Abstract
Stem cell transplantation procedures using intraparenchymal injections cause tissue injury in addition to associated surgical risks. Intravenous cell administration give engraftment in parenchymal lesions although the method has low efficacy and specificity. In pathological conditions with inflammation, such as traumatic brain injury, there is a transient up-regulation of ICAM-1 and VCAM-1 which might provide environmental cues for migration of stem cells from blood to parenchyma. The aim of this study was to i) analyze the effect of intra-arterial administration on cellular engraftment, ii) compare engraftment and side effects between three different stem cell systems, and iii) analyze gene expression in these three systems. We performed specific intra-arterial transplantations with human mesenchymal stem cells (hMSCs), human neural progenitor cells (hNPCs), and rat neural progenitor cells (rNPCs) in a rat model of traumatic brain injury. These results were compared to the intravenous route for each cell type, respectively. Analysis of engraftment and recipient characterization was performed by immunohistochemistry. We further characterized the different types of cells by microarray and RT-qPCR analysis. Specific intra-arterial transplantations produced significantly higher engraftment compared to intravenous transplantation with hMSCs and rNPCs. No engraftment was detected after intra-arterial or intravenous administration of hNPCs. Characterization of integrin expression indicated that CD49dVCAM-1 and possibly ICAM-1 interactions through CD18 and CD11a, respectively, are important for engraftment after intravascular cell administration. No side effects, such as thromboembolic complications, were detected. When translating stem cell therapies to clinical practice, the route of transplantation and the properties of the cell lines (homing, diapedesis, and migration) become important. This study supports the use of selective intra-arterial transplantation for improving engraftment after traumatic brain injury. In addition, we conclude that careful analysis of cells intended for local, intra-arterial transplantation with respect to integrin expression is important.
Keywords
Introduction
Emerging stem cell therapies aimed at treatment of different central nervous system (CNS) disorders show great promise (7). Different stem cell lines have been transplanted in a wide spectrum of pathological conditions, both in preclinical animal models and in a few clinical phase I and II studies (4,26). Cell-based therapies are in clinical trials in, for example, Parkinson's disease (13,14), ischemic stroke (4,21), and spinal cord lesions (29). When comparing intra-arterial and intravenous transplantations in clinical trials following spinal cord injuries patient outcome seems to favor an intra-arterial approach (29). Evidence is gathering for both open surgical and different intravascular methods of transplantation to the CNS (4) and there is an ongoing discussion of routes of delivery in cell transplantation (31), as illustrated by a recent publication comparing intracerebral and intravenous administration (18). This holds true especially for stem cell cultures aimed at intravascular administration. Studies comparing different methods of transplantation show that the largest engraftment was obtained via the intrastriatal route as compared to intravenous and intraventricular methods (19) and via the selective intra-arterial route as compared to an intravenous method (20). For all catheter-based and needle-based injections the effect on cell cultures on the passage through the systems appears to be limited (17). In contrast to the hypothesis that transplanted cells have their effect in a damaged area in situ, some studies show that engraftment might not even be important for reduction in stroke lesion size and functional outcome (8). Engraftment is, however, a necessity when a curative treatment with neural network integration is the desired result.
Moreover, when translating cell treatment to clinical trials, the surgical risks of intraparenchymal CNS transplantation are presumably higher than intravenous or specific intra-arterial methods. In this context, it is interesting to compare the intra-arterial route of administration with intraparenchymal injection, which has a reported aggregated risk due to burr holes and placement of electrodes from deep brain stimulation. Diverging studies report complication rates in the range from very low to 8% risk of hemorrhage (6,12). Notably, it is probably necessary to establish a number of burr holes to accommodate for several injection trajectories, thereby multiplying the patient risk. On the other hand, selective intra-arterial transplantation technique coupled with preinterventional imaging-based planning creates the possibility to selectively cover an injured volume supplied by several target vessels (2). This targeting could thereby mitigate the surgical risks of a high number of injection trajectories following, for example, ischemic stroke. Even though the anticipated risks for endovascular transplantation also need to be taken into consideration, these would probably be lower than the surgical risks. Cell transplantations through the vertebral artery showed no adverse events in a smaller number of patients (29). In the clinical setting, different methods will probably be assessed from patient to patient.
For all intravascular routes of transplantation, the properties of the cells themselves are important for engraftment. The integrin families expressed in transplanted cells have implications for the actual diapedesis and/or migration within the parenchyma of the receiving organ as well as outcome parameters (16). CD49d has been implicated in cellular homing from the blood stream, as the multiple sclerosis pharmaceutical monoclonal antibody drug Natalizumab that target it (28). With intravascular approaches to stem cell transplantation, fluorescence-activated cell sorting (FACS) for CD49d yields an improved engraftment and better neurological outcome (16). It seems important with a first passage through the target organ in order to secure the largest possible engraftment (23). This is an even more delicate consideration if the source of transplantable cells are limited or associated with difficult ethical considerations.
This study was designed to investigate transplantation and consequent engraftment of three different stem cell systems, human mesenchymal stem cells (hMSCs), human neural precursor cells (hNPCs), and rat neural precursor cells (rNPCs) in a model of traumatic brain injury (TBI) in the rat. We have then characterized and compared the different cell types to further elucidate different target integrins that could be pivotal for engraftment after intravascular transplantation.
Materials and Methods
Ethical Statement
All human cell isolations were approved separately by either the Southern or the Northern Regional Ethics Committee of Stockholm. All animal handling and protocols were approved by the Northern Regional Ethics Committee of Stockholm.
Cell Preparation
hMSCs were isolated from bone marrow taken from the iliac crest of two healthy female volunteers. Heparinized bone marrow was mixed with a double volume of phosphate-buffered saline (PBS), centrifuged at 900 × g, resuspended, and layered over a Percoll gradient (1.073 g/ml, Sigma-Aldrich, St. Louis, MO, USA). The mononuclear cells were collected from the interface, washed, and resuspended in hMSC medium consisting of Dulbecco's modified Eagle's medium-low glucose (DMEM-LG) (Life Technologies, Gaithersburg, MD, USA), supplemented with 10% fetal bovine serum (Sigma-Aldrich) and 1% antibiotic-antimycotic solution (Life Technologies). The serum lot was selected on the basis of optimal MSC growth and differentiation. The cells were plated in culture flasks (Becton Dickinson Biosciences, Bedford, MS, USA) at a density of 160,000 cells/cm2. Nonadherent cells were removed after 48–72 h and the adherent cells were cultured. When >70% confluence was reached, the cells were detached by trypsin and ethylenediaminetetraacetic (EDTA) (Gibco BRL, Grand Island, NY, USA) and replated at a density of 4000 cells/cm2. The cells were harvested in passage 3 and classified as hMSCs based on their ability to differentiate into bone, fat, and cartilage and by flow cytometric analysis (positivity for CD73, CD90, CD105, and CD166, negativity for CD14, CD31, CD34, CD45, and CD80) (15).
hNPCs were derived from the subcortical forebrain of human first trimester embryos according to the method of Piao et al. (24) with a small modification according to Carpenter et al. (9). The embryonic tissue was retrieved from elective routine abortions performed at the Department of Gynecology, Karolinska University Hospital, after written informed consent by the pregnant woman. The embryonic brain was identified in the rinsing fluid and the subcortical forebrain was isolated. Mitotically active hNPCs, with the majority immunoreactive for the progenitor marker nestin, a smaller population expressing markers of more differentiated neurons (β-tubulin III) or astrocytes (glial fibrillary acidic protein) and also displaying the corresponding morphology underwent mechanical dissociation with a glass Teflon homogenizer in DMEM/F12 (1:1, Life Technologies); 2–4 million cells were cultured in an 80-cm2 flask in DMEM/F12 with the addition of 0.6% glucose (Sigma), 5 mM HEPES (Life Technologies), 2 μg/ml heparin (Sigma), 1% N2 supplement (Life Technologies), 20 ng/ml basic fibroblast growth factor (FGF), 20 ng/ml epidermal growth factor, and 10 ng/ml ciliary neurotrophic factor (all from R&D systems) in a humidified 37°C atmosphere with 5% CO2. After formation of free-floating neurospheres, the cells were passaged every 7–10 days using incubation in TrypLE Express (Invitrogen) for 4–5 min at 37°C followed by gentle trituration. At passage, trypan blue staining was used as viability test and for cell counting, and cells were reseeded at the original density. Before transplantation, cells were mechanically dissociated and prepared to a final concentration of 50,000 cells/μl.
For generation of rNPCs, cortices from rats at embryonic day 15 were dissected and mechanically dispersed in a modified serum-free, HEPES-free N2-supplemented DMEM/F12 medium (Invitrogen, Carlsbad, CA, USA). The primary cells were plated on dishes (35, 60, 100 mm) coated with poly-L-ornithine and fibronectin (Sigma). The cells were treated with human recombinant basic FGF2 at 10 ng/ml every 24 h and the N2 medium was replaced every 48 h. Cells were labeled with 20 μM bromodeoxyuridine (BrdU) for 24 h and transferred to Hank's balanced salt solution prior to engraftment.
Animal Preparation
A total number of 34 female Sprague-Dawley rats (BW 250 g; B&K Universal AB, Stockholm, Sweden) were included in the study. They were anesthetized with an intramuscular injection of 0.2 ml Hypnorm-Dormicum [1:1:2; Hypnorm (fentanyl citrate 0.315 mg/ml, fluanisone 10 mg/ml, Janssen Pharmaceutical, Beerse, Belgium)/Dormicum (midazolam 1 mg/ml, Roche AB, Stockholm, Sweden)/dH2O]. Prior to skin incision, 0.1 ml Xylocaine (5 mg/ml, Astra, Södertälje, Sweden) was injected subcutaneously in the sagittal midline of the skull or in the midline of the neck. TBI surgery was conducted in a stereotactic frame.
The animals were randomly assigned to intra-arterial, hMSC (n = 5), hNPC (n = 5), rNPC (n = 6) or intravenous hMSC (n = 5), hNPC (n = 6), rNPC (n = 5) stem cell transplantation. Within these groups, each animal received 2,500,000 cells during the transplantation procedure. The animals were then euthanized at 1 day posttransplantation.
Contusions were performed by using a weight-drop model, according to procedures previously described (11). Briefly, the animal was mounted in a stereotactic device, a craniotomy was performed 3 mm posterior and 2.5 mm lateral to bregma. A 24-g weight was dropped, guided by a piston, from a height of 9.3 cm to a foot plate placed on the exposed dura, leading to a maximum compression of 3 mm. After impact, the scalp was sutured and the animals were allowed to recover in their home cages. The contusion model used in this study does not cause any gross neurological symptoms, and the animals show normal behavior.
Transplantation
One day following TBI, the animal was reanesthetized and a neck exploration was performed. The transplantation procedure for intra-arterial administration included ligation of the ipsilateral external carotid artery. Thereafter, via the stump of the external carotid artery, cannulation of the internal carotid artery was performed with a nitinol alloy catheter (nitinol superelastic tube with outer diameter 0.193 ± 0.0127 mm, inner diameter 0.104 ± 0.0127 mm, and length 300 mm (Tube NiTi SE 508, ground surface, Euroflex GmbH, Pforzheim, Germany) connected to a microsyringe. After securing access to the vascular system, the nitinol catheter was advanced in the internal carotid artery, passing the origin of the pterygopalatine artery towards the skull base. Cell suspensions were then infused during 2 min. Blood flow in both the common carotid and internal carotid artery was preserved during the entire transplantation procedure and only the external carotid artery remained ligated after surgery. For intravenous transplantation, the nitinol catheter was inserted into the ipsilateral internal jugular vein via a neck incision and advanced through the superior thoracic aperture past the subclavian vein. The neck approach for intravenous administration was chosen to mimic the surgical trauma of the intra-arterial administration. Two hours postoperatively, animals were scored according to the Bederson scale for stroke classification (5). The Bederson scale is a scale from 0 to 3, where 0 stands for no neurological deficit, 1 stands for forelimb flexion when suspended in the tail, 2 stands for diminished resistance to lateral pushing, and 3 stands for circling behavior.
Tissue Handling and Immunohistochemistry
The animals were anesthetized and decapitated 1 day after the transplantation, the brain was removed and quickly frozen in isopentanedry ice before cryosectioning. The lower left lobe of the lung, the right liver lobe, and the spleen were also dissected and frozen in the same manner. Coronal 14-μm cryosections of the brain and the other organs were taken using a Leica cryostat (CM 3000, Leica Instruments GmbH, Nussloch, Germany). The sections were thaw mounted onto Super Frost/Plus object glasses (Menzel-Gläzer, Braunschweig, Germany) and stored at −20°C prior to use. Frozen sections were air dried, rehydrated in 1× PBS, fixed in 4% buffered paraformaldehyde for 10 min at room temperature (RT), rinsed in 1× PBS, and blocked with normal horse serum 15 μl/ml in 1% bovine serum albumin and 0.3% Triton X-100. Human nuclear antigen (HuN), a protein found in human cell nuclei but not in rodent nuclei, primary antibodies (MAB1281, Chemicon International, Temecula, CA, USA), or BrdU antibodies (Dako A/S, Denmark) were added for incubation overnight at +4°C. For internal negative controls, slides were incubated with PBS. The following day, sections were rinsed in 1× PBS and incubated with biotinylated anti-mouse IgG (Vector Laboratories, CA, USA) for 1 h at RT. After washing in 1× PBS, the sections were incubated with ABC kit solution (Vector Laboratories) for 1 h at RT. Following another rinsing in 1× PBS, 3,3′-diaminobenzidine (DAB) (Vector Laboratories) was added for 3 min at RT. Cell counting was performed by light microscopy with a Leica DM 4000 B (Leica Instruments GmbH).
For fluorescent immunohistochemistry (IHC) staining, frozen sections were air dried, rehydrated in 1× PBS, fixed in 4% buffered paraformaldehyde for 10 min at RT, rinsed in 1× PBS, and blocked with normal goat serum 15 μl/ml in 1% bovine serum albumin and 0.3% Triton X-100. Vascular cell adhesion molecule-1 (VCAM-1) or intracellular adhesion molecule-1 (ICAM-1) antibodies were added and sections were stored overnight. In negative controls, slides were incubated with PBS. Sections were washed in 1× PBS and incubated with fluorescent Alexa488 goat anti-rabbit 1:500 (Immunogen, USA) for 1 h at RT. After washing in 1× PBS slides were mounted in PBS/glycerol (1:3).
The brain of each animal was screened for transplanted cells. This was performed by analyzing cryosections from the whole brain with immunohistochemistry. Frontal cryosections were obtained along the rostro-caudal axis. For analyzing the number of cells in the perilesional zone, six different sections with 140-μm intervals, starting from the lesion center, were analyzed. All transplanted cells in the whole ipsi- and contralateral hemisphere of each analyzed section were counted. Only rare cells were detected at distances more than 3 mm from the lesion border. For lung, spleen, and liver, four randomly chosen high power fields (corresponding to 36 mm2) from three different slides spaced 88 μm apart were counted.
For analysis of adverse effects by the transplantation such as thromboembolic complications, hematoxylin and eosin staining was performed according to Mayers protocol. Briefly, formalin fixated slides were dipped in Mayers hematoxylin rinsed in tap water and then rinsed in distilled water followed by staining by eosin. Slides were further rinsed in distilled water and then dehydrated through ethanol dilution series. Stained slides were evaluated using light microscopy.
Microarray and RT-qPCR
Cellular mRNA was extracted from cell suspensions with Qiagen RNeasy kit (Qiagen, Sweden) according to the manufacturer's protocol. Agilent bioanalyzer 2100 (Agilent AB, Sweden) was then used for mRNA quality control. After quality control mRNA was amplified and prepared using Illumina RNA sample prep kit (Illumina Inc., USA) and then finally applied to an Illumina whole human genome chip (HT12) (Illumina Inc.), scanned, normalized, and analyzed through Illumina Beadarray system with standard Beadarray software (Illumina Inc.). RT-qPCR reactions were performed in triplicates for two biological replicates of each cell type using Platinum Quantitative PCR SuperMix-UDG (Invitrogen), according to the manufacturer's instructions. To ensure generation of specific PCR products, annealing temperatures were set in line with the recommendations of the Harvard primer bank (http://pga.mgh.harvard.edu/primerbank/) and the formula according to Rychlik et al. (25) as Ta≈Tm, thereby increasing primer-template binding stringency (27). The resulting thermocycling program was used: 50°C for 2 min, 94°C for 2 min, and then 40 cycles of 94°C for 30 s, 59/60°C for 30 s, and 72°C for 30 s on the ABI PRISM 207000 Detection System (PE Applied Biosystems, Foster City, CA, USA). A melting curve was obtained for each PCR product after each run, in order to confirm that the SYBR Green signal corresponded to a unique and specific amplicon of the calculated Tm. Indeed, the obtained melting curves were well defined and displayed a predicted product Tm, thus representing a single PCR product per primer set, validating the primer design. Then, the ΔΔCt method was used for quantifying mRNA expression levels relative to TATA-binding protein (TBP) mRNA levels as endogenous reference. Gene-specific primers were achieved from Universal ProbeLibrary Assay Design Center (Roche Applied Science) or designed with the Primer3 software. The specificity of PCR primers was determined by BLAST run of the primer sequences. All primers were purchased from MWG Biotech. The following primers were used: human CD11a, forward GATCCTCGTCCAAGTGATCG and reverse GCAGAGGCTGAACATGGAA; rat cd11a, forward TCTCCTTCCGAAAAGTGGAG and reverse CCTCGCAGCTCACAGGTATT; human CD18, forward GCTGTCCCCACAAAAAGTG and reverse CCGGAAGGTCACGTTGAA; rat cd18, forward TCCACAAAAAGTGACCCTTAACT and reverse CGTCGGAAAGTCACATTGAA; human CD29, forward CGATGCCATCATGCAAGT and reverse ACACCAGCAGCCGTGTAAC; rat cd29, forward ATCATGCAGGTTGCAGTTTG and reverse CGTGGAAAACACCAGCAGT; human CD49d, forward GGAATATCCAGTTTTTACACAAAGG and reverse AGAGAGCCAGTCCAGTAAGATGA; rat cd49d, forward CGGAAAGAATCCAAACCAGA and reverse GTCTTCCCACAGGGTTCTCC.
Statistical Analysis
Comparisons between all groups were carried out through Kruskall-Wallis analysis followed by analysis for differences between transplantation method; that is, intravenous versus specific intra-arterial by Mann-Whitney U-test for the three cell systems with subsequent Bonferroni correction for each comparison (C = 3). Nonparametric tests were chosen due to the high variability in the rNPC group and the zero values in the hNPC groups. Microarray p-value for signal is generated as an internal control in Beadstudio through internal controls randomly placed on the chip. A value of p < 0.05 was considered significant.
Results
We performed transplantations of three different types of stem cells to a rat model of traumatic brain injury. The transplantations were carried out through endovascular selective intra-arterial administration and were compared to the intravenous route. All rats survived and were included in the study. The specific intra-arterial method was significantly more efficient for rNPCs and hMSCs (Fig. 1). Neither intra-arterial nor intravenous administration of hNPCs yielded detectable engraftment.

Engrafted cells were counted per section and are reported with median (marker), quartiles (box), and max–min (whiskers). In (A) a significant difference was detected between engraftment levels per section following selective intra-arterial and intravenous transplantation in the ipsilateral hemisphere of the rat neural progenitor cell (rNPC) group. In (B) a significant difference was determined between engraftment levels per section following selective intra-arterial and intravenous transplantation in the ipsilateral hemisphere of the human mesenchymal stem cell (hMSC) group and finally in (C) a panel of all engraftment levels per section in the ipsilateral hemispheres of all groups.
Selective Intra-Arterial Transplantation Is More Efficient Than Intravenous Transplantation
Results of engraftment levels were obtained through IHC methods by counting engrafted cells in sectioned brains (Fig. 2). The first analysis aimed for comparison of the impact of specific intra-arterial transplantation to intravenous controls for the three different cell lines. We show that specific intra-arterial transplantation created significantly higher engraftment levels when compared to the intravenous route in cell line per cell line comparison. rNPC levels were more than fivefold higher than the control group (p = 0.068) and hMSCs were more than 15-fold higher than the control group in absolute values (p = 0.021) with a large spread within the intra-arterial groups (Fig. 1). To further increase the understanding of cell line properties, all groups transplanted with different cell lines, through either intra-arterial or intravenous routes, were analyzed together. This analysis showed that there were statistically significant differences between the different cell lines (Fig. 1).

Microscope images of immunohistochemical staining of engrafted cells in (A) hMSCs in the perilesional area stained with HuN (human nuclei) and in (B) rNPCs. Scale bar: 50 μm.
The data reported are represented with medial values and quartiles since a large variability was observed in the rNPC group. Two possible explanations for the engraftment variability are an interindividual variability in the outbred recipient strain or variability in the cell suspensions with respect to expression of adhesion molecules. To address the possible difference in the cell cultures, a detailed analysis of different methodological problems with respect to transplantational procedures was also performed. Following each session of transplantation, the leftover suspensions were passed through the catheter and live/dead ratios were calculated. No significant cell death was recorded suggesting that the cell handling, prior to transplantation, did not cause the variability observed (data not shown).
In this study, we were not able to perform a successful transplantation of hNPCs despite a high survival ratio of cells leaving the catheter system. These results prompted further analysis of both the recipient rat brain and the cell cultures used with respect to interaction between the transplanted cells and the host model system.
IHC and Gene Expression Profiling by Microarray Experiments of Host and Graft
A possible mechanism for homing and engraftment is likely to include regulation of different factors in the endothelium within the TBI-exposed brains. To investigate the CNS response to the TBI injury, we performed VCAM-1 IHC staining and thus confirmed previous data (10) suggesting that our TBI model results in upregulation of VCAM-1 in the ipsilateral hemisphere following trauma (Fig. 3). These findings indicate that the injured CNS parenchyma provides cues for diapedesis and migration of engrafted cells in similar ways as immunological cell respond to inflammatory cues.
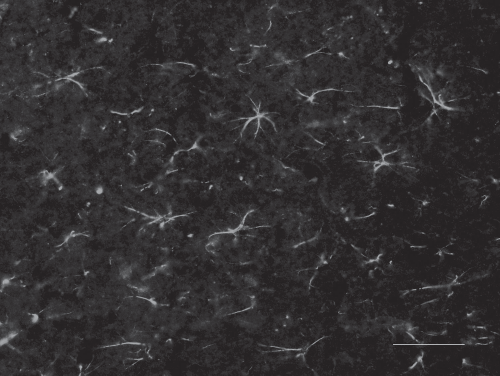
Microscope image of immunohistochemical staining with vascular cell adhesion molecule-1 (VCAM-1) upregulated 1 day after the traumatic brain injury in the model at the time of transplantation. Scale bars: 50 μm.
As a screening method we performed microarray on the human cells. This posed a bioinformatical problem since the hMSC and hNPC cultures are very different. We chose to perform an active in-depth screening of signals corresponding to mRNA levels of proteins known to be pivotal parts of immune system diapedesis and migration. rNPCs were not included in the microarray analysis due to problems with cross-species comparisons in the chips used. hMSCs showed a broad expression of integrins commonly expressed by immune system cells that are important for diapedesis through the vessel endothelia and subsequent migration into the parenchyma. Specifically, analysis of the heterodimers forming receptors for ICAM-1 and VCAM-1 were analyzed. The most interesting finding was the statistically significant CD49d signal of 68 in hMSC as opposed to 0.4 in hNPC (p = 0.0047 for fold change value), indicating that expression might be pivotal for reaching a robust intravascular engraftment. In summary, integrin receptors matching ICAM-1 and VCAM-1 were significantly higher in the hMSCs compared to the hNPCs (Table 1). To confirm microarray analysis and to perform cross-species analysis, RT-qPCRs were performed.
Signal Levels From Microarray Chip With p-Values and Indicating the Ligand of Each
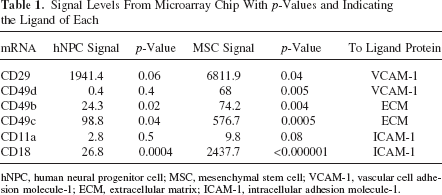
hNPC, human neural progenitor cell; MSC, mesenchymal stem cell; VCAM-1, vascular cell adhesion molecule-1; ECM, extracellular matrix; ICAM-1, intracellular adhesion molecule-1.
Gene Expression Analysis of Cells by RT-qPCR
We next examined the gene expression by RT-qPCR by analyzing normalized mRNA levels of gene of interest relative to the mRNA levels of the ubiquitous TATA box binding protein (TBP) (3). Confirming the microarray data, average mRNA levels of CD49d were highest in the hMSCs (0.98) followed by the rNPCs (0.0057) and lowest in hNPCs (0.0012) (Fig. 4). Furthermore, hMSCs also displayed the highest expression of CD29 mRNA (175.18) followed by hNPCs (12.19) (Fig. 4). Thus, the very late antigen 4 (VLA-4) heterodimer is expressed in falling order from hMSCs, rNSCs, and hNPCs. Furthermore, CD11a mRNA was detected in hNPCs (0.0029) but low CD18 mRNA (0.00025), suggesting that CD11a–CD18–ICAM-1 interaction may be dispensable for engraftment. In contrast, hMSCs displayed high CD18 mRNA levels (0.43) but no detectable CD11a expression, suggesting negligible ICAM-1-dependent engraftment in the hMSCs. The data are given as relative expression of integrin mRNA to TBP mRNA levels and were based on double biological replicates.
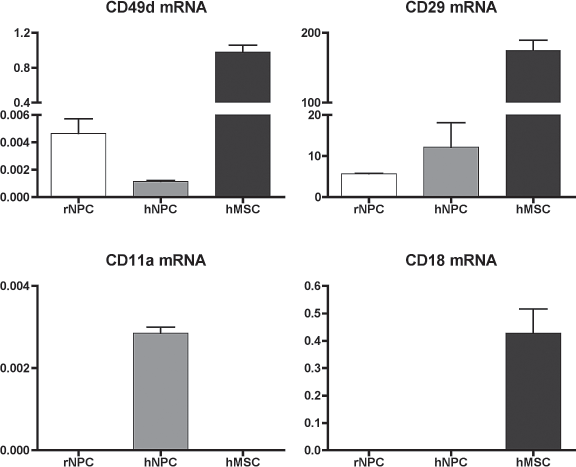
Bars represent normalized levels of integrin CD49d, CD29, CD11a, and CD18 mRNA expression in rNPCs, hNPCs, and hMSCs relative to endogenous TAT box binding protein (TBP) mRNA levels as measured by RT-qPCR (see Materials and Methods for details). Error bars represent the distribution between the biological replicates.
Analyses of possible cell suspension emboli were also performed. All rats were scored by the method of Bederson et al. (5) and no rats showed clinical signs of cerebral infarction. To further evaluate the possibility of small infarctions, hematoxylin-eosin stainings were performed. No ischemic lesions were found in the brain parenchyma.
Cell suspensions were also tested by in vitro passage through the catheter system showing that more than 90% of the cells survived the shear stress load that passage through the system generates.
Analysis of other possible sites of engraftments was performed to assess spread of transplanted cells to other organs outside the designated target site within the CNS. IHC stainings were performed on sections of lung, liver, and spleen. hMSCs were detected only in the spleen whereas, interestingly, almost no hNPCs or rNPCs were found in the spleen or in any other organ investigated.
Discussion
In this study we show that targeted intra-arterial transplantation of rNPCs and hMSCs produces a significantly higher engraftment compared to intravenous administration. The method is easily adoptable for standard interventional microcatheter techniques with minimal invasive strategies, and encompasses a possibility for repeated transplantations. The third cell type used, hNPCs, surprisingly produced no detectable engraftment thereby showing the importance of cell type characterization prior to intravascular transplantation studies. As we have described previously, when isolating and expanding endogenous hNPCs we achieved robust results with a population of mitotically active NPCs. The majority of cells were immunoreactive for the progenitor marker nestin, while a smaller population expressed markers of more differentiated neurons (β-tubulin III) and astrocytes (glial fibrillary acidic protein) and also displayed the corresponding morphology (24).
These findings are even more interesting since the hNPCs used in this study have been transplanted using open surgical technique with robust engraftment in the rat (1,30). Considering both previous studies and the current results, we started a broad approach analysis of properties of both cell cultures and recipient parenchyma.
The results from both the microarray and RT-qPCR analysis suggest, albeit indirectly, that CD29–CD49d–VCAM-1 signaling might be one of the pivotal interactions that are necessary for intraluminal transplantation. The observation that hNPCs gave no engraftment suggests that an intraluminal transplantation technique requires an active process from the cells transplanted with respect to diapedesis.
The microarray approach when comparing hMSCs and hNPCs presents a bioinformatical problem with respect to data management. We chose to perform data mining on receptors known to be active in endothelial adhesion to get an indication which receptors might be lacking on the hNPCs, thereby indirectly implicating them in the engraftment process for the hMSCs. The indirect method used in the present study confirmed published results implicating CD49d in intravascular engraftment (16), which gives some weight to the model. Projects with either FACS sorting of cell culture or knock-in methods would shed further light on the actual cell and vessel endothelium interaction. This study further suggests that the inflammatory activation in the CNS occurring in several different pathological conditions, such as TBI, ischemic stroke, and primary inflammatory diseases, is necessary for effective intravascular transplantation.
A possibly contributing factor may be the inherent heterogeneity of hNPC cultures. As we have described previously (23), a small but significant proportion of cells differentiate in vitro into early neurons and astrocytes. It is possible that the more mature neural cells have even less propensity to migrate through the vessel wall. As a result, the number of cells available for engraftment may be 30–40% lower than the actual cell counts. However, this cannot explain the complete absence of engrafted hNPCs. Another possible explanation to the lack of engraftment with hNPCs might be the xenobarrier. Pitted against that hypothesis is the fact that the hMSCs resulted in a robust engraftment and that the same hNPCs have been transplanted in an open surgical fashion. These surprising results may also be explained by the fact that the hNPCs are more susceptible to the hostile environment of the blood stream than the other cell systems analyzed. In vivo imaging techniques such as MRI tracing of magnetically labeled cells are probably needed for better evaluation of the destiny of these cells.
Nevertheless, when transplanting a cell type that gives engraftment it is still possible to increase efficiency ratios through intra-arterial transplantation. In clinical practice this could be achieved by placement of a microcatheter just upstream of a target site. In the present study on the rat we were not able to navigate the catheter closer to the target than the ipsilateral carotid artery. In the clinical setting, today's catheter technique would make highly selective administration of cells possible such as previously shown for hMSCs in the rat (23). When translating cell-based regenerative medicine, the actual route of transplantation will be very important. For operative techniques there is a problem with scalability and for intravenous techniques there is a problem with efficacy. We show that by selective intra-arterial methods it is possible to increase the level of engraftment (23). Further, not all cells are optimal for intraluminal transplantation. It is previously known that insulin-producing cells lack the capability to perform diapedesis and we now show that hNPCs, a cell system previously transplanted by open surgical means (30), also lack the capability to perform diapedesis. For these cell systems we have developed the Extroducer as a means to establish direct, minimally invasive, parenchymal access in hard to reach organs (22).
As previously described (23), the absence of adverse effects in transplanted animals suggests that in the short term, this method is a safe method for delivering high concentrations of different cell types intra-arterially. It could be argued that the higher engraftment rates in our intra-arterial groups could be a consequence of microembolization of cells. In that scenario we would, however, have detected histological changes.
In a small TBI in a rodent model, a surgical burr hole approach for transplantation might give sufficient engraftment as shown previously, with effects on biological outcome parameters (30). In larger trauma or following ischemic events, both the intravenous and intra-arterial routes are less invasive for the suggested patients. These results are more easily translated into clinical practice than comparable intraparenchymal injections in rat or other small rodent models since the increase in absolute disease-ridden volume in many human CNS conditions (e.g., ischemic stroke) dictates a large number of burr holes and subsequent injection trajectories.
When translating stem cell transplantation to clinical practice we consider this study to strengthen the hypothesis that both the method of transplantation and the properties of the cells aimed for transplantation must be carefully considered. Both the current study and others, indicating CD49d from a more direct approach, imply that for intravascular administration, the integrin expression by cell lines is very important. When planning the actual operative procedure, we consider this study to further strengthen the evidence of a first passage effect when performing targeted intra-arterial administrations as opposed to intravenous transplantation. This is especially interesting in ischemic stroke when an endovascular procedure might already be planned, such as a mechanical thrombectomy, and the possibility to perform transplantations in the same procedure further decreases patient risk.
Footnotes
Acknowledgments
The authors wish to thank BEA, Karolinska for excellent technical assistance with the microarray analysis. This study was supported by grants from the Swedish Research Council, Karolinska Institutet funds, the Swedish Society of Medicine and Strokefonden. The authors declare no conflict of interest.
