed as previously described (37,38). The cDNA samples were subjected to real-time PCR by a 7300 Real Time PCR System (Applied Biosystems, Foster City, CA) with TaqMan probes (Applied Biosystems) as listed in Table 1. Nine housekeeping genes (HKGs), including 18S rRNA (18S), β-actin (ACTB), β-glucuronidase (GUSB), glyceraldehyde-3-phosphate dehydrogenase (GAPDH), hypoxanthine phosphoribosyltransferase (HPRT1), peptidylprolyl isomerase A (PPIA), ribosomal protein L4 (RPL4), TATA box binding protein (TBP), and transferrin receptor (TFRC), amplified by real-time RT-PCR were analyzed by statistical algorithms using two computer programs, geNorm (39) and NormFinder (1), for determination of reference genes as described previously (37,38).
Characteristics of Target Genes Evaluated by Real-Time RT-PCR

Statistical Analysis
All data were expressed as mean ± SD. With the use of InStat software (Version 3, GraphPad Software, San Diego, CA), an unpaired Student t-test was performed to compare with two groups. A probability value of p < 0.05 was considered to be significant.
Results
The average surface area of the harvested Ex-form was significantly larger (7.3 ± 0.5 cm2, n = 4) than that of Sh-form (1.2 ± 0.2 cm2, n = 4) (Fig. 1A, B). The smaller surface area of Sh-form was due to the shrinkage of the cell cluster in the culture medium. In spite of these differences, both hepatocyte sheet forms retained their morphologies regardless of the harvesting methods (Fig. 1C–H).

The morphologies of extended sheet form (Ex-form) and shrinking sheet form (Sh-form). (A) The hepatocyte sheet attached to a culture dish before harvesting as a hepatocyte sheet (Ex-form). (B) The hepatocyte sheet was peeled by gentle pipetting and floated in culture medium, resulting in Sh-form. H&E staining of sections of the Ex-form (C) and Sh-form (D) at the time of harvesting. The thickness (E) and the horizontal axis (F) of hepatocytes of the harvested Ex-form (n = 4) and Sh-form (n = 5) were measured. The open and the closed bars show the thickness and the horizontal axes of Ex- and Sh-forms, respectively. Ex-form (G) and Sh-form (H) on day 2 after reculture were observed by fluorescence confocal microscope. Scale bars: 10 mm (A, B) and 50 μm (C, D, G, H). *p < 0.001.
Histological examination of the harvested hepatocyte sheets revealed that the hepatocytes in Ex-form showed a flat and horizontally extended morphology (Fig. 1C). In marked contrast, the hepatocytes in Sh-form showed a thick and cuboidal morphology (Fig. 1D). The thickness of the hepatocytes was 7.6 ± 0.5 and 14.5 ± 0.9 μm in Ex-form (n = 4) and Sh-form (n = 5), respectively (p < 0.001) (Fig. 1C–E). The horizontal axis of hepatocytes was 52.2 ± 4.5 and 23.4 ±1.2 μm in Ex-form (n = 4) and Sh-form (n = 5), respectively (p < 0.001) (Fig. 1C, D, F). The ultrastructural examination of the harvested hepatocyte sheets by transmission electron microscope showed hepatocyte-specific intercellular structures (e.g., bile canaliculi and gap junctions), numerous mitochondria, and abundant glycogen storage granules (Fig. 2A, B).
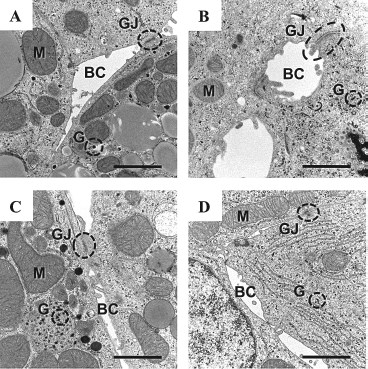
Ultrastructures of the harvested and recultured hepatocyte sheets observed by a transmission electron microscope. The liver-specific microstructures of extended sheet form (A, C) and shrinking sheet form (B, D). (A, B) At harvest. (C, D) Day 2 after reculture. BC, bile canaliculi; GJ, gap junctions; M, mitochondria; G, glycogen. Scale bar: 1 μm.
To evaluate their functions including their adherent ability, protein secretion, drug metabolization, and hepatocyte-specific mRNA expressions, the harvested sheets were transferred to Matrigel-coated 60-mm dishes for reculture. Both of the harvested hepatocyte sheet forms adhered well to the Matrigel-coated dishes while retaining their cell shape morphologies (Fig. 1G, H). The thickness of the hepatocytes of the 2-day cultured Ex- and Sh-forms were 16.7 ± 0.9 and 23.9 ± 4.2 μm, respectively (p < 0.001). Ultrastructural examination on the 2-day cultured sheet forms revealed that hepatocyte-specific microstructures including bile canaliculi and gap junctions were retained in both type of forms (Fig. 2C, D).
Hepatocyte-specific protein (albumin and hA1AT) production and metabolizing activity of the recultured hepatocyte sheets were quantified (Fig. 3). The total amount of albumin secreted from both sheet forms for 24 h showed no significant differences (4.8 ±1.0 and 4.6 ± 0.6 μg/1 × 106 of hepatocytes in Ex- and Sh-forms, respectively) (Fig. 3A). The total amount of secreted hA1AT for 24 h shows no significant differences between the two sheet forms (Fig. 3B). When the albumin and hA1AT production levels were recalculated per sheet versus per square centimeter of sheet surface demonstrated that Sh-form showed significantly higher production levels than Ex-form. (Fig. 3E, F).
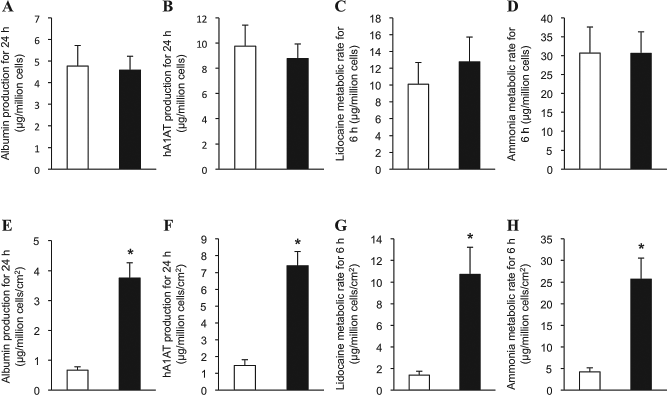
Production levels of albumin and human α1-antitrypsin (hA1AT), and the abilities of drug metabolism of lidocaine and ammonia of the recultured hepatocyte sheets. The amount of albumin (A, E) and hA1AT (B, F) secreted into the culture medium from the recultured hepatocyte sheets for 24 h were measured by ELISA (n = 5 for both groups). The amount of lidocaine (C, G) (n = 7 for both groups) and ammonia (D, H) (n = 4 for both groups) metabolized by the hepatocyte sheets for 6 h after 42 h reculture. The upper row (A–D) shows the protein production levels and the drug metabolic rate per hepatocyte sheet. The lower row (E–H) shows the protein production levels and the drug metabolic rate per square centimeter of the hepatocyte sheet. The open and the closed bars show extended sheet form and shrinking sheet form, respectively. *p < 0.001.
For the assessment of the drug metabolic ability of the recultured hepatocyte sheets, the metabolized amounts of lidocaine and ammonia in the culture medium for 6 h were quantified. The metabolic rate of lidocaine of both sheet forms showed no significant differences (10.1 ± 2.6 and 12.8 ± 3.0 μg/sheet forms for Ex- and Sh-forms, respectively) (Fig. 3C). The metabolic rate of ammonia of both sheet forms showed no significant differences (Fig. 3D). Recalculation of these metabolized amounts per sheet versus per square centimeter of sheet surfaces demonstrated that the function of Sh-form was significantly higher than that of Ex-form by 7.7 times and 6.1 times for lidocaine and ammonia metabolism, respectively (Fig. 3G, H).
Next, the hepatocyte-specific gene expression levels of the two sheet forms were analyzed by real-time PCR. To obtain reliable gene expression data, the expression levels of nine commonly analyzed HKGs were assessed in advance, and the most statistically stable HKGs for this study were determined by a geNorm and NormFinder softwares (1,39). As a result, PPIA and HPRT1 were identified to be the most suitable reference gene set (data not shown). The normalization of the mRNA expression levels of albumin (ALB), hA1AT, ATP-binding cassette, subfamily B (MDR/TAP), member 11 (ABCB11), cytochrome P450, family 3, subfamily a, polypeptide 13 (CYP3A13), hepatic nuclear factor 4α (HNF-4α), coagulation factor VIII (F8), and coagulation factor IX (F9) through the geometric mean of PPIA and HPRT1 gave no significant differences between the two sheet forms in any gene analyzed (Fig. 4).
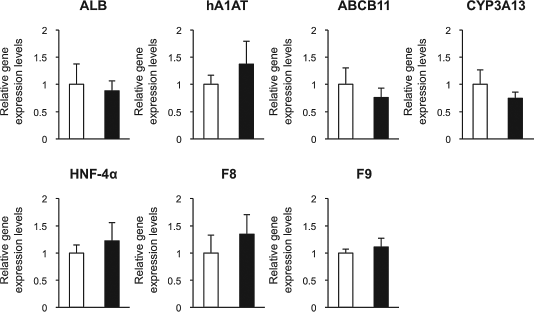
mRNA expression levels of the target genes of the recultured extended sheet form and shrinking sheet form determined 48 h after the reculture. The mRNA expression levels of albumin (ALB), human α1 antitrypsin (hA1AT), adenosine triphosphate-binding (ATP) cassette subfamily B, member 11 (ABCB11), cytochrome P450 family 3, subfamily a, polypeptide 13 (CYP3A13), hepatic nuclear factor-4α (HNF-4α), coagulation factor VIII (F8), and coagulation factor IX (F9) were determined by real-time PCR. The data were expressed as relative ratios to the extended sheets. The open and closed bars show extended sheet form and shrinking sheet form, respectively. n = 5 for both groups.
Discussion
Based on the cell sheet engineering technologies, the present study established hepatocyte sheet harvesting procedures that provide us two different hepatocyte sheet forms: extended sheet form (Ex-form) and shrinking sheet form (Sh-form). Total surface area of Sh-form was significantly smaller than that of Ex-form. We showed that both forms were able to favorably attach to Matrigel-coated culture surfaces in the reculture experiments. Functionalities of the harvested hepatocyte sheets were assessed by the reculture experiments, and there was practically no difference between Ex- and Sh-forms in terms of proteins expressions, chemical metabolizing activities, and mRNA expression levels of hepatocyte-specific genes. Since the protein production levels and the chemical metabolizing activities were similar levels per sheet basis for both forms, the recalculation of these values per unit area of sheet surface demonstrated that the function of Sh-form was significantly higher than that of Ex-form.
Although the original morphology of hepatocytes in vivo is cuboidal shape, primary cultured hepatocytes show a gradual change in cell shape, and become flat and elongate as time progresses (11,27,31). Since the present primary hepatocyte culture system was used PIPAAm dishes, reducing temperature from 37°C to 20°C for a short period of time gave a complete detachment of the hepatocytes from the culture surfaces and allowed the cells to float in the culture medium and become a uniform sheet. This floating process associated with a shrunk of sheet resulting in a drastically smaller size (Sh-form). During this shrink process, the hepatocyte shape changed from flatly extended form to cuboidal form. In contrast, the coverage of support membrane over the hepatocyte cluster prior to reducing culture temperature to 20°C allowed us to harvest a hepatocyte sheet (Ex-form) while retaining the same cell morphology (flat and elongation shape). It is of note that hepatocyte-specific microstructures (e.g., bile canaliculi and gap junctions), numerous mitochondria, and abundant glycogen storage granules were equally observed in both forms. These findings indicated that hepatocyte sheet shrinkage during the sheet floating process gave no significant internal structural alterations of the hepatocyte in the sheet in spite of allowing the cell shape to become cuboidal form.
It was reported that hepatocytes cultured on a basement membrane matrix, Engelbreth-Holm-Swarm gel (available under the commercial name Matrigel), showed cuboidal shape and maintained the expression levels of liver-specific genes such as HNF-4α and albumin (10, 19,23). HNF-4α is a liver-enriched transcription factor that regulates numerous liver-expressed genes including albumin and several cytochrome P450 (CYP) genes (20,21). The present findings, which were similar hepatocyte-specific protein production levels and chemical metabolizing activities observed in Ex- and Sh-forms, could be the results of similar HNF-4α expression levels in both forms, which are regulated by interactions between the hepatocytes and Matrigel. In contrast, it has been reported that hepatocytes cultured on type I collagen showed less HNF-4α induction levels, which closely associated with impaired expression levels of several liver-specific genes compared with those of cells cultured on Matrigel (10). In this regard, further functional analyses of Ex- and Sh-forms of hepatocytes recultured on type I collagen or other extracellular matrices (ECMs) await future investigation (4).
Cellular functions are regulated by ECM geometry as well as the component of ECM. Matrigel sandwich configuration, which was made by overlaying ECM, increased the number of clear cuboidal-shaped cells and induced an increase in the expression of liver-specific function compared to cultures on a single ECM-coated surface (2). Singhvi et al. (35) reported that hepatocyte functions were regulated by the degree of contact between cell and culture surfaces. Sandwiching Ex- and Sh-forms between Matrigels and altering the contact surface between cell and culture surface such as patterned surface with varying degrees of surface charge and resisting protein adsorption might provide new insights to various associations among cell shape, function, and culture surfaces.
Hepatocyte-based therapeutic approaches, either based on cell transplantation or tissue engineering, have clinically or experimentally highlighted their ability to provide supportive hepatic functions to the individual (7,8, 17,24,27,30,36,41). In the clinic, hepatocyte transplantation has been applied to nearly hundred of patients suffering from various types of liver diseases (7,27). Recently, Meyburg et al. (16) have carried out hepatocyte transplantation to four children (ages 1 day to 3 years) with severe neonatal urea cycle disorders. Clinical and metabolic stabilization was noted in all four patients. The fact that the four children were treated with hepatocytes isolated from a single donor liver demonstrates the potential of hepatocyte-based therapies to overcome donor shortage in organ transplantation (16). Two children (ages 3 months and 2 years) with congenital coagulation factor VII deficiency had also successfully achieved phenotypic corrections by hepatocyte transplantation (6). These clinical successes in hepatocyte transplantation prompted investigators to bioengineer functional liver system using isolated hepatocytes. To maximize the functional engraftment rate of the hepatocytes at ectopic sites for the creation of a functional liver system, our group has established several innovative experimental approaches (9,12,24–31,41). One of the key technologies in our series of developments is the creation of a uniform hepatocyte sheet using the PIPAAm culture dishes (31,40). Since transplantation of hepatocyte sheet can be performed under minimally invasive procedures (e.g., local anesthesia and small skin incision), this type of approach could reasonably be adopted to newborn and baby patients. It is important to establish in the present study that a hepatocyte sheet could be shrunk in size (less than 1/6 of original size) without losing functionalities. Shrunk hepatocyte sheets would be valuable for liver tissue engineering in areas that are limited in transplantable space. In order to make the cell sheet-based tissue engineering approach to be less minimally invasive, a laparoscopic device specific for delivering a harvested sheet has already been developed by our group (15).
In conclusion, this study demonstrates that the change of the shape of cultured hepatocytes from flatly extended form to cuboidal form could be clinically feasible. Functional laminar assembled hepatocytes (Sh-form) composing of hepatocytes with cuboidal form can be harvested. Sh-form is small in size but expresses the same functionality as nonshrinking Ex-form based on in vitro experiments. We believe that the functional small-sized hepatocyte sheet could serve as a valuable medical device for establishing a new generation therapies toward liver diseases.
Footnotes
Acknowledgments
The authors thank Ms. Kazuko Kawahara and Ms. Chinatsu Kohno for technical assistance in tissue sampling and histological assessment, and Dr. Norio Ueno for critical reading of the manuscript. The authors declare no conflict of interest. This study was supported in part by the Special Coordination Funds for Promoting Science and Technology from the Ministry of Education, Culture, Sports, Science and Technology of Japan (K.O. and T.O.) and Global Center of Excellence Program (K.O), Novartis Foundation Japan (K.O.), and the Bayer Hemophilia Award Program (K.O.).
