Abstract
Since current treatments for both acute and chronic lung diseases are less than ideal, there has been recent interest in the use of cell-based therapies for inflammatory lung disease. Specifically, human amnion epithelial cells (hAECs) have been shown to reduce bleomycin-induced lung injury and prevent subsequent loss of respiratory function, primarily through modulation of the host immune response. The precise mechanisms of this effect remain unclear. We aimed to investigate the potential of hAECs to mitigate bleomycin-induced lung injury in surfactant protein C deficient (Sftpc-/-) mice, which are highly susceptible to pulmonary injury as a result of impairment of macrophage function. Primary hAECs were administered to wild-type (Sftpc+/+) and Sftpc-/- mice 24 h after exposure to bleomycin. Compared to Sftpc+/+ mice receiving bleomycin alone, Sftpc+/+ mice administered hAECs 24 h after bleomycin exposure had decreased expression of proinflammatory genes, decreased macrophage and neutrophil infiltration, fibrosis, collagen content, and α-smooth muscle actin as well as a significant improvement in lung function. Compared to Sftpc-/- mice given bleomycin alone, Sftpc-/- mice administered hAECs 24 h after bleomycin did not have a decrease in inflammatory gene expression or a reduction in macrophage pulmonary infiltration. Subsequently, Sftpc-/- mice did not show any decrease in pulmonary fibrosis or improvement of lung function after hAEC administration. The ability of hAECs to mitigate bleomycin-induced lung injury is abolished in Sftpc-/- mice, suggesting that hAECs require normal host macrophage function to exert their reparative effects.
Introduction
The World Health Organization predicts that chronic respiratory disorders, such as idiopathic pulmonary fibrosis (IPF) and chronic obstructive pulmonary disease (COPD), will become the third most common cause of death worldwide (44). While the initiating causes of respiratory diseases differ, the resultant lung damage, namely chronic inflammation, fibrosis, and scarring leading to loss of functional lung tissue, is similar (3,41,43). Current therapies for respiratory disease include nonspecific anti-inflammatory agents (8,11,17) as well as targeted anti-inflammatory therapies (9,37). Thus far, these therapies have not proven adequate in the long term and are often associated with significant side effects. Accordingly, there is an increasingly urgent need for the development of novel, effective treatments of chronic lung disease. With this in mind, there has been recent interest in the reparative/regenerative potential of cell-based therapies, including cells derived from the bone marrow, umbilical cord, and placental tissues (24,35,36).
With such a cell therapy in mind, we have recently shown that human amnion epithelial cells (hAECs) offer promise as a cell therapy for lung disease. Specifically, we have shown that hAECs have the ability to differentiate into lung epithelial-like cells in vitro, expressing thyroid transcription factor Nkx2.1 (also known as TTF-1) and pulmonary surfactant proteins A, B, C, and D (23,31). We have also shown that the administration of hAECs in vivo reduces bleomycin-induced lung inflammation and fibrosis and facilitates the regeneration of normal lung architecture in both immune-compromised (31) and immune-competent (33) mice. Importantly, these protective and reparative effects are associated with mitigation of the loss of lung function induced by bleomycin (33). However, the precise mechanism(s) by which hAECs exert their effects in this model of lung injury remain uncertain. In particular, it is not clear whether hAECs exert their effects via differentiating into lung epithelial cells in vivo, as we have shown occurs in immune-compromised mice (31), or whether the principle mechanism is via modulation of the host response to injury. Based on a study undertaken in immune-competent mice where we observed no in vivo differentiation of hAECs and yet mitigation of lung injury, we have recently suggested that the latter is the more likely mechanism (33).
To explore the mechanisms of action of hAECs, we undertook the current study to investigate the ability of hAECs to both differentiate into lung epithelial cells in vivo and to mitigate bleomycin-induced lung injury in surfactant protein C-deficient (Sftpc-/-) mice. We chose to undertake the work using this strain for mice for two reasons. First, if we were able to demonstrate the production of surfactant protein C following hAEC administration then this would confirm that hAECs were differentiating in vivo into functional lung epithelial cells. Second, Sftpc-/- mice are known to have a heightened susceptibility to lung injury secondary to impaired macrophage function, including reduced phagocytic activity and increased metalloproteinase activity (18,19,26). This afforded an opportunity to examine one aspect of hAEC–host immune response interaction.
Materials and Methods
Animals and Experimental Groups
All experimental procedures were approved by Monash Medical Centre Animal Ethics Committee and were conducted in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (2006). Surfactant protein C knockout (Sftpc-/-) and wild-type (Sftpc+/+) mice on a 129/S6 background were sourced from Cincinnati Children's Hospital Medical Center, USA. A total of 61 Sftpc-/- and 69 Sftpc+/+, 6–8-week-old female mice weighing 18–21 g were randomly divided into four treatment groups: Saline—mice received intranasal instillation (IN) of saline and an intraperitoneal (IP) injection of 400 μl saline 24 h later (Sftpc-/- n = 14, Sftpc+/+ n = 14);bleomycin—mice received IN bleomycin and an IP injection of saline 24 h later (Sftpc-/- n = 14, Sftpc+/+ n = 15);saline + hAECs—mice received IN saline and an IP injection of 4 million hAECs 24 h later (Sftpc-/- n = 16, Sftpc+/+ n = 17);bleomycin + hAECs—mice received IN instillation bleomycin and an IP injection of 4 million hAECs 24 h later (Sftpc-/- n = 17, Sftpc+/+ n = 23).
All mice were culled at either day 7 (for scoring of inflammation) or day 14 (for scoring of fibrosis) following IN bleomycin or saline. These time points for the various analyses were selected based on previous work that identified that inflammation was maximum at day 7 and fibrosis maximum at day 14 following bleomycin treatment (33). Body weight, core temperature, and basal lung function were recorded prior to commencement of experiments and at days 3, 7, and 14.
Bleomycin Induction of Pulmonary Fibrosis
Intranasal administration of bleomycin to rodents is considered a useful model for the histological changes seen in human pulmonary fibrosis (42). Mice were anesthetized with an IP injection of xylazine (2 mg/kg) and ketamine (100 mg/kg), allowing administration of an IN dose of bleomycin (4 U/kg for Sftpc-/- mice or 7 U/kg for Sftpc+/+ mice) in a total volume of 24–28 μl saline. The administered doses for each mouse strain were determined by dose titration to induce comparative levels of pulmonary fibrosis in each strain as determined by Ashcroft scoring with minimal animal mortality (data not shown). Animals in the saline and hAEC-treated groups received 21–25 μl IN saline alone.
hAEC Isolation, Labeling, and Injection
Isolation of hAECs suitable for clinical use was performed as described previously (34) using good manufacturing practice (GMP) processes. Prior to IP injection, hAECs were labeled with carboxyfluorescein succinimidyl ester (CFSE). Briefly, hAECs were resuspended at 15 million cells/ml in 5 μM CFSE and incubated at 37°C for 10 min. hAECs were washed three times in PBS and resuspended in sterile saline (10 million cells/ml).
Lung Function Testing
Lung function was assessed using unrestrained whole body plethysmography as previously described (29), at days 0, 3, 7, and 14 for each group of animals. Briefly, the plethysmograph is a single cylindrical Perspex chamber (150 × 50 mm) fitted with a temperature and relative humidity probe (Vaisala, Finland) and a volumetric pressure transducer (Grass Instrument Co., USA). Change in pressure caused by the tidal movement of gas within the chamber (as the mouse breathes) is measured by the pressure transducer, amplified (Octal Bridge Amp; Powerlab, AD Instruments, Australia), and recorded (Chart 5.1, AD Instruments, Australia) using a standard desktop PC. The pulmonary function of each mouse was recorded, while the animal was stationary and calm, for an average of 2 min. Wave form analysis was used to analyze the respiratory trace obtained from each animal, including the measurement of the change in pressure caused by the tidal movement of gas within the chamber (PT), respiration rate (breaths/min), total breathing cycle time (Ttot; s), inspiration time (Ti; s), and expiration time (Te; s). Tidal volume (VT; ml) was calculated as described by Drorbaugh and Fenn (13). Minute volume (VE; ml/min), inspiratory duty cycle (Ti/Ttot; %), inspiratory flow rate (VT/T i; ml/s), and the ratio of inspiration time to expiration time (Ti/Te ratio) were also calculated.
Tissue Collection
Animals were culled by an IP injection of 150 mg/kg Lethobarb (Virbac, Australia). The right lung was ligated at the right mainstem bronchus, excised, weighed, and processed for RNA isolation. Upon excising the right lung, the trachea was then exposed, cannulated, and the left lung was instilled with 4% paraformaldehyde (PFA). The left lung was then excised, immersed in 4% PFA for 48 h, and processed for histology. Flow cytometric analysis for CFSE-labeled hAECs was performed at day 7 on whole lungs (right and left) from six Sftpc-/- and six Sftpc+/+ (saline + hAECs, n = 2; bleomycin + hAECs, n = 4). No other outcomes were collected from these 12 animals.
Assessment of Lung Fibrosis, Collagen, and α-Smooth Muscle Actin Deposition
Histological sections from throughout each left lung were cut and sequential fields of view (200x magnification) were independently scored, in triplicate, by three researchers, each blinded to treatment group, for the severity of parenchymal fibrosis according to the semiquantitative method previously described by Ashcroft (1). An overall mean (±SEM) score was calculated from the triplicate scores from all three scorers.
Collagen quantification was performed using Sirius red as previously described (25). Tissue sections were stained with Sirius red and collagen was quantified by digital image analysis using the Image J software package [National Institutes of Health (NIH)]. For each tissue sample, five images were captured using brightfield light microscopy at 200x magnification using a light microscope (AxioSkop, Zeiss, Germany) with a camera attachment (AxioCam ICc 3, Zeiss, Germany). The percentage of area positive for Sirius red was calculated as previously described (38). Each image was converted to a 16-bit black-and-white image and threshold values adjusted to include only stained areas. Threshold values were maintained for all samples. The active area score (percentage of stained pixels per field of view) was determined and analyzed using statistical analysis software (GraphPad Prism, Graphpad Software Inc., USA). Tissue sections were incubated with primary antibodies against α-smooth muscle actin (α-SMA) (1:100 ab5694, Abcam, USA) at room temperature for 1 h, followed by incubation with a biotinylated secondary antibody and peroxidase-labeled streptavidin (LSAB+ System HRP, Dako, Denmark). Stained cells were visualized using the liquid DAB substrate with hematoxylin counterstain.
Detection of CFSE-Labeled Cells
We used two approaches to detect CFSE-labeled hAECs in mouse lungs. First, left and right lung lobes were washed to remove residual blood, minced, and passed through a 70-μm filter to obtain a single cell suspension, and then analyzed using a BD FACSCanto II Flow cytometry system (BD Biosciences, USA). Gates were set based on the fluorescent profile of CFSE-labeled and unlabeled hAECs and which excluded dead cells as well as small cells such as red blood cells. Second, to detect CFSE-labeled hAECs histologically, sections from throughout each left lung were cut and incubated with primary antibodies [1:500 rabbit polyclonal anti-Pro-Surfactant C (ProSP-C), AbCam, USA; 1:1000 anti-fluorescein, Invitrogen, USA] for 1 h at room temperature. Sections were then incubated with secondary antibody for ProSP-C (AlexaFluor® 568 donkey anti-rabbit IgG, Invitrogen, USA) for 1 h at room temperature. Slides were counter stained with DAPI (Sigma, USA) and mounted with Dako Fluorescent Mounting Medium (Dako, Denmark). Slides were visualized with the Leica DMR HCS microscope system to determine presence of SP-C (red) and CFSE-positive cells (green).
Assessment of Lung Inflammation
To assess pro- and anti-inflammatory cytokine responses to bleomycin ± hAECs, we isolated RNA for quantitative PCR. Briefly, total RNA was isolated from snap frozen lungs using TRIzol (Invitrogen) and converted to cDNA using the Thermoscript Reverse Transcription System (Invitrogen) according to the manufacturer's instructions. Quantitative PCR was performed using Lightcycler 480 SYBR Green I Master Mix (Roche, Switzerland) and the Corbett Research Rotor-Gene 3000 (QIAGEN, Germany). Primers were directed against interleukin (IL)-1β, IL-6, macrophage inhibitory factor (MIF), transforming growth factor-β (TGF-β), eosinophil chemotactic factor 1 (also known as Ym1 and chitinase 3-like 3), arginase (Arg1), and 18S. Primer sequences and annealing temperatures are available upon request. Gene expression of each marker was normalized to 18S.
We assessed lung inflammation by counting macrophage and neutrophils in mouse lungs. To do this, we identified macrophages by immunolabeling the F4/80 antigen and we identified neutrophils with myeloperoxidase. Briefly, tissue sections were incubated with primary antibodies against F4/80 (1:100, MCA497R, AbD Serotec, Germany) or myeloperoxidase (1:200, ab45977, Abcam) at room temperature for 40 min. Tissue sections were then incubated in biotinylated secondary antibody (1:200, AP183β, Chemicon, USA) before detection using the EnVision detection system (LSAB+ System HRP, Dako) and liquid DAB substrate (Dako). Tissues were counterstained with hematoxylin.
For each tissue sample, five images were captured using brightfield light microscopy at 200x magnification using a light microscope (AxioSkop, Zeiss, Germany) with a camera attachment (AxioCam ICc 3, Zeiss, Germany). Each image was converted to an RGB image and threshold values for nuclei (blue) or DAB (brown) were adjusted to include only stained areas using Meta-Morph bioanalytical software (Molecular Devices, USA). Minimum and maximum cell size thresholds were also determined. Threshold values were maintained for all samples. F4/80- or myeloperoxidase-stained cells were counted using the bioanalytical software and expressed as a fold change from the saline control group.
Pulmonary Macrophage Isolation
Lung injury was induced in Sftpc-/- and Sftpc+/+ mice as described above. Mice were culled 24 h following bleomycin and lungs minced using a McIlwain tissue chopper (Campden Instruments, USA). Lung tissues were then digested in Waymouths medium (Invitrogen, USA) containing 25 mg/ml collagenase I, 25 mg/ml collagenase IA, 2.5 mg/ml DNase, 25 mM HEPES, and 10% heat-inactivated chicken serum for 15 min at 37°C. Pulmonary macrophages were sorted using antibodies targeting CD45 (1:150, #560457, BD Biosciences, USA) and F/480 (1:50, AB60343, Abnova, Taiwan) with a Mo-Flo XDP high-speed sorter (Dako, Denmark). Macrophages were cultured in RPMI-1640 media (Invitrogen) containing 20% fetal bovine serum (FBS), 1% insulin transferrin selenium (ITS), nonessential amino acids (NEAA), and 10% L929 conditioned medium for 7 days.
Chemotactic Macrophage Migration Assay
Macrophage migration was assessed as previously described (27). Briefly, chemotactic assays were performed with 6.5-mm diameter, 3.0-μm pore size, 24-well cell culture inserts (BD Biosciences, USA). The bottom chamber was filled with either 0.2 ml RPMI-1640 or RPMI-1640 containing 600 ng/ml of recombinant mouse macrophage inflammatory protein (rMIP)-2 (R&D Systems, USA). Macrophages (1 × 105), with or without hAEC-conditioned medium (CM), were added to the top chamber and plates were incubated at 37°C for 4 h. Cell culture insert membranes were stained with Diff-Quick (Lab Aids, Australia) and the number of migrated cells determined using light microscopy.
Data Analysis and Statistics
Data were expressed for each experimental group as mean ± SEM and statistical significance determined using statistical analysis software (GraphPad Prism, Graphpad Software Inc., USA). Body weight and lung function testing data were analyzed using the repeated measures analysis of variance (ANOVA) with the Bonferroni post hoc test. Confidence intervals of 95% were assumed to be significant. Ashcroft scores, collagen content, Western blot, and qPCR data were analyzed using the one-way ANOVA with the Bonferroni post hoc test and a 95% confidence interval was assumed to be significant.
Results
Effect of Bleomycin and hAEC Administration on Body Weight
Compared to saline controls, administration of bleomycin to Sftpc+/+ mice resulted in a significant reduction in body weight from day 7 (19.2 ± 0.4 vs. 15.6 ± 0.5 g, p ≤ 0.0001) (Fig. 1A). Compared to bleomycin-alone animals, administration of hAECs to bleomycin-treated Sftpc+/+ mice resulted in significant improvement in body weight at day 7 and 14 (day 7, 15.6 ± 0.5 vs. 17.2 ± 0.3 g, p ≤ 0.05; day 14, 16.9 ± 0.6 vs. 18.9 ± 0.4 g, p ≤ 0.05). Compared to saline controls, administration of bleomycin to Sftpc-/- mice resulted in a significant reduction in body weight from day 7 (day 7, 18.7 ± 0.5 vs. 16.4 ± 0.9 g, p ≤ 0.05) (Fig. 1B). No significant differences in body weight were observed at any time point between bleomycin-only-treated Sftpc-/- mice and Sftpc-/- mice that received hAECs 24 h after bleomycin.

Effect of hAEC administration on body weight of Sftpc+/+ (A) and Sftpc-/- (B) mice. Administration of bleomcyin resulted in a significant reduction in body weight in both strains, compared to saline control mice. Administration of hAECs to bleomcyin-treated Sftpc+/+ mice resulted in a partial restoration of body weight (day 7 and 14). Administration of hAECs to bleomcyin-treated Sftpc-/- mice did not result in a significant improvement in body weight. Significant differences between bleomycin and bleomycin + hAEC groups: *p ≤ 0.05, **p ≤ 0.001.
Effect of Bleomycin and hAEC Administration on Lung Function
In both Sftpc+/+ mice and Sftpc-/- mice bleomycin administration significantly altered lung function. Compared to control animals, bleomycin-treated mice had a significantly increased minute volume in both Sftpc+/+ (day 7, 1,909 ± 188 vs. 2,582 ± 208 ml/kg, p ≤ 0.05; day 14, 1,789 ± 167 vs. 2,891 ± 443 ml/kg, p ≤ 0.001) (Fig. 2A) and Sftpc-/- mice (day 7, 1,905 ± 137 vs. 2,402 ± 130 ml/kg, p ≤ 0.05; day 14, 1,618 ± 81 vs. 2,686 ± 248 ml/kg, p ≤ 0.0001) (Fig. 2B), as well as a significantly increased respiratory rate (breaths/min) in both Sftpc+/+ (day 14, 287 ± 11 vs. 403 ± 43, p ≤ 0.001) (Fig. 2C) and Sftpc-/- mice (day 14, 269 ± 10 vs. 378 ± 30, p ≤ 0.001) (Fig. 2D). In Sftpc+/+ mice, compared to control animals, bleomycin-treated mice had a significantly increased expiration time (day 3, 0.13 ± 0.01 vs. 0.20 ± 0.01 s, p ≤ 0.0001) (Fig. 2E). No significant differences were observed between Sftpc-/- control and bleomycin-treated groups for expiration time (Fig. 2F). Compared to control animals, bleomycin-treated mice had a significantly decreased inspiration time in both Sftpc+/+ (day 7, 0.1 ± 0.01 vs. 0.07 ± 0.003 s, p ≤ 0.0001) (Fig. 2G) and Sftpc-/- mice (day 7, 0.09 ± 0.003 vs. 0.07 ± 0.01 s, p ≤ 0.0001; day 14, 0.09 ± 0.002 vs. 0.06 ± 0.01 s, p ≤ 0.001) (Fig. 2H).

Effect of hAEC administration on lung function of Sftpc+/+ and Sftpc-/- mice. Sftpc+/+ mice in the bleomycin-treated group had significantly increased minute volume (A), respiration rate (breaths/min) (C), and expiration time (E) and decreased inspiration time (G) compared to saline control animals. Sftpc-/- mice in the bleomycin-treated group had significantly increased minute volume (B), respiration rate (breaths/min) (D), and decreased inspiration time (H) compared to control animals. No significant difference was detected for expiration time (F) between the bleomycin-treated group and saline control animals. Administration of hAECs to bleomycin-treated Sftpc+/+ mice resulted in partial restoration of physiological lung function with significant improvement in minute volume (day 14), respiration rate (breaths/min) (day 14), expiration time (day 3), and inspiration time (day 7) compared to bleomycin-treated control animals. Administration of hAECs to bleomycin-treated Sftpc-/- mice did not result in any significant improvement in body weight, minute volume, respiration rate, or inspiration time compared to bleomycin-treated control animals at any time point.
In Sftpc+/+ mice, but not in Sftpc-/- mice, the administration of hAECs following bleomycin was associated with mitigation of impaired lung function. Compared to bleomycin alone animals, administration of hAECs to bleomycin-treated Sftpc+/+ mice resulted in a significantly lower minute volume (day 14, 2,891 ± 443 vs. 2,034 ± 80.4 ml/kg, p ≤ 0.05) (Fig. 2A), lower respiration rate (breaths/min) (day 14, 403 ± 43 vs. 310 ± 10, p ≤ 0.001) (Fig. 2C), lower expiration time (day 3, 0.20 ± 0.01 vs. 0.17 ± 0.01 s, p ≤ 0.05) (Fig. 2E), and a longer inspiration time (day 7, 0.07 ± 0.01 vs. 0.08 ± 0.01 s, p ≤ 0.05) (Fig. 2G). In contrast, no significant differences in any lung function parameter were observed between Sftpc-/- bleomycin-alone and bleomycin + hAECs animals.
Effect of Bleomycin ± hAEC Administration on Lung Fibrosis, Collagen Deposition, and Activated Myofibroblasts
Pulmonary fibrosis is characterized by the obliteration of alveolar spaces followed by the deposition of a matrix of fibronectin, collagen, and α-smooth muscle actin, a product of activated myofibroblasts (41). Compared to saline-treated controls, pulmonary fibrosis at day 14, as assessed by Ashcroft scoring, was increased by bleomycin treatment in both Sftpc+/+ (0.8 ± 0.1 vs. 5.0 ± 0.3, p ≤ 0.0001) (Fig. 3A) and Sftpc-/- mice (0.8 ± 0.1 vs. 4.2 ± 0.6, p ≤ 0.0001) (Fig. 3B). The extent of fibrosis induced in Sftpc+/+ and Sftpc-/- mice was similar, as expected from the dose–titration studies. Administration of hAECs after bleomycin treatment significantly reduced fibrosis in Sftpc+/+ mice (5 ± 0.3 vs. 3.5 ± 0.4, p ≤ 0.05) (Fig. 3A) but not in Sftpc-/- mice (4.2 ± 0.6 vs. 5.1 ± 0.6, p = 0.34) (Fig. 3B).

Effect of hAEC administration on lung fibrosis of Sftpc+/+ and Sftpc-/- mice. At day 14, bleomycin-treated mice had significantly higher Ashcroft scores of fibrosis compared to saline control mice (A, B). Administration of hAECs to bleomycin-treated mice resulted in significantly lower Ashcroft scores of fibrosis for Sftpc+/+ mice (A) but not for Sftpc-/- mice (B). At day 14, bleomycin-treated Sftpc+/+ and Sftpc-/- mice had increased lung collagen content compared to saline control mice (C, D). Administration of hAECs to bleomycin-treated mice resulted in a significantly lower percentage of collagen per area of lung for Sftpc+/+ mice (C) but not for Sftpc-/- mice (D). Administration of bleomcyin resulted in a significant increase in α-SMA (smooth muscle actin) in Sftpc+/+ and Sftpc-/- mice compared to saline control mice (E, F). Administration of hAECs to bleomycin-treated mice resulted in a significantly decreased levels of α-SMA for Sftpc+/+ mice (E) but not for Sftpc-/- mice (F). *p ≤ 0.05, **p ≤ 0.001, ***p ≤ 0.0001.
Compared to control animals, bleomycin administration increased collagen content at day 14 in both Sftpc+/+ (2.54 ± 0.45% vs. 15.13 p ≤ 2.58%, p ≤ 0.0001) (Fig. 3C) and Sftpc-/- mice (1.70 ± 0.15% vs. 9.06 ± 1.44%, p ≤ 0.0001) (Fig. 3D). In the Sftpc+/+ mice, this effect of bleomycin was significantly mitigated by hAEC administration (15.13 ± 2.58% vs. 9.11 ± 1.22%, p ≤ 0.05) (Fig. 3C), an effect not observed in the Sftpc-/- mice (9.06 ± 1.44% vs. 9.38 ± 1.32%, p = 0.88) (Fig. 3D).
Similarly, compared to control animals, bleomycin administration increased day 14 levels of α-SMA in both Sftpc+/+ (76 ± 24-fold change compared to control, p ≤ 0.01) (Fig. 3E) and Sftpc-/- mice (84 ± 21-fold change compared to control, p ≤ 0.05) (Fig. 3F). While the administration of hAECs following bleomcyin mitigated increases in pulmonary α-SMA in Sftpc+/+ mice (21 ± 9-fold change compared to control, p ≤ 0.05) (Fig. 3E), this effect was not in seen in Sftpc-/- mice (day 14, 71.99 ± 25.20-fold change compared to control) (Fig. 3F). Representative images of histological sections are shown in Figure 4. And representative images of Sirius red-stained histological sections are shown in Figure 5.

Representative images at day 14 of haematoxylin and eosin-stained histological sections from Sftpc+/+ and Sftpc-/- mice: saline (A), saline + hAECs (B), bleomycin (C), and bleomycin + hAECs (D). Histological sections were scored for levels of fibrosis as described in Materials and Methods. Scale bars: 50 μm.

Representative images at day 14 of Sirius red-stained histological sections from Sftpc+/+ and Sftpc-/- mice: saline (A), saline + hAECs (B), bleomycin (C), and bleomycin + hAECs (D). Lung collagen content was determined using Sirius red-stained photomicrographs that were quantified by digital image analysis as described in Materials and Methods. Scale bars: 50 μm.
Analysis of Lung Tissue for hAECs
We were unable to detect any CFSE labeled cells, by either flow cytometry or immunohistochemistry, in any of our groups of animals. A sample of CFSE-labeled cells utilized as a positive control retained the CFSE label and were viable for the duration of the experiment. Surfactant protein C expression was only detected in the endogenous lung epithelium of Sftpc+/+ mice (data not shown).
Effect of Bleomycin and hAEC Administration on Lung Inflammation
Compared to control animals, bleomycin significantly increased the expression of proinflammatory genes in the lungs of both Sftpc+/+ mice (IL-1β: 1.3 ± 0.2 vs. 52.2 ± 29.6, p ≤ 0.05; IL-6: 0.4 ± 0.2 vs. 21 ± 7.6, p ≤ 0.001; MIF: 1.7 ± 0.4 vs. 13.2 ± 3.5, p ≤ 0.05; TGF-β: 1.2 ± 0.4 vs. 8 ± 2.3, p ≤ 0.05) (Fig. 6) and Sftpc-/- mice (IL-1β: 0.4 ± 0.3 vs. 1.8 ± 0.5, p ≤ 0.05; IL-6: 0.70 ± 0.2 vs. 8.5 ± 4, p ≤ 0.05; TGF-β: 1.6 ± 0.3 vs. 8.6 ± 1.5, p ≤ 0.05) (Fig. 6). There was no effect of bleomycin on MIF expression in Sftpc-/- mice (Fig. 6).
The administration of hAECs to bleomycin-treated Sftpc+/+ mice was associated with significantly lower proinflammatory pulmonary gene expression than in bleomycin-alone animals (IL-1β: 4 ± 2.3 vs. 52.2 ± 29.6, p ≤ 0.05; IL-6: 7.4 ± 2.1 vs. 20.7 ± 7.6, p ≤ 0.05; MIF: 5.5 ± 1.9 vs. 13.2 ± 3.5, p ≤ 0.05; TGF-β: 3.3 ± 0.7 vs. 8 ± 2.3, p ≤ 0.05) (Fig. 6). In contrast, in Sftpc-/- animals the administration of hAECs 24 h after bleomycin was associated with significantly lower pulmonary gene expression only for IL-1β (0.5 ± 0.3 vs. 1.8 ± 0.5, p ≤ 0.05) and TGF-β (0.6 ± 0.1 vs. 8.6 ± 1.5, p ≤ 0.05) (Fig. 6).

Effect of hAEC administration on lung inflammation in Sftpc+/+ and Sftpc-/- mice. Analysis of inflammatory cytokine gene expression was performed by qPCR at day 7. Bleomycin-treated Sftpc+/+ mice had significantly increased gene expression for interleukin-1β (IL-1β) (A), IL-6 (C), macrophage inhibitory factor (MIF) (E), and transforming growth factor-β (TGF-β) (G) compared to saline control animals. Bleomycin-treated Sftpc-/- mice had significantly increased gene expression for IL-1β (B), IL-6 (D), and TGF-β (H) compared to saline control animals. No significant change in gene expression was observed for MIF (F) in bleomycin-treated Sftpc-/- mice. Administration of hAECs to bleomycin-treated mice resulted in a significant decrease in gene expression of IL-1β, IL-6, MIF, and TGF-β in Sftpc+/+ mice, while only IL-1β and TGF-β were significantly reduced in Sftpc-/- mice. *p ≤ 0.05, **p ≤ 0.001, ***p ≤ 0.0001.
Effect of Bleomycin and hAEC Administration on Leukocyte Number
Changes to the leukocyte number in the lung were determined on day 7 in order to evaluate hAEC-mediated effects at a time where maximal inflammation would be observed in the bleomycin model. Compared to control animals, in both Sftpc+/+ mice and Sftpc-/- mice bleomycin administration was associated with an increased number of both lung macrophages [Sftpc+/+: 28.8 ± 4.4-fold change compared to control, p ≤ 0.0001 (Fig. 7A); Sftpc-/-: 24.8 ± 9.1-fold change compared to control, p ≤ 0.05 (Fig. 7B)] and neutrophils [Sftpc+/+: 44.4 ± 11.3-fold change compared to control, p < 0.0001 (Fig. 7C); Sftpc-/-: 31.3 ± 8.5-fold change compared to control, p ≤ 0.0001 (Fig. 7D)]. In Sftpc+/+ mice, administration of hAECs to bleomycin-treated animals partially mitigated the bleomycin-induced increase in both macrophages and neutrophils (macrophages: 10.80 ± 4.496-fold change compared to control, p ≤ 0.001; neutrophils: 13.6 ± 4.6-fold change compared to control, p ≤ 0.001). However, while the administration of hAECs to bleomycin-treated Sftpc-/- mice partially mitigated the neutrophil influx (14.3 ± 3.3-fold change compared to control, p ≤ 0.001), there was no significant effect on macrophage number (26 ± 5.8-fold change compared to control).

Quantitation of F4/80 and myeloperoxidase (MPO) immunohistochemical staining in Sftpc+/+ and Sftpc-/- mice. F4/80 and myeloperoxidase positive cells were quantified at day 7. Bleomycin-treated Sftpc+/+ and Sftpc-/- mice had significantly increased numbers of F4/80 expressing macrophages (A, B) and myeloperoxidase expressing neutrophils (C, D) at day 7. Administration of hAECs to bleomycin-treated Sftpc+/+ mice resulted in a significant decrease in the numbers of infiltrating F4/80 (A)- and myeloperoxidase (C)expressing cells. Administration of hAECs to bleomycin-treated Sftpc-/- mice resulted in a significant decrease in the numbers of infiltrating myeloperoxidase (D)-expressing cells, while no significant difference was observed in the numbers of F4/80-expressing cells (B). *p ≤ 0.05, **p ≤ 0.001, ***p ≤ 0.0001.
Gene expression for Ym1 and arginase-1, markers of the alternatively activated macrophage phenotype, was measured at day 7. Compared to control Sftpc+/+ mice, bleomycin significantly increased gene expression of Ym1 (8.53 × 10−6 ± 8.40 × 10−6 vs. 269.6 ± 140.4, p ≤ 0.001) and arginase-1 (0.081 ± 0.045 vs. 31,709 ± 14,215, p ≤ 0.05). Compared to bleomycin-treated Sftpc+/+ mice, mice receiving hAECs 24 h after bleomcyin had further increased gene expression for Ym1 (269.6 ± 140.4 vs. 1,895 ± 406.1, p ≤ 0.001) and arginase-1 (31,709 ± 14,215 vs. 1.11 × 106 ± 0.556 × 106, p < 0.05). No significant differences were observed for Arg1 or Ym1 gene expression between Sftpc-/- groups (Fig. 8).
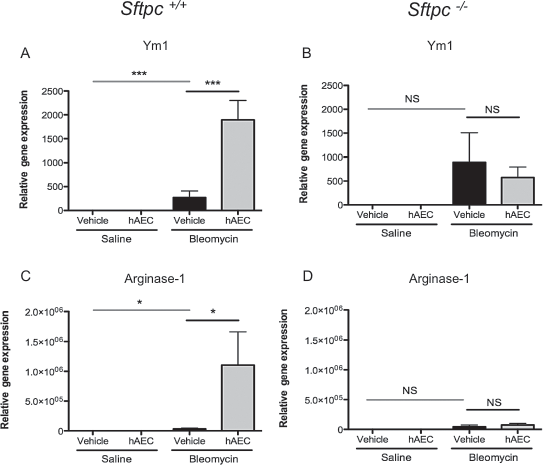
Effect of hAEC administration on alternatively activated macrophage phenotype in Sftpc+/+ and Sftpc-/- mice. Gene expression of arginase-1 (Arg1) and Ym1, markers of alternatively activated macrophages, was measured by qPCR at day 7. Bleomycin-treated Sftpc+/+ mice had significantly increased gene expression for Ym1 (A) and Arg1 (C), compared to saline control animals. Mice receiving hAECs 24 h after bleomcyin had significantly increased gene expression for Arg1 (A) and Ym1 (B). No significant differences were observed for Arg1 or Ym1 gene expression between Sftpc-/- groups. *p ≤ 0.05, ***p ≤ 0.0001.
Effect of hAEC Supernatant on Macrophage Migration In Vitro
An in vitro chemotactic migration assay was used to determine the effect of hAEC conditioned medium (CM) on the migration of macrophages from Sftpc+/+ and Sftpc-/- mice. A macrophage cell line derived from C57B16 bone marrow used as a positive control migrated towards the MIP-2 chemotactic gradient (9.0 ± 0.7-fold change compared to control, p ≤ 0.001). Migration was significantly inhibited by the addition of hAEC CM (1.1 ± 0.7-fold change compared to control, p ≤ 0.001). Macrophages from Sftpc+/+ mice also migrated towards the MIP-2 chemotactic gradient (4.7 ± 0.6-fold change compared to control, p ≤ 0.01) and migration was significantly inhibited by the addition of hAEC CM (1.7 ± 0.4-fold change compared to control, p ≤ 0.05) (Fig. 9). Macrophages from Sftpc-/- mice did not have a significant increase in migration towards the MIP-2 chemotactic gradient (2.7 ± 1.4-fold change compared to control) and there was no significant change with the addition of hAEC CM (3.2 ± 1.0-fold change compared to control).

Effect of hAEC conditioned medium (CM) on macrophage migration in vitro. An in vitro chemotactic migration assay was used to determine the effect on hAEC CM on the migration of macrophages from C57Bl6 bone marrow and from Sftpc+/+ and Sftpc-/- lungs. Macrophages from C57B16 and Sftpc+/+ mice migrated towards the MIP-2 chemotactic gradient and migration was significantly inhibited by the addition of hAEC CM. No significant differences were observed in macrophage migration between Sftpc-/- groups. **p ≤ 0.001, ***p ≤ 0.0001.
Discussion
In this study we investigated the ability of hAECs to mitigate acute bleomycin-induced lung injury by both differentiation into lung epithelial cells in vivo and by modulation of host lung inflammation and subsequent structural and functional lung changes. We demonstrated that administration of hAECs reduced bleomycin-induced injury, both inflammation and fibrosis, and loss of lung function in Sftpc+/+ mice, but not in Sftpc-/- mice. We were unable to demonstrate any integration or differentiation of hAECs into the lungs of either mouse strain. Instead, the ability of hAEC therapy to mitigate lung injury appeared related to effects on macrophage recruitment and polarization.
We and others have shown that placental and amnion-derived cells are effective in mitigating and repairing bleomycin-induced lung injury (5,31,33). However, precisely how hAECs exert these effects remains uncertain. While studies in immune compromised mice have shown that both hAECs (31) and amniotic fluid-derived cells (6) are able to integrate into the host epithelium and differentiate in vivo into lung epithelial cells following bleomycin lung injury (6), we have not been able to show this in immune competent mice in either a previous study (33) or in this current study. Acknowledging that FACS analysis, one of the approaches we use for cell detection, excludes dead cells and has a lower level of sensitivity compared to other techniques (33), in this study we utilized both FACS analysis and immunohistochemistry to identify CFSE-labeled cells in host tissue. As before, we were unable to detect any hAECs in either the injured or uninjured lung. In light of our inability to show either integration or differentiation of hAECs in immune competent mice, and yet still show mitigation of bleomycin injury by hAEC administration, we believe that differentiation of hAECs into lung epithelial cells is not likely to be the key mechanism underlying the prevention and/or repair of acute lung injury. Rather, we suggest that the hAECs primarily exert their effects by reducing acute inflammation, both leukocyte infiltration and cytokine responses, and thereby mitigating subsequent pulmonary fibrosis.
A wide array of inflammatory and fibrotic signaling pathways are activated by bleomycin, including key players in lung injury such as TGF-β (2), the inflammasome (12,30,46), and inflammatory cytokines such as IL-1β, IL-6, interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α) (39). In this regard, hAEC administration has been shown to significantly decrease profibrotic gene expression in bleomycin-treated mice (33), a finding confirmed in the current study where TGF-β gene expression was significantly reduced following hAEC administration in both Sftpc+/+ and Sftpc-/- mice. We also noted that while hAEC administration significantly reduced IL-1β expression in both Sftpc+/+ and Sftpc-/- mice, the relative expression of IL-1β in bleomycin-treated Sftpc+/+ mice was 30-fold greater than in Sftpc-/- mice. This suggests that the IL-1β response to bleomycin in Sftpc-/- mice is compromised. Further, hAEC administration significantly reduced IL-6 and MIF gene expression in Sftpc+/+ mice, an effect that was not observed in Sftpc-/- mice. As IL-6 and MIF are involved in the recruitment of macrophages into the lung following bleomycin injury (40), these data suggest that one difference in the effect of hAEC administration in Sftpc+/+ and Sftpc-/- mice may involve the recruitment of macrophages to the site of injury. In this regard, impaired macrophage function (altered morphology, reduced phagocytic activity, and increased metalloproteinase activity) in Sftpc-/- mice is thought to underlie this strain's increased susceptibility to respiratory tract infections and, we suggest, underlie heightened and prolonged fibrosis following bleomycin insult (18–20,26).
Specifically, Sftpc-/- macrophages express markers of alternative activation, such as increased arginase activity, which contribute to matrix remodeling and collagen synthesis (15,22), as well as abundant expression of chitinase protein Ym1, which has been associated with epithelial inflammation and asthma (7,14). Further, production of matrix metalloproteinases 2 and 9 are increased in alveolar macrophages of Sftpc-/- mice, indicating that macrophages in these mice may have an altered role in the process of inflammation and the development of fibrosis (18).
The observation that administration of hAECs decreased lung macrophage number after bleomycin in Sftpc+/+ mice but not Sftpc-/- mice supports our suggestion that the lack of effect of hAECs in mitigating injury in Sftpc-/- mice may be due to an inability of hAECs to modify macrophage behavior in that mouse strain. Our in vitro observation that hAEC conditioned media reduced migration of a macrophage cell line and lung macrophages derived from Sftpc+/+ mice but did not alter migration of macrophages from Sftpc-/- mice supports our suggestion that hAECs exert their protective effects via macrophage modulation. Further, in wild-type mice hAECs induced polarization of macrophages towards the alternatively activated phenotype whereas they did not alter macrophage phenotype in Sftpc-/- mice. Taken together, these in vitro and in vivo observations suggest that the macrophage of Sftpc-/- mice are not able to respond to hAECs in a manner that Sftpc-/- macrophages do and that this lack of response underlies, at least in part, the inability of hAECs to reduce bleomycin-induced lung injury. Specifically, we believe that the inability of hAECs to influence the Sftpc-/- macrophage phenotype leads to the continued presence of inflammatory signals for further extracellular matrix deposition, perpetuating the fibrotic process.
Of course, this study was not designed to explore how hAECs influence macrophage recruitment and function. Nonetheless, to guide future research it is useful to consider likely pathways. Alternatively, activated macrophages respond to stimuli such as IL-4 and IL-13, which are produced and released by the human placenta in significant amounts (4,10,45). In response to such stimuli, they release cytokines including TGF-β and IL-10 (21,32) to aid in the resolution of inflammation and wound healing. IL-4 and IL-13 also augment expression of IL-1 decoy receptor and the IL-1 receptor α-chain in vitro and in vivo, counteracting the proinflammatory actions of IL-1 (16,28). A recent paper by Zhang and colleagues reported that human gingival mesenchymal stem cells (MSCs) aided repair in a murine model of cutaneous wound healing (47) by encouraging the polarization of macrophages to the alternatively activated phenotype. Taken together with the results from the current study, we believe that hAECs may function via similar mechanisms, and as such depend on normally functioning macrophages for their injury prevention effects.
In summary, the findings from this study suggest a need for adequate macrophage recruitment and polarization in the modulation of bleomycin-induced lung injury by hAECs in immune competent mice. Further studies are needed to elucidate precisely the nature of the interaction between therapeutically transplanted cells and the host's immune system.
Footnotes
Acknowledgments
We would like to thank Professor Jeffrey A. Whitsett for providing us with colonies of Sftpc+/+ and Sftpc-/- mice, Ashley Mansell for providing us with the C57Bl6 macrophage cell line and L929 conditioned medium, and Rutu Acharya for technical assistance. This research was funded by a project grant from the National Health and Medical Research Council of Australia (NHMRC Project Grant 606483)and supported by the Victorian Government's Operational Infrastructure Support Program. The authors declare no conflicts of interest.
