Abstract
Cardiovascular disease is the leading cause of death globally, and stem cell therapy remains one of the most promising strategies for regeneration or repair of the damaged heart. We report that human placenta-derived multipotent cells (hPDMCs) can modulate cardiac injury in small and large animal models of myocardial ischemia (MI) and elucidate the mechanisms involved. We found that hPDMCs can undergo in vitro cardiomyogenic differentiation when cocultured with mouse neonatal cardiomyocytes. Moreover, hPDMCs exert strong proangiogenic responses in vitro toward human endothelial cells mediated by secretion of hepatocyte growth factor, growth-regulated oncogene-α, and interleukin-8. To test the in vivo relevance of these results, small and large animal models of acute MI were induced in mice and minipigs, respectively, by permanent left anterior descending (LAD) artery ligation, followed by hPDMC or culture medium-only implantation with follow-up for up to 8 weeks. Transplantation of hPDMCs into mouse heart post-acute MI induction improved left ventricular function, with significantly enhanced vascularity in the cell-treated group. Furthermore, in minipigs post-acute MI induction, hPDMC transplantation significantly improved myocardial contractility compared to the control group (p=0.016) at 8 weeks postinjury. In addition, tissue analysis confirmed that hPDMC transplantation induced increased vascularity, cardiomyogenic differentiation, and antiapoptotic effects. Our findings offer evidence that hPDMCs can modulate cardiac injury in both small and large animal models, possibly through proangiogenesis, cardiomyogenesis, and suppression of cardiomyocyte apoptosis. Our study offers mechanistic insights and preclinical evidence on using hPDMCs as a therapeutic strategy to treat severe cardiovascular diseases.
Keywords
Introduction
Cardiovascular disease is the leading cause of death globally (14). A major obstacle in treating cardiovascular disorders is the limited intrinsic regenerative ability of the human adult heart. One of the most promising treatment strategies is cell transplantation to regenerate or facilitate repair of the damaged heart. Several different kinds of stem cells have been used for treatment of cardiovascular disorders with limited success, since the mechanisms underlying cell therapy to enhance myocardial recovery have mainly been through vasculogenesis and paracrine mechanisms, with only a few reports showing regeneration or replacement of actual cardiomyocytes (24).
Recent data suggest that the mesenchymal stem cell fraction of bone marrow may help with cardiac repair (2,34,35). However, bone marrow mesenchymal stem cells (BMMSCs) are rare progenitors, and their numbers further decrease with age (33). Placental tissue, on the other hand, originates from the early stage of embryonic development and contains fetal cells, which may retain the plasticity of early embryonic cells; moreover, the placenta is an immune-privileged site maintaining fetomaternal tolerance during pregnancy (7). Emerging evidence indicates that human term placenta-derived multipotent cells (hPDMCs) have the capacity of multilineage differentiation into cells with ectodermal, mesodermal, and endodermal phenotypes (9,41). These hPDMCs can be obtained without ethical concerns or the need for invasive procedures. Also, their strong immunosuppressive properties make these multilineage cells very good candidates for cell therapy to treat damaged organs (3,8).
Given this background, we hypothesized that hPDMCs would be good cellular candidates for cardiac repair. We found that hPDMCs constitutively express cardiomyogenic transcription factors. In vitro, hPDMCs can differentiate into cardiomyocyte-like phenotype and enhance angiogenesis by secreting proangiogenic factors. We further documented that transplantation of these cells into two in vivo models—a small animal mouse model and a large animal pig model—of myocardial infarction (MI) resulted in functional improvement. Tissue analysis confirmed increased vascularity, cardiomyogenesis, and suppression of cardiomyocyte apoptosis after hPDMC treatment. Our findings provide a rationale for further investigation into the utility of hPDMCs for the treatment of severe cardiovascular diseases.
Materials and Methods
Cell Culture
hPDMCs (three donors: two males, one female) were isolated and expanded as previously reported (41). Briefly, term (38-10 weeks’ gestation) placentas from healthy donor mothers were obtained with informed consent approved according to the procedures of the institutional review board. The specimens were washed several times in phosphate-buffered saline (PBS; Gibco-Invitrogen Corp., Grand Island, NY, USA) and then mechanically minced and enzymatically digested with 0.25% trypsin-EDTA (Gibco-Invitrogen) for approximately 10 min at 37°C. The cell suspension was cultured in Dulbecco's modified Eagle's medium (DMEM; Gibco-Invitrogen) supplemented by 10% fetal bovine serum (FBS; selected lots; HyClone, Logan, UT, USA), 100 U/ml penicillin, and 100 g/ml streptomycin (Sigma-Aldrich, St. Louis, MO, USA). Cell cultures were maintained at 37°C with a water-saturated atmosphere and 5% CO2. When cells were approximately 80% confluent, they were recovered with 0.25% trypsin/EDTA and replated at a dilution of 1:3. BMMSCs were purchased from Cambrex (three donors; East Rutherford, NJ, USA) and cultured as previously reported (29). Immunophenotyping and trilineage differentiation was performed as previously reported (41,42). Fibroblasts (Hs68) were obtained from American Type Cell Culture (ATCC, Manassus, VA, USA). Adult mouse cardiomyocytes (mCardio) were isolated from 6- to 8-week-old C57BL/6 mouse hearts (three males) after the animals received CO2 euthanasia. The hearts were dissected and immediately digested with collagenase solution (10 mg/ml collagenase A and 10 mg/ml collagenase B, both from Roche Diagnostics, Indianapolis, IN, USA) in 10 mM HEPES-buffered solution in 20% FBS (HEPES obtained from Sigma-Aldrich) at 37°C. After addition of IMDM high-glucose supplemented with 20% FBS, the cells were centrifuged at 1,000 rounds per minute for 3 min. The pellet was then resuspended in differentiation medium consisting of IMDM high glucose, 20% (FBS), 5,000 IU/ml penicillin/streptomycin, 200 mM L-glutamine, 0.15 mM 1-thioglycerol (all from Gibco-Invitrogen), and 50 μg/ml ascorbic acid (Sigma-Aldrich) and incubated on culture dishes for 1 h to separate cardiomyocytes from cardiac fibroblasts. Next, the floating cardiomyocytes were collected (12). Conditioned medium (CM) of cells was collected after 48 h of culture.
Animal Experimentation
The investigation conforms with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). Severe combined immunodeficiency (SCID) mice and C57BL/6 mice were obtained from the Laboratory Animal Center at National Taiwan University College of Medicine (Taipei, Taiwan). Minipigs (Lanyu breed) were obtained from Taitung Animal Propagation Station (Taitung, Taiwan). All animal experimentation was approved by Animal Resources at National Taiwan University College of Medicine and Far Eastern Memorial Hospital. The preliminary experiments for both mice and pigs included sham operation, which did not affect cardiac parameters as measured by ultrasound (data not shown).
Acute MI was induced in SCID mice (male, 3 months old) by permanent ligation of left anterior descending (LAD) coronary artery with 7-0 silk suture (Ethicon, Somerville, NJ, USA) at about 2 mm beneath the left atrial appendage after the animal underwent anesthetization via intraperitoneal injection of ketamine/xylazine combination (100 mg/kg, 10 mg/kg; both from Sigma-Aldrich), intubation with ventilator support, and left thoracotomy. Adequacy of anesthesia was monitored by lack of reflex to foot pinch. Infarct size (%) in the mouse heart was defined as (sum of epicardial and endocardial infarct lengths)/(sum of epicardial and endocardial lengths of a heart section) (38). Infarct size (%) in the pig heart was defined as (infarct area × left ventricular thickness) (cm3)/heart weight (g). After MI induction, a total of 106 hPDMCs in 25 μl culture medium (experimental group) or 25 μl culture medium only without cells (control/vehicle group) was injected into the border zone area in 5-μl portions immediately after MI induction. The wound was then closed layer by layer. At 29 days after MI, the mice were sacrificed by CO2 euthanasia, and the hearts were harvested and cryoembedded.
Lanyu minipigs (~1 year old, 45 to 70 kg) were sedated (atropine 0.04 mg/kg IM; Sigma-Aldrich), anesthetized (tiletamine/zolazepam combination: 2.2 mg/kg, 2.2 mg/kg IM; Boehringer Ingelheim Agrovet A/S, Hellerup, Denmark), intubated, and ventilated with isoflurane (MAC = 1.2-2.0% IH; Sigma-Aldrich). The animals underwent experimental MI by permanent ligation of LAD coronary artery just beyond the first diagonal branch after the chest was opened. A total of 107 hPDMCs in 2 ml culture medium (experimental group; six male pigs) or 2 ml culture medium only without cells (control/vehicle group; six male pigs) was injected into the border zone area in 100-μl portions immediately after MI induction. The wound was then closed layer by layer. At 8 weeks after MI, the animals were sacrificed by blood depletion after general anesthesia with tiletamine/zolazepam combination (2.2 mg/kg, 2.2 mg/kg IM). The hearts were harvested, fixed in 10% formalin, and embedded in paraffin.
Echocardiography
Echocardiography on mice was performed using a GE Vivid 7 Ultrasound machine (General Electric Company, Fairfield, CT, USA) with GE I13L probe (5.8 to 14.0 MHz) after the animal underwent anesthetization via intraperitoneal injection of ketamine/xylazine combination (100 mg/kg, 10 mg/kg) at days 1, 8, and 29 days post-MI. Echocardiography on minipigs was performed using Acuson Cypress with 3V2c probe (5 to 7 MHz; Siemens AG, Berlin, Germany) after the animal underwent general anesthesia with tiletamine/zolazepam combination (2.2 mg/kg, 2.2 mg/kg IM) at day 0 (before and immediately after MI), 4 weeks, and 8 weeks post-MI. The operators were blinded to the treatment strategies. Mean left ventricular ejection fraction (LVEF), end-diastolic dimension, and end-systolic dimension were obtained from M mode in the mid-left ventricular line after repeated measurement for three times.
Coculture of hPDMCs with Cardiomyocytes
Neonatal mouse cardiomyocytes were isolated from the hearts of neonatal (less than 3 days old) C57/BL6 mice after the animals received cervical dislocation. The hearts were minced and digested using collagenase solution (collagenase A 10 mg/ml and B 10 mg/ml from Roche Diagnostics in 10 mM HEPES buffered solution from Sigma-Aldrich in 20% FCS from Gibco-Invitrogen) at 37°C. After addition of IMDM high-glucose supplemented with 20% FBS, the cells were centrifuged at 500 × g for 3 min. The cells were suspended in differentiation medium containing IMDM, 20% FCS, 5,000 IU/ml penicillin/streptomycin, 200 mM L-glutamine (all from Gibco-Invitrogen), 1.5 × 10-4 M 1-thioglycerol (Sigma-Aldrich), and 50 μg/ml ascorbic acid (Sigma-Aldrich) (41), and preplated to allow preferential attachment of noncardiomyocytes. After 1 h, the nonadherent cells (predominantly cardiomyocytes) were collected (28) and mixed with hPDMCs at the ratio of 4:1. The mixed cells were cultured onto six-well culture dishes in differentiation medium and incubated for 8 days to study the cardiomyogenic differentiation potential of the hPDMCs.
Analysis of Gene Expression by RT-qPCR
RT-qPCR (reverse transcription-quantitative polymerase chain reaction) was performed as previously reported (42) using an ABI 7500 Real-Time PCR System and SYBR green (both from Applied Biosystems, Foster City, CA, USA). Dissociation curve analyses were performed using the instrument's default setting immediately after each PCR run according to the standard protocol provided by manufacturer. Primers were as follows: GATA4, forward primer: 5'-TCATCTCACTACGGGCACAG-3’, reverse primer: 5'-GGGAAGAGGGAAGATTACGC-3’; and HAND2, forward primer: 5'-GGCTCAGACTCGCCCCTGGA-3’, reverse primer: 5'-CCTCTCGTGCTCATACAAAGG-3’.
Adenoviral Transduction of hPDMCs
hPDMCs (104 cells per well of a 24-well plate) were transduced with the eGFP-encoding, first-generation adenovirus vector hAd5/F50.CMV.eGFP (20) using 100, 200, 300, or 500 μl of vector stock. After incubation for 2 h at 37°C the inoculum was replaced by 500 μl of culture medium, and the cells were further incubated at 37°C for 3 days. Approximately 80% of hPDMCs could be seen to be GFP+ after transduction.
Immunohistochemistry and Immunofluorescence
Murine hearts were cryoembedded, cut into 10-μM-thick cross-sections, and fixed with 4% paraformaldehyde in PBS. The primary antibodies were directed against von Willebrand factor (vWF; 1:200; Abcam, Cambridge, UK) or human lamin A/C antibody (1:200; Vector Laboratories, Burlingame, CA, USA) using Alexa Fluor-conjugated secondary antibodies (Gibco-Invitrogen) for visualization. Swine hearts were fixed with 10% formalin and embedded in paraffin prior to 5 μM sectioning. The sections were stained with antibodies specific for human HLA class 1 ABC (1:200; Abcam), cardiac troponin T (1:200; Santa Cruz Biotechnology, Dallas, TX, USA), connexin-43 (1:200; Sigma-Aldrich), and vWF (1:200; Abcam) using Alexa Fluor-conjugated secondary antibodies (Gibco-Invitrogen) for visualization. Cultured cells were fixed with 4% paraformaldehyde (Sigma-Aldrich) in PBS and stained with antibodies specific for human lamin A/C (1:200; Vector Laboratories), α-sarcomeric actinin (Sigma-Aldrich) using Alexa Fluor-conjugated secondary antibodies (Gibco-Invitrogen) for visualization. 4’, 6-Diamidino-2-phenylindole dihydrochloride (DAPI; Sigma-Aldrich) was used for visualization of cell nuclei.
Cytokine Array
Cytokine array experiments were performed as recommended by the manufacturer. Conditioned medium (1 ml obtained from 10 ml of medium used to culture 1 × 106 cells in a T75 flask for 48 h) obtained from culturing various types of cells [BMMSCs, hPDMCs, mouse cardiomyocytes (mCardio)] was mixed with a cocktail of biotinylated detection antibodies and then incubated on a qualitative cytokine array (RayBiotech, Norcross, GA, USA) for 2 h. Next, the array was incubated with streptavidin–horseradish peroxidase followed by chemiluminescent detection. After detection, the signals on the arrays were quantified to generate a protein profile using ImageJ software (US National Institutes of Health, Bethesda, MD, USA).
Angiogenesis Assay
Human umbilical vein endothelial cells (HUVECs; obtained from Bioresource Collection & Research Center, Hsinchu, Taiwan) were plated in 96-well plates (4 × 104 cells/well; all culture dishes/plates from Corning Life Sciences, Corning, NY, USA) coated with extracellular matrix gel (Sigma-Aldrich) in medium only, CM mix (1:1 M200:CM), or medium supplemented with hepatocyte growth factor (HGF; 20 ng/ml), interleukin-8 (IL-8; 50 ng/ml), growth-regulated oncogene a (GRO-α; 50 ng/ml), tissue inhibitor of metalloproteinase 1 (TIMP-1; 0.5 μg/ml), tissue inhibitor of metalloproteinase 2 (TIMP-2; 0.5 μg/ml; all from Peprotech, Rocky Hill, NJ, USA) as well as with certain combinations of these recombinant human growth factors as described. HUVEC tube formation was quantified according to published methods by counting the total number of branch points (5,13).
Tdt-Mediated dUTP-Biotin Nick-End Labeling (TUNEL) Staining and Quantification
Swine hearts were fixed with 10% formalin and embedded in paraffin prior to 5 μM sectioning. The paraffin sections were deparaffinized by xylene, absolute and graded series of ethanol, and washed. TUNEL staining was performed using In Situ Cell Death Detection Kit, POD (Roche) as per the manufacturer's protocol. The apoptotic cardiomyocytes were quantified as TUNEL+ cardiomyocytes % on heart sections at the border zone of infarcted hearts (31). Six different border zone areas were analyzed per heart.
Statistical Analysis
Numerical data are presented as mean ± standard error of the mean (SEM). We used two-tailed t-test to compare the mean between groups and used ANOVA with Bonferroni's post hoc to make multiple comparisons. Wilcoxon rank sum test was used to compare serial ejection fraction change between the hPDMC-treated and control murine hearts. Statistical analysis was performed using SPSS 13.0 software (IBM, Armonk, NY, USA). Values of p < 0.05 were considered statistically significant.
Results
hPDMCs Express Genes Involved in Cardiomyogenesis
hPDMCs have previously been shown to possess a surface marker profile similar to BMMSCs and the potential to differentiate into endodermal, mesodermal, and ectodermal cells (9,41) (Fig. 1A, B). To test whether hPDMCs have the potential to differentiate into cardiomyocytes, we first analyzed the expression in these cells of genes involved in cardiomyogenesis using RT-qPCR. For comparison, RNA from the human fibroblasts and from BMMSCs was analyzed in parallel (Fig. 1C). The expression levels of the cardiomyogenic transcription factors GATA4 and HAND2 (37) were significantly higher in hPDMCs than in fibroblasts (negative control) and BMMSCs, indicating the possible cardiomyogenic differentiation potential of hPDMCs (Fig. 1C). Thus, we hypothesized that hPDMCs might help modulate cardiac injury by differentiating into cardiomyocytes.

Characterization of hPDMCs and cardiomyogenic differentiation potential. (A) Surface marker expression profile and (B) trilineage differentiation potential of hPDMCs cultured in control/expansion medium compared with the respective differentiation medium. Staining for Alizarin red to detect calcium deposition for osteogenesis, Oil red O for oil droplet formation in adipogenesis, and Alcian blue for glucosaminoglycans detection in chondrogenesis was performed. (C) RT-qPCR analysis of GATA4 and HAND2 expression levels in, from left to right, human fibroblasts, human BMMSCs, and hPDMCs (three samples in each group). hPDMCs express high levels of the cardiomyogenic transcriptional factor genes GATA-4 and HAND2. (D) Transduction of hPDMCs with 500 μl of hAd5/F50.CMV.eGFP. (E) Coculture of eGFP-labeled hPDMCs with mouse neonatal cardiomyocytes. No beating eGFP+ cells were identified up to 14 days after coculture. (F) Immunostaining of representative hPDMCs (labeled with an antibody specific for human nuclear lamin A/C: green) for α-sarcomeric actinin (red) after 8 days of coculture with mouse neonatal cardiomyocytes (arrow heads). hPDMCs (arrows) stained positive for α-sarcomeric actinin with striation.
hPDMCs Can Differentiate In Vitro Into Cardiomyocyte-Like Cells
Because hPDMCs express high levels of certain cardiomyogenic transcription factors, we hypothesized that these cells could potentially differentiate into cardiomyocytes. To test this, hPDMCs were labeled genetically with enhanced green fluorescent protein (eGFP) using an adenovirus vector (20) (Fig. 1D) and cocultured with mouse neonatal cardiomyocytes, but no spontaneous beating could be seen in GFP+ cells (Fig. 1E) (32). However, after 8 days of coculture with mouse neonatal cardiomyocytes, approximately 20% of hPDMCs (labeled with human nuclear lamin A/C-specific antibodies: green) expressed the striated cardiomyocyte marker, a-sarcomeric actinin (red) (Fig. 1F). These data offered in vitro evidence that hPDMCs may have cardiomyogenic differentiation potential.
hPDMCs Promote Angiogenesis In Vitro
Because the placenta is a highly vascularized tissue, we hypothesized that hPDMCs might mediate proangiogenic effects through secreting paracrine factors. To address this question, we assessed the tube formation ability of HUVECs cultured in M200 medium (endothelial cell culture medium), DMEM (i.e., BMMSC expansion culture medium), CM of hPDMCs, BMMSCs, or adult mouse cardiomyocytes (mCardio). We found that tube formation by endothelial cells (HUVECs) was highest in hPDMC-conditioned medium (Fig. 2A), yielding 2,759 ± 106 branch points per cm2 (Fig. 2B). The branch point number of HUVEC tube formation was 1,710 ± 80 in M200 medium (p = 0.001 vs. hPDMCs), 840 ± 381 in DMEM (p = 0.008 vs. hPDMCs), 1,749 ± 194 in BMMSC-conditioned medium (p = 0.010 vs. hPDMCs), and 1,532 ± 289 in mCardio-conditioned medium (p = 0.016 vs. hPD MCs) (Fig. 2B). These results indicate that paracrine factors secreted by hPDMCs are likely involved in promoting angiogenesis (n=3 in each group; all data shown as mean ± SEM).

Proangiogenic effects of hPDMCs and identification of angiogenic factors secreted by hPDMCs. (A) Representative images of HUVECs maintained in M200 medium (i.e., HUVEC culture medium), DMEM, or conditioned medium of BMMSCs, mCardio, or hPDMCs. (B) Quantification of HUVEC tube formation ability in the indicated media. The propensity to form tubular networks is highest for HUVECs exposed to hPDMC-conditioned medium (n = 3 in each group; error bars ± SEM). (C) Cytokine array analysis for secreted factors by BMMSCs and hPDMCs. Complete medium was used as control. (D) Relative fold change of secreted factors in mouse cardiomyocyte-conditioned medium (mCardio), BMMSC-conditioned medium, and hPDMC-conditioned medium. Urokinase receptor (uPAR), glucocorticoid-induced tumor necrosis factor receptor (GITR), HGF, GRO-α, human CC chemokine-4 (HCC-4), and interleukin-6 receptor (IL-6R) were secreted only by hPDMCs. (E) Quantification of tube formation by HUVECs in single recombinant protein-containing medium, in comparison to DMEM and hPDMC-conditioned medium. Tube formation ability in HGF- or GRO-α-containing medium is higher (p < 0.05) than that in DMEM. (F) Quantification of tube formation by HUVECs in multiple recombinant protein-containing medium, in comparison to DMEM and hPDMC-conditioned medium. The medium containing HGF, GRO-α, and IL-8 significantly enhanced tube formation compared to DMEM, indicating an important proangiogenic role of these three factors, which are secreted in high amounts by hPDMCs (n = 3 in each group; error bars ± SEM).
hPDMCs Mediate Proangiogenesis Through Secretion of Hepatocyte Growth Factor (HGF), Interleukin-8 (IL-8), and Growth-Regulated Oncogene-α (GRO-α)
To identify which factors produced by hPDMCs could promote angiogenesis, we used a cytokine/chemokine array to identify proangiogenic factors in hPDMC-conditioned medium (Fig. 2C). For comparison, BMMSC-conditioned medium and complete medium only were also analyzed. The most abundantly secreted factors in hPDMC-conditioned medium were HGF, IL-8, GRO-α, TIMP-1, and TIMP-2 (Fig. 2D). Of these factors, only TIMP-1 and TIMP-2—reported to have antiangiogenic effects (17)—were also found in BMMSC-conditioned medium (Fig. 2D). To study the proangiogenic effects of the individual cytokines and their combined effects, we performed HUVEC tube formation assays using recombinant versions of the five most abundant factors in hPDMC-conditioned medium. In comparison to DMEM (branch point number: 1,689 ± 87; mean ± SEM), HGF- and GRO-α-treated HUVECs (2,262 ± 180, p = 0.046 compared to DMEM and 2,464 ± 228, p = 0.034, respectively) showed significantly higher tube-forming ability, whereas HUVECs treated with IL-8 did not (1,757 ± 140, p = 0.703) (Fig. 2E). Addition of TIMP-1, on the other hand, significantly decreased HUVEC tube formation ability (843 ± 183, p = 0.030), whereas TIMP-2 did not have any significant effects. To explore whether the hPDMC-secreted proangiogenic factors had any synergistic effects, we used different combinations of the three most highly secreted cytokines—HGF, GRO-α, and IL-8—to induce tube formation in HUVECs. Quantification of HUVEC tube formation revealed that the simultaneous addition of all three factors enhanced tube formation most significantly (1,735 ± 201 vs. 1,035 ± 73 branch points in DMEM only, p = 0.031; data shown as mean ± SEM). Collectively, HGF, GRO-α, and IL-8, which are the major cytokines secreted by the hPDMCs, appear to promote angiogenesis synergistically (Fig. 2F) (n=3 in each group; all data shown as mean ± SEM).
hPDMCs Modulate Cardiac Injury in a Mouse MI Model
To demonstrate that hPDMCs may promote heart repair, we induced MI in SCID mice by performing LAD coronary artery ligation. Immediately after injury, we either injected hPDMCs or culture medium only (vehicle) into the border zone area and assessed for cardiac function in vivo by performing echocardiograms immediately, 8, and 29 days postsurgery (Fig. 3A). A total of 18 SCID mice (nine receiving hPDMCs injection and nine receiving culture medium/vehicle injection) underwent experimental MI, with five dying soon after MI. Finally a total of seven SCID mice receiving hPDMC treatment and a total of six SCID mice receiving culture medium treatment survived. There was no difference in the infarct size between the cell treatment group (30.9 ± 10.1%) compared to vehicle-treatment group (30.0 ± 12.0%). The ejection fraction of post-MI hearts was less attenuated in the hPDMC-treated mice than in the control group during the 29-day follow-up (Fig. 3B, C), suggesting that hPDMCs may modulate cardiac injury.

Improvement of cardiac function in mouse MI model following hPDMC transplantation. (A) Schematic diagram of heart function evaluation after artificial MI induction, with subsequent intramyocardial hPDMC or culture medium only injection in SCID mice. (B) Left ventricular ejection fraction (LVEF) measured immediately and 29 days following MI in mice treated with hPDMCs (solid line) or injected with culture medium only (dotted line). (C) Comparison of LVEF change (i.e., ejection fraction at the measured time – ejection fraction immediately post-MI) after MI in mice treated with hPDMCs (solid line) or injected with culture medium only (dotted line) (p = 0.02) (n = 6 to 7 in each group; error bars ± SEM).
hPDMC Transplantation Enhances Vascularity in Post-MI Mice Hearts
Our mouse MI model studies showed functional improvement of heart function after hPDMC injection. To determine the underlying mechanism for the functional benefits of hPDMCs in cardiac repair, we analyzed heart sections from the post-MI SCID mice after either culture medium only (vehicle; n = 6) or hPDMC (n = 7) injection. Using human nuclear lamin A/C-specific antibodies, we did not detect hPDMCs in murine hearts at 29 days after MI induction (data not shown). Since increased vascularity has been shown as one of the mechanisms for improvement of cardiac function after cell therapy and our in vitro data also showed that hPDMCs might enhance angiogenesis (Fig. 2), we assessed for changes in blood vessel density in the heart sections of post-MI mice by staining for vWF, a marker for mature endothelial cells. Five sections at different parts of the hearts were analyzed. We found that in the heart sections of hPDMC-treated mice, vascularity was higher than in those of control/culture medium-treated animals at regions near the MI, non-MI, as well as the border zone (Fig. 4A). The vascularity (capillaries/ mm2) was 1,077 ± 128 versus 407 ± 21 (p = 0.003) in the MI zone, 1,630 ± 484 versus 351 ± 69 (p = 0.04) in the non-MI zone, and 1,736 ± 250 versus 600 ± 158 (p = 0.005) in the border zone for sections of the hPDMC- and culture medium-treated animals, respectively (data shown as mean ± SEM) (Fig. 4B). These results demonstrate the proangiogenic effects of hPDMCs in vivo and their therapeutic potential for ischemic heart disease.
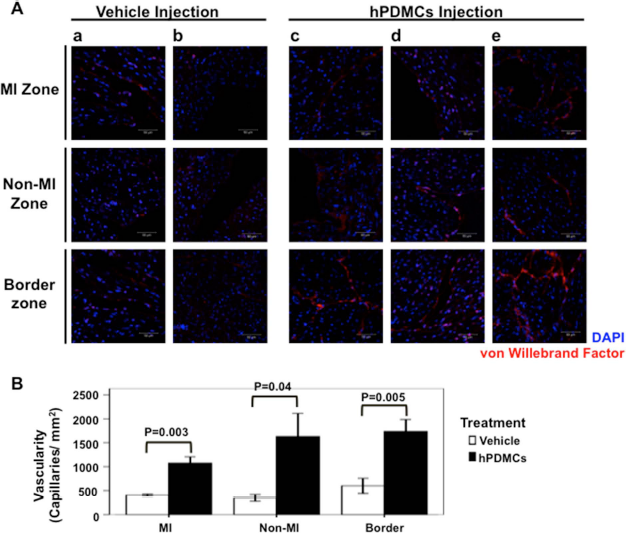
Proangiogenic effects of hPDMCs in post-MI murine hearts. (A) Representative heart sections from mice injected post-MI with culture medium only or hPDMCs were stained for vWF in the infarction zone, noninfarcted area, and border zone. (a and b) Different mice receiving culture medium injection; (c-e) different mice receiving hPDMCs injection. (B) Quantitative analysis of capillary density (capillaries per mm2) in heart sections of culture medium-treated (white) and hPDMC-treated (black) mice. From left to right: MI, non-MI, and border zone areas. The vascular density is significantly higher in the hPDMC-treated hearts than in those injected with culture medium, indicating the proangiogenic effects of hPDMCs (n = 6 to 7 in each group; error bars ± SEM).
hPDMCs Modulate Cardiac Injury in a Pig MI Model
We have shown that hPDMCs have cardiomyogenic potential and proangiogenetic effects in vitro and demonstrated that transplantation of hPDMCs in a mouse MI model might attenuate adverse cardiac remodeling. To apply this therapeutic strategy preclinically, we induced MI in 1-year-old Lanyu minipigs by permanent coronary ligation of the LAD artery just beyond the first diagonal branch after open chest surgery. hPDMCs or culture medium only was then injected intramyocardially into the border zone area. Echocardiography was performed immediately, 4 weeks, and 8 weeks after MI and hPDMCs or culture medium-only treatment (Fig. 5). Coronary artery ligation resulted in anterior wall hypokinesia/akinesia or dyskinesia (Fig. 5B, C). A total of 17 minipigs underwent experimental MI, with five dying during or soon after operation. Finally, a total of six pigs receiving hPDMC treatment and six pigs receiving culture medium-only treatment survived. The LVEF at baseline and immediately after MI in each group was similar (Fig. 5D). Heart contractility in the culture medium-only treatment group was seen to deteriorate over time. By contrast, heart contractility in the hPDMC treatment group improved with time. At 8 weeks post-MI, LVEF in the hPDMC treatment group was significantly improved compared to the culture medium-only treatment group (53.8 ± 4.4% vs. 39.3 ± 2.3%, p =0.016). These data support that hPDMCs modulate cardiac injury in a large animal model.

Enhancement of heart contractility in pig MI model after hPDMC treatment. (A) Schematic diagram of heart function evaluation after experimental MI induction with subsequent intramyocardial hPDMC or culture medium injection in a minipig model (left). Scar formation (arrowheads) at anterior wall and interventricular septum was noted in the heart at 8 weeks post-MI (right). (B) Permanent ligation of left anterior descending coronary artery resulted in anterior wall dyskinesia with erythematous change (arrowhead). (C) Echocardiogram at 4 weeks after MI showing anterior wall akinesia (arrows). (D) A serial measurement of left ventricular ejection fraction (LVEF) at baseline before MI, immediately, 4 weeks, and 8 weeks after MI in minipigs treated with hPDMCs (solid line) or injected with culture medium only (dotted line) (p = 0.016) (n = 6 in each group; error bars ± SEM).
hPDMCs Transplantation Promotes Angiogenesis in the Post-MI Pig Hearts
We have shown that the heart function improved significantly after hPDMC injection into post-MI pig hearts. To explore the underlying mechanism involved in the modulation of cardiac injury by hPDMCs in the swine MI model, we harvested the hearts from the post-MI minipigs with culture medium-only or hPDMC treatment at 2 months post-MI (n = 6 in each group) (Fig. 6A). To exclude the possibility that the improvement of cardiac function in the hPDMC treatment group was due to the different infarct size between the culture medium-only and hPDMC treatment groups, we measured the infarct size in culture medium-only and hPDMC-treated hearts and found that there were no significant differences between the two groups (20.1 ± 6.8% in culture medium treatment group vs. 20.1 ± 8.0% in hPDMC treatment group, p = 0.87) (Fig. 6B). MI was confirmed by staining with Masson's trichrome stain, in which cardiac fibrosis can be seen as staining blue (Fig. 6C). To assess if hPD-MCs promote angiogenesis in the post-MI swine model, we analyzed for changes in vascularity by assessing for capillary density in the heart sections of post-MI pigs with staining for vWF. Five sections at different parts of the heart border zone area were analyzed. We found that in the heart sections of hPDMC-treated pigs, vascularity was higher than in those of culture medium-treated animals at the border zone (Fig. 6D). The vascularity (capillaries/ mm2) was 2,618 ± 145 versus 738 ± 134 (p < 0.001) in the border zone for sections of the hPDMC- and culture medium-treated animals, respectively (data shown as mean ± SEM) (Fig. 6E). We did not find that the endothelial cells (vWF+ cells) stained positive for human HLA-ABC antibody, however, implicating that these proangiogenic effects were likely mediated through paracrine factors. Collectively, these results demonstrate the proangiogenic effects of hPDMCs in the post-MI hearts of large animals.

hPDMC treatment promotes angiogenesis in post-MI pig hearts. (A) Representative figures of pig heart slices after culture medium only or hPDMC intramyocardial injection at 8 weeks post-MI. (B) Quantitative analysis of heart infarct size at 8 weeks post-MI in the culture medium- and hPDMC-treated pigs (p = 0.687). (C) Masson's trichrome stain in post-MI pig hearts for fibrosis. Representative figures of pig heart slices after vehicle or hPDMCs intramyocardial injection at 8 weeks post-MI. Cardiac fibrosis was stained blue. Note in the heart treated with hPDMCs, viable myocardium existed among fibrosis tissue. (D) Representative heart sections from pigs injected post-MI with culture medium only or hPDMCs were stained for vWF in the border zone area (green, labeled by anti-vWF antibody). (a-d) Different pigs receiving culture medium only treatment; (e-h) different pigs receiving hPDMC treatment. (E) Quantitative analysis of capillary density (capillaries per mm2) in heart sections of culture medium-treated (white) and hPDMC-treated (black) pigs. The capillary density is significantly much higher in the hPDMC-treated hearts than in those injected with culture medium only, implying the proangiogenic effects of hPDMCs (p < 0.001) (n = 6 in each group; error bars ± SEM).
In Vivo Evidence That Implanted hPDMCs Engraft in Pig Heart and Differentiate Into Cardiomyocyte-Like Phenotype
To confirm if hPDMCs can contribute to cardiomyogenesis in the post-MI pig heart, we analyzed the heart sections from the post-MI minipigs with hPDMC treatment at 2 months post-MI using anti-human HLA-ABC antibody to detect hPDMCs. We found that hPDMCs were retained in the pig heart at 8 weeks post-MI in the hPDMC treatment group (Fig. 7A). To further characterize if hPDMCs differentiate into cardiomyocytes, we costained the sections with cardiac troponin T (cTnT)—a marker for striated cardiomyocytes—and found that the hPDMCs within the pig heart stained positively for this marker (Fig. 7B). Moreover, staining for the gap junction protein, connexin-43, between implanted and neighboring/host cells was positive (Fig. 7C), suggesting that the injected hPDMCs are integrated with the surrounding swine cardiomyocytes. These data suggest that hPDMCs can engraft long term in injured swine hearts and differentiate—or at least contribute—to cardiomyogenesis.

hPDMCs contribute to cardiomyogenesis in pig hearts. (A) Representative heart sections from post-MI, hPDMC-injected pigs (green fluorescence, labeled by anti-human HLA-ABC antibody; arrowheads) shown with retained hPDMCs at 8 weeks after MI induction and hPDMCs injection. Cell nuclei visualized with DAPI staining. (B) In vivo evidence that hPDMCs might contribute to cardiomyogenesis. Colocalization of human HLA-ABC antibody (green), representing hPDMCs, and cardiac troponin (cTnT; red fluorescence), representing cardiomyocytes, from representative heart sections of post-MI, hPDMC-injected pigs. (C) Representative heart sections from post-MI, hPDMC-injected pigs (green fluorescence, labeled by anti-human HLA-ABC antibody; arrows showing staining for the gap junction protein connexin-43 (red fluorescence; arrowheads) between retained hPDMCs and neighboring host cells at 8 weeks after MI induction and cell injection. Cell nuclei visualized with DAPI staining.
hPDMCs Inhibit Apoptosis in Porcine Cardiomyocytes
To investigate if proangiogenesis and neomyogenesis of hPDMCs might result in suppression of cardiomyocyte apoptosis, we used TUNEL staining to detect fragmented DNA in cardiomyocytes after hPDMCs or culture medium-only treatment in the pig myocardial infarction model. We found that the percentage of TUNEL-positive cardiomyocytes at the border zone area of pig hearts at 8 weeks after MI was lower in the hPDMC treatment group (Fig. 8A). Quantification analysis showed TUNEL-positive cardiomyocytes (%) was 5.40 ± 0.8% versus 12.22 ± 1.0% (p < 0.001) in the border zone for sections of the hPDMC- and culture medium-treated pigs, respectively (data shown as mean ± SEM) (n = 6 in each group) (Fig. 8B).

hPDMCs suppress cardiomyocyte apoptosis in pig hearts. (A) Representative TUNEL stain at border zone area of pig heart slices after culture medium only or hPDMC intramyocardial injection at 8 weeks post-MI. (a, b) Different pigs receiving culture medium-only treatment; (c-d) different pigs receiving hPDMC treatment. (B) Quantitative analysis of TUNEL-positive cardiomyocyte percentage at border zone area of pig hearts at 8 weeks post-MI. The TUNEL-positive cardiomyocytes were significantly lower in the hPDMC-treated group than in the culture medium-treated group (p < 0.001) (n = 6 in each group; error bars ± SEM).
Discussion
Improvements in infectious agent control and the continued westernization of the developing world have propelled ischemic heart disease and heart failure to become leading causes of morbidity and mortality worldwide. Myocardial infarction, the most common cause of such diseases, results in profound cardiomyocyte loss, which currently is without cure. Current treatments do not reverse the loss of myocardium but are mainly aimed at reducing risk factors, including hypertension and hypercholesterolemia, or at ameliorating disease severity with the use of antiplatelet agents, β blockers, aldosterone antagonists, or angiotensin-converting enzyme inhibitors/angiotensin receptor blockers. Cardiomyocyte turnover has been recently documented in human by 14C labeling, with very low annual turnover rates of 0.2% to 2% (6). This limited ability of cardiomyocytes to regenerate is further decreased with age and clearly insufficient to compensate for the severe myocardial loss resulting from disease, such as MI. Thus, there is continued interest in the use of stem cells to treat ischemic heart diseases and/or heart failure to remedy this critical issue. Several types of cells including stem cells and progenitor cells such as skeletal myoblasts (25) and bone marrow mononuclear cells (2,10) have been applied in MI patients. However, myoblast implantation in infarcts was shown to increase ventricular arrhythmia events with failure to improve cardiac function (4). Meta-analyses of bone marrow mononuclear cell injection in MI patients showed a small 2-4% statistically significant improvement in left ventricular function, levels, which are unclear with regard to clinical relevance (11,18,44). Moreover, in these studies in which the use of stem cells/progenitor cells have demonstrated cardiac function improvement, direct evidence to elucidate exact mechanisms involved were often not clearly shown (24,30), even in trials involving BMMSCs (1,15). In this study, we show that the use of hPDMCs, an ethically compliant source of multilineage cells, can result in functional improvement in two animal models of MI: a small animal mouse model and a large animal swine model. Overall, the beneficial effects of hPDMC treatment likely involve cardiomyogenic, proangiogenic, and antiapoptotic effects, as supported by in vitro and in vivo evidence.
In comparison to adult bone marrow mononuclear cells and BMMSCs, which have been used in animal studies and widely applied in clinical trials to treat post-MI heart failure, hPDMCs potentially have several advantages over adult cells. First of all, the expansion capacity and proliferation rate of hPDMCs are much higher than similar stem cells from bone marrow (16), making cell preparation much more efficient and feasible for clinical use in which high cell volumes are necessary. Second, hPDMCs show stronger paracrine proangiogenic effects than BMMSCs, as assessed by endothelial cell tube formation assay (Fig. 2B), and accumulating evidence suggests that improvement of angiogenesis may be the dominant mechanism by which bone marrow stem cell therapy results in functional improvement post-MI (21,43). hPDMCs highly secrete HGF and IL-8, both well-known proangiogenic factors (26,39), as well as GRO-α (also known as CXCL1), which we show to be strongly angiogenic as well. While we found that both hPDMCs and BMMSCs secrete TIMP-1, a factor reported to have antiangiogenic effects (17), the high levels and synergistic proangiogenic effects of hPDMC-secreted HGF, IL-8, and GRO-α strongly enhanced vascularity overall. In addition to the paracrine effect of proangiogenesis, we found that undifferentiated hPDMCs highly express a number of cardiomyocytic transcription factors, compared to BMMSCs (Fig. 1). Indeed, one report has shown that human placenta-derived stem cells may differentiate in vitro into cardiomyocytes using coculture systems (27), results that we have also found based on in vitro expression of α-sarcomeric actinin. More evidence in terms of expression of other cardiomyogenic markers such as Nkx2.5 or spontaneous beating would be necessary to establish definitive in vitro differentiation of hPDMCs into cardiomyocytes. While two reports have also shown placenta-source MSCs to have therapeutic effects in animal models of MI, mechanisms of action were not clearly dissected, and pretreatment of transplanted cells with specific agents was necessary for sarcomeric-like features or connexin-43 positivity to occur (36,40). We speculate that these differences from our data could be due to isolation method differences between ours and these studies, resulting in the isolation of populations of placenta-derived stem cells that are functionally distinct (3,41). Collectively, the various distinctive characteristics of hPDMCs may explain their in vitro capacities and ability to modulate cardiac injury in vivo as we demonstrated in both small and large animal models of MI. The data strongly support that hPDMCs may be particularly suited for use in cardiac diseases.
In this study, we reveal a novel role for hPDMCs in the inhibition of cardiomyocyte apoptosis in vivo using a pig MI model. The result may be due to direct antiapoptotic effect or indirect effect by proangiogenetic effect of hPDMCs. Future research is needed to identify the direct antiapoptotic targets of hPDMCs on suppression of cardiomyocyte apoptosis.
For both our animal models, we chose to inject hPDMCs immediately post-MI, rather than several days later as has been commonly done. The optimal timing of stem cell therapy for MI has long been debated, and a recent study has shown that only the delivery of therapeutic agents immediately post-MI resulted in any efficacy (22). Our data indicate that the therapeutic effects of hPDMCs in the swine model were still evident up to 2 months, which is a time point significantly longer than that found in most other large animal studies (19,22,23). This finding of such dramatic LVEF improvement—a nearly 15% improvement in LVEF with hPDMC treatment—in a large animal model beyond 1 month's time is truly exciting and has strong implications for the use of this cell type in a clinical setting for heart diseases.
In summary, our findings strongly implicate that cardiomyogenesis, proangiogenesis, and antiapoptotic effect on cardiomyocytes of hPDMCs might play important roles in modulating cardiac injury. We offer here evidence from bench to small and large animal study demonstrating that hPDMC implantation can result in dramatic and significant functional cardiac improvement. Our data suggest that hPDMC therapy potentially offers a new therapeutic strategy for cardiac protection and treatment of ischemic cardiovascular diseases and heart failure.
Footnotes
Acknowledgments
This study was supported by grants from the National Science Council of Taiwan (NSC 98-3111-B-002-009, NSC 99-3111-B-002-005, NSC 99-3114-B-002-006, NSC 100-2314-B-002-052, NSC 101-2314-B-002-093-MY3, NSC 101-2321-B-002-058) to M.-L.Y. and grants of Far Eastern Memorial Hospital (FEMH-2012-C-011, FEMH-2012-SCRM-A-005) to Y.-H.L. The authors declare no conflicts of interest.
