Abstract
Chronic liver injury and inflammation lead to hepatic fibrosis, cirrhosis, and liver failure. Embryonic and mesenchymal stem cells have been shown to reduce experimental liver fibrosis but have potential limitations, including the formation of dysplastic precursors, tumors, and profibrogenic cells. Other stem-like cells may reduce hepatic inflammation and fibrosis without tumor and profibrogenic cell formation. To test this hypothesis we transplanted human amnion epithelial cells (hAEC), isolated from term delivered placenta, into immunocompetent C57/BL6 mice at week 2 of a 4-week regimen of carbon tetrachloride (CCl4) exposure to induce liver fibrosis. Two weeks following hAEC infusion, intact cells expressing the human-specific markers inner mitochondrial membrane protein and human leukocyte antigen-G were found in mouse liver without evidence of host rejection of the transplanted cells. Human albumin, known to be produced by hAEC, was detected in sera of hAEC-treated mice. Human DNA was detected in mouse liver and also spleen, lungs, and heart of some animals. Following hAEC transplantation, CCl4-treated animals showed decreased serum ALT levels and reduced hepatocyte apoptosis, compared to controls. hAEC-treated mouse liver had lower TNF-α and IL-6 protein levels and higher IL-10 compared to animals given CCl4 alone. Compared to CCl4 controls, hAEC-treated mice showed fewer activated collagen-producing hepatic stellate cells and less fibrosis area and collagen content. Reduced hepatic TGF-β levels in conjunction with a twofold increase in the active form of the collagen-degrading enzyme matrix metalloproteinase-2 in hAEC-treated mice compared to CCl4 controls may account for the reduction in fibrosis. hAEC transplantation into immunocompetent mice leads to cell engraftment, reduced hepatocyte apoptosis, and decreased hepatic inflammation and fibrosis.
Keywords
Introduction
The liver responds to persistent inflammation from multiple causes, including viral infection, alcohol excess, and hepatic steatosis, with a wound-healing, fibrogenic response that in some patients can lead to cirrhosis, portal hypertension, hepatocellular carcinoma, and liver failure. These serious disorders are associated with significant mortality risk and impose considerable financial burdens on health care systems worldwide. Liver transplantation is the only curative therapy for end-stage disease, but access to sufficient donors is problematic, the procedure costly, and life-long immune suppression is required.
Strategies to reduce hepatic fibrosis and avoid the need for organ transplantation have focused on hepatic stellate cells (HSC), the principal collagen-producing cell in the liver. Many antifibrotic therapies have been explored but none have become available yet for clinical use (35). Recent reports have shown that transplantation of embryonic stem cells (ESC) and adult hematopoietic and mesenchymal stem cells (MSC) into animals with experimental liver injury can result in transdifferentiation of these cells into hepatocytes and in reduced fibrosis (4,10,11,21,29,32), although not all authors have demonstrated positive results (8). Adverse outcomes, such as tumor formation by ESC-derived hepatocytes (4) and fusion of MSC with host cells to form dysplastic precursors, have been observed (37). In addition, there is considerable evidence that bone marrow is a source of profibrogenic cells in the liver and that bone marrow-derived MSC transdifferentiate into cells capable of supporting hepatic fibrogenesis (13).
Alternatively, there are stem-like cells that may overcome the disadvantages of ESC and MSC. We and others have shown that human amnion epithelial cells (hAEC) display features of pluripotent/multipotent stem cells and can transdifferentiate into several lineages derived from each of the primary germ layers, including hepatocytes (14,20). The cells are plentiful; in excess of 100 million hAEC can be obtained noninvasively from the amnion membrane of term delivered placenta, a hugely abundant and noncontentious source of stem cells for potential cellular therapies. Interestingly, primary undifferentiated hAEC produce a number of hepatocyte specific proteins, including serum albumin (24,30), and can engraft in the murine liver as demonstrated in a study exploring the use of hAEC as a transgene carrier for the treatment of inherited liver diseases (27).
Importantly, unlike ESC, hAEC do not form teratomas or tumors following xenotransplantation (14,20). These cells have also been successfully transplanted into allogeneic recipients because they appear to lack key allo-recognition molecules that would elicit host immune responses (15). Further, anti-inflammatory properties have been attributed to hAEC and amnion membrane and employed clinically as topical therapy in ophthalmic and dermal applications to promote wound healing and reduce scarring (31). A recent study investigating the effects of a mixed cell population consisting of hAEC and amnion-derived MSC reported a reduction in fibrosis in lungs of bleomycin-injured mice (6). However, whether the anti-inflammatory properties of hAEC can be exploited following cell transplantation to reduce fibrosis remains largely unknown. In the present study, we transplanted hAEC into immune competent C57/BL6 mice with carbon tetrachloride (CCl4)-induced hepatic fibrosis and show that hAEC xenografts survive, continue to secrete albumin, and reduce hepatic inflammation and fibrosis.
Materials and Methods
Human Amnion Tissue
Amnion membranes were obtained from healthy women with a normal singleton pregnancy undergoing cesarean section at term (37–40 weeks gestation; n = 16). Informed written consent was obtained from each woman. The study was approved by the Southern Health Human Research Ethics Committee.
Animals
Six-week-old male C57/BL6 mice were purchased from Monash Animal Services, Melbourne, Australia. The study was approved by the Animal Ethics Committee, Monash University. Animals received humane care under the Australian code of practice for the care and use of animals for scientific purposes.
Cell Isolation
hAEC were isolated from amnion membranes and their purity assessed as described previously (14). Briefly, the tissue was digested twice in 0.25% trypsin/EDTA solution (Gibco, Grand Island, NY) for 20 min with agitation at 37°C. Trypsin was inactivated with fetal calf serum (Invitrogen, Carlsbad, CA) and dispersed cells washed in DMEM-F12 medium (Gibco). Batches >99% positive for the epithelial markers cytokeratin-7, 8/18 (Dako, Glostrup, Denmark) by flow cytometry (14) were used in the experiments described below.
Induction of Hepatic Fibrosis and Cell Transplantation
Mice were given intraperitoneal injections with CCl4 (1 μl/g body weight diluted 1:10 in olive oil) twice weekly for 4 weeks. CCl4 administration induces necroinflammatory liver injury that reproducibly causes hepatic fibrosis. Midway through the CCl4 treatment, animals received hAEC (2 × 106 in 250 μl saline) via the tail vein. Previous dose-response experiments using 1–4 × 106 cells showed that 2 × 106 was the optimal cell dose for infusion (data not shown). Control groups consisted of mice given CCl4 only, CCl4 + saline (vehicle control), and healthy mice that did not undergo any treatment. Each cohort consisted of 8–10 mice. Animals were culled 4 weeks after commencing the experiment and blood and liver tissue collected. Spleen, heart, and lungs were also collected from mice given hAEC.
Detection of Human Cells by DNA PCR
To determine if hAEC were present in murine organs, DNA was extracted from liver, lung, spleen, and heart tissue using phenol chloroform and amplified by PCR using primers specific for human Alu repeat sequences (9). DNA extracted from healthy mouse liver served as a control.
Detection of Human Serum Albumin
Albumin secreted by primary, undifferentiated hAEC has been measured in sera of SCID mice harboring amnion tissue transplants (27). As an indirect measure of hAEC survival and functionality, sera collected from C57/BL6 mice (hAEC treated and controls; n = 10/group) were tested for human serum albumin using an ELISA specific for the human protein (Bethyl Laboratories, Montgomery, TX), following the manufacturer's instructions.
Measurement of Alanine Aminotransferase (ALT) and Total Bilirubin
Serum ALT and bilirubin were measured by Southern Cross Pathology, Monash Medical Centre using reagents and calibrators from Beckmann-Coulter (Sydney, Australia).
Hepatic Cytokine Concentrations
Liver tissue was homogenized in 50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100, 0.5% Tween-20, 0.1% SDS, 1 mM EDTA, and protease inhibitor cocktail (Roche), sonicated, and centrifuged at 14,000 × g for 15 min to remove debris. Interleukin (IL)-6, IL-10, tumor necrosis factor-α (TNF-α), and monocyte chemoattractant protein (MCP)-1 were measured by the Cytometric Bead Array (BD Biosciences, San Jose, CA) and IL-1 and transforming growth factor-β (TGF-β) by ELISA (R&D Systems, Minneapolis, MN). Total protein content in tissue lysates was determined by the BCA assay (Thermo Scientific, Rockford, IL) and cytokine concentrations normalized against the total protein content.
Immunohistochemistry
hAEC present in the liver were identified using human inner mitochondrial membrane (IMM) protein and human leukocyte antigen (HLA)-G. For IMM, 4% paraformaldehyde-fixed paraffin-embedded liver tissue sections 5 μm in thickness were dewaxed, rehydrated, and antigen retrieval carried out by boiling the sections in citrate buffer. Endogenous peroxidase activity was quenched in 0.6% H2O2 for 15 min. HLA-G immunohistochemistry was performed on OCT-embedded frozen liver tissue sections, 4 μm in thickness, fixed in 2% paraformaldehyde for 10 min. Protein block (Dako, Glostrup, Denmark) was applied for 15 min followed by IMM (1:100, Chemicon, Boronia, Australia) or HLA-G antibodies (1:50, BD Biosciences) and incubated at 37°C for 1 h. Human term fetal membranes served as a positive control. Isotype-matched mouse IgG was applied to sections serving as negative controls. Sections were then incubated with biotinylated goat anti-mouse secondary antibodies followed by Envision kit reagents (Dako). Antibody binding was detected using DAB chromogen (Sigma-Aldrich, St. Louis, MO).
To identify apoptotic hepatocytes, caspase-3 staining was carried out. Paraffin-embedded liver tissue sections were dewaxed, rehydrated, and antigens retrieved by boiling in citrate buffer. Following incubation in 0.6% H2O2 and protein block solution, sections were incubated overnight at 4°C with 1:100 diluted caspase-3 antibody (Promega, Madison, WI). After several washes sections were incubated sequentially with biotinylated goat anti-mouse secondary antibodies, ABC kit reagents (Vector Laboratories Burlinghame, CA); antibody binding was detected with DAB.
To localize α-smooth muscle actin (α-SMA), present in activated HSC, liver tissue sections were placed in an autostainer (Dako) and incubated sequentially with: peroxidase block (Dako), CAS protein block (Zymed, San Francisco, CA), 1:5000 diluted α-SMA antibody (Sigma-Aldrich), 1:300 diluted rabbit anti-mouse IgG2a (Invitrogen), ABC reagents (Vector Laboratories), and DAB (Dako). Incubations were carried out at room temperature for 15 min except primary antibody (30 min) and DAB (5 min). Slides were counterstained with hematoxylin, dehydrated, and mounted in DPX.
Picro Sirius Red Staining
Deparaffinized liver tissue sections were stained with Picro Sirius Red to identify the extent of histological fibrosis. Sections were incubated for 90 min in Picro Sirius Red stain, washed in acetic acid and water (5:1000), dehydrated in ethanol, and mounted in DPX. Fibrosis area was measured by computer-assisted morphometry. Fifteen consecutive nonoverlapping fields were acquired from each mouse liver; the images were digitized and fibrosis area analyzed by Scion Image for Windows (vAlpha 4.0.3.2, Scion Corporation, Frederick, MD).
Hydroxyproline Assay
Hepatic collagen content was measured by the hydroxyproline assay, as described previously (25), with minor modifications. Briefly, liver tissue was hydrolyzed in 6N HCl and absorbance measured at 558 nm. The hydroxyproline content was calculated from a standard curve prepared with trans-4-hydroxy-L-proline (Sigma-Aldrich), data were multiplied by a factor of 6.94 to convert the hydroxyproline measurement to collagen content and expressed per milligram of wet tissue weight.
Gelatin Zymography
Matrix metalloproteinases (MMPs) were extracted from mouse liver tissue (n = 4/group). MMP-2 and MMP-9 activity was assessed by gelatin zymography as described previously (36). Densitometry of the active MMP bands was performed using a GS-710 Calibrated Imaging Densitometer (Bio-Rad, Hercules, CA) and Quantity-One software (Bio-Rad).
Statistical Analysis
Data were expressed as mean ± SEM. Statistical significance of planned comparisons was determined by one-way ANOVA with the Newman-Keuls post test for multiple comparisons (GraphPad Prism 5.0b for Mac OS X, GraphPad Software, San Diego, CA). Values of p < 0.05 were considered statistically significant.
Results
hAEC Engraft in CCl4-Injured Liver
We and others have shown that hAEC express pluripotent and some mesenchymal stem cell markers including Oct-4, Sox-2, Nanog, SSEA-4, Tra-1-60, Tra-1-80, GCTM2, CD29, CD73, and CD166, have low levels of HLA class IA antigens, high levels of the class IB antigen HLA-G, and lack HLA class II molecules. hAEC are negative for the hematopoietic lineage markers CD31, CD34, and monocytic marker CD45 (14,20,24,26). These cells were injected intravenously into immune competent C57/BL6 mice 2 weeks after exposure to CCl4 and culled 2 weeks later. Intact IMM and HLA-G-positive cells were detected in liver sections from 8 of 10 mice tested and were observed around portal tracts and spreading into surrounding hepatic lobules (Fig. 1A). Due to limitations in IMM antibody binding to paraffin sections and HLA-G to frozen sections, colocalization using serial sections could not be carried out. hAEC lining amnion of term fetal membranes were positive for IMM and HLA-G. Liver tissue sections from mice receiving CCl4 only and those incubated with mouse IgG lacked IMM and HLA-G staining.

hAEC engraft in the injured liver. Cells positive for human specific IMM and HLA-G (arrows) were found in portal triads and adjacent parenchyma. HLA-G and IMM staining of hAEC lining human fetal membranes is also shown. Sections from mice given CCl4 only and those incubated with isotype-matched IgG lacked staining. Scale bars: 50 μm (A). Murine sera collected 2 weeks after hAEC transplantation contained human albumin providing further evidence of hAEC engraftment and survival (B).
DNA PCR for human Alu repeat sequences was carried out to detect human cells in liver of CCl4-treated mice. Human DNA was detected in mouse liver of 4 of 5 hAEC-treated animals. In order to determine if hAEC had engrafted in other organs, spleen, heart, and lung tissues were also examined. Human DNA was detected in spleen and lungs of 3 of 5 mice and heart tissue of 1of 5 mice tested.
As an indirect measure of the presence and functionality of hAEC, we also tested for human albumin 2 weeks post-cell transplantation. Human albumin was detected in murine sera from all CCl4 + hAEC-treated animals and was absent in sera of mice treated with CCl4 or saline vehicle, confirming the specificity of the ELISA (Fig. 1B).
hAEC Transplantation Reduces Serum ALT
Elevations in bilirubin and ALT generally reflect hepatocyte injury and these proteins increased significantly in animals given CCl4 compared to healthy controls (p < 0.05 and p < 0.001, respectively) (Fig. 2). Serum ALT was significantly lower in mice injected with hAEC compared to CCl4 alone (p < 0.01). Although serum bilirubin was lower in mice receiving hAEC compared to CCl4 alone, this reduction was not statistically significant.
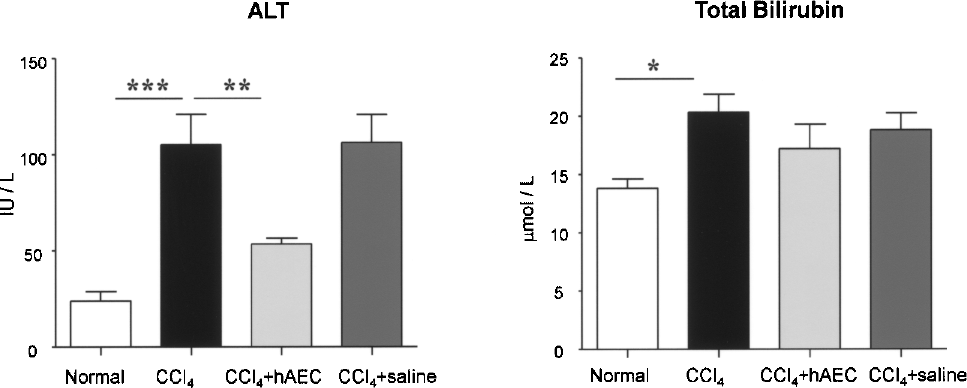
hAEC transplantation reduces liver injury. Alanine aminotransferase (ALT) and total bilirubin were measured in sera of C57/BL6 mice given cells or saline plus CCl4, healthy mice, and those given CCl4 alone. Bilirubin and ALT were elevated in CCl4-treated animals compared to normal mice (*p < 0.05 and ***p < 0.001, respectively). Mice transplanted with hAEC had reduced serum ALT compared those given CCl4 alone (**p < 0.01).
Hepatocyte Apoptosis Is Reduced with hAEC Transplantation
Engulfment of apoptotic hepatocytes by Kupffer cells and HSC augments hepatic inflammation and fibrosis so we asked whether reduced hepatocyte apoptosis could contribute to the reduction in inflammatory indices such as serum ALT, an outcome demonstrated in other forms of experimental liver injury (18). Caspase-3-positive cells were significantly more frequent in CCl4-treated mice compared to healthy animals (p < 0.001) (Fig. 3A. Mice given CCl4 + hAEC showed significantly fewer apoptotic hepatocytes (p < 0.05). Staining of serial sections showed the presence of IMM-positive hAEC in these regions (Fig. 3B). We also investigated whether hAEC transplantation induced hepatocyte proliferation. While PCNA-positive cells were present throughout hepatic lobules and portal tracts, significant differences were absent among treatment groups (data not shown).

Hepatocyte apoptosis is reduced with hAEC transplantation. Caspase-3 immunohistochemistry in liver tissue sections shows increased numbers of immunopositive nuclei in CCl4-treated animals. The number of apoptotic hepatocytes declined significantly in mice treated with hAEC (*p < 0.05; A). Serial sections showed that areas with IMM-positive hAEC had few apoptotic hepatocytes (B). Scale bars: 50 μm.
hAEC Decrease Hepatic Inflammation
Given the reduction in hepatocyte apoptosis, we then asked whether there might also be hAEC-associated changes in pro- and anti-inflammatory cytokines. Liver tissue lysates from healthy mice and animals given CCl4 with or without hAEC were analyzed using a flow cytometry-based assay or ELISA. Mice transplanted with hAEC had significantly reduced IL-6 and TNF-α and increased IL-10 hepatic protein content compared with mice given CCl4 alone (p < 0.01, p < 0.0001, and p < 0.01, respectively) (Fig. 4). IL-1 and MCP-1 protein levels did not alter with hAEC treatment.
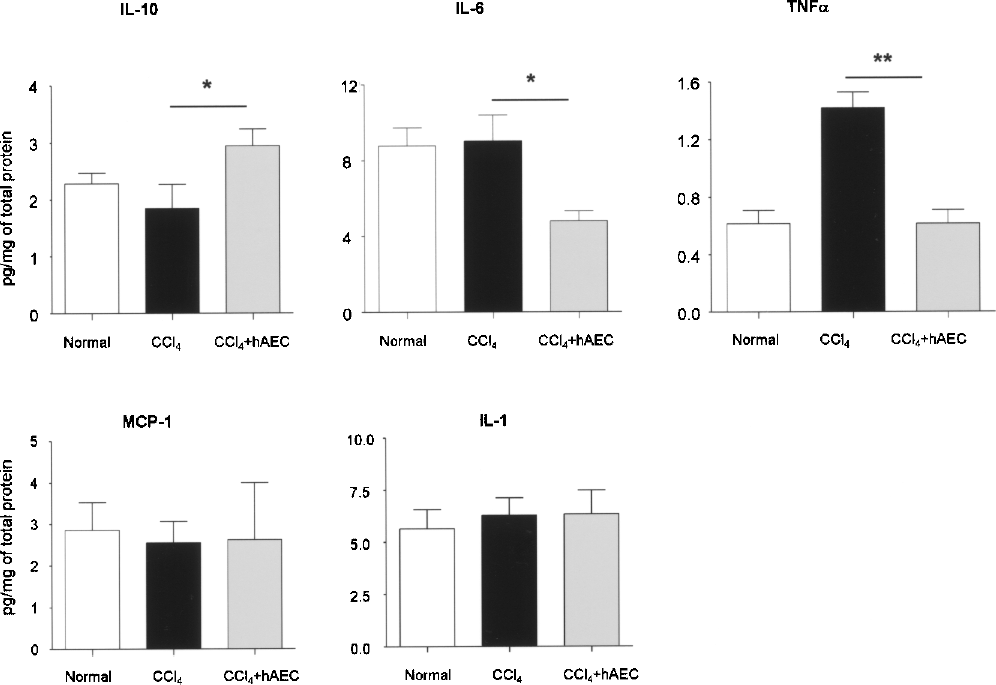
Hepatic cytokine levels are altered with hAEC transplantation. Cytokine concentrations in hepatic tissue lysates were determined through a multiplex bead array or ELISA (IL-1). Compared to mice exposed to CCl4, IL-6 and TNF-α were lower in animals treated with CCl4 + hAEC (*p < 0.01, **p < 0.0001). IL-10 increased in livers of hAEC-treated mice (*p < 0.01). MCP-1 and IL-1 remained unaltered.
hAEC Reduce HSC Activation
Activated HSC and Kupffer cells release inflammatory cytokines including TNF-α and IL-6. Because hAEC transplantation led to a reduction in TNF-α and IL-6, we investigated whether HSC activation was similarly reduced. As expected, the number of α-SMA-positive cells was significantly elevated in CCl4-treated mice compared with healthy controls (p < 0.001) (Fig. 5). CCl4-treated mice given hAEC demonstrated significantly fewer α-SMA-positive cells compared with CCl4 alone (p < 0.001). Using serial sections, IMM-positive hAEC were found in close proximity to residual α-SMA-stained cells (Fig. 5B).
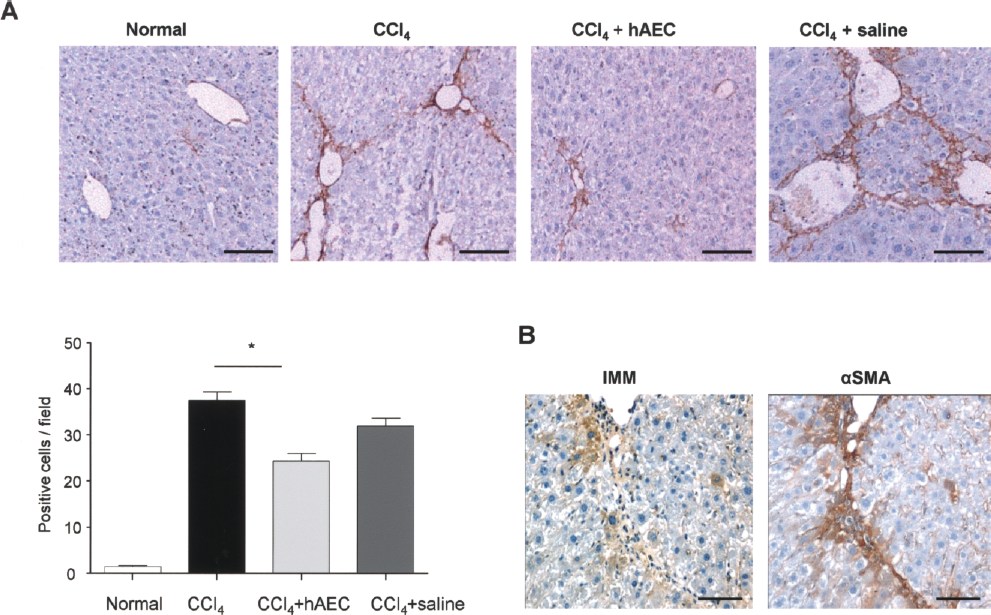
Hepatic stellate cell activation declines with hAEC transplantation. α-Smooth muscle actin (α-SMA) immunohistochemistry was performed on liver tissue sections and the number of immunopositive cells showed that activated HSC declined in mice injected with CCl4 + hAEC compared to CCl4 alone (*p < 0.001; A). Serial sections showed IMM-positive hAEC in close proximity to α-SMA-positive cells (B). Scale bars: 50 μm.
hAEC Reduce Hepatic Fibrosis Area and TGF-β Content
Because HSC are the principal collagen-producing cells in the liver, we next examined whether reduced HSC activation was associated with reduced hepatic fibrosis demonstrated by Sirius red staining and quantitation by computer-assisted morphometry. After 4 weeks of CCl4 treatment, fibrotic bands were clearly evident by Sirius red staining and fibrosis area was significantly increased in CCl4-treated animals compared to untreated controls (p < 0.001) (Fig. 6A). However, mice given hAEC showed significantly less fibrosis area compared with CCl4 alone (p < 0.001). IMM-positive cells were found adjacent to some of the fibrotic bands that were still evident in hAEC-treated mice (Fig. 6B).

hAEC transplantation reduces hepatic fibrosis. Fibrosis in liver tissue sections taken from C57/BL6 mice was visualized by Sirius red dye staining and fibrosis area quantified by computer morphometry. Animals treated with hAEC had reduced fibrosis area (*p < 0.001; A). Staining of serial sections showed IMM-positive hAEC closely associated with Sirius red staining in mice treated with cells (B). Scale bars: 50 μm.
We also assessed whether the histological changes seen in fibrosis area were reflected in the hepatic collagen content. Hepatic collagen concentration, measured by the hydroxyproline assay, was significantly elevated in CCl4-treated mice compared to healthy controls (p < 0.001) (Fig. 7). hAEC-treated animals showed significantly lower hepatic collagen content compared with CCl4 alone (p < 0.05).

hAEC treatment reduces hepatic collagen content and TGF-β levels and increases gelatinase activity. Hepatic collagen content was measured by the hydroxyproline assay and TGF-β by ELISA (A, B). Collagen and TGF-β content were lower in CCl4-treated mice transplanted with hAEC compared to CCl4 alone. Densitometric analysis of gelatin zymograms showed that active MMP-2 increased in mice given hAEC compared with CCl4-treated mice (C). (*p < 0.05, **p < 0.01).
To explore mechanisms that could lead to a reduction in HSC activation, we measured the profibrotic cytokine TGF-β in hepatic tissue lysates by ELISA normalized by protein content. TGF-β content increased significantly in response to CCl4 compared to normal controls (p < 0.001) but was significantly lower in mice treated with CCl4 + hAEC (p < 0.01) (Fig. 7B). We also measured changes in enzymes that alter extracellular matrix composition by degrading collagen. Active MMP-2 increased twofold in mice treated with hAEC compared to mice given CCl4 alone (p < 0.05) (Fig. 7C). Active MMP-9, however, was not significantly altered with hAEC treatment, compared to that measured in the CCl4 group alone (Fig. 7C).
Discussion
In this study we have shown that hAEC engraft in livers of immunocompetent mice and reduce CCl4-induced hepatic fibrosis. hAEC engraftment was demonstrated by the presence of intact human IMM and HLA-G-positive cells 2 weeks posttransplantation while the presence of human albumin, a protein known to be secreted by the transplanted cells (24,30), in mouse serum suggests that the hAEC remain viable. Analysis for human DNA in several murine organs showed that the intravenously transplanted hAEC traffic primarily to the injured liver. Consistent with these findings, Bailo et al. reported the presence of human DNA in swine and rodent organs several weeks after the transplantation of hAEC together with human amnion MSC (2). These cells were also found in lungs of bleomycin-injured C57/BL6 mice, 2 weeks post-cell transplantation through cytokeratin-19 immunohistochemistry to identify the human cells (6). Although factors regulating the migration and engraftment of the transplanted hAEC remain unknown, chemokines, adhesion molecules, and liver-specific factors such as sphingosine 1-phosphate may play a role (19).
We did not observe any histological evidence of host rejection of transplanted hAEC, which were present in portal tracts and adjacent parenchyma without the neutrophil or lymphocyte infiltration typical of immune-mediated rejection. Similar observations have been made following transplantation of hAEC into bonnet monkeys with spinal cord injury (28). This may be due to factors such as low levels of expression of HLA class IA and II molecules and costimulatory molecules (3,14) or active inhibition of host T-cell proliferation (2,34). hAEC are known to express HLA-G, which serves not only as a marker to follow the fate of the transplanted cells but is also likely to play an important role in subverting the host immune response to these cells. HLA-G is a non-classical, nonpolymorphic class I molecule that was first described in choriocarcinoma cells (7) and is expressed in fetal–placental cells that invade the maternal uterus and thus may modulate the maternal immune response against the fetus (17). In addition to inhibiting proliferation of alloreactive T cells (1), HLA-G may also constrain host immune responses by stimulating IL-10 production by regulatory T cells (22). In this context it is interesting to note that we found increased IL-10 protein levels in hepatic tissue lysates from hAEC-treated mice.
Multipotent stem cells can also be harvested from human umbilical cord. Recently, human umbilical cord MSC (hUMSC) have been shown to decrease CCl4-induced hepatic fibrosis in rodent models. Jung et al. (16) derived hUMSC from cord blood samples and showed a decrease in markers of hepatic fibrogenesis that was most prominent 4 weeks following tail vein infusion. Tsai et al. (33) injected hUMSC obtained from Wharton's jelly directly into the liver and showed decreased hepatic collagen and TGF-β content in treated rats almost to control levels. Reduced hepatic inflammation and release of bioactive factors and cytokines in association with Wharton's jelly cell transplantation were implicated in fibrosis reduction. However, while engraftment of a large number of Wharton's jelly MSC and fibrosis reduction was demonstrated with this technique, the authors noted that direct hepatic injection may be complicated by postprocedural bleeding that could potentially impair fibrosis reduction by inducing platelet-derived growth factor leading to HSC activation.
The antifibrotic properties of hAEC have not been previously investigated in experimental liver fibrosis. In chronic liver injury, activation of HSC by TGF-β, the prototypical fibrogenic cytokine, leads to production of collagen and other extracellular matrix components. We found that hAEC infusion in CCl4-treated immunocompetent mice was associated with a significant decrease in hepatic TGF-β content with fewer α-SMA-positive cells, suggesting decreased HSC activation. In addition, CCl4-treated mice showed less histological fibrosis and lower hepatic collagen content following hAEC infusion compared to controls given CCl4 alone. The development of this fibrolytic phenotype may occur through reduction of proinflammatory signals that activate HSC and through the increased activity of collagen degrading enzymes following hAEC transplantation.
CCl4-treated mice infused with hAEC showed lower hepatic tissue levels of the proinflammatory cytokines IL-6 and TNF-α with increased expression of the anti-inflammatory cytokine IL-10. This antifibrotic cytokine profile may reflect a direct interaction between hAEC and HSC in which hAEC-derived factors regulate HSC activation. In support of this hypothesis, a recent study showed that IL-6 expression by cultured HSC induced MSC secretion of IL-10 leading to decreased HSC collagen production (23). Alternatively, hAEC may alter profibrogenic activation signals for Kupffer cells and HSC by reducing hepatocyte apoptosis. In the process of removing damaged cells, engulfment of apoptotic hepatocytes by Kupffer cells and HSC leads to their activation with increased TGF-β expression, TNF-α secretion, and death-receptor expression (5) and can lead to liver fibrosis (38). Prevention of hepatocyte apoptosis by inhibiting death-receptor-mediated pathways can prevent experimental liver fibrosis (40). In this context, we found that CCl4-treated mice infused with hAEC showed significantly fewer apoptotic hepatocytes than animals given CCl4 alone. Thus, inhibition of hepatocyte apoptosis, which is a potent stimulus for hepatic fibrogenesis, may be an additional mechanism leading to hAEC-induced fibrosis reduction.
Increased expression of collagen-degrading enzymes such as matrix metalloproteinases is thought to be an important mechanism underlying fibrosis regression (12). Our finding that hAEC infusion was associated with a twofold increase in MMP-2 activity compared to that seen in mice treated with CCl4 alone supports the development of a hAEC-associated fibrolytic state and is consistent with increased MMP-2 activity identified by others during fibrosis regression in CCl4-treated rats (39). The cellular source of MMP-2 during fibrosis regression is unclear because HSC secrete MMP-2 yet HSC apoptosis is an important mechanism of fibrosis resolution (39), and we identified a significant decrease in activated HSC following hAEC transfusion. In our study, MMP-2 expression was significantly greater in hAEC-treated animals than controls, suggesting a specific effect of this cell population, although whether as the primary source or through interaction with other resident liver cells remains unclear.
In conclusion, we found that hAEC transplantation effectively reduces experimental hepatic fibrosis by reducing proinflammatory and profibrotic cytokines and inducing a collagen-degrading phenotype without eliciting host immune rejection of the xenotransplanted cells. hAEC are readily available, highly abundant and non-controversial stem-like cells that may represent a potential new therapeutic approach for the treatment of hepatic fibrosis and cirrhosis. We are currently testing this hypothesis in a long-term model of CCl4 exposure with a multiple hAEC dosing regimen.
Footnotes
Acknowledgments
We thank E. M. Wallace for providing amnion tissue, M. Hickey for helpful discussions, and Chongxin Zhao for assistance with gelatin zymography. U.M. is supported by Project Grants #436836 and #491145 from the Australian National Health and Medical Research Council (NHMRC). This study was funded by NHMRC project grant #606473 to U.M. and W.S. C.S.S. is supported by a National Heart Foundation of Australia/NHMRC RD Wright Fellowship (#454636). E.D.W. is supported by a NHMRC Biomedical Career Development Award (#519539).
