Abstract
Human amnion epithelial cells (hAECs) have attracted recent attention as a promising source of cells for regenerative therapies, with reports that cells derived from human term amnion possess multipotent differentiation ability, low immunogenicity, and anti-inflammatory properties. Specifically, in animal models of lung disease characterized by significant loss of lung tissue secondary to chronic inflammation and fibrosis, the transplantation of hAECs has been shown to reduce both inflammation and subsequent fibrosis. To further explore the mechanisms by which hAECs reduce pulmonary fibrosis and enhance lung regeneration, we utilized a bleomycin-induced model of pulmonary fibrosis and investigated the ability of hAECs to reduce fibrosis and thereby improve pulmonary function. We aimed to determine if hAECs, injected into the peritoneal cavity could migrate to the lung, engraft, and form functional lung epithelium, and whether hAECs could modulate the inflammatory environment in the bleomycin-injured lung. We demonstrated that, compared to bleomycin alone, IP administration of hAECs 24 h after bleomcyin, decreased gene expression of the proinflammatory cytokines TNF-α, TGF-β, IFN-γ, and IL-6 and decreased subsequent pulmonary fibrosis with less pulmonary collagen deposition, reduced levels of α-smooth muscle actin and decreased inflammatory cell infiltrate. We also showed that hAECs are able to prevent a decline in pulmonary function associated with bleomycin-induced lung damage. We were unable to detect any significant engraftment of hAECs in injured, or uninjured, lung after administration. The findings from this study support the further investigation of hAECs as a potential cell therapy for inflammatory and fibrogenic diseases.
Introduction
Respiratory disease is a leading cause of morbidity and mortality. Mainly caused by tobacco smoking, occupational irritant exposure, and pollution, the disease burden of respiratory disease continues to grow, and is projected to become the third most common cause of death worldwide by the World Health Organization (47). While the causes of respiratory diseases differ, the end resultant organ damage is similar—chronic inflammation, fibrosis, and scarring—leading to loss of functional lung tissue (8,43,45). Current treatments, such as oral corticosteroids, have only limited effectiveness and are not without significant long-term adverse effects, including cardiac complications, glucose intolerance, and adrenal suppression (11,21,23,42,48). Hence, there is an increasingly urgent need for the development of novel effective treatments of lung disease, including cell- and gene-based therapies (7,10).
In this regard, the use of cells isolated from human term placenta for regenerative medicine holds great promise (9). Recently, human amnion epithelial cells (hAECs) have attracted attention as a potential source of cells for regenerative therapies (37), with reports that epithelial cells derived from human term amnion possess multipotent differentiation ability (26), low immunogenicity (5), and anti-inflammatory functions (30). Importantly, while hAECs have similar properties to embryonic stem cells, they are not sourced from human embryos but rather from term placentae that are usually discarded following birth. With a view to future applications, we have recently demonstrated that hAECs can be collected, isolated, and stored in a manner suitable for clinical use (36).
We have previously demonstrated that freshly isolated hAECs express the thyroid transcription factor Nkx2.1 (also known as TTF-1) (26). This transcription factor is one of the earliest lineage-specific markers of the developing lung and is involved in the regulation of branching morphogenesis. Further, lung epithelial-specific gene expression can be induced in primary hAECs following culture in small airway growth media (SAGM) (36)—a media developed to maintain primary culture of small airway cells to induce differentiation of ESCs into type II alveolar epithelial cells (1).
Supporting these in vitro observations, Carraro and colleagues have recently demonstrated that a mixed population of cells derived from amniotic fluid, including but not exclusively hAECs, were able to differentiate in vivo into distinct lung cell types, dependent on the nature of lung injury induced (14). In addition, the acute administration of a mixture of hAECs and human amnion/chorion-derived mesenchymal stem cells into mice with bleomycin-induced pulmonary fibrosis has been shown to decrease both inflammatory cell infiltration and fibrosis (13). Most recently, we have shown that the delayed administration of hAECs to mice with established bleomycin-induced lung fibrosis significantly reduced that fibrosis and facilitated the regeneration of normal lung architecture (35). Thus, it appears that hAECs may be able to either prevent lung damage and/or enhance lung repair following damage via one or both of two possible mechanisms. First, the ability of hAECs to differentiate into lung epithelial-like cells both in vitro and in vivo suggests that when these cells migrate to areas of alveolar injury and incorporate into the host lung, they directly contribute to its functional restoration. Second, the ability of hAECs to modulate immune responses both in vitro (25) and in vivo (25,46) is consistent with them being able to modify the host responses to tissue injury. We undertook this current study to further explore the mechanisms by which hAECs may afford repair of pulmonary fibrosis and to assess whether hAECs were able to improve lung function in a model of lung injury. Specifically, using the bleomycin-induced model of pulmonary fibrosis in immune competent mice, we aimed to determine if hAECs, administered by IP injection, could 1) migrate to the lung, incorporate into the host tissue, and form functional lung epithelium; 2) modulate the lung inflammatory response to bleomycin injury; and 3) rescue bleomycin-induced pulmonary dysfunction.
Materials and Methods
Animals and Experimental Groups
All experimental procedures were approved by Monash University School of Biomedical Sciences Animal Ethics Committee and were conducted in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes. A total of 62, 6-week-old female C57BL/6 mice weighing 16–18 g were used in this study. Of these, 12 mice were used solely for flow cytometry (see below). The remaining 50 mice were randomly divided into four treatment groups: Saline—mice received an intranasal (IN) instillation of saline and then an IP injection of saline 24 h later (n = 10); Bleomycin—mice received an IN instillation of bleomycin and then an IP injection of saline 24 h later (n = 14); Saline + hAECs—mice received an IN instillation of saline and then an IP injection of 4 million hAECs 24 h later (n = 12); Bleomycin + hAECs—mice received an IN instillation of bleomycin and then an IP injection of 4 million hAECs 24 h later (n = 14). Mice were culled either 7 or 14 days following IN bleomycin or saline instillation, divided equally between time points. Body weight, core temperature, and basal lung function were recorded prior to commencement of experiments and at day 3, 7, and 14.
Bleomycin Induction of Pulmonary Fibrosis
Intranasal administration of bleomycin to rodents is considered to reproduce the histological changes observed in human pulmonary fibrosis (44). Mice were anesthetised by IP injection of xylazine (2 mg/kg) and ketamine (100 mg/kg), allowing administration of an IN dose of bleomycin (8 U/kg) in a total volume of 21–25 μl saline. This dose was shown to be optimal to induce pulmonary fibrosis without animal mortality as determined by dose titration study (data not shown). Animals in the saline + hAEC-treated groups received 21–25 μl IN saline alone.
hAEC Isolation, Labeling, and Injection
Isolation of hAECs suitable for clinical use was performed using good manufacturing practice (GMP) processes, as previously described (36). Prior to IP injection, hAECs were labeled with carboxyfluorescein succinimidyl ester (CFSE). Briefly, hAECs were resuspended at 15 million cells/ml in 5 μM CFSE and incubated at 37°C for 10 min. To prevent CFSE toxicity, hAECs were then washed three times in PBS and resuspended at 10 million cells/ml in sterile saline. Cell viability was assessed by trypan blue exclusion.
Lung Function Testing
Lung function was assessed using unrestrained whole body plethysmography, as previously described (33), at days 0, 3, 7, and 14 for each group of animals. Briefly, the plethysmograph is a single cylindrical Perspex chamber (150 × 50 mm) fitted with a temperature and relative humidity probe (Vaisala, Finland) and a volumetric pressure transducer (Grass Instrument Co., USA). Change in pressure caused by the tidal movement of gas within the chamber (as the mouse breathes) is measured by the pressure transducer, amplified (Octal Bridge Amp; Powerlab, AD Instruments, Australia), and recorded (Chart 5.1, AD Instruments, Australia) using a standard desktop computer. The pulmonary function of each mouse was recorded while the animal was stationary and calm for an average of 2 min. Waveform analysis was used to analyze the respiratory trace obtained from each animal, including the measurement of the change in pressure caused by the tidal movement of gas within the chamber (PT), respiration rate (breaths/min), total breathing cycle time (Ttot; s), inspiration time (Ti; s), and expiration time (Te; s). Tidal volume (<VT; ml), was calculated as described by Drorbaugh and Fenn (19). Minute volume (VE; ml/min), inspiratory duty cycle (Ti/Ttot;%), inspiratory flow rate (VT/Ti; ml/s), and the ratio of inspiration time to expiration time (Ti/Te ratio) were also calculated.
Tissue Collection
Animals were culled by an IP injection of 150 mg/kg Lethobarb (Virbac, Australia). The right lung was ligated at the right mainstem bronchus, excised, weighed, and processed either for RNA (middle lobe), protein (inferior lobe), or for the measurement of hydroxyproline (superior lobe). Upon excising the right lung, the trachea was then exposed and cannulated, allowing the left lung to be instilled with 4% paraformaldehyde (PFA). The left lung was then excised, immersed in 4% PFA for 48 h, and processed for histology. Flow cytometric analysis for CFSE-labeled hAECs was performed on whole lungs (right and left) from 12 animals (Saline, n = 2; Bleomycin, n = 2; Saline + hAECs, n = 4; Bleomycin + hAECs, n = 4). No other outcomes were collected from these 12 animals.
Ashcroft Score of Fibrosis and Semiquantitative Morphological Index
Histological sections from throughout each left lung were cut and 20–25 sequential fields of view (200× magnification) were independently scored in triplicate by three of the authors (S.M., R.L., R.A.), each blinded to treatment group, for the severity of parenchymal lesions induced by bleomycin treatment according to the semiquantitative method previously described by Ashcroft (4). An overall mean (±SEM) score was calculated from the triplicate scores from all three scorers.
Hydroxyproline Assay and Sirius Red for Collagen
Collagen content was assessed by the measurement of hydroxyproline (OHPRO) and by Sirius red immunostaining. Briefly, OHPRO was measured by an established in-house assay. For each animal, the superior lobe of the right lung was incubated at 110°C for 16 h in glass pyrex tubes containing 2.5 ml 6 M HCl. Samples were allowed to cool and vortexed to ensure complete acidification of tissue. Hydrolysates were diluted 1:1 with ddH2O and centrifuged at 5000 × g for 10 min. A 1-ml aliquot of each sample was removed and neutralized to pH 7.0 by addition of 10 M NaOH and made up to a total volume of 2 ml with ddH2O. To measure OHPRO concentration, 100 μl of each sample added to a chloramine T solution [7% (w/v) chloramine-T in dH2O with 4 parts acetate/citrate buffer pH 6.0] and incubated for 5 min at room temperature. Diaminobenzidine (DAB) was used for color formation and the absorbance was read at 558 nm (Spectramax Plus, Molecular Devices, USA). Hydroxyproline concentration was expressed as a proportion of wet lung weight (jig hydroxyproline/mg wet weight).
Collagen staining was performed using Sirius Red as previously described (27), with minor modifications. Specifically, following Sirius red staining, collagen was quantified by digital image analysis using the Image J software package (National Institutes of Health). For each tissue sample, five images were captured using brightfield light microscopy at 200× magnification using a light microscope (AxioSkop, Zeiss, Germany) with a camera attachment (AxioCam ICc 3, Zeiss, Germany). The percentage of area positive for Sirius red was calculated as previously described (39). Briefly, each image was converted to a 16-bit black-and-white image and threshold values adjusted to include only stained areas. Threshold values were maintained for all samples. The active area score (percentage of stained pixels per field of view) was determined and analyzed using statistical analysis software (GraphPad Prism, Graphpad Software Inc., USA).
Western Blotting
Total protein was extracted from snap-frozen lungs and concentration determined using a Pierce BCA protein assay kit (Thermo Fisher Scientific, USA). Protein (40 μg) was subjected to a reducing SDS-PAGE and transferred onto a PVDF membrane (Immobilon-P, Millipore, USA). Membranes were blocked with 5% skim milk in PBS/0.5% Tween-20 (PBS/T) before incubation with primary antibodies overnight at 4°C [1:2000 rabbit polyclonal anti-α-smooth muscle actin (αSMA), Abcam, USA.; 1:2000 mouse monoclonal anti-β-actin, Santa Cruz Biotechnology Inc., USA]. Membranes were incubated with secondary antibodies (1:5000 goat anti-rabbit HRP, Santa Cruz Biotechnology Inc., USA; 1:5000 goat anti-mouse HRP, Serotec, UK) at room temperature for 1 h. Bands were visualized using an Immobilon™ Western Chemiluminescent HRP substrate (Millipore, USA) and quantitated using the Quantity One™ quantitation analysis software (Bio-Rad Laboratories, USA).
Flow Cytometry
To detect CFSE-labeled hAECs, lung lobes were washed to remove residual blood, minced, and passed through a 70-μm filter to obtain a single cell suspension, and then analyzed using a BD FACSCanto II Flow cytometry system (BD Biosciences, USA). Gates were set based on the fluorescent profile of CFSE-labeled and unlabeled hAECs and excluded dead cells as well as small cells such as red blood cells.
RNA Isolation and Quantitative PCR
Total RNA was isolated from snap-frozen lungs using TRIzol (Invitrogen, USA) and converted to cDNA using the Thermoscript Reverse Transcription System (Invitrogen, USA) according to the manufacturer's instructions. Quantitative PCR was performed using Lightcycler 480 SYBR Green I Master Mix (Roche, Switzerland) and the Corbett Research Rotor-Gene 3000 (QIAGEN, Germany). Primers were directed against IL-1β, IL-2, IL-6, TNF-α, IFN-γ, IL-10, MIF, MIP-1α, TGF-β, and 18S. Primer sequences and annealing temperatures are listed in Table 1. Gene expression of each marker was normalized to 18S.
Primer Sequences and Annealing Temperatures
Immunohistochemistry for Macrophages and Neutrophils
To assess macrophage and neutrophil infiltration tissue sections were incubated with primary antibodies against either F4/80 (1:100, MCA497R, AbD Serotec, Germany) or myeloperoxidase (1:200, ab45977, Abcam, USA), respectively, at room temperature for 40 min. Tissue sections were then incubated in biotinylated secondary antibody (1:200, AP183β, Chemicon, USA) before detection using the EnVision detection system (LSAB+ System HRP, Dako, Denmark) and liquid DAB substrate (Dako, Denmark). Tissues were counterstained with hematoxylin. For each tissue sample, five images were captured using brightfield light microscopy at 200× magnification using a light microscope (AxioSkop, Zeiss, Germany) with a camera attachment (AxioCam ICc 3, Zeiss, Germany). Each image was converted to an RGB image and threshold values for nuclei (blue) or DAB (brown) were adjusted to include only stained areas using MetaMorph bioanalytical software (Molecular Devices, USA). Minimum and maximum cell size thresholds were also determined. Threshold values were maintained for all samples. F4/80- or myeloperoxidase-stained cells were counted using the bioanalytical software and expressed as a percentage of nucleated cells for each field of view.
Data Analysis and Statistics
Data were expressed for each experimental group as mean ± SEM and statistical significance determined using statistical analysis software (GraphPad Prism, Graphpad Software Inc., USA). Body weight and lung function testing data were analyzed using the repeated measures analysis of variance (ANOVA) with the Bonferroni post hoc test. Ashcroft scores, collagen content, Western blot, and qPCR data were analyzed using the one-way ANOVA with the Bonferroni post hoc test. Statistical significance was accorded when p < 0.05.
Results
Effect of Bleomycin and hAEC Administration on Body Weight and Lung Function
Compared to control mice (saline), administration of bleomycin resulted in a significant reduction in body weight over 3 days (17.57 ± 0.30 vs. 16.51 ± 0.13 g, p ≤ 0.01) (Fig. 1A). No significant difference was observed between bleomycin-treated mice and mice that received hAECs 24 h after bleomycin treatment groups at any time point.

Effect of hAEC administration on body weight and lung function. Administration of bleomycin resulted in a significant reduction in body weight over a period of 3 days compared to control mice (A). Mice in the bleomycin-treated group had significantly increased expiration time (B), decreased minute volume (C), lower respiration rate (D), and decreased inspiratory duty cycle (E) compared to control animals. Administration of hAECs to bleomycin-treated mice resulted in a partial restoration of physiological lung function with significant improvement in expiration time, minute volume, respiration rate, and inspiratory duty cycle compared to bleomycin-treated control animals. No significant difference was detected in inspiration time or tidal volume between bleomycin-treated and control animals at any time point (F, G) (*p ≤ 0.05, **p ≤ 0.001, ***p ≤ 0.0001).
Compared to control animals, bleomycin-treated mice had a significantly increased expiration time (day 3, 0.097 ± 0.002 vs. 0.214 ± 0.01 s; day 7, 0.107 ± 0.003 vs. 0.176 ± 0.011 s; day 14, 0.092 ± 0.002 vs. 0.162 ± 0.013 s; p ≤ 0.0001) (Fig. 1B), a decreased minute volume (day 3, 39.55±1.19 vs. 25.41 ± 0.92 ml, p ≤ 0.0001; day 7, 45.67 ± 2.42 vs. 31.79 ± 2.12 ml, p ≤ 0.05) (Fig. 1C), a lower respiratory rate (breaths/min) (day 3, 361.6 ± 9.2 vs. 212.8 ± 7.1, p ≤ 0.0001; day 7, 382.6 ± 12.7 vs. 256.3 ± 13.3, p ≤ 0.0001; day 14, 376.2 ± 12.3 vs. 281.1 ± 17.8, p ≤ 0.05) (Fig. 1D), and a decreased inspiratory duty cycle (day 3, 40.9 ± 0.4% vs. 24.9 ± 1.4%, p ≤ 0.0001; day 7, 37.9 ± 1.2% vs. 29.3 ± 1.8%, p ≤ 0.001; day 14, 41.4 ± 0.8% vs. 26.1 ± 1.4%, p ≤ 0.0001) (Fig. 1E). No significant differences were detected in inspiration time or tidal volume between bleomycin-treated and control animals at any time point (Fig. 1F, G).
Compared to bleomycin-alone animals, administration of hAECs to bleomycin-treated mice resulted in a partial restoration of physiological lung function with significant differences in expiration time (day 3, 0.214 ± 0.01 vs. 0.128 ± 0.007s, p ≤ 0.0001; day 7, 0.176 ± 0.011 vs. 0.113 ± 0.005 s, p ≤ 0.0001; day 14, 0.162 ± 0.013 vs. 0.115 ± 0.011 s, p ≤ 0.001), minute volume (day 3, 25.41 ± 0.92 vs. 35.86 ± 1.58 ml, p ≤ 0.0001; day 7, 31.79 ± 2.12 vs. 46.59 ± 4.34 ml, p ≤ 0.001), respiration rate (breaths/min) (day 3, 212.8 ± 7.1 vs. 286.5 ± 12.2, p ≤ 0.0001; day 7, 256.3 ± 13.3 vs. 340.8 ± 16.1, p ≤ 0.0001), and inspiratory duty cycle (day 3, 24.9 ± 1.4% vs. 35.4 ± 1.7%, p ≤ 0.0001; day 7, 29.3 ± 1.8% vs. 36.5 ± 1.6%, p ≤ 0.001; day 14, 26.1 ± 1.4% vs. 33.5 ± 2.0%, p ≤ 0.05) (Fig. 1).
Effect of Bleomycin and hAEC Administration on Lung Fibrosis and Collagen Content
Pulmonary fibrosis is characterized by the obliteration of the alveolar space followed by the deposition of a matrix of fibronectin and collagen. Wet lung weight (expressed as g/kg body weight), Ashcroft score of fibrosis, collagen content, and αSMA content provided an indication of the degree of pulmonary fibrosis in the mice from each experimental group at day 7 and 14. At day 7, compared to control mice, bleomycin-treated mice had an increased wet lung weight (4.55 ± 0.24 vs. 6.42 ± 0.58 g/kg, p ≤ 0.01), an effect not altered by hAEC administration (data not shown). No significant differences in lung weight between any groups were observed at day 14 (data not shown). Compared to control mice, bleomycin-treated mice had significantly higher Ashcroft scores of fibrosis at both day 7 and 14 (day 7, 0.49 ± 0.04 vs. 2.30 ± 0.45, p ≤ 0.001; day 14, 0.38 ± 0.04 vs. 3.00 ± 0.31, p ≤ 0.0001) (Fig. 2A, B). The increased fibrosis scores for bleomycin-treated animals were significantly reduced in those that also received hAECs 24 h after bleomycin (day 7, 2.30 ± 0.45 vs. 0.98 ± 0.37, p ≤ 0.05; day 14, 3.00 ± 0.31 vs. 1.04 ± 0.27, p ≤ 0.0001).

Effect of hAEC administration on lung fibrosis. At days 7 and 14, bleomycin-treated mice had significantly higher Ashcroft scores of fibrosis compared to control mice (A, B). We observed significantly lower Ashcroft scores of fibrosis for animals that received hAECs 24 h following bleomycin administration at day 7 and 14 compared to the bleomycin-treated group. Mice from the bleomycin-treated group had increased lung collagen content at day 14 compared to control mice (D). Mice that received hAECs 24 h after bleomycin had a significantly lower percentage of collagen per area of lung at day 14. Administration of bleomcyin resulted in a significant increase in αSMA compared the saline control group (E). Mice receiving hAECs following bleomcyin had decreased levels of αSMA compared to the bleomcyin-treated mice (*p ≤ 0.05, **p ≤ 0.001, ***p ≤ 0.0001).
Lung collagen content was determined using Sirius red staining and expressed as a percentage of collagen per area of lung. Compared to controls, mice from the bleomycin-treated group had increased lung collagen content at day 14 (1.24 ± 0.17% vs. 8.15 ± 1.78%, p ≤ 0.0001). Compared to bleomycin-treated mice, the mice that received hAECs 24 h after bleomycin treatment had significantly less collagen at day 14 (8.15+ 1.78% vs. 4.42 ± 0.36%, p ≤ 0.05) (Fig. 2D) but not at day 7. Lung hydroxyproline (OHPRO) content was determined at days 7 and 14 and expressed as a proportion of wet lung weight. There were no significant differences in OHPRO content between groups at either time point (data not shown).
An increase in αSMA-expressing myofibroblasts is a characteristic of pulmonary fibrosis (34,40). Compared to control animals, bleomycin-treated animals had significantly more pulmonary αSMA, as determined by Western blot densitometry (0.57 ± 0.14 vs. 1.34 ± 0.12, p ≤ 0.05) (Fig. 2E), an effect prevented by administration of hAECs following bleomycin (0.43 ± 0.10, p < 0.001) (Fig. 2E).
Representative images of hematoxylin and eosin-stained sections from each of the groups are shown in Figure 3A–D. Sections were scored as described in Materials and Methods. Representative images of Sirius red-stained sections from each of the groups are shown in Figure 4A–D. Collagen content was measured as described in Materials and Methods.
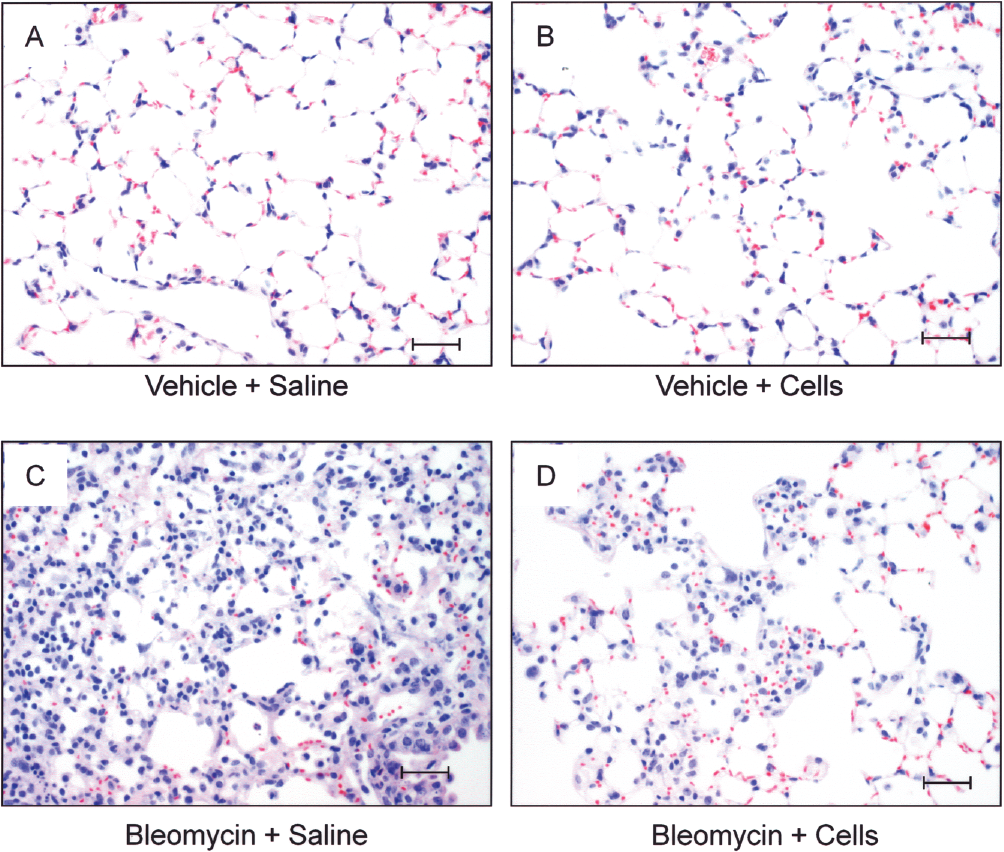
Representative images at day 14 of haematoxylin and eosin-stained histological sections from each group; Vehicle + Saline (A), Vehicle + Cells (B), Bleomycin + Saline (C), and Bleomycin + Cells (D). Histological sections were scored for levels of fibrosis as described in Materials and Methods. Scale bars: 50 μm.

Representative images at day 14 of Sirius red-stained histological sections from each group; Vehicle + Saline (A), Vehicle + Cells (B), Bleomycin + Saline (C), and Bleomycin + Cells (D). Lung collagen content was determined using Sirius red-stained photomicrographs, which were quantified by digital image analysis as described in Materials and Methods. Scale bars: 50 μm.
Flow Cytometric Analysis of Lung Tissue for hAECs
To determine whether hAECs had migrated to the site of injury following IP injection, we used flow cytometry to detect the presence of CFSE-labeled hAECs in the lung 7 and 14 days after cell administration. We did not detect any CFSE-positive cells by FACS analysis in any organ studied, including lung, brain, heart, spleen, liver, and kidneys (data not shown).
Effect of Bleomycin and hAEC Administration on Lung Inflammation
To determine the effect of hAEC administration on lung inflammation, qPCR was performed at day 7 using probes specific for a number of pro- and anti-inflammatory cytokines. Gene expression was expressed as fold change relative to 18S. Compared to control animals, the mice receiving bleomycin had significantly increased pulmonary gene expression for several proinflammatory cytokines: TNF-α (1.9 ± 0.8 vs. 116.2 ± 55.1, p ≤ 0.001) (Fig. 5A), TGF-β (1.6 ± 0.3 vs. 2874 ± 1668, p ≤ 0.05) (Fig. 5B), IFN-γ (3.5 ± 1.3 vs. 12.8 ± 4.5, p ≤ 0.05) (Fig. 5C), and IL-6 (0.4 ± 0.2 vs. 106.8 ± 41.1, p ≤ 0.0001) (Fig. 5D). Compared to bleomycin-treated animals, the administration of hAECs 24 h after bleomycin was associated with significantly lower pulmonary gene expression for TNF-α (116.2 ± 55.1 vs. 21.2 ± 10.5, p ≤ 0.05) (Fig. 5A), TGF-β (2874 ± 1668 vs. 193 ± 50, p ≤ 0.05) (Fig. 5B), IFN-γ (12.8 ± 4.5 vs. 4.5 ± 0.7, p ≤ 0.05) (Fig. 5C), and IL-6 (106.8 ± 41.1 vs. 8.8 ± 3.3, p ≤ 0.0001) (Fig. 5D). No significant differences between groups were detected for MIP1, MIF, IL-1β, IL-2, and IL-10 (data not shown).

Effect of hAEC administration on lung inflammation. Analysis of pro- and anti-inflammatory cytokine gene expression was performed by qPCR at day 7. Administration of hAECs 24 h after bleomycin resulted a significant decrease in the gene expression for TNF-α (A), TGF-β (B), IFN-γ (C), and IL-6 (D) compared to bleomycin control mice (*p ≤ 0.05, **p ≤ 0.001, ***p < p ≤ 0.0001).
To determine the effect of hAEC administration on macrophage and neutrophil pulmonary infiltration, we performed immunohistochemical staining for F4/80 and myeloperoxidase at day 7. At day 7, compared to control animals, there were significantly more macrophages (1.9 ± 0.6% vs. 15.5 ± 3.2%, p ≤ 0.001) (Fig. 6A) and neutrophils (0.4 ± 0.1% vs. 13.0 ± 4.9%, p ≤ 0.05) (Fig. 6B) in the lungs of bleomycin-treated animals, an effect reduced by the administration of hAECs (macrophages: 15.5 ± 3.2% vs. 5.3 ± 2.5%, p ≤ 0.05; neutrophils: 13.0 ± 4.9% vs. 1.1 ± 0.3%, p ≤ 0.05) (Fig. 6A, B).

Quantitation of F4/80 and myeloperoxidase immunohistochemical staining at day 7. Administration of hAECs 24 h after bleomycin resulted in a significant decrease in the percentage of F4/80 (A) and myeloperoxidase (B) positive cells in the lung compared to bleomycin control mice (*p ≤ 0.05, **p ≤ 0.001).
Discussion
We undertook this study to further explore the mechanisms by which hAECs may be able to abrogate bleomycin-induced lung injury. We have confirmed that hAECs can modulate the host inflammatory response, reduce fibrosis and collagen deposition, and prevent loss of lung function in this mouse model of lung injury. We show, at least in this model, that hAECs do not mediate this effect by being incorporated into host tissue and differentiating into alveolar cells. These observations extend previous reports of the use of hAECs in lung injury (13,35) by confirming the ability of hAECs to reduce fibrosis and by showing, for the first time, that hAECs can mitigate the inflammatory cytokine response in an immune-competent mouse and, again for the first time, that these effects result in improved lung function.
In this study we confirmed that the acute administration of hAECs can reduce bleomycin-induced lung fibrosis, as has been shown previously by us (35) and by others (13). However, there are some important differences between each of these studies. In our previous study we used immune-deficient (SCID) mice as a “proof-of-principle” model for hAECs in lung injury repair. In that study, we chose SCID mice to avoid the possibility that the xenogenic use of human amnion cells in immune-competent mice might induce an immune rejection that would mask any effects of hAECs on lung injury. While we thought that a xenogenic rejection was unlikely, as hAECs express low levels of HLAs (26) and have been shown by others to be immune privileged (29), the only previous report of the use of human amniotic fluid derived cells that included hAECs and other cell types in lung injury was in another immune-deficient (nude) mouse (14). This is important because it is the host immune/inflammatory response to bleomycin that underlies the resultant fibrotic injury (11). Thus, we thought it necessary to confirm that hAECs could mitigate bleomycin-induced fibrosis in an immune competent (C57BL/6) mouse with an intact host immune response. We chose the C57BL/6 mouse as this strain has been shown to be susceptible to bleomycin-induced lung injury (11). Cargnoni et al. (13) also used C57BL/6 mice. However, they administered as mixture of 50% hAECs and 50% human amnion-derived mesenchymal stem cells (hAMSCs)/human chorion-derived mesenchymal stem cells (hCMSCs), rather than hAECs alone. Therefore, it was not possible to determine from their study whether it was the mesenchymal stem cells (either hAMSCs or hCMSCs), hAECs, or both cell types that were effecting the mitigation of lung injury. Our study definitively shows that hAECs alone are able to mitigate injury in immune competent animals. Further work will be required to determine whether MSCs alone also possess this ability.
While hAEC administration clearly reduced the bleomycin-induced lung injury, it is worthwhile to comment on the apparent discrepancy in our collagen deposition results, as determined by Sirius red staining and hydroxyproline content. While bleomycin treatment increased collagen deposition and hAECs prevented this, as determined by Sirius red staining, we observed no changes in hydroxyproline content—a measure of collagen content. We believe that this apparent discrepancy is due to the patchy nature of the fibrotic lung injury induced by bleomycin. While some areas of lung are badly damaged, other areas are relatively uninjured. The Sirius red staining and scoring was undertaken on whole left lung whereas the hydroxyproline assays were performed only on the superior lobe of the right lungs. We believe that, being a more comprehensive assessment of whole lung, of the two methods, the Sirius red staining is likely to be a more accurate reflection of lung injury.
It is also pertinent to highlight that we observed minor increases in fibrosis (Ashcroft score), collagen content, and inflammatory cell infiltrate in the lungs of the control mice (i.e., no bleomycin) that received hAECs. This suggests that the administration of hAECs to otherwise healthy immune-competent mice induces an inflammatory response that might have detrimental effects. Such an effect was not observed by Cargnoni et al. (13), whether the transplanted cells were allogeneic or xenogeneic, but their cells were a mixed population of amnion and mesenchymal cells. Further investigation is certainly merited to verify our observation for hAECs alone. If confirmed, the extrapolation of our findings to the clinical setting would be that hAECs may not be suitable as a preventative therapy in otherwise uninjured individuals but rather reserved for management during acute injury.
As the therapeutic applications of hAECs are developed it will be important to understand how they exert their effects. Several studies have suggested an anti-inflammatory role for hAECs in the treatment of disease. It has been proposed that soluble factors produced by hAECs have anti-inflammatory effects in the cornea (25) and act to inhibit both the innate and adaptive immune systems (30). This effect appears to be independent of direct administration of hAECs into the site of injury or their presence in host tissue. Because TGF-β, TNF-α, and IL-6 have been shown to play significant roles in the progression of fibrosis through the mediation of the epithelial to mesenchymal transition (3,12,22,32,41,50), one of the mechanisms through which the hAECs mediate their effects is likely to be through the direct regulation of cytokine levels and signaling.
The lung fibrosis induced by bleomycin is thought to relate, at least in part, to the recruitment of macrophages into the lung, possibly by increased MIP-1, which is induced by elevated IL-6 and TNF-α (43). In this regard, in this current study hAECs reduced the expression of TNF-α, IFN-γ and IL-6, although not MIP-1, 7 days following bleomycin administration. This supports our previous observations in SCID mice where we showed that hAECs reduced downstream MIP-1 expression 14 days following bleomcyin (35). While we have not measured MIP-1 at day 14 in this current study, by assessing inflammatory cell infiltration we did explore whether one mechanism whereby hAECs afforded protection against bleomycin-induced injury might be the modulation of macrophage recruitment. Consistent with the lower TNF-α and IL-6 levels at day 7, and by implication subsequent MIP-1 levels (35), the administration of hAECs following bleomycin-induced injury significantly reduced inflammatory cell infiltration, specifically macrophages and neutrophils. This supports the observation made by Cargnoni and colleagues (13) that mixed hAEC/hAMSC/hCMSC administration reduced macrophage recruitment after lung injury. Of course, neither our study nor that of Cargnoni et al. (13) specifically explored whether this mitigating effect of hAEC, or hAEC/hAMSC/hCMSC, on macrophage recruitment is mediated via a modified cytokine response or directly on the leukocytes. Neither has either study asked whether reducing macrophage recruitment is required for the protective (antifibrotic) effect of the hAECs. These important mechanistic questions require further investigation.
Another possible mechanism by which hAECs may reduce bleomycin-induced fibrosis is by preventing an increase in TGF-β. TGF-β is profibrogenic, driving myofibroblast differentiation (24), leading to αSMA and collagen deposition. While we did not assess myofibroblast differentiation directly, the mitigation of increased TGF-β by hAECs was accompanied by a reduction in lung αSMA and collagen content, consistent with reduced myofibroblast differentiation. Further support for hAECs being able to modulate myofibroblast function is the observation that amnion membrane extract can prevent and reverse myofibroblast differentiation in vitro (31).
We were also keen to better understand the contribution to tissue injury repair afforded by the integration of hAECs into the damaged lung epithelium and their subsequent differentiation into alveolar cells, as we have previously reported in a SCID mouse (35). To our surprise, in this current study we were unable to identify any hAECs in any major organs, including injured or uninjured lung. There are a number of reasons that might explain the apparent discrepancies between these findings with those of previous studies. First, in the current study we administered the hAECs IP rather than IV, as has been the route of choice in most other stem cell studies using lung injury animal models (14,28,35,49). However, Cargnoni et al. (13) reported no differences in engraftment rates between IV, intratracheal, or IP routes of placenta-derived cells, which argues against this as a likely explanation. Second, this study utilized a purified population of term hAECs, while Cargnoni et al. (13) used a mixed population of mesenchymal and epithelial cells, and Carraro et al. (14) used a population of cells derived from amniotic fluid that would have included hAECs, other epithelial cells, and mesenchymal cells. It is certainly feasible that the cells detected in the host tissue in these previous two studies were not hAECs but rather other cell populations, such as mesenchymal cells. It is also possible that our ability to demonstrate small numbers of hAEC in the lungs SCID mice, but our inability to detect hAEC presence in this study where we used immune competent C57Bl/6 mice is due to xenogenic cell clearance in the immune-competent mice. However, we do not believe this to be a likely explanation as Cargnoni et al. (13) have shown that the cell source (allogenic or xenogenic) does not affect either cell presence in the lung or the efficacy of reducing bleomycin-induced fibrosis in immune-competent animals. Rather, we believe that the most likely explanation is that the methods of cell detection used in other amnion cell studies were different to those that we used in this current study. We used FACS to detect the presence of hAECs in host tissue, and gating was applied to exclude dead cells in the analysis. In previous studies, “human cells” were detected by either PCR or in situ hybridization for human DNA, or immunohistochemistry for cell markers (13,14,35). None of these methods are able to discern between dead human cells and live human cells resident in the host tissue. We acknowledge that FACS has a lower level of sensitivity compared to molecular techniques such as PCR. Nonetheless, we believe that it remains reasonable to conclude that there was not evidence of retention of significant numbers of live hAECs in the host tissue in this current study.
Regardless of the reasons underlying the differences between the studies in the detection of cell presence in host tissue, we believe that the current study shows that the presence or functional engraftment of hAECs in host tissue is not actually required for the mitigation of bleomycin-induced lung injury, as we previously suggested (35). In this regard, this current study strongly suggests that the observed effect of hAECs on the reduction of lung injury is most likely mediated via their anti-inflammatory properties, either TGF-β or inflammatory cell modulation.
In addition to these structural effects, this study has shown for the first time that hAECs can protect against loss of lung function following bleomycin. Previously, we have used whole body barometric plethysmography to characterize the changes in respiratory function in C57BL/6 mice following bleomycin administration (unpublished data). In the present study, we observed a similar loss in respiratory function following bleomycin, characterized by the development of an asymmetric breathing pattern and a decreased respiration rate. This breathing pattern was characterized by a proportionately longer expiration time than inspiration time. Expiration is normally a passive process and the observed increase in expiration time following bleomycin treatment is likely to have been attributed to the greater tissue resistance in the bleomycin-injured lungs. This resistance is overcome during inspiration as it is an active process; therefore, bleomycin-treated mice were able to maintain a normal inspiration time. However, the proportion of the respiratory cycle attributed to inspiration was decreased significantly due to the prolonged duration of expiration. Although the volume of air per breath (tidal volume) remained normal in the bleomycin-treated mice, they had a significantly lower respiration rate, due to prolonged expiration, that resulted in a decreased volume of air breathed per minute (minute volume). Thus, the overall effect on respiratory function of the structural changes induced by bleomycin was the development of an asymmetric breathing pattern due to a longer expiration time and a resultant decreased respiration rate. However, this bleomycin-induced loss of lung function was attenuated by the administration of hAECs, presumably by mitigation of underlying inflammation and structural damage. This shows that the improvement in the biochemical and histological outcome measurements afforded by hAECs are associated with functional improvement—a key outcome for any future therapy.
Importantly, this study is the first to demonstrate the efficacy of hAECs that have been isolated using good manufacturing practice (GMP) processes suitable for clinical use. Such isolation and storage processes will be required if hAECs are to become further developed as a cell-based clinical therapy. Certainly in the field of lung disease, the experimental data suggest that hAECs offer much promise. This is exciting because there is currently no effective treatment for diseases such as idiopathic pulmonary fibrosis (IPF) and chronic obstructive pulmonary disease (COPD) (2,6,15). For example, nonspecific anti-inflammatory treatments such as corticosteroids and cytotoxics have not proven useful in the long term and are associated with significant side effects (16,18,20). Even targeted anti-inflammatory therapies such as etanercept, a TNF-α blocking agent, or imatinib, a tyrosine kinase inhibitor that interferes with TGF-β and PDGF signaling, have not proven useful (17,38). Thus, while studies on hAECs as a therapy for pulmonary fibrosis remain experimental, there is an urgent need for an effective therapy and they offer much promise.
In summary, the findings from this study suggest that hAECs can modify bleomycin-induced lung injury, reducing lung inflammation and fibrosis, most likely via modulation of the host's injury response. The next steps in exploring hAECs as a cell-based therapy for lung disease will be to assess whether they can rescue/repair established lung fibrosis, as has been suggested (35), and whether primary, undifferentiated cells or hAECs partially differentiated into lung progenitor cells afford the best results. In the absence of any other effective therapy for pulmonary fibrosis we believe that such questions need to be answered with some urgency before progressing to clinical trials.
Footnotes
Acknowledgment
This research was funded by the National Health and Medical Research Council of Australia.
