Abstract
This study was to investigate the effect of trichostatin A (TSA), a histone deacetylase (HDAC) inhibitor, on cardiac differentiation of bone marrow mesenchymal stem cells (MSCs) in vitro. Rat MSCs were isolated and divided into six groups: 1) control; 2) 5-azacytidine treatment (5-aza, 10 μM); 3) treatment with TSA (100, 300, and 500 nM); 4) treatment with 5-aza followed by incubation with TSA; 5) coculture with neonatal cardiomyocytes (CMs); and 6) treatment with TSA then coculture with CMs. HDAC activity was significantly inhibited in TSA-treated cells with the maximal inhibition after 24 h of exposure to TSA at 300 nM. No changes in HDAC activity were observed in control, 5-aza-treated, or coculture groups. Following 7 days of differentiation, the expression of early cardiac transcription factors GATA-4, NKx2.5, MEF2c, and cardiac troponin T (cTnT) was increased by 6—8 times in the cells in 5-aza-treated, coculture, or TSA-treated groups over control as determined using real-time PCR, immunofluorescence staining, and Western blotting. However, the percent cTnT-positive cells were dramatically different with 0.7% for control, 10% for 5-aza-treated, 25% for coculture, and 4% for TSA-treated group (500 nM). TSA treatment of the cells pretreated with 5-aza or cocultured with CMs dramatically increased the expression of GATA-4, NKx2.5, and MEF2c by 35—50 times over control. The cTnT protein expression was also significantly increased by over threefold by TSA treatment (500 nM) in both 5-aza-treated and coculture group over control. The percent cTnT-positive cells in both 5-aza-pre-treated and coculture groups were significantly increased by TSA treatment after 1 week of differentiation by up to 92.6% (from 10.3% to 19.8%) and 23.9% (from 24.5% to 30.2%), respectively. These data suggested that TSA enhanced the cardiac differentiation of MSCs after 5-aza induction or during coculture with CMs through a mechanism beyond the inhibition of HDAC activity.
Keywords
Introduction
Cell-based therapy remains an attractive and viable option for tissue regeneration and repair after injuries like myocardial infarction (9,36,50). Bone marrow mesenchymal stem cells (MSCs) are one of the main sources for cell therapy for cardiac repair and regeneration since these cells are easily obtainable without ethical concerns, and have little (if any) inherent immunogenicity for any adverse immune reactions (5,42). However, the efficacy of MSC differentiation into cardiomyocyte has been very inconsistent in both in vivo and in vitro studies. In some in vitro experiments, only a very small portion of MSCs turned positive phenotypically for cardiomyocyte during MSC differentiation that clearly limited their potential clinical benefits for cardiac repair. Some in vivo studies showed no cardiac differentiation from bone marrow-derived multipotent progenitor cells after transplantation in the infarction area (3,15,32). Therefore, it is critically important to establish an effective method to enhance the differentiation of MSCs into cardiomyocytes.
There are two major approaches to the induction of MSCs to differentiate into cardiac myocytes in vitro: chemical induction with 5-azacytidine (5-aza) or amphotericin (AMPH) and coculture with mature cardiomyocytes (31,40,48). However, the efficacy of these techniques on inducing cardiac differentiation of MSCs is limited and very variable (34). Recently, we demonstrated that suberoylanilide hydroxamic acid (SAHA), a histone deacetylase (HDAC) inhibitor, significantly enhanced MSC cardiomyocyte differentiation with increased expression of cardiomyocyte-specific genes and proteins (14). Like SAHA, trichostatin A (TSA) is a potent HDAC inhibitor that suppresses the activities of HDACs, including all 11 known human class I and class II HDACs. Inhibition of HDAC by SAHA and TSA leads to differentiation, cell cycle arrest, and apoptosis in many tumor cells (4,10). Treatment of human MSCs with TSA stimulates their osteogenic differentiation with increased expression of alkaline phosphatase and mineralization in vitro (39). TSA also enhances the differentiation of MSCs into hepatocyte-like cells with increased expression of hepatocyte-specific proteins including hepatocyte nuclear factor 3β (HNF3β), α-fetoprotein (AFP), albumin (ALB), HNF1α, multidrug resistance-associated protein 2 (MRP2), and CCAAT-enhancer-binding protein α (C/EBPα) (12).
The present study was designed to determine if TSA could enhance the differentiation of MSCs into cardiomyocytes when the cells were pretreated with the inducing agent 5-aza or cocultured with neonatal cardiomyocytes (CMs). Indeed, we observed that either treatment with 5-aza or coculture with CMs induced cardiac specification of MSCs in vitro. However, addition of TSA substantially enhanced the cardiomyocyte differentiation of MSCs with increased expression of cardiomyocyte-specific genes and proteins in the cells pretreated with 5-aza or cocultured with CMs.
Materials and Methods
Culture, Expansion, and Characterization of Rat MSCs
Adult Wistar rats 8 weeks old (40 ± 10 g) and neonatal Wistar rats (0—3 days old) were purchased from the Experimental Animal Center at Chongqing Medical University, China. The experimental protocol was approved by the Animal Usage and Welfare Committee at Chongqing Medical University. MSCs were prepared from adult Wistar rats, and cultured in DMEM/F12 Medium (Invitrogen, USA) at 37°C with 5% CO2 and 95% air as described (7). After 48 h, the media were changed to remove nonadherent cells. At 90% confluence, the cells were subcultured for four passages and harvested for phenotypic characterization and differentiation. For cell phenotypic characterization, the cells were fixed and prepared for immunofluorescence staining for CD71 and CD105 as described (8). The cell preparations were exposed to mouse anti-CD71 (1:100, Abcam, Britain) and rabbit anti-CD105 (1:100, Abcam). Goat anti-mouse tetramethyl rhodamine isothiocyanate (TRITC) antibody (1:50) and goat anti-rabbit fluorescein isothiocyanate (FITC) antibody (1:50) were used as the secondary antibodies, respectively. The cells were subsequently analyzed using a fluorescence microscope (Olympus BX51, Japan).
Preparation of Rat Neonatal Cardiac Myocytes
Primary cultures of ventricular cardiac myocytes (CMs) were prepared from 0—3-day-old neonatal Wistar rats using the method previously described with minor modifications (26). Briefly, CMs were dispersed from the neonatal rat ventricles by a series of incubation at 37°C in D-Hanks solution containing 1.0 mg/ml trypsin (1:250, Difco Laboratories). The dispersed cells were then cultured at a density of 3 × 105 cells/cm2 for 90 min (95% air/5% CO2 at 37°C) to eliminate the fibroblasts. The unattached cells were transferred to, and cultured on collagen-coated plates at a density of 1.25 × 105 cells/ cm2 in DMEM/F12 (50:50 by volume) media supplemented with 10% fetal bovine serum (FBS, Hyclone) and 1% penicillin/streptomycin solution (Gibco). After 48 h of culture, over 99% of the cells in culture were CMs as estimated by immunocytochemical staining for the sarcomeric protein α-actin. The cells were then transferred to a Transwell culture plate for coculture with MSCs without direct contact.
Cardiac Differentiation of Rat MSCs
To induce cardiomyocyte differentiation, rat MSCs were cultured at a density of 1 × 105 cells/ml on a 30-cm2 culture flask in 5 ml basal media supplemented with 10% FBS (v/v) as described previously (16,45). The basal media consisted of 50% (v/v) DMEM and 50% (v/ v) F12 medium (Invitrogen, USA). At 100% confluence, the cells were exposed to 5-aza (10 μM) for 24 h, and then transferred to the complete media with 10% FBS (without 5-aza) with media change every 3 days at 37°C with 5% CO2, and 95% air for up to 2 weeks. To evaluate the effect of TSA on cardiac differentiation of MSCs, the cells were incubated with different concentrations of TSA (0, 100, 300, and 500 nM; Sigma, St Louis, MO, USA) for another 48 h after 24 h of culture with 5-aza. The expression of cardiomyocyte-specific genes and proteins including GATA binding protein 4 (GATA-4), NK2 transcription factor related, locus 5 (NKx2.5), myocyte enhancer factor 2c (MEF2c), cardiac troponin T (cTnT), and myosin heavy chain (MHC) was determined with real-time PCR, immunofluorescence staining, and Western blot analysis at different time points during differentiation.
Rat MSCs (passage 4) were also induced to cardiomyocytes using coculture system with neonatal CMs as described previously (16,24). MSCs were first treated with TSA (0, 100, 300, and 500 nM) for 48 h, then transferred to the Transwell culture plate for coculture with neonatal CMs at a cell density of 2 × 105 cells per well without direct contact between MSCs and CMs. The cultures were maintained in the complete media with 10% FBS (without TSA) with media change every 48 h for up to 2 weeks at 37°C with 5% CO2 and 95% air. The expression of cardiomyocyte-specific genes and proteins including GATA-4, NKx2.5, MEF2c, cTnT, and MHC was determined with real-time PCR and Western blot analysis at different time points during differentiation.
HDAC Assay
HDAC enzyme activity (HDAC I) in the cells at different time points during differentiation were determined using HDAC Assay Kit (Upstate, USA) as per manufacturer's protocol. The nuclear extract of the cells (20 μg) was prepared as described by the Fluorometric HDAC activity assay (Upstate) and colorimetric HDAC activity assay (Biovision, Mountain View, CA, USA) were performed according to the manufacturer's instructions. Results were reported as fluorescent units (FU), and incubated at room temperature with 1 mM HDAC Assay Substrate for 60 min. The reactions were terminated with HDAC Activator Solution. The samples were read at 405 nm. All samples were prepared in duplicates.
Immunofluorescence Staining
The cells at different stages of differentiation were fixed and prepared for immunofluorescence staining to evaluate the expression of cardiomyocyte-specific protein cTnT as described previously (44). The cell preparations were incubated with the primary antibody against cTnT (1:100, Abcam, Britain), followed by exposure to the secondary antibody as recommended by the manufacturer (FITC-conjugated IgG, 1:150, Santa Cruz, USA). Cells incubated only with appropriate secondary antibody with the same dilution factor were used as negative controls. The cells were also exposed to 4′,6-diamidino-2-phenylindole (DAPI; Molecular Probe, USA) for nuclear staining. The preparations were then examined and analyzed using a fluorescence microscope. The percentage of cTnT-positive cells was determined from 10 randomly selected views for every preparation in each group.
Quantitative Real-Time PCR Analysis
Quantitative real-time RT-PCR assay was used to assess the transcriptional expression of cardiomyocyte-specific genes including GATA-4, NKx2.5, and MEF2c. Total RNAs were extracted from the cells and reverse-transcribed with a kit from Takara (TAKARA, Japan) for the assay using SYBR Green dye kit (TIANGEN, China) as described previously (1). The primer sequences were: for GATA4—-5′-ctgtggcctctaccacaagat-gaa-3′ (forward) and 5′-gtctggcagttggcacagga-3 (reverse); for NKx2.5—5′-accctcgggcggataagaa-3′ (forward) and 5′-gacaggtaccgctgttgcttga-3′ (reverse); for Mef2c—5′-cacgacagagtggcagatgg-3′ (forward) and 5′-ggggaggagat ttggcttg-3′ (reverse); and for β-actin—5′-ggagattactgcc ctggctccta-3′ (forward) and 5′-gactcatcgtactcctgcttgctg-3′ (reverse). All the primers were designed by the software Primer 5.0, and synthesized by Dalian Bao Biological Genes Corp. Limited (Dalian, China). Amplification reagent RNA PCR (AMV) VER 3.0 was bought from Dalian Bao Biological Genes Corp. The mRNA levels were normalized using β-actin as housekeeping gene, and analyzed by 2−ΔΔCt ± SD (27).
Western Blot Analysis
Cell lysates were prepared from the undifferentiated and differentiating MSCs at different time points for Western immunoblot analysis to determine the expression level of cardiomyocyte-specific protein cTnT as described (25). A total of 20 μg protein was loaded on 8% sodium dodecyl sulfate (SDS)-polyacrylamide gel, and subsequently transferred to nitrocellulose membranes after electrophoresis. The preparations were then blocked with 5% non-fat dry milk for 1 h at room temperature, followed by incubation with primary antibody mouse anti-rat cTnT monoclonal antibody (dilution factor 1:200, Abcam, USA) or mouse anti-rat β-actin monoclonal antibody (dilution factor 1:200, Abcam, USA) at 4°C for 12 h. The preparations were then incubated with the secondary antibody (goat anti-mouse IgG, 1:100, Santa Cruz Biotechnology, USA) in blocking buffer for 1 h, followed by exposure to the Enhanced Chemiluminescence Luminal reagent (Santa Cruz Biotechnology) for 1 min for analysis with software Image-Pro Plus 5.1 (Media Cybernetics, Inc. USA).
Statistical Analysis
All the data were expressed as mean ± SEM. Statistical comparison of the data was performed using the t-test for comparison between two groups or one-way ANOVA, and post hoc Tukey's test for multiple comparisons between more than two groups. Data were analyzed using an SPSS statistical package for Windows (SPSS Inc., Chicago, IL). A value of p < 0.05 was considered statistically significant.
Results
TSA Effectively Inhibited HDAC Activity In MSCs
We first examined HDAC activity in MSCs in the first 48 h during differentiation. There was a detectable level of HDAC activity in the undifferentiated MSCs that remained stable during the first 48 h of differentiation (Fig. 1). No change in HDAC activity was observed in the cells when treated with 5-aza or cocultured with neonatal CMs during the first 48 h (Fig. 1A). When the cells were incubated with TSA, the HDAC activity was significantly decreased in a dose- and time-dependent manner as expected (Fig. 1A). The HDAC activity started to decrease in MSCs after 6 h of exposure to TSA at the concentration of 300 or 500 nM. The maximal inhibition of HDAC activity occurred after 24 h of incubation with 300 nM TSA with a significant reduction of the activity by 59.5% (p < 0.05, n = 5). No further reduction of HDAC activity was observed when the cells were exposed to higher dose of TSA (500 nM) and/ or longer duration (48 h) (Fig. 1). Pretreatment of the cells with 5-aza (10 μM) had no effect on HDAC activity in MSCs in the presence of TSA (Fig. 1B). These data suggested that the dosage and duration of TSA intervention used in the present study were adequate. These results also indicated that no interaction between TSA and 5-aza on the HDAC activity was present in MSCs.

Time- and dose-dependent inhibition of histone deacetylase (HDAC) activity by trichostatin A (TSA) in mesenchymal stem cells (MSCs). HDAC activity in MSCs in the first 48 h during differentiation after treatment with 5-azacytidine (5-aza) or TSA or coculture with neonatal cardiomyocytes (CMs) was determined. There was a detectable level of HDAC activity in the undifferentiated MSCs that remained stable during the first 48 h of differentiation. No change in HDAC activity was observed in the cells when treated with 5-aza or cocultured with neonatal CMs during the first 48 h. When the cells were incubated with TSA, the HDAC activity was significantly decreased in a dose- and time-dependent manner as expected. The HDAC activity started to decrease in MSCs after 6 h of exposure to TSA at the concentration of 300 or 500 nM. The maximal inhibition of HDAC activity occurred after 24 h of incubation with 300 nM TSA with a significant reduction of the activity. No further reduction of HDAC activity was observed when the cells were exposed to higher dose of TSA (500 nM) and/or longer duration (48 h) (A). Pretreatment of the cells with 5-aza (10 μM) (B) or coculture with CMs (C) had no effect on HDAC activity in MSCs in the presence of TSA (100, 300, and 500 nM). MSCs: MSCs without treatment or coculture; 5-aza: MSCs treated with 10 μM 5-aza for 24 h; Coculture: MSCs cocultured with neonatal CMs; 100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively; 5-aza+100, 300, 500: MSCs treated with 10 μM 5-aza for 24 h, followed by treatment with 100, 300, and 500 nM TSA, respectively; Coculture+100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively, then cocultured with CMs.
Cardiac Differentiation of MSCs with 5-Aza Induction, Coculture, or TSA Treatment
To compare the efficacy of cardiac differentiation of MSCs under different conditions, the expression profiles of the early cardiac-specific transcription factor genes including GATA-4, NKx2.5, and MEF2c were determined in the cells during differentiation after treatment with 5-aza or TSA or coculture with neonatal CMs. The transcriptional expression of these genes was significantly increased by 6—8 times in the cells in all three treatment groups after 1 week of differentiation compared to untreated ones as shown in Figure 2. Interestingly, the mRNA levels of these genes were similar in the cells that were either treated with 5-aza or TSA or cocultured with CMs. These data indicated that these three methods had similar potential to induce cardiac differentiation of MSCs in vitro.
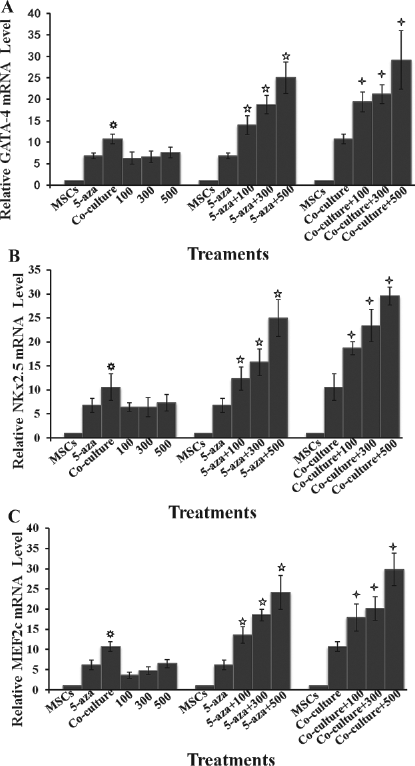
The expression profiles of GATA binding protein 4 (GATA-4), NK2 transcription factor related, locus 5 (NKx2.5), and myocyte enhancer factor 2c (MEF2c), after 1 week of differentiation. The transcriptional expression of the early cardiac-specific transcription factor genes GATA-4, NKx2.5, and MEF2c were determined in the cells using real-time PCR during differentiation after treatment with 5-aza or TSA or coculture with neonatal CMs. The mRNA levels for GATA-4 (A), NKx2.5 (B), and MEF2c (C) were similarly increased in the cells after 1 week of differentiation after exposure to 5-aza or TSA or coculture with CMs. There was no difference in their mRNA levels in the cells between 5-aza group and TSA treatment groups. Interestingly, the mRNA levels of these three genes were significantly higher in the cells cocultured with CMs than those in 5-aza- or TSA-treated cells. Treatment of the cells with TSA dramatically enhanced the expression of the cardiac-specific genes GATA-4, NKx2.5, and MEF2c in a dose-dependent manner with as much as 35—40-fold increase in their mRNA levels when the 5-aza-treated cells were exposed to 500 nM TSA compared to undifferentiated MSCs. Similarly, the transcriptional expression of GATA-4, NKx2.5, and MEF2c was substantially upregulated by 40—50 times in the cells of coculture system when the cells were first treated with 500 nM TSA. MSCs: bone marrow mesenchymal stem cells without treatment or coculture; 5-aza: MSCs treated with 10 μM for 24 h; Coculture: MSCs cocultured with CMs; 100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively; 5-aza+100, 300, 500: MSCs treated with 10 μM for 24 h, followed by treatment with 100, 300, and 500 nM TSA, respectively; Coculture+100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively, then cocultured with CMs. p < 0.05 compared with 500 nM TSA group; p < 0.05 compared with 5-aza group; p < 0.05 compared with coculture group.
Similarly, the expression of cardiac protein cTnT was also increased in the cells after 1 week of differentiation following induction with 5-aza or TSA or coculture with CMs as evaluated using immunofluorescence staining (Fig. 3) and Western blot analysis (Fig. 4). No apparent difference in the level of cTnT protein was observed in the cells that were induced with any one of the methods. No cTnT protein was present in the undifferentiated MSCs. These results were consistent with the data on the transcriptional expression of cardiac specific genes GATA-4, NKx2.5, and MEF2c, and further suggested that the efficacy of MSC cardiac differentiation was comparable for the three induction methods.
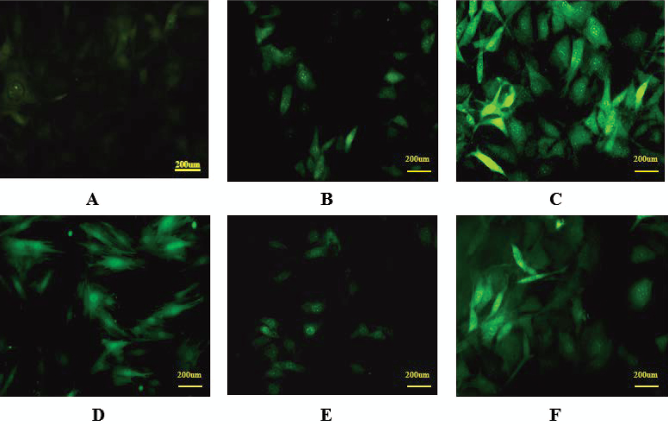
Immunofluorescence staining for cardiac troponin T (cTnT). The expression of cTnT was evaluated using immunofluorescence staining in the cells during cardiomyocyte differentiation. There was no detectable cTnT in undifferentiated MSCs as expected (A). The cells stained positive for cTnT after 1 week of coculture with CMs (B). Significantly increased level of cTnT was observed in the cells when treated with TSA at 500 nM (C) for 48 h, then cocultured with CMs (400x). There were detectable cTnT in the cells 1 week after 5-aza induction (D). There was only minimal expression of cTnT with weak immunofluorescence staining in the cells treated with TSA with the concentration up to 500 nM 1 week after differentiation (E). The expression of cTnT was dramatically increased with substantially enhanced intensity of fluorescence in the cells that was initially treated with 5-aza for 24 h, followed by exposure to TSA at the concentration of 500 nM (F). Scale bar: 200 μm.

The expression of cardiomyocyte-specific protein cTnT during differentiation. The expression profile of cardiomyocyte-specific protein cTnT was evaluated using Western blotting analysis in the cells during cardiomyocyte differentiation of MSCs. There was no detectable cTnT in undifferentiated MSCs as expected. When the cells were treated with 5-aza or TSA, there was a low, but detectable, level of cTnT in the cells after 1 week of differentiation. There was a concentration-dependent increase in cTnT level in the cells when treated with TSA alone (100, 300, and 500 nM) (A). There was a significantly increased protein level of cTnT in the cells that were pretreated with 5-aza and exposed to TSA (100, 300, and 500 nM) (B). Similar results on cTnT expression were observed in the cells cocultured with CMs. Treatment of the cells with TSA (100, 300, and 500 nM) significantly enhanced the expression of cTnT in these cells after 1 week of differentiation (C). The relative expression of cTnT in the cells with different treatments is summarized in (D). MSCs: bone marrow mesenchymal stem cells without treatment or coculture; 5-aza: MSCs treated with 10 μM for 24 h; Coculture: MSCs cocultured with CMs; 100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively; 5-aza+100, 300, 500: MSCs treated with 10 μM for 24 h, followed by treatment with 100, 300, and 500 nM TSA, respectively; Coculture+100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively, then cocultured with CMs. p < 0.05 compared with MSCs group; p < 0.05 compared with 5-aza group; p < 0.05 compared with coculture group.
However, there were significant differences in the percentages of cTnT-positive cells between the groups with different treatments as shown in Figure 5. In the control group, there were less than 1% of the differentiating cells that were cTnT positive after 1 week of culture. When the cells were pretreated with 5-aza, a little more than 10% of cells were positive for cTnT after 1 week of differentiation. On the other hand, there were close to 25% of cells stained positive for cTnT after 1 week of coculture with CMs. Interestingly, only up to 4% of cells were cTnT-positive after 1 week of differentiation in the group that were pretreated with up to 500 nM TSA. These data suggested that only a small portion of the cells differentiated into the cells that exhibited cardiac phenotype when induced with TSA or 5-aza, or cocultured with CMs in vitro alone.

The percentage of cTnT-positive cells after 1 week of differentiation. The percent cells positive for cTnT were determined in each group after 1 week of differentiation. In the control group, there were less than 1% of the cells that were cTnT positive after 1 week of culture. When the cells were pretreated with 5-aza, a little more than 10% of cells were positive for cTnT after 1 week of differentiation. On the other hand, there were close to 25% of cells stained positive for cTnT after 1 week of coculture with CMs. Interestingly, less than 5% of cells were cTnT positive in the group that was pretreated with up to 500 nM TSA. There was a concentration-dependent increase in the percentage of cTnT-positive cells when the cells were treated with 500 nM TSA in 5-aza-pretreated group (from 10.3% to 19.8%) and coculture group (from 24.5% to 30.2%) after 1 week of differentiation. These data suggested that TSA significantly enhanced the cardiac differentiation of MSCs after 5-aza induction and during coculture with CMs. MSCs: bone marrow mesenchymal stem cells without treatment or coculture; 5-aza: MSCs treated with 10 μM for 24 h; Coculture: MSCs cocultured with CMs; 100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively; 5-aza+100, 300, 500: MSCs treated with 10 μM for 24 h, followed by treatment with 100, 300, and 500 nM TSA, respectively; Coculture+100, 300, 500: MSCs treated with 100, 300, and 500 nM TSA, respectively, then cocultured with CMs. p < 0.05 compared with MSCs group; p < 0.05 compared with 5-aza group, p < 0.05 compared with coculture group.
TSA Significantly Enhanced Cardiac Differentiation of Mscs Treated with 5-Aza or Cocultured with CMs In Vitro
To evaluate the interaction between TSA and 5-aza or CM coculture on cardiac specification of MSCs, the cells were initially treated with 5-aza, then exposed to different concentrations of TSA at the beginning of differentiation, or first treated with different concentrations of TSA prior to coculture with CMs. The reason to treat the cells with TSA first in the coculture system was to avoid the exposure of CMs to TSA. As shown in Figures 2—5, the expression profiles of cardiac-specific genes and proteins including GATA-4, NKx2.5, MEF2c, and cTnT were similar in the cells during cardiac differentiation after induction with 5-aza or TSA or coculture with CMs alone, although the percentage of cTnT-positive cells was different. Interestingly, treatment of the cells with TSA dramatically enhanced the expression of the cardiac-specific genes and protein in a dose-dependent manner with as much as 35—40-fold increase in their mRNA levels when the 5-aza-treated cells were exposed to 500 nM TSA compared to undifferentiated MSCs. Similarly, the transcriptional expression of GATA-4, NKx2.5, and MEF2c was substantially upregulated by 40—50 times in the cells of coculture system when the cells were first treated with 500 nM TSA. The cTnT protein expression was also significantly increased in the cells by over threefold by TSA treatment (500 nM) in both 5-aza-treated group and coculture group as compared to the groups without TSA treatment. Increased intracellular cTnT level in the cells treated with TSA was also demonstrated using immunofluorescence staining in both 5-aza-treated group and coculture group (Fig. 3).
Treatment of the cells with TSA also significantly increased the percent cTnT-positive cells in both 5-aza-pretreated and coculture groups (Fig. 5). There was a concentration-dependent increase in the percentage of cTnT-positive cells by up to 92.6% (from 10.3% to 19.8%) and 23.9% (from 24.5% to 30.2%) when the cells were treated with 500 nM TSA in 5-aza-pretreated group and coculture group after 1 week of differentiation, respectively (p < 0.05). These data suggested that TSA significantly enhanced the cardiac differentiation of MSCs after 5-aza induction and during coculture with CMs.
Discussion
MSCs can be induced to differentiate into cardiac myocytes in vitro and in vivo. Currently, there are mainly two methods for inducing the commitment of MSCs into the cells that exhibit some degree of cardiomyocyte phenotypically: treatment with inducing agents like 5-aza or amphotericin and coculture with cardiac muscle cells (31,34,40). Unfortunately, neither method has been good enough to generate mature functioning cardiac myocytes from MSCs at significant levels of frequencies for cell-based therapies clinically (16,28). In the present study, we demonstrated that treatment of MSCs with TSA significantly enhanced the cardiac specification of MSCs with increased levels of expression of cardiac-specific genes and proteins, and frequencies of cardiac-committed cells when the cells were pretreated with 5-aza or cocultured with neonatal CMs.
Cardiac differentiation of MSCs is a very complex process that is currently under extensive investigation. The existing data suggest that microenvironment and cell-to-cell communications (both direct and indirect) are important factors that define the cardiac specification of MSCs (30,49). Coculture of MSCs with CMs is a typical setting to create an environment for the cells to interact with other cells and microenvironmental factors for cardiac differentiation. The data from the present study also demonstrated that coculture of MSCs without direct cell-to-cell contact with CMs was a reasonable option for this purpose. The demethylating agent 5-aza has been shown to promote the cardiac commitment of MSCs both in vitro and in vivo (6,31). Its low efficacy on inducing cardiac differentiation of MSCs is considered possibly due to the reduced response to other factors produced in the microenvironment that could be involved in the process of cardiac differentiation after the treatment of 5-aza (6).
Microenvironment and intercellular communications are critical to the differentiation of MSCs into cardiac lineage and maturation (19,46). Direct cell-to-cell contact has been shown to be important for the differentiation of MSCs into cardiomyocytes in vitro (35). Coculture of MSCs with neonatal CMs without direct cell-to-cell contact is reported to be insufficient to induce cardiac specification of MSCs in vitro (43). However, in the present study, we observed that a significant portion of the cells (over 24%) exhibited cardiac phenotype with expression of cardiac-specific genes and proteins after 1 week of differentiation during coculture of MSCs with neonatal CMs without direct cell-to-cell contact. Our results were consistent with a recent observation that demonstrated that 12.5% of MSCs became cardiomyocytes phenotypically after indirect coculture with CMs, while over 30% of the cells that expressed cTnT in direct coculture system with CMs (30). The reason(s) for the apparent differences in the outcomes of the experiments is unknown at this point. It might be related to different experimental conditions.
The balance of acetylation and deacetylation of nucleosomal histones is tightly regulated by histoneacetyltransferases and HDAC, and is critical to the modulation of chromatin structure and function as well as the regulation of gene expression (29). Inhibition of HDAC by SAHA and TSA leads to differentiation, cell cycle arrest, and apoptosis of many tumor cells (4,13,39). Recently, we observed that SAHA was more potent in promoting the cardiac differentiation of MSCs than 5-aza (12). However, cardiac differentiation of MSCs was not further improved by SAHA when the cells were pretreated with 5-aza. Like SAHA, TSA is a potent HDAC inhibitor that suppresses the activities of HDACs, including all 11 known human class I and class II HDACs (18,33). Treatment of human MSCs with TSA stimulates their osteogenic differentiation with increased expression of alkaline phosphatase and mineralization in vitro (11). TSA also enhances the differentiation of MSCs into hepatocyte-like cells with increased expression of hepatocyte-specific proteins including HNF3β, AFP, ALB, HNF1α, MRP2, and C/EBPα (38), and facilitate the differentiation of embryonic stem cells to cardiac myocytes and promote cardiomyogenesis (20,21).
In the present study, HDAC enzymatic activity was maximally inhibited by TSA at the concentration of 300 nM in MSCs. No further inhibition of HDAC activity was observed in the cells when TSA concentration was increased to 500 nM. However, unlike SAHA, treatment of MSCs with TSA alone had very limited efficacy on inducing their differentiation into cardiomyocytes with only 4% of the cells positive for cTnT after 1 week of differentiation. In addition, treatment of MSCs with combined SAHA and 5-aza did not show any improvement in their cardiac commitment as compared to the cells exposed to SAHA alone (14). In contrast, MSCs that were pretreated with 5-aza and then exposed to TSA exhibited a dramatic increase in the expression of cardiac genes and proteins, as well as cell population with cardiomyocyte phenotype. Clearly, these two agents were different in the induction of cardiac differentiation of MSCs, and the beneficial effects of TSA on cardiac differentiation of MSCs were beyond the inhibition of HDAC activity. It is, however, possible that SAHA and TSA may inhibit different isoforms of HDAC in MSCs. It has been shown that inhibition of HDACII suppressed the expression of genes for cardiac hypertrophy, while blocking HDACI attenuated the expression of the genes that were involved in the process of preventing myocardial hypertrophy (17,23). Recently, TSA has been shown to increase the expression of telomerase reverse transcriptase (TERT) in human MSCs in association with increased level of acetylation of histone H3 and H4 (37). It is known that TERT plays a critical role in the function of both human MSCs and embryonic stem cells including proliferation and differentiation (41,47). Further studies are needed to investigate the underlying mechanism(s) on the interaction between TSA and 5-aza and the differences between SAHA and TSA on cardiac differentiation of MSCs.
In the coculture experiments, TSA intervention significantly increased the expression of the early transcription factors of cardiac muscles GATA-4, NKx2.5, and MEF2c, as well as cTnT, while only slightly increased the percentage of cTnT-positive cells after 1 week of differentiation of MSCs. These data suggested that the predominant role of TSA in the cardiac differentiation of MSCs was likely to promote the process of maturation, not differentiation in the coculture system. Little is known on the maturation of the newly differentiated cardiomyocytes from MSCs or embryonic stem cells. Clearly, it was not related to HDAC activity since TSA had minimal effects on the expression of cardiac genes and proteins during cardiac differentiation of MSCs at the concentration of 300 nM with maximal inhibition of HDAC activity in the cells. It is possible that the early cardiac transcription factors NKx2.5 and GATA-4 play an important role in the maturation process in the cells treated with TSA (2). Further investigation is certainly warranted to elucidate the mechanism(s). One may argue that the small increase in the number of differentiated cardiomyocytes from MSCs above coculture after TSA treatment may have limited relevance to the actual cellular therapies clinically. However, the increased expression of cardiac specific genes and proteins in the cells may enhance the function of these cells on cardiac repair. Further studies (both in vitro and in vivo) are needed to evaluate the functional status of these MSC-derived cells after different treatment. In the present study, we did not evaluate the cardiac differentiation of MSCs that were pretreated with 5-aza in the setting of coculture since addition of 5-aza to the coculture system with neonatal CMs did not affect the outcome of cardiac commitment of MSCs (22). However, it could be interesting to determine if the combination of 5-aza and TSA treatments could promote the cardiac differentiation of MSCs in the setting of coculture with CMs.
In summary, the present study demonstrated that TSA significantly enhanced the cardiac differentiation of MSCs that were pretreated with 5-aza or cocultured with neonatal CMs with significant increase in the expression of cardiac genes and proteins, as well as the percentage of cTnT-positive cells after 1 week of differentiation with a mechanism beyond the inhibition of HDAC activity. The data suggested that combination of 5-aza induction with TSA intervention or pretreatment with TSA plus coculture with neonatal CMs was an effective method to generate cardiomyocytes from MSCs in vitro.
Footnotes
Acknowledgments
This study was supported by the National Science Foundation of China (Grant Number: 30973219), and the Foundation of Chongqing Science & Technology Commission (Grant Number CSTC, 2009BA5084), and partially supported by NIH R01 HL094650-01A2 (ZL). The authors declare no conflicts of interest.
