Abstract
Cell-based therapies for liver disease rely on a high-quality supply of hepatocytes and a means for storage during transportation from site of isolation to site of usage. Unfortunately, frozen cryopreservation is associated with unacceptable loss of hepatocyte viability after thawing. The purpose of this study was to optimize conditions for cold storage of rat hepatocyte spheroids without freezing. Rat hepatocytes were isolated by a two-step perfusion method; hepatocyte spheroids were formed during 48 h of rocked culture in serum-free medium (SFM). Spheroids were then maintained in rocked culture at 37°C (control condition) or cold stored at 4°C for 24 or 48 h in six different cold storage solutions: SFM alone; SFM + 1 mM deferoxamine (Def); SFM + 1 μM cyclosporin A (CsA); SFM + 1 mM Def + 1 μM CsA, University of Wisconsin (UW) solution alone, UW + 1 mM Def. Performance metrics after cold storage included viability, gene expression, albumin production, and functional activity of cytochrome P450 enzymes and urea cycle proteins. We observed that cold-induced injury was reduced significantly by the addition of the iron chelator (Def) to both SFM and UW solution. Performance metrics (ammonia detoxification, albumin production) of rat hepatocyte spheroids stored in SFM + Def for 24 h were significantly increased from SFM alone and approached those in control conditions, while performance metrics after cold storage in SFM alone or cold storage for 48 h were both significantly reduced. A serum-free medium supplemented with Def allowed hepatocyte spheroids to tolerate 24 h of cold storage with less than 10% loss in viability and functionality. Further research is warranted to optimize a solution for extended cold storage of hepatocyte spheroids.
Introduction
Hepatocytes of high viability and high functionality are required in the fields of in vitro drug toxicity, metabolism testing, and development of cell-based therapies for liver disease (19,25). Primary hepatocytes form tissue-like multicellular aggregates of spherical geometry (spheroids) under rocked suspension culture conditions (26). Hepatocyte spheroids serve as an efficient three-dimensional liver tissue construct (17). High viability and the ability to maintain high levels of liver-specific functions (albumin production, urea synthesis, and cytochrome P450 activity) are important features of hepatocyte spheroids (6,20). These features allow hepatocyte spheroids to serve as an alternative source of liver tissue for ex vivo research and therapeutic applications such as the bioartificial liver (BAL) (1,18).
Ex vivo applications often require transfer of hepatocytes, including hepatocyte spheroids, to a remote site for use. The hepatocytes must remain viable during the transport process and retain their differentiated functions when called on for use at the remote site. Cryopreservation of hepatocytes by freezing has been utilized for clinical application of first-generation BAL devices, including a randomized multicentered clinical trial of porcine hepatocytes to treat acute liver failure reported by Demetriou et al. (9). Freezing allowed batching of pig hepatocytes in the Demetriou device, which was convenient and reduced the cost of safety testing. However, freezing resulted in a loss of viability and functionality upon rewarming to 37°C. Reduced functionality of hepatocytes after thawing adversely impacted efficacy of the Demetrious liver support device and likely contributed to its lack of FDA approval.
As a result, alternatives to freezing have been considered such as short-term cold storage at chilled temperatures above freezing. Short-term cold storage in solutions developed to minimize ischemia–reperfusion injury is the preferred option for preservation and transportation of transplantable livers (4). Several preservation solutions have been developed and marketed (8,11,13,22). The most successful and well known of these products is University of Wisconsin (UW) solution. Naturally, these commercial solutions, as well as a handful of customized solutions, have been considered for cold storage of isolated hepatocytes, both in suspension (14,28,29) and in collagen gel culture (16,21). In contrast, the cold storage of hepatocyte spheroids is poorly studied (16).
Cold-induced injury of liver tissue and isolated hepatocytes is associated with an increase in the intracellular chelatable iron pool followed by formation of reactive oxygen species (ROS) (31). A second, iron-independent but chloride-dependent, pathway has also been observed after cold storage of rat hepatocytes (32). We hypothesize that the cold storage of hepatocyte spheroids could be improved by supplementation of the preservation solution with an iron chelator (deferoxamine; Def) and an inhibitor of the mitochondria permeability transition (cyclosporin A; CsA) to inhibit the iron-dependent and chlorine-dependent pathways, respectively (14,27,35).
In this study, we examined the effect of cold storage treatment on the viability and liver-specific functionality of rat hepatocyte spheroids. The studies were designed to evaluate the beneficial effects of Def and CsA in two preservation solutions: UW solution (gold standard) and a custom serum-free medium (SFM) recently developed for culture of hepatocyte spheroids (3). Along with optimizing conditions for cold storage of hepatocyte spheroids, a second purpose of this study was to determine the duration of safe cold storage in terms of integrity and functionality of spheroids under these conditions.
Materials and Methods
Materials
Chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA) unless stated otherwise. All animal procedures were performed under the guidelines set forth by, and with the approval of, the Mayo Foundation Animal Care and Use Committee in accordance with those set forth by the National Institutes of Health.
General Description
Our set of experiments consisted of measuring performance metrics of rat hepatocyte spheroids during control conditions of continuous rocked culture at 37°C versus continuous rocked culture at 37°C after 24 and 48 h of cold storage (4°C), as outlined in Figure 1. Six cold storage media were tested (UW alone, UW + 1 mM Def, SFM alone; SFM + 1 mM Def, SFM + 1 mM Def + 1 μM CsA, SFM + 1 μM CsA). Culture conditions were standardized as follows: T75 flasks (BD Falcon, Bedford, MA, USA) containing 20 ml of SFM media incubated in 5% CO2 environment at 37°C. An oscillating rocker plate (Bellco Technology, Vineland, NJ, USA) was utilized for spheroid formation and continuous culture. All measurements of functionality were based on 24 h of continuous culture in fresh medium. Six T75 flask cultures were included in each group. All cells within an experiment were obtained from the same harvest. Harvests were repeated in triplicate to confirm reproducibility of the data.

Experimental design. Freshly isolated rat hepatocytes were suspended in serum-free media (SFM) and rocked continuously at a frequency of 10 cycles per minute to induce spheroid formation. After 48 h of rocking, the spheroids were washed with SFM and then divided into control and cold storage (CS) conditions. Control conditions consisted of spheroids in rocked culture at 37°C without cold storage. Fresh medium was changed every 24 h. Four cold storage media were tested (SFM alone; SFM + Def; SFM + Def + CsA; and SFM + CsA). After cold storage (24 or 48 h), the spheroids were washed and placed in 20 ml SFM for rocked cell culture in a 5% CO2, 37°C incubator. Final hepatocyte concentrations of 5 × 105 cells/ml were used for all measurements. Bold arrows (→) correspond to culture intervals used for sample collection during day 4 and day 5 after hepatocyte isolation. Def, deferoxamine; CsA, cyclosporin A.
Spheroid Cultures and Conditions
Hepatocytes were isolated from male Sprague–Dawley rats (300–400 g; Harlan, Indianapolis, IN, USA) by a two-step perfusion method as previously described (33). All harvests yielded hepatocytes with viability exceeding 95% by trypan blue dye exclusion.
Freshly isolated hepatocytes were suspended in SFM, composed of William's E supplemented with 0.2 U/ml insulin, 6 μg/ml transferrin, 100 U/ml penicillin G, 100 mg/ ml streptomycin, 3 g/L human albumin, 2.2 g/L sodium bicarbonate, 1 g/L l-carnitine, 2.0 mM l-glutamine, 100 nM dexamethasone, 40 ng/ml glucagon, 20 ng/ml Gly-His-Lys, 1,000 U/L heparin, 1 mg/L warfarin, and 5 ng/ ml of mouse epidermal growth factor (EGF) (3).
The cells, suspended in SFM, were placed in a spheroid box (33 × 28 × 6 cm) custom-made of polycarbonate by Mayo Division of Engineering and siliconized with Sigmacote for 30 min (20) and gently rocked continuously at a frequency of 10 cycles per minute (0.17 Hz) to induce spheroid formation and to maintain spheroids in suspension. Final hepatocyte concentration was 5 × 105 cells/ml per spheroid box. All culture conditions were maintained in a 5% CO2, 37°C incubator as previously described (20). Spheroids were centrifuged and resuspended in fresh culture media every 24 h.
Cold Storage and Rewarming
After 48 h of continuous rocking, the spheroids were washed with SFM at room temperature. The culture medium was changed to the cold storage medium (UW alone, UW + 1 mM Def; SFM alone; SFM + 1 mM Def; SFM + 1 mM Def + 1 μM CsA; SFM + 1 μM CsA) at room temperature. Spheroids were left in rocked culture without cold storage as control. Spheroids intended for cold storage were rocked in glass dishes (10 × 8 × 2 cm) custom-made by Mayo Division of Engineering and siliconized with Sigmacote for 30 min and then transferred into 50-ml conical tubes (BD Falcon; 1 × 106cells/ml, 20 ml in total) and put on ice for 1 h. Of note, spheroid box and glass dishes differ in size (i.e., volume capacity) but possess similar properties of spheroid formation and culture. Finally, tubes of spheroids were placed in a refrigerator to be cold stored at 4°C. After 24 or 48 h of cold storage treatment, the spheroids were centrifuged at 50 × g for 5 min, and the supernatant fluid was removed. Warm SFM (20 ml) at 37°C was added to each tube. The tubes were mixed gently prior to adding 20 ml of cell suspension to each glass culture dish and continued to rocking culture. Cultures were maintained in a humidified incubator at 37°C with a 5% CO2 atmosphere.
Viability of Rat Hepatocytes
Viability of rat hepatocyte spheroids was evaluated by inverted epifluorescence microscopy (Axioscope, Carl Zeiss, Inc., Thornwood, NY, USA) using the Fluoroquench™ fluorescence viability stain (One Lambda, Canoga Park, CA, USA). Green fluorescein isothiocyanate images were captured using a 450-nm excitation filter and merged with in-plane acridine orange images captured using a 546-nm emission filter.
Quantification of Albumin, Ureagenesis, Diazepam Metabolism
On days 3, 4, and 5 of culture, SFM was supplemented with 20 μM diazepam and 2.5% v/v deuterium-enriched heavy ammonia gas (15ND3) (1.12 mM; Cambridge Isotope Laboratories, Inc., Andover, MA, USA). Supernatant medium was collected and stored at −20°C prior to analysis. The concentration of rat albumin in medium was determined by enzyme-linked immunosorbent assay (ELISA) kit (Bethyl, Montgomery, TX, USA; #E80-129). The concentration of total urea was measured using a QuantiChrom™ DIUR-500 Urea Assay Kit (BioAssay System, Hayward, CA, USA) according to the manufacturer's instructions. Production of urea through the complete urea cycle was determined by the conversion of heavy ammonia (15ND3) to native and deuterium-enriched urea. Isotopes of urea were quantified by capillary gas chromatography/mass spectrometry. Concentrations of diazepam and its two major metabolites (nordiazepam and temazepam) were determined by high-performance liquid chromatography (HPLC) with mass spectrometry detection in the CTSA Metabolomics Core lab at Mayo Clinic. Samples were deconjugated in β-glucuronidase/ arylsulfatase (600 Fishman units and 4800 Roy units/ml, respectively; Roche, Indianapolis, IN, USA) and sodium acetate buffer (0.1 M) for 2 h. Reactions were quenched with ethanol. Data acquisition was completed by Chrom Perfect® software (Denville, NJ, USA). All assays were performed in triplicate.
Transmission Electron Microscopy (TEM)
Spheroids were examined by TEM on days 4 and 5 after primary hepatocyte isolation. Briefly, samples of spheroids were preserved in Trump's fixative (1% glutaraldehyde and 4% formaldehyde in 0.1 mol/L phosphate buffer, pH 7.2) and sectioned (90 nm) by the Electron Microscopy Core Facility (Mayo Clinic, Rochester, MN, USA). Sections were placed on 200-nm mesh copper grids and stained with lead citrate. Micrographs were taken with a JEOL 1200 EXII electron microscope (Peabody, MA, USA) operating at 60 kV.
Quantitative Reverse Transcription Polymerase Chain Reaction (QRT-PCR)
Total RNA was extracted from cultured spheroids at each time point using an RNeasy Plus MiniKit (Qiagen, Valencia, CA, USA) according to the manufacturer's instructions. All PCR were performed in 96-well optical reaction plates (Applied BioSystems, Foster City, CA, USA) in 20 μl reaction volume using SYBR Green PCR Master Mix (Applied Biosystems, No. 4389986). Reactions were carried out on the ABI PRISM® 7900HT Sequence Detection System (SDS) (Applied Biosystems), and data were analyzed with the SDS Version 2.4 software. Triplicate amplifications were carried out for each target gene. Quantification was performed using the comparative Ct method normalized with the expression levels of the endogenous reference gene hypoxanthine guanine phosphoribosyl transferase. Expression of each target gene was normalized to its level in normal culture spheroids on day 4. Primer sequences are listed in Table 1 (2,20).
Primer Sequences for RT-PCR

Statistical Methods
Results are expressed as mean values ± SEM. Statistical analysis was done by one-way ANOVA–Dunnett' s test; a value of p ≤ 0.05 was considered significant.
Results
Cold Storage of Hepatocyte Spheroids in UW Versus SFM
We were first interested in the influence of preservation solution (UW vs. SFM) on viability of rat hepatocyte spheroids and their albumin production as a measure of functionality. Cell viability and albumin concentration were measured after cold storage of 24 h/day 4 and 48 h/ day 5 in UW alone, SFM alone, UW + Def, and SFM + Def. Results of viability and albumin studies, summarized in Table 2, indicated a similar effect of cold preservation in SFM and UW solution. An equivalent rise in cell viability and albumin concentration was observed after Def supplementation of SFM and UW solution. These results were reproduced in studies on two additional rat harvests. Therefore, SFM was used as the base preservation solution in subsequent cold storage experiments.
Cold Storage in UW Solution Versus SFM

Results are mean ± SEM of 10 measurements from a single liver harvest; UW, University of Wisconsin solution; SFM, serum-free medium; Def, deferoxamine.
Viability and Ultrastructure of Hepatocyte Spheroids After Cold Storage
We were next interested in the influence of the duration of cold storage on the viability and integrity of rat hepatocyte spheroids. Four SFM-based storage media were compared to the control condition (Fig. 2). Representative fluorescence images from all five conditions on both day 4 and day 5 cultures are summarized in Figure 2A. Fluoroquench™ data showed that hepatocyte spheroids tolerated cold storage for 24 h, but decreased cell viability and increased necrosis within spheroids was observed after 48 h of cold storage. In Figure 3, TEM images of spheroids obtained from the control group and SFM + Def + CsA group showed good hepatocyte health as demonstrated by their abundant, smooth-appearing, few edematous mitochondria, and well-demarcated nuclei. However, TEM images of spheroids stored in SFM show signs of necrosis and impending cell death, especially after 48 h of cold storage. Mitochondria in these images were markedly edematous or ballooned. In addition, hepatocytes maintained in these conditions showed many lipofuscin bodies and lipolysosome, which were commonly seen in hepatocytes that have died by necrotic cell death. These findings contrast TEM images obtained from spheroids stored in SFM + Def + CsA on day 5, which continued to show mitochondria, a few lipolysosomes, and distinct nuclei.
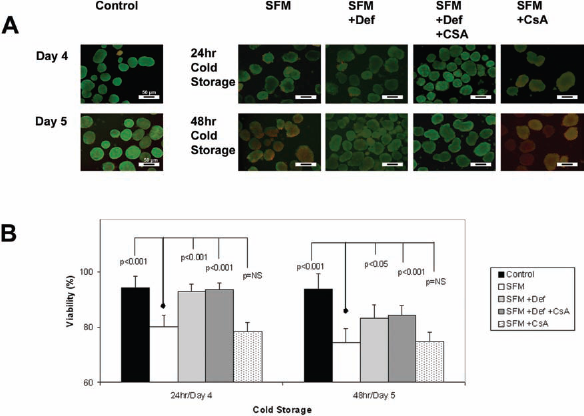
Vital staining and percent viability of hepatocyte spheroids. (A) Representative Fluoroquench™ stained images of spheroids from each of the five culture conditions obtained on day 4 and day 5 after isolation: Control (no cold storage, left column), SFM, SFM + Def, SFM + Def + CsA, and SFM + CsA (far right column). Fluorescence images were obtained using an inverted 40× lens. Fluoroquench™-stained hepatocytes were viable (green) or necrotic (red/orange). These images were used to determine the cell viability for each cold condition after 24 or 48 h. Scale bars: 50 μm. (B) The mean viability of hepatocyte spheroids was calculated from red and green fluorescence and the average of >10 spheroids per measurement. p Values were determined from comparison to the SFM condition on the corresponding day. Viability = (mean green fluorescence/mean red fluorescence + mean green fluorescence) × 100. Note that the y-axis starts at 60%.

Transmission electron microscopy (TEM) of hepatocyte spheroids. Representative low-power TEM and high-power TEM images of spheroids from three culture conditions obtained on day 4 and day 5 after isolation: Control (no cold storage, left column), SFM and SFM + Def + CsA (far right column). Low-power TEM images were captured at 2,500×; high-power images were at 6,000×. TEM images show the cell viability for each cold condition after 24 or 48 h. Scale bars: 20 and 5 μm, respectively.
These observations are shown graphically in Figure 2B, which summarizes the viability measured using Fluoroquench™ staining. Viability was greatest in three groups on day 4: control (94.26%), SFM + Def (92.92%), and SFM + Def + CsA (93.71%), the latter two after 24 h of cold storage. These three values were significantly higher than spheroids maintained under SFM and SFM + CsA (p < 0.001). The viability on day 5 was also similar in the control and cold storage conditions of SFM + Def and SFM + Def + CsA.
Albumin Production and Gene Expression by Rat Hepatocyte Spheroids
As illustrated in Figure 4A, albumin production was greatest on day 4 and similar in the control and cold storage conditions of SFM + Def and SFM + Def + CsA. In contrast, SFM + CsA and SFM alone conditions were similarly low on both day 4 and day 5. Albumin gene expression, shown in Figure 4B, confirms the pattern of albumin production of Figure 4A.

Albumin production and gene expression. (A) The concentration (mg/dl) of rat albumin was determined by enzyme-linked immunosorbent assay (ELISA). Spheroids cold stored under SFM + Def and SFM + Def + CsA conditions for 24 or 48 h produced significantly higher rates of albumin (p < 0.01) compared to spheroids maintained under SFM or SFM + CsA conditions. Error bars are the standard deviation of the average of three calculated rates for each condition. (B) Expression of albumin gene was examined for each of the groups by qRT-PCR during the day 4 and day 5. Control group day 4 values served as calibrators to determine the relative expression of gene at each time point and group.
Ammonia Clearance and Urea Cycle Activity
Ammonia Clearance
We were next interested in the influence of cold storage treatment and spheroid culture on the ammonia detoxification activity of rat hepatocyte spheroids. Figure 5A demonstrates that the ammonia clearance of SFM + Def and SFM + Def + CsA group was 71.9%, 77.4% on day 4, and 54%, 55.2% on day 5, while ammonia clearance was similar to the control group (78.6% on day 4, 62.7% on day 5) after cold storage of 24 and 48 h. All three measurements were significantly higher than spheroids cold stored in SFM alone (p < 0.001).

Ammonia clearance and urea production. (A) Detoxification of ammonia was reported as the percentage of ammonia clearance from baseline levels. A significant difference was observed between the SFM group versus control, SFM + Def, SFM + Def + CsA, and SFM + CsA after both 24 and 48 h of cold storage, p < 0.05. (B) Average urea production for each condition was measured on day 4 and day 5 using the commercially available QuantiChrom™ DIUR-500 urea assay kit. Heavy ammonia (2.5% v/v) was added to culture media, and levels of deuterium-enriched urea were determined by gas chromatography/mass spectrometry on samples collected at 24 h. Percentages indicate the proportion of urea labeled with deuterium (sum of all isotopes) compared to total urea in each sample. The rate of heavy and total urea production by control group, SFM + Def group and SFM + Def + CsA group were significantly greater than the other two groups, p < 0.05.
Formation of Heavy Urea
Production of urea and the percentage produced as heavy urea in medium supplemented with heavy ammonia are summarized in Figure 5B. All four groups demonstrated intact urea cycle activity by the conversion of deuterium-enriched (heavy) ammonia to isotopes of heavy urea. The percentage of total urea produced as heavy urea by spheroid hepatocytes ranged from 45% to 64% in control, SFM + Def and SFM + Def + CsA groups. These values were greater than spheroids stored in SFM and SFM + CsA, which ranged from 12% to 31% (p < 0.05). Conversely, spheroids stored in SFM and SFM + CsA had higher production of nonheavy urea. Appearance of nonheavy urea in the medium likely represented a single enzymatic step as arginase leaked from injured hepatocytes and converted arginine to ornithine + urea in the media.
Expression of Urea Cycle Genes
Expression of the six urea cycle genes [arginase 1 (Arg1), arginosuccinate lyase (Asl), arginosuccinate synthetase 1 (Ass1), carbamoyl-phosphate synthetase 1, mitochondrial (Cps1), N-acetylglutamate synthase (Nags), and ornithine transcarbamylase (Otc)] on day 4 and day 5 are summarized in Figure 6. Expression values are reported as fold change relative to the level measured in control cultures on day 4. Urea cycle gene expression levels decreased under all cold storage conditions from day 4 to day 5. SFM + Def and SFM + Def + CsA groups had higher expression levels of most (five of six) urea cycle genes (Arg1, Ass1, Cps1, Nags, and Otc) compared to spheroids cold stored in SFM and SFM + CsA (p < 0.05). Expression of Asl in the SFM group was similar to others on day 4 but decreased rapidly after 48 h of cold storage on day 5.

Expression of liver urea cycle genes. Expressions of the six genes of the urea cycle [arginase 1 (Arg1), arginosuccinate lyase (Asl), arginosuccinate synthetase 1 (Ass1), carbamoyl-phosphate synthetase 1, mitochondrial (Cps1), N-acetylglutamate synthase (Nags), and ornithine transcarbamylase (Otc)] were examined by qRT-PCR for each of the four groups on day 4 and day 5. Data are reported as relative gene expression of each group and at each time point compared to the control group-day 4.
Functionality and Expression of Cytochrome P450 Genes
Diazepam Metabolism: Influence of Cold Storage
The rate of diazepam metabolism to its primary metabolites (nordiazepam, temazepam) was studied in cultures of rat hepatocyte spheroids to assess cytochrome P450 (Cyp) activity. As illustrated in Figure 7A, nordiazepam formation was obviously higher in control group compared to the cold storage groups on day 4 and day 5 (p < 0.01), indicating decreased Cyp family 2, subfamily C (Cyp2c) activity after cold storage of 24 or 48 h. The levels of nordiazepam formation in the SFM + Def group and the SFM + Def + CsA group were higher than corresponding levels in the SFM group and SFM + CsA group (p < 0.01). Similar to nordiazepam, formation of temazepam was highest in the control group (p < 0.01), indicating a loss of Cyp3a activity after cold storage (Fig. 7B). Temazepam levels on day 4 were higher in the SFM + Def group and SFM + Def + CsA group compared to the SFM group and the SFM + CsA group (p < 0.01).

Clearance of diazepam and formation of its two major metabolites. (A) Nordiazepam formation of hepatocyte spheroids was highest in the control group (p < 0.05), indicating loss of cytochrome P450 family 2 subfamily C (Cyp2c) activity after 24 and 48 h of cold storage. (B) Temazepam formation of hepatocyte spheroids was highest in the control group (p < 0.01), indicating a loss of Cyp3a activity after 24 and 48 h of cold storage, but temazepam levels were higher in the SFM + Def group and SFM + Def + CsA group of SFM compared to the other two groups on day 4 (p < 0.01). (C) Diazepam elimination was calculated in all day 4 and day 5 cultures. The clearance of diazepam was highest in control group. As expected, a significant difference was observed between the control group, SFM + Def group, and SFM + Def + CsA group compared to the SFM alone group and SFM + CsA group at each time point, p < 0.01. No significant difference was observed between the SFM group and SFM + CsA group. Data are represented as mean ± standard deviation. Note that the y-axis starts at 20%.
As illustrated in Figure 7C, diazepam elimination rate in the control group was 69% on day 4, which was slightly higher than the SFM + Def group (64.67%) and SFM + Def + CsA group (67.67%). On day 5, the rate of diazepam elimination in control group (77.33%) was higher than the other four groups (p < 0.05). However, the rates of diazepam elimination in the SFM + Def group and SFM + Def + CsA group were higher than the SFM group and SFM + CsA group at both day 4 and day 5 (p < 0.01).
Expression of Metabolism Genes After Cold Storage
To further evaluate the functionality of rat hepatocyte spheroids maintained in four cold solutions of 24- and 48-h duration, we examined the expression of six liver-related genes [Cyp family1, subfamily A, polypeptide 1 (Cyp1a1), Cyp1a2, Cyp3a1, Cyp3a2, Cyp2c6, and hepatocyte nuclear factor 6 (HNF6)]. Expression levels of these genes are summarized in Figure 8. These genes include Cyp2c6 involved in nordiazepam production and Cyp3a1 and Cyp3a2 involved in temazepam production from diazepam, respectively. Five of six liver special gene (Cyp1a2, Cyp3a1, Cyp3a2, Cyp2c6, and HNF6) expression levels decreased under all cold storage conditions compared to control levels. The expression of Cyp1a1 increased slightly in the SFM + Def group and SFM + Def + CsA group after cold storage of 24 h on day 4, but decreased after 48 h of cold storage (day 5). All levels of gene expression were higher in both groups containing Def (SFM + Def and SFM + Def + CsA) compared to the other two groups, which did not contain Def.

Expression of liver-specific genes. Expression of the six liver-specific genes [cytochrome P450 family 1, subfamily A, polypeptide 1 (Cyp1a1), Cyp1a2, Cyp3a1, Cyp3a2, Cyp2c6, and hepatocyte nuclear factor 6 (HNF6)] was examined by qRT-PCR on day 4 and day 5 for each of the four cold storage groups and control group. Expression levels were normalized to the control group day 4.
Discussion
Severe liver failure is common and carries a high risk of mortality in patients with either acute liver failure or acute-on-chronic liver failure (12). Extracorporeal support with a cell-based therapy (i.e., bioartificial liver) can be employed as a strategy for bridging these severely ill patients to transplantation or to recovery (24). Providing access to a high-quality supply of hepatocytes or hepatocyte spheroids for cell-based therapy is a major objective of several research programs, including our own (37). Success of a cell-based therapy program, such as our spheroid reservoir bioartificial liver program (5), requires the transport of hepatocyte spheroids from the site of production to the patient's bedside, which is often a remote site. A major challenge of implementing a cell therapy program involving hepatocyte spheroids is to devise a method of prolonging shelf life of the spheroids with minimal loss in viability and functionality prior to use.
A popular method of storing cells ex vivo is control-rate freezing followed by storage at very low temperatures (36). While this method has worked successfully for small, nonanchorage-dependent cell types, such as blood cells and transformed cell lines, freezing is more damaging to large, mitochondria-rich, anchorage-dependent cell types such as hepatocytes. In a recent report, frozen cryopreservation of human hepatocytes was associated with 52% viability and 48% attachment after thawing (34). In contrast, our current study indicates that cold storage of rat hepatocyte spheroids for 24 h at 4°C was associated with greater than 90% viability, less than 10% loss in functionality (ammonia detoxification, albumin production), and avoided the inconvenience of controlled-rate freezing. It is important to point out that differences exist between hepatocytes from rats and humans. Therefore, our results may differ if applied to human hepatocytes.
The current study also addressed the preferred composition of a preservation solution for cold storage of hepatocyte spheroids. For over 20 years, the leading solution for cold storage of transplantable livers has been UW solution (8). Similarly, UW solution is a commonly tested solution for cold storage of hepatocytes (14,32). In our initial set of studies, we compared cold storage of hepatocyte spheroids in UW solution versus a SFM that we developed for culture of hepatocyte spheroids (3). Our SFM is based on William's E medium supplemented with an antioxidant (carnitine) and a physiological concentration of human albumin. The iron chelator Def was evaluated since it has been shown to inhibit the occurrence of apoptotic morphology in hepatocyte culture (14,32). We also evaluated CsA because of its properties of stabilizing mitochondrial permeability transition after ischemia–reperfusion injury (30). In our initial studies, we observed similar viability and albumin production by hepatocyte spheroids stored in SFM versus UW solution and in SFM + Def versus UW + Def. Therefore, our cheaper SFM solution was used in the remainder of experiments. We acknowledge that our SFM may not be equivalent to UW solution for cold preservation of hepatocyte spheroids since UW solution was not evaluated in all studies. We can conclude that the beneficial effects of Def were significant compared to those of CsA alone when evaluated in the SFM.
The liver is a major site of detoxification, and ammonia is arguably the most important endogenous toxin requiring clearance by the liver (10). A functioning urea cycle must be present for ammonia to be efficiently detoxified and eliminated from the body. Therefore, ammonia removal and urea production are important considerations for cold storage of spheroid for use in a BAL. Our data indicate similar rates of both ammonia detoxification and ureagenesis by hepatocyte spheroids under control conditions and after cold storage for 24 h in SFM supplemented with Def (Fig. 5).
Expression analysis of genes involved in hepatocyte metabolism, including the urea cycle genes, were also best maintained after 24 h of cold storage in SFM supplemented with Def (Fig. 6).
The Cyp enzymes are also important to detoxification of endogenous and exogenous wastes (7,15). Our cold storage spheroids expressed higher levels of Cyp1a1 mRNA after 24 h of cold storage in SFM with Def compared to control. Furthermore, mRNA expression of Cyp1a2, Cyp3a1, Cyp3a2, and Cyp2c6 in cold-stored spheroids was greater in storage medium supplemented with Def (Fig. 8).
In conclusion, UW solution and SFM supplemented with Def provided a beneficial effect in reducing cellular damage and maintaining functionality of hepatocyte spheroids during cold storage. For the most part, hepatocyte spheroids tolerated 24 h of cold storage in SFM supplemented with Def remarkably well, but some cellular activities were reduced to unacceptable levels after 48 h of cold storage. Posthypothermic rates of ammonia detoxification approached that of control spheroids at corresponding time points after spheroid formation. The expression levels of genes involved in ammonia detoxification and albumin production were lower in spheroids cold stored for both 24 and 48 h than corresponding untreated control spheroids, indicating that cold storage was associated with some degree of damage. The molecular mechanism of this damage remains unclear but likely involves multiple pathways (14,23,32). These findings suggest that although further research is still needed in this area, the use of hepatocyte spheroids at distant user sites may be possible when transported in cold storage.
Footnotes
Acknowledgments
This work was funded by grants from NIH (RO1-DK56733), the Marriott Foundation, the Wallace H. Coulter Foundation, the 12th Five-Year National Science and Technology Major Project for Infectious Diseases, China (No. 2012 ZX 10002004-005, Hongling Liu), and Jiangsu province government, China (Yue Yu). The authors declare no conflicts of interest.
