Abstract
Cancer often arises in a background of chronic tissue damage. It is also increasingly appreciated that such an injured tissue microenvironment might foster the selective emergence of altered cells, leading to neoplasia. Accordingly, reversal of chronic tissue damage could represent a potential strategy to counteract neoplastic disease. In these studies, we aim to investigate whether transplantation of normal cells in the context of an injured, neoplastic-prone microenvironment might impact on the evolution of the carcinogenic process. A rat model of chemically induced hepatocarcinogenesis was used. Animals were given a single dose of diethylnitrosamine (DENA), followed by two injections of retrorsine (RS), a pyrrolizidine alkaloid that imposes a persistent block on hepatocyte cell cycle. At the end of this protocol, rats were either given no further treatment or injected, via the portal circulation, with 4 million normal hepatocytes isolated from a syngenic donor. After 3 months, rats given DENA+RS alone displayed numerous discrete nodular lesions (up to 30 per liver), ranging 1 to 3 mm in size. On the other hand, in animals receiving DENA+RS and transplantation, donor hepatocytes were able to repopulate over 50% of the host liver, as expected. Most importantly, both the number and the size of hepatocyte nodules were greatly reduced in these animals (percent nodular area was 1.8 ± 0.3, down from a control value of 8.5 ± 2.8). The above data indicate that strategies aimed at reestablishing a normal tissue microenvironment might be relevant to the management of neoplastic disease.
Keywords
Introduction
The pathway towards cancer in many organs, including the liver, is often inextricably linked to chronic disease processes that hamper the functional capacity of the tissue, and also its regenerative potential (7,9). For example, liver cirrhosis, the single most important risk factor for hepatocellular carcinoma, is associated with impaired liver regeneration both in humans (20,21) and in experimental animals (5,11). Thus, it is reasonable to hypothesize that such a growth-impaired background could provide a favorable competitive landscape for the selective emergence of rare cells with altered/preneoplastic phenotype (4,8,18,26). Consistent with this interpretation, studies from our laboratory have indicated that hepatocytes isolated from chemically induced liver nodules can rapidly proliferate and progress to cancer following transplantation into a growth-constrained liver microenvironment, while they are unable to grow to any significant extent when injected into the liver of a normal, syngenic recipient (16).
An important corollary of the hypothesis above is that strategies aimed at normalizing a chronically injured and/or growth-constrained tissue environment might be of value in the management of neoplastic disease (18). The present study was designed to test this possibility. Normal hepatocytes were transplanted in the setting of an irreversibly injured, neoplastic-prone microenvironment; donor-derived cells were able to extensively repopulate the host liver and this effect was associated with a significant decrease in the incidence of chemically induced hepatocyte nodules.
Materials and Methods
Animals and Treatments
Liver carcinogenesis was induced using a sequential exposure to diethylnitrosamine (DENA) and retrorsine (RS), a pyrrolizidine alkaloid that imposes a persistent block on normal hepatocyte cell cycle (14,25). We predicted that a population of DENA-initiated hepatocytes might be able to withstand the mitotic block imposed by RS and selectively proliferate vis-à-vis the surrounding parenchymal cells; such prediction was also based on data available for other alkaloids related to RS (10).
Male Fischer 344 rats, 4 weeks of age, were injected with DENA (160 mg/kg, IP), followed by two doses of RS (30 mg/kg each, IP), given at 2 and 4 weeks after DENA administration. Two weeks after the inducing protocol, animals were divided into two groups of eight rats each: group 1 received no further treatment, while group 2 was transplanted with 4 × 106 hepatocytes isolated from a normal syngenic donor. All animals were killed 4 months after DENA administration. All experiments were approved by the University of Cagliari Ethical Committee for Animal Experimentation; all animals received humane care in accordance with NIH Guidelines for the care and use of animals.
Hepatocyte Isolation and Transplantation
Hepatocytes for transplantation were isolated according to a two-step collagenase perfusion technique (2,22). Cell viability, determined by trypan blue exclusion at the end of the isolation procedure, was >90%. Hepatocytes were suspended in PBS (1 × 107/ml) and were injected via a mesenteric vein.
Animals were anesthetized and a small incision (about 1 cm) was performed in the upper abdominal wall; hepatocytes were then delivered through a branch of the mesenteric veins, using a syringe with a 26-gauge needle. The fate of donor-derived cells in the recipient liver was followed using the F344-dipeptidyl-peptidase type IV (DPP-IV)-deficient model for cell transplantation (24). Donor hepatocytes were isolated from animals expressing the marker enzyme (DPP-IV positive), while DPP-IV-deficient rats were used as recipients. Since the Fischer 344 rat is a syngenic strain, no immunosuppression was required for successful cell transplantation.
Histochemical and Immunohistochemical Methods
After sacrifice, livers were removed and examined for the presence of hepatic nodules or any other evident lesions. Samples were taken from each lobe and were either frozen for cryostat sections or fixed in buffered formalin for standard histological analysis and immunohistochemistry.
The incidence of preneoplastic lesions was evaluated using immunohistochemical detection of the marker enzyme glutathione-
Statistical Analysis
Results are presented as mean ± SE. Two-tailed Student's
Results
The Induction of Hepatic Nodules by Sequential Exposure to DENA+RS
As predicted, sequential exposure to DENA and RS resulted in the occurrence of numerous hepatocyte nodules, both on the liver surface (up to 30/liver, mean 18 ± 5) (Fig. 1A) and on sectioning (Fig. 1B). Nodular lesions were white-grayish in color and resembled those induced by other classical protocols of chemical hepatocarcinogenesis in the rat, both on macroscopic appearance and upon histological analysis (Fig. 1C, D). They also expressed the GST 7-7 marker enzyme (Fig. 1B), further supporting their putative preneoplastic nature.

Liver samples of rats exposed to diethylnitrosamine plus retrorsine ((DENA+RS) and killed 4 months later (see text for details). (A) At least three white-grayish nodules (arrows) in the caudate lobe. (B) A whole cryostat section of a liver samples from the same group processed for immunohistochemical detection of glutathione-
The surrounding liver was mainly occupied by enlarged (megalocytic) hepatocytes, with rare clusters of very small hepatocytes that have been referred to as regenerative nodules (19); mild to intense proliferation of oval cells was also observed (Fig. 1E, F).
Liver Repopulation by Transplanted Cells in DENA+RS-Treated Liver
We have repeatedly shown that preexposure to RS sets the stage for extensive repopulation of the host liver by transplanted normal hepatocytes (18). However, no studies had been published so far on the effect of a combined treatment with DENA and RS on liver repopulation.
Percent of host liver replacement by donor-derived cells was therefore evaluated in these experiments using a computer-assisted image analyzer (17). High levels of repopulation were observed in all transplanted animals, with an average of 53 ± 7% of total liver replacement by DPP-IV-expressing hepatocytes (Fig. 2A, B). The distribution of transplanted cells clusters was generally homogeneous throughout the recipient lobes, although some areas displayed lower levels of repopulation. Combined histochemical-immunohistochemical analysis for DPP-IV and GST 7-7 revealed that no overlapping was present between the two markers (i.e., none of the donor-derived hepatocytes clusters expressed the GST 7-7 enzyme), as previously observed (15).

Liver samples of rats exposed to DENA+RS followed by hepatocyte transplantation and killed 4 months later (see text for details). (A) A whole cryostat section of a liver samples processed for dual histochemical-immunohistochemical detection of F344-dipeptidyl-peptidase Type IV (DPP-IV) activity (orange-rust) and GST 7-7 expression (dark blue) (see text for details). Note the extensive repopulation of the host liver by DPP-IV-positive, donor-derived hepatocytes. (B) A microphotograph of a transplanted liver is shown: only residual endogenous megalocytes were present (thin arrow).
The Effect of Hepatocyte Transplantation on the Incidence of Nodules in the Liver of DENA+RS-Treated Rats
We next aimed to determine whether transplantation of normal hepatocytes and the associated extensive liver repopulation had any influence on the growth of nodules induced by sequential exposure to DENA and RS. Results are reported in Figure 3 and 4. The number of GST 7-7-positive focal lesions was greatly reduced in rats receiving DENA+RS and transplantation compared to the control group given DENA+RS only (3 ± 2 vs. 19 ± 8 lesions/cm2) (Fig. 4A; see also Fig. 3A and C vs. 3B and D for comparison). This was also reflected in the relative area occupied by nodular lesions, below 2% in the group receiving DENA+RS and transplanted cells (1.8 ± 0.3%), down from a control value of 8.5 ± 2.8%. This represents a net decrease of almost 80% between group 1 and group 2 (Fig. 4B). Furthermore, the mean size of the three largest GST 7-7-positive nodules found in each animal of the two groups was reduced by a factor of about 5 in transplanted versus nontransplanted rats (Fig. 4C).
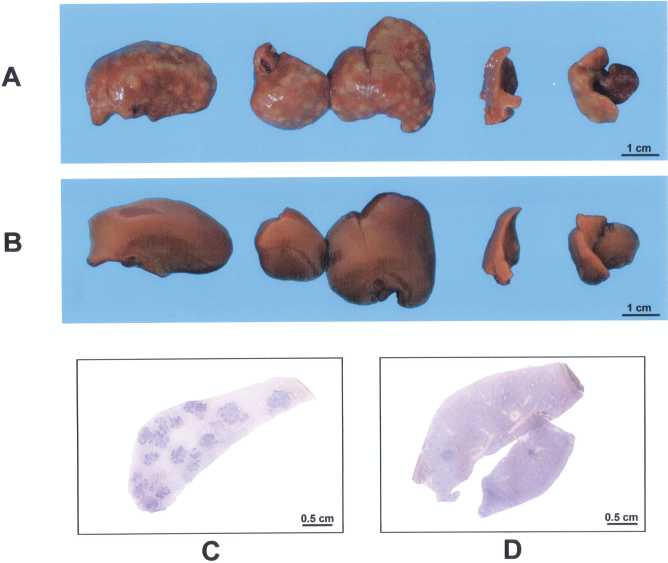
Liver samples of rats exposed to DENA+RS (A, C) or DENA+RS+Tx (transplant) (B, D) and killed 4 months later (see text for details). (A) DENA+RS: each liver lobe shows numerous white-grayish nodules, similar to Figure 1A; however, no such lesions are present in liver lobes in (B), taken from a rat exposed to DENA+RS+Tx. (C, D) Whole cryostat sections of the same livers processed for immunohistochemical detection of GST 7-7 (see text for details). Note the presence of several GST 7-7-positive lesions (dark blue) in the liver in (C) (DENA+RS), while few such lesions are present in (D) (DENA+RS+Tx).

The incidence of GST 7-7-positive nodules was evaluated in both DENA+RS and DENA+RS+Tx groups at 4 months after starting the experiment. (A) The number and size distribution of GST 7-7 expressing lesions is compared. (B) The percent area occupied by nodular lesions in each group. (C) The three largest GST 7-7-positive nodules from each animal were considered, and their mean size was compared in the two groups. (D) The extent of liver repopulation by transplanted cells in rats exposed to DENA+RS+Tx. Data are mean ± SE. Significantly different from control: *
Interestingly, DPP-IV-positive hepatocyte clusters were occasionally seen inside nodular, GST 7-7-expressing lesions (Fig. 5A), suggesting the possibility that donor-derived normal hepatocytes might integrate into hepatic nodules and disrupt their continuity.

(A) Combined histochemical-immunohistochemical staining for DPP-IV (orange-rust) and GST 7-7 (dark blue) on cryostat liver sections from a rat exposed to DENA+RS+Tx. Note that clusters of donor-derived cells appear to be present around and/or in the context of GST 7-7-positive nodular lesions. The surrounding host liver is unstained. No overlapping was ever observed between DPP-IV and GST 7-7 reactivity. (B) The effect of hepatocyte transplantation on liver histology is shown: only residual RS-induced megalocytes were present (thin arrow), while proliferation of bile ductular cells was greatly reduced.
The histology of transplanted and repopulated livers was also remarkably different compared to the nontransplanted group. Only residual chords of megalocytes were seen in between clusters of donor-derived cells. The latter displayed normal size, normal histology, and were perfectly integrated into the host liver, with no evident demarcation from resident hepatocytes (Figs. 2B, 5B). Proliferation of oval cells was also greatly reduced in livers receiving hepatocyte transplantation (Fig. 5B).
Parameters related to liver size and liver DNA content in both transplanted and nontransplanted groups were also measured. No significant differences were seen in final body weight and liver weight. Interestingly, a slight increase in liver DNA content was observed in the group receiving transplantation (6.42 ± 0.12 vs. 5.64 ± 0.34 mg DNA/liver/per 100 g b.w.,
Discussion
The unequivocal information that emerges from these studies is that transplantation of normal hepatocytes in the setting of a neoplastic-prone liver microenvironment is able to exert a significant impact on the development of nodular preneoplastic lesions. Injection of 4 × 106 hepatocytes, isolated from a normal syngenic donor, into the liver of a recipient animal preexposed to a carcinogenic protocol, resulted in a sharp delay and/or reduction in the incidence of hepatocyte nodules. This effect was accompanied by an extensive repopulation of the host liver by transplanted cells, resulting in a profound modification of the neoplastic-prone tissue landscape.
It is being increasingly appreciated that carcinogenesis is not a cell-autonomous process; rather, the phenotypic behavior of preneoplastic and even neoplastic cell populations appears to be critically dependent on specific cues provided by the surrounding microenvironment (3,6,13). This emerging paradigm is of particular relevance to liver cancer, given the fact that hepatocellular carcinoma most often arises in a background of chronic injury, with the ensuing inflammation, repair, fibrosis, and cirrhosis (1). Such a reactive microenvironment is likely to contribute critical factors for the selective emergence of cells with altered phenotype on the pathway towards cancer. Within this context, transplantation studies conducted by our group have firmly established the essential role of the microenvironment in determining the growth behavior of altered/nodular hepatocytes, as referred to in the Introduction (18).
The present studies add to those findings and provide a “proof of principle” that an irreversibly altered tissue microenvironment does in fact represent a driving force during early stages of carcinogenesis. Most importantly, our data indicate that the role of the microenvironment is potentially amenable to modulation. In fact, transplantation of normal cells was able to reverse, at least in part, tissue alterations induced by the carcinogenic protocol and this effect was associated with a sizeable reduction in the incidence of preneoplastic nodules.
No definitive mechanism(s) can be proposed at this time to explain this phenomenon. One attractive possibility is that the extensive structural remodeling of the host liver resulting from normal hepatocyte transplantation may help renormalize the neoplastic-prone microenvironment induced by DENA and RS, as suggested by stem cell transplantation studies (12,23). This in turn would translate into a decreased selective pressure for the emergence of altered cells. Alternately, or in combination, transplanted normal cells may compete with resident altered hepatocytes for the availability of critical growth factors that are essential for both cell types, thereby limiting the growth of the latter. In a recent review we discussed the intriguing analogies existing between liver repopulation and carcinogenesis, and suggested that in fact these two processes may share common pathogenetic mechanisms (10). Within this conceptual framework, normal cells could possibly be used to competitively counteract the selective growth of altered cells during early stages of carcinogenesis, as described in the present studies.
In summary, we provide evidence to suggest that strategies aimed at normalizing the microenvironment of a chronically and/or irreversibly injured liver tissue prone to cancer may help reduce the risk of neoplastic disease.
Footnotes
Acknowledgments
