Abstract
Hepatocyte transplantation is regarded as a promising option to correct hereditary metabolic liver disease. This study describes a novel method involving regional transient portal ischemia (RTPI) in combination with hepatic irradiation (IR) as a preparative regimen for hepatocyte transplantation. The right lobules of rat livers (45% of liver mass) were subjected to RTPI of 30–120 min. Liver specimens and serum samples were analyzed for transaminase levels, DNA damage, apoptosis, and proliferation. Repopulation experiments involved livers of dipeptidylpeptidase IV (DPPIV)-deficient rats preconditioned with RTPI (60–90 min) either with or without prior partial hepatic IR (25 Gy). After reperfusion intervals of 1 and 24 h, 12 million wild-type (DPPIV positive) hepatocytes were transplanted into recipient livers via the spleen. RTPI of 60–90 min caused limited hepatic injury through necrosis and induced a distinct regenerative response in the host liver. Twelve weeks following transplantation, small clusters of donor hepatocytes were detected within the portal areas. Quantitative analysis revealed limited engraftment of 0.79% to 2.95%, whereas control animals (sham OP) exhibited 4.16% (determined as relative activity of DPPIV when compared to wild-type liver). Repopulation was significantly enhanced (21.43%) when IR was performed prior to RTPI, optimum preconditioning settings being 90 min of ischemia and 1 h of reperfusion before transplantation. We demonstrate that RTPI alone is disadvantageous to donor cell engraftment, whereas the combination of IR with RTPI comprises an effective preparative regimen for liver repopulation. The method described clearly has potential for clinical application.
Keywords
Introduction
Currently, whole organ transplantation remains the only therapy available for a number of end-stage liver diseases, with the need for suitable donor livers increasing continuously (31). The search for an alternative to improve liver function or to correct genetically related defects is therefore of central importance (10). The transplantation of isolated cells (hepatocytes) into the diseased organ constitutes a promising alternative and has already been performed for various indications (26, 33). Inherited metabolic liver disease would be the primary target for strategies of liver cell therapy, because donor cells engraft preferably via portal vein infusions into the recipient livers, of which the architecture and circulation remains widely unharmed. However, hepatocyte transplantation into unconditioned recipient liver results in only low rates of cell engraftment (1). Furthermore, donor hepatocytes are known to lose the battle with endogenous cells, which proliferate preferentially in response to regeneration stimulus in the host liver. One solution to overcome this major obstacle to liver cell therapy consists of repetitive infusions aiming to deposit sufficient numbers of donor hepatocytes into the recipient liver (26). Although documented results of serial hepatocyte transplantations are encouraging, donor organ shortage greatly inhibits the availability of isolated donor cells, of which the number and quality is restricted more than ever (33). Therefore, several strategies have been developed to prime the host liver for efficient donor cell engraftment and subsequent selective expansion (11).
To date, most of the protocols significantly enhancing the outcome of hepatocyte transplantation in animal models have been based on the application of toxins or even carcinogens (e.g., pretreatment with pyrrolizidine alkaloids or monocrotaline) and are therefore not applicable in clinical settings (15,19,40). Hence, novel concepts of liver repopulation have to be established, utilizing methods and techniques currently employed both safely and routinely in a clinical setting. In a recent study of the workgroup, substantial liver repopulation was achieved using regional external beam irradiation to reduce the mitotic capacity of host hepatocytes followed by partial hepatectomy (PH). These techniques combined acted as a strong selective proliferation stimulus prior to donor cell transplantation (5,16,17). However, PH is a large operative procedure associated with a significant risk of operative complications, such as impairment of liver function and biliary leakage (9,37), which cannot be easily considered within a preconditioning regimen. Consequently, there is an obvious need for less or even noninvasive preconditioning protocols, especially when considering patients with severe hereditary liver disease, which are most commonly children (26).
Materials and Methods
Reagents
Chemicals, reagents, and antibodies were supplied by Sigma-Aldrich (Munich, Germany) unless otherwise specified.
Isolation of Primary Hepatocytes
Fisher rat hepatocytes were isolated employing two-step in situ collagenase perfusion of the liver first described by Seglen (32). The obtained hepatocyte suspension was further purified using Percoll® density gradient centrifugation (Pharmacia, Uppsala, Sweden). Freshly isolated hepatocytes (purification grade approx. 98%) displaying a vitality of greater than 90% (tested with trypan blue exclusion) and cell attachment greater than 70% proved to be sufficient for further transplantation experiments.
Animals and Design of Preconditioning and Transplantation Experiments
As recipients, a strain of DPPIV-deficient Fisher 344 rats was established in the animal care facilities of the University Medical Centre Goettingen, Germany. Syngeneic donor Fisher 344 rats were purchased from Charles River Germany. The animals were housed under 12/12-h light/dark cycles with standard rodent feed and water available ad libitum. All animal breeding, care, and experimentation procedures were in accordance with German national legislation on animal protection. Irradiation and all surgical procedures were performed under constant sevofluran/oxygen inhalation. Buprenorphin (0.1 mg/kg body weight) was applied intraperitoneally during anesthesia, and was repeated subcutaneously 8–12 h later.
In preliminary studies, male and female rats with a mass of 250–300 g received RTPI lasting 30, 60, 90, and 120 min through median laparotomy. The portal branches of the right superior anterior and right liver lobules 1–3 (45% of liver mass) were isolated and occluded with a Yasargil clamp (Fig. 1A). Sham-operated animals served as control group. The laparotomy incision was closed by continuous suture with Vicryl® (Ethicon, Norderstedt, Germany). Rats were sacrificed for tissue and serum analysis at different time intervals of reperfusion (6, 24, and 72 h and 7 days). Liver tissue was fixed in 4% formalin, dehydrated in ethanol, embedded in paraffin, and sectioned at a thickness of 2 μm. In addition, liver samples were stored in RNAlater® (Qiagen, Hilden, Germany) until processing.

RTPI through temporary clamping of the right portal branches supplying the right liver lobules 1–3 (45% of liver mass) (A) and illustration of the experimental design of liver repopulation following preconditioning RTPI either with or without prior partial liver IR (B). To demonstrate long-term liver repopulation (12 and 24 weeks), reporter cells (DPPIV positive) were transplanted via the spleen into DPPIV-negative rats either Δt = 1 or 24 h after termination of ischemia.
In the transplantation experiments, rats were subjected to 60 and 90 min of RTPI either alone or in combination with external beam, computed tomography-based partial liver IR (25 Gy) of the right liver lobules (45% of hepatic mass) 4 days prior to RTPI (Fig. 1B) as described previously (5). Following reperfusion intervals of 1 or 24 h, hepatocyte were transplanted through relaparotomy. After mobilization of the spleen, 12 × 106 freshly isolated hepatocytes were slowly injected over 3 min into the parenchyma, from where they are known to migrate via the portal vein into the recipient liver (all liver lobules). Control animals underwent hepatocyte transplantation without RTPI (sham OP) or IR alone. Both morbidity and mortality in all IR and/or RTPI groups prior to and following hepatocyte transplantation was overall low; only single animals died as a result of narcotic or surgical complications. Rats were sacrificed for tissue analysis at long-term time intervals (12 and 24 weeks following transplantation). Tissue samples from each liver lobe were excised and snap frozen in 2-methylbutane at −70°C. Cryosections of 5 μm in thickness were fixed in ice cold acetone for 10 min.
Detection of Serum Transaminase Levels
Hepatic injury was assessed by analyzing serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels. Analyses were completed according to standard procedures in blood samples at 6, 24, and 72 h and 7 days following RTPI.
Immunohistological and Immunofluorescence Analysis
Immunohistological analysis and immunofluorescence colocalization studies were performed as described previously (5,17). The primary antibodies [antiproliferating cell nuclear antigen (PCNA) from Neomarkers, anti-Connexin 32 (Cx32) (Sigma-Aldrich, Munich, Germany), and anti-DPPIV (BD Transduction Laboratories, Germany)] were used at dilutions of 1:500, 1:5000, and 1:100, respectively.
In preparation of bromodeoxyuridine (BrdU) staining, rats received an intraperitoneal injection of 50 mg/kg body mass BrdU 2 h prior to harvesting. BrdU incorporation was detected by a mouse anti-BrdU antibody (Dako GmbH, Hamburg, Germany). BrdU- and PCNA-positive hepatocytes were counted in 20 high-power fields (original magnification of 400×). Cell counts were performed blindly and the labeling index was expressed as a percentage of total hepatocytes counted.
Luminescence
Tissue was stored at −80°C after organ harvesting. Tissue samples of 100 mg were homogenized in 1 ml ice-cold HBSS with a Potter Elvehjem homogenizer (Braun, Melsungen). After centrifugation at 13,000 rpm for 10 min at 4°C (Biofuge pico from Heraeus, Osterode, Germany), the supernatant was stored again at −80°C. The quantitative analysis of DPPIV content was carried out using the DPPIV-Glo™ Protease Assay (Promega, Madison, WI, USA) according to the manufacturer's instructions. The liver sample supernatants were diluted 1:30 in HBSS and transferred in duplicates to a 96-well plate. After addition of 50 μl DPPIV-Glo™ reagent into each well, samples were measured in the microplate reader Tecan GENios (Mississauga, Ontario, Canada). As internal standard, all samples were normalized to samples from DPPIV-negative as well as DPPIV-positive (wild-type) rat liver. The latter was set as 100% for the evaluation of transplanted liver samples.
Statistical Analysis
We performed analysis of variance (ANOVA) to investigate treatment effects, differences between controls and treatment modalities were furthermore subjected to Dunnett's post hoc test. A value of p < 0.05 was taken as significant.
Results
Regional Transient Portal Ischemia Causes Hepatic Injury and Induces a Distinct Regenerative Response
Preliminary experiments aimed to examine the influence of ischemic damage on liver tissue. Therefore, RTPI of the right lobules of the liver was provoked by means of a small temporary clamp. After various periods of ischemia (30, 60, 90, and 120 min), the blood flow to these lobes was restored. The extent of liver damage (hepatocyte necrosis) was difficult to assess through morphometric analysis of the tissue alone and was therefore estimated more precisely by measuring systemic serum transaminase levels after reperfusion periods of 6, 24, and 72 h, and 7 days after RTPI (Fig. 2). Whereas sham-operated control rats only exhibited a minor augmentation of ALT and AST after 6 h of reperfusion, RTPI from 30 up to 120 min resulted in a remarkable increase in serum transaminase levels in a time-dependent manner. From 24 h of reperfusion onwards, the levels began to decrease gradually, which suggests that the necrotic period was tapering off. Although there was a dramatic drop in serum levels between 6 and 24 h, the parameters were still elevated after 72 h and only finally recovered to physiologically normal levels after 7 days.

Time course of aspartate aminotransferase and alanine aminotransferase serum levels following RTPI of the right liver lobules. The x-axis indicates the time of reperfusion after RTPI from 6 h (h) to 7 days (d). Serum transaminase levels were clearly elevated shortly after ischemia and declined to base levels within 7 days. The data are from 3–4 animals per group.
The extent of liver regeneration was assessed implementing two independent markers (expression of PCNA and incorporation of BrdU into nuclear DNA). Because DNA production peaks after 24 h in regenerating rat livers (2), tissues were harvested at the time point of 24 h but also after 72 h and 7 days of reperfusion. Positive hepatocytes were subjected to morphometric analysis. RTPI induced a distinct regenerative response, as seen after 24 h, at which point in time multiple hepatocytes in the parenchyma displayed PCNA staining (9.8% after 120 min of ischemia) (Fig. 3A). It must be noted that there were also a few positive endothelial cells in the portal vessels (Fig. 3B). As expected, the number of PCNA-positive hepatocytes dropped with shorter periods of ischemia (5.3% after 90 min and 2.6% after 60 min), and 30 min resulted in only slightly elevated numbers (0.7%). Fast liver reconstitution could be documented between 72 h and 7 days with ever decreasing numbers of PCNA-positive hepatocytes in all ischemic groups. The findings were in line with BrdU incorporation (Fig. 3C, D). After a noticeable increase in the number of positive hepatocytes 24 h after ischemia intervals of 60–120 min, the labeling index continuously declined, reaching background levels after 1 week (~0.3%).
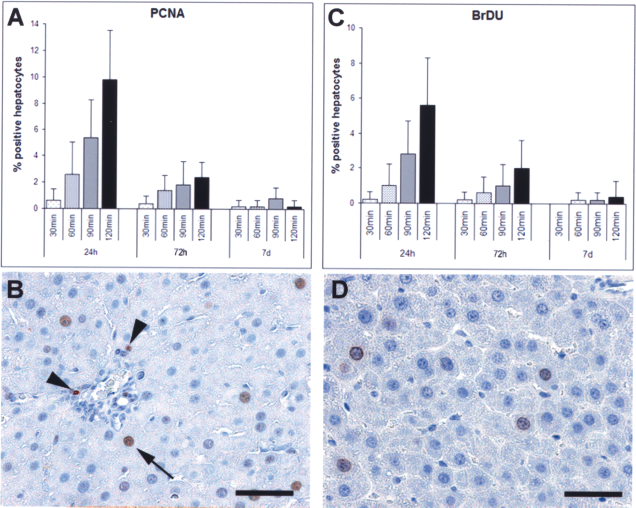
Liver regeneration assessed by labeling indices of PCNA staining (A) and BrdU incorporation (C) in rat liver after RTPI (30, 60, 90, and 120 min) and different time intervals of reperfusion (24 and 72 h, 7 days). Representative photomicrographs of lobule 1 following 90 min of ischemia are presented at the bottom (B and D, respectively). Arrow: positive hepatocyte, arrowheads: positive endothelial cells. Scale bar in (D): 50 μm.
Examining the type of liver injury following RTPI, we could not detect any significant extent of apoptosis in the different ischemic lobules (cell death detection ELISA, data not shown). However, large areas of necrosis were clearly detectable in close proximity to the portal areas as demonstrated by abundant hepatocytic staining of iNOS (inducible nitric oxide synthase) along with marked transcript expression in tissue homogenates (data not shown).
Ischemia with Prior Irradiation Results in Major Liver Repopulation
Encouraged by our findings that RTPI causes mild hepatic injury and induces a distinct regenerative response, our aim was to develop a clinically feasible protocol of host liver pretreatment. We considered ischemia periods of 60–90 min to be efficient, and more importantly, to be safe, to initiate the necessary regenerative response. Furthermore, we evaluated two different time intervals of reperfusion (1 and 24 h) before hepatocyte transplantation, assuming that hepatic RTPI results in an acute inflammatory response that may differently impact donor cell engraftment within the first 24 h (4). Liver repopulation was assessed using luminescence analysis of liver homogenates (Fig. 4). Both ischemic and nonischemic liver lobules displayed engrafted donor cells. However, the extent of repopulation in untreated lobules never exceeded those of the pretreated liver lobules (data not shown). Following RTPI of 60 min and 1 h of reperfusion before hepatocyte transplantation, quantitative analysis revealed 2.95% and 4.28% of repopulation after 12 and 24 weeks, respectively. When choosing 24 h of reperfusion, liver repopulation was even less, 1.5% and 3.52%, respectively. RTPI of 90 min and 1 h reperfusion resulted in likewise small repopulation degrees of 1.87% and 2.11%, respectively, which were even lower in the group of 24-h reperfusion. Control (sham OP) animals (none = “no” pretreatment group) exhibited overall significantly higher degrees of repopulation of 4.16% after 12 weeks and 5.52% after 24 weeks (p < 0.05). In all groups, there was an increase in repopulation from 12 to 24 weeks, indicating ongoing slow repopulation. However, there were no significant differences in donor cell expansion when comparing hepatic ischemia of 60 and 90 min, nor when considering the two different reperfusion intervals of 1 and 24 h. Moreover, our repopulation experiments clearly demonstrated that all modalities of RTPI were disadvantageous to donor cell proliferation within the host liver when compared to hepatocyte transplantation in untreated liver.

Quantitative analysis of liver repopulation by luminescence assay detecting DPPIV activity from donor cells. Pretreatment with RTPI (60 or 90 min) and reperfusion intervals prior to transplantation (1 and 24 h) as indicated. Control group “none” = transplantation in untreated liver, and control group “IR” = transplantation in irradiated liver. Fluorescence intensity was determined as relative activity of DPPIV when compared with wild-type liver normalized to 100%. The combined pretreatment of IR and RTPI led to significant liver repopulation represented by high levels of DPPIV activity (∗15.09% and ∗∗21.43%) following preconditioning of 60 and 90 min of ischemia, respectively, and 1-h reperfusion. With respect to 24 h of reperfusion, repopulation was only significant (***8.99%) in terms of 90 min of RTPI. ∗, ∗∗, ***Significant difference (ANOVA with Dunnett's post poc pest, p < 0.05) versus control group IR. Each bar represents the mean ± SEM. The data are from 3–4 animals per group.
In animals pretreated with IR alone, liver repopulation was to some extent higher after 12 weeks (5.86%) when compared to the control of no pretreatment (4.16%). However, when IR was performed prior to RTPI, donor hepatocytes were selectively amplified in the experimental groups of 1-h reperfusion (15.09% after 60-min ischemia and to 21.43% after 90-min of ischemia) (p < 0.05 vs. IR alone). With respect to the reperfusion interval of 24 h, there was only a significant increase when IR was followed by RTPI of 90 min, whereas 60 min could improve the repopulation efficiency, albeit with no significant impact in the combined pretreatment setting.
Immunofluorescence colocalization studies assessed the quality and confirmed the extent of liver repopulation detected by the luminescence studies. In the long term following transplantation, the overall liver structure appeared to be undamaged and without any sign of inflammation in all animals. Recipient livers pretreated with ischemia alone only displayed individual DPPIV-positive cells or scattered clusters of maximum five donor cells in diameter as seen after 12 weeks following 90 min of RTPI and 1 or 24 h of reperfusion, respectively (Fig. 5A, B). The combined stimulus of irradiation and ischemia clearly enhanced donor cell proliferation with formation of large clusters, which were up to 40–50 cells in diameter (Fig. 5D, E). In more detail, donor cells and their descendents expressed DPPIV in a bile canalicular pattern, which was flanked by well-dotted staining of Cx32 (major hepatic gap junction protein, enabling direct cell to cell communication) (inset of Fig. 5D). Both expression patterns are unique to fully differentiated and metabolically active hepatocytes.

Repopulation of the host liver by transplanted DPPIV-positive hepatocytes. Minimal repopulation (12 weeks) of recipient liver following RTPI of 90 min with reperfusion intervals of 1 and 24 h (A, B). IR prior to RTPI (60 and 90 min) enabled preferential proliferation of engrafted hepatocytes resulting in large clusters (D, E). Transplanted cells were identified by donor specific DPPIV immunofluorescence staining (red), colocalization with the differentiation marker Cx32 (green), and nuclear counterstaining with DAPI (blue), Transplantation into sham-operated animal (C) and into irradiated rat liver as controls (F). Original magnification 100×. Scale bar in (F): 100 μm.
Discussion
Hepatocyte transplantation has been used in many animal models of metabolic disease such as Wilson's disease, Crigler-Najjar syndrome, tyrosinemia, or hypercholesterolemia. In these experimental models, preconditioning of the host liver prior to cell transfer is regularly used to enhance proliferation of the transplanted cells, up to near total repopulation of the host liver (11). In contrast, only a few studies have been taken out on naive livers, yielding dramatically lower repopulation rates (27,38,39). This corresponds with the results of clinical use in human metabolic disorders, in which the amelioration of metabolic parameters has been demonstrated in most of the cases, but no patient has actually been cured in the long term so far (26). Thus, an appropriate minimally invasive preparative regimen of the host liver needs to be considered for human hepatocyte transplantation aiming to optimize the efficacy of cell therapy. We therefore decided to focus our research on RTPI alone or in combination with percutaneous external beam liver IR, two methods usable within reason in adults as well as in children with congenital metabolic disorders.
Ischemic preconditioning is well known as a therapeutic strategy in whole organ liver transplantation (14). It usually consists of a short period of both portal and arterial inflow occlusion followed by reperfusion. This improves the primary function and regenerative capacity of the transplanted organ (36). Whereas studies in the Watanabe hereditary hypercholesterolemia rabbit have demonstrated that regional ischemia-reperfusion can substantially increase donor cell engraftment (3), this technique would still require a surgical approach to the liver hilum, which we regarded as too invasive for clinical use. As an alternative to complete arterial/portal ischemia, selective portal ischemia has been suggested as a method of hepatic preconditioning prior to hepatocyte transplantation. However, most studies used permanent ligation (8,28), which would result in substantial liver damage. Ilan et al. demonstrated in the Gunn rat (animal model for Crigler-Najjar syndrome) that also transient clamping of a portal vein branch through laparotomy could improve the efficacy of cell transplantation (13). In human hepatocyte transplantation, surgical or interventional vascular access to the portal vein has to be established anyway for cell application (25), and it is tempting to use this catheter to induce transient regional portal ischemia additionally by blocking a portal vein branch with a balloon prior to cell application.
First of all, we had to evaluate the feasibility and effects of transient portal ischemia, as previous studies with permanent portal vein ligation only resulted in extensive necrosis of the corresponding lobules (22,29,30). Our results on RTPI revealed that all ischemia times from 30 to 120 min caused remarkable elevation of transaminases with a peak at 6 h and a slower decline from 24 to 72 h. However, the response to the damage was fast regeneration, the extent of which was proportional to the ischemia period. From data in the literature, it is not clear whether portal ischemia would result in necrosis or apoptosis (6,34). We therefore compared the extent of necrotic and apoptotic changes in the ischemic lobules (12). All parameters indicated that necrosis induced by RTPI was the predominant form of cell death, whereas apoptosis did not play a relevant role. With these data as a basis, we regarded ischemia times of 60 and 90 min, respectively, as most suitable for the transplantation experiments with respect to histological findings as well as any potential clinical application.
We chose a reliable transplantation model (hepatocytes isolated from wild type Fisher 344 rats were transplanted into DPPIV-negative hosts) to assess the safety of preconditioning and to quantify liver repopulation by donor cells (18). From our findings that RTPI caused mild necrotic injury of the liver and a distinct regenerative response that was terminated after 1 week, we thought that engraftment of transplanted hepatocytes would also be facilitated. Surprisingly, pretreatment with RTPI alone actually worsened transplantation efficacy when compared with transplantation in untreated liver. However, liver repopulation was enhanced up to 5.2-fold using the combination of RTPI and partial liver IR. To note, the morphology of engrafted donor hepatocytes and their descendents indicated full integration into the host parenchyma and metabolic integrity (distinct bile canalicular expression pattern of DPPIV and well-dotted Cx32 staining). Ischemia times of 90 min produced better results than 60 min, suggesting that the effects of IR were more pronounced in direct proportion to the extent of ischemic liver damage and mediated regeneration stimulus.
Combining liver IR with RTPI, two previous studies have already demonstrated great benefits for donor cell engraftment and/or proliferation (20,21). In contrast to the present study, these experiments used very high doses of 50 Gy delivered to the whole liver by laparotomy. Furthermore, the duration of RTPI varied within the studies, and the time interval between RTPI and cell transplantation was considerably longer (24–72 h) than in our study. From our findings, time intervals of 1 h were clearly favorable when compared to 24 h. This would evidently facilitate the clinical approach of preconditioning and subsequent transplantation in one combined treatment session. We may have to speculate that some beneficial effects of RTPI such as cytokine release and proinflammatory response might aid cell engraftment during this early reperfusion period (23).
Reports on the effect of RTPI alone on hepatocyte transplantation efficiency in the literature are inconsistent. Our data suggest that engraftment and long-term repopulation was lower in animals that received RTPI alone compared to control animals. In both studies published by Malhi et al. (20,21), single ischemic preconditioning resulted in low liver repopulation efficiencies (<2%) or limited therapeutic effects (3 responders out of 11), which was within the range or only slightly better when compared to the controls of untreated liver. The workgroup of Weber and colleagues reported a higher degree of repopulation, which was approximately 6–7% in the long-term period following portal temporary occlusion (embolization). To note, these studies were performed under the additional stimulus of partial hepatectomy and in nonhuman primates (7), two conditions that would explain the different results well.
Our results contribute to the assumption that liver repopulation in animals requires two fundamental conditions. Firstly, the regeneration capacity of the recipient liver needs to be impaired by blocking the cell cycle of endogenous hepatocytes, for example, by using IR (17). Secondly, some mitotic stimulus has to be introduced, such as RTPI. Here, we report for the first time that combining the preparative regimen of external beam limited dose IR and moderate RTPI prior to single dose hepatocyte transplantation results in selective donor cell proliferation of >15%, which is known to be sufficient for the correction of genetic disorders and treatment of liver failure (24). Reinforcing our concept, it has already been demonstrated in a retrospective study on children undergoing hepatic IR after liver transplantation for uncontrollable rejection that external IR was tolerated well in the long run (18 years) (35). Therefore, we consider our combined pretreatment approach to be fully suitable for direct transfer into clinical use. Moreover, our studies demonstrate that clinically feasible preconditioning methods have to be evaluated carefully, as a mild preparative regimen may require the exact planning of individual stimuli, their duration, and chronological order.
Footnotes
Acknowledgments
The authors would like to thank Sabine Wolfgramm and Sabrina Goldmann for their excellent work in preparing the immunolabeling figures for this article. We would furthermore like to express our appreciation to Andrew Entwistle for his critical review of the manuscript. The study was supported by the Cytonet GmbH & Co. KG, Weinheim, Germany and partially by the Federal Ministry of Education and Research BMBF (Grant-Number 01GN0537).
