Abstract
Human islets are subjected to a number of stresses before and during their isolation that may influence their survival and engraftment after transplantation. Apoptosis is likely to be activated in response to these stresses. Apoptosis due to intrinsic stresses is regulated by pro- and antiapoptotic members of the Bcl-2 family. While the role of the Bcl-2 family in apoptosis of rodent islets is becoming increasingly understood, little is known about which of these molecules are expressed or required for apoptosis of human islets. This study investigated the expression of the Bcl-2 family of molecules in isolated human islets. RNA and protein lysates were extracted from human islets immediately postisolation. At the same time, standard quality control assays including viability staining and β-cell content were performed on each islet preparation. Microarrays, RT-PCR, and Western blotting were performed on islet RNA and protein. The prosurvival molecules Bcl-xl and Mcl-1, but not Bcl-2, were highly expressed. The multidomain proapoptotic effector molecule Bax was expressed at higher levels than Bak. Proapoptotic BH3-only molecules were expressed at low levels, with Bid being the most abundant. The proapoptotic molecules BNIP3, BNIP3L, and Beclin-1 were all highly expressed, indicating exposure of islets to oxygen and nutrient deprivation during isolation. Our data provide a comprehensive analysis of expression levels of pro- and antiapoptotic Bcl-2 family members in isolated human islets. Knowledge of which molecules are expressed will guide future research to understand the apoptotic pathways activated during isolation or after transplantation. This is crucial for the design of methods to achieve improved transplantation outcomes.
Introduction
Type 1 diabetes (T1D) is the result of autoimmune destruction of the insulin-producing pancreatic β-cells by cells of the immune system, resulting in lifelong dependence on insulin injections. One approach to correcting blood glucose levels in T1D is allogeneic human islet transplantation. In this procedure pancreases obtained from organ donors are processed to yield isolated islets by a combination of mechanical and enzymatic digestion followed by density gradient purification. Islets are transplanted into the portal circulation of recipients who receive immunosuppressive medication to inhibit rejection (60).
The requirement for islets from more than one donor to reverse diabetes in most recipients is one limitation to the wider application of the procedure. This is due to losses of islets in the isolation process and following transplantation, when many islets die prior to engraftment even in syngeneic models (6). Islets are exposed to many insults during their isolation, including mechanical stress, hypoxia, and variations in temperature and collagenase blends (3,5,31,43). After transplantation other factors have been identified that may influence the viability and function of transplanted islets over the short and long term. These include an instant blood-mediated inflammatory reaction (4,39), toxicity of immunosuppressive drugs to the β-cells (16,24,28), transplant rejection, and reoccurrence of autoimmunity (7,56,57,62).
Islet cell death during and after isolation may be at least in part due to apoptotic mechanisms. The pro- and antiapoptotic members of the Bcl-2 family act in a tightly regulated manner to control cell death (58). Proapoptotic BH3-only molecules (Bim, Bid, Bad, Puma, Noxa, Bmf, Bik, and Hrk) initiate apoptosis signaling either by binding to the prosurvival molecules (Bcl-2, Bcl-xL, Mcl-1, Bcl-w, and A1) or by direct interaction with the proapoptotic multidomain molecules Bax and Bak (58). This signaling results in loss of mitochondrial outer membrane potential, release of cytochrome c, and activation of caspases.
We aimed to examine expression of genes associated with cell death, with particular focus on pro- and antiapoptotic members of the Bcl-2 family. We reasoned that high expression of proapoptotic molecules may indicate that islets have experienced more stress during isolation and may therefore be less likely to survive after transplantation. We have found consistent expression of some genes in most islet preparations possibly indicating apoptotic pathways activated by islet isolation.
Materials and Methods
Human Islet Isolation
Human pancreata were obtained, with informed consent from next-of-kin, from heart-beating, brain-dead donors by the Australian Islet Transplant Consortium. The project was approved by the human ethics committees of the hospitals involved and the Red Cross. Human islets were purified as previously described (54) by intraductal perfusion and digestion of the pancreas with collagenase followed by purification using ficoll density gradients. Purified islets were cultured in CMRL 1066 media (Invitrogen, Grand Island, NY, USA) supplemented with 10% human serum albumin, 100 U/ml penicillin, 100 mg/ml streptomycin, and 2 mmol/l L-glutamine (complete CMRL), in a 37°C, 5% CO2 humidified incubator. Islet purity was assessed by light microscopy after staining with dithizone (Sigma Chemical, St. Louis, MO, USA). For assays involving monodispersed islet cells, the islets were incubated in Accutase solution (Chemicon, Billerica, MA, USA) for 10 min at 37°C, then mechanically dispersed using a pipette. Islet cells were washed and recovered in complete CMRL for 1 h at 37°C.
Human Islet Quality Analysis
Islets were stained with dithizone (Sigma Chemical) and analyzed by light microscopy at 40x magnification, and purity was visually estimated. Islet viability was quantified by fluorescence microscopy, after staining with 254 μM ethidium bromide and 81 μM acridine orange (Sigma Chemical). Islet preparations were routinely assessed for viability by simultaneously staining monodispersed islet cells for necrosis with 5 μg/ml 7-aminoactinomycin D (7-AAD), β-cell subsets with 10 μM Newport green (NPG), and apoptosis with 1 μM tetramethylrhodamineethylester (TMRE) (Molecular Probes, Invitrogen, Grand Island, NY), and analyzing by flow cytometry following the method of Ichii et al. (25). The β-cell viability index (BCVI) was calculated by multiplying the percentage of NPG+ live cells, by the percentage of TMRE+/NPG+ cells from the flow cytometry histogram gates. Routine glucose-stimulated insulin secretion assays were performed to determine the insulin secretory capacity of the isolated islets. Islets were stimulated at low (3 mM) and high (20 mM) glucose for 30 min at 37°C. Islet supernatants were collected and the insulin content measured using an ELISA kit (Mercodia, Uppsala, Sweden), following the manufacturer's instructions. The stimulation index (SI) was calculated by dividing the insulin stimulated at 20 mM glucose by the insulin stimulated at 3 mM glucose. In vivo functionality for each preparation was also tested by transplanting 2,000 human islet equivalents (IEQ) under the kidney capsule of each of at least three diabetic nonobese diabetic severe combined immundeficient rat insulin promoter H-2Kb class I heavy chain overexpressing (NOD.SCID.RIPKb) (59) mice, and monitoring blood glucose levels for 4 weeks posttransplant. The mice were nephrectomized and blood glucose tested to ensure the normalization of blood glucose levels was due to the islet graft. Mice were considered diabetic when they had a nonfasting blood glucose concentration of ≥ 15 mmol/L on two consecutive measurements.
RNA Extraction and Quantification
Total cellular RNA was extracted from approximately 3,000 IEQ using the Macherey-Nagel (Duren, Germany) NucleoSpin® RNA II kit, following the manufacturer's instructions. RNA was extracted within 12 h of isolation. The RNA quality and relative composition was assessed using an RNA 6000 nanochip on the Agilent 2100 Bioanalyzer using (Agilent Technologies, Palo Alto, CA, USA).
Microarrays
Total RNA was extracted from the postisolation human islets from nine normal cadaveric pancreas donors using the RNeasy Plus Mini Kit (Qiagen, CA, USA) according to the manufacturer's instructions. RNA quality was assessed using an Agilent 2100 Bionalyzer (Agilent Technologies), and all samples had an RNA integrity number (RIN) > 8. cRNA from each donor was hybridized onto an Affymetrix HG-U133+2 whole genome microarray (Affymetrix, Santa Clara, CA, USA) at the Ramaciotti Centre for Gene Function analysis (UNSW, Sydney, Australia), according to manufacturer protocols. Raw data were normalized using robust multichip analysis (RMA) (26) on the log (base 2) scale, and probes were collapsed to unique gene symbols based on the probe with the largest average expression level. Sample H43 had aberrantly low levels of INS1 and was excluded; we present the results based upon eight donors. Normalized expression levels for the Bcl family genes were extracted, ranked by abundance, and represented as a heatmap, with a global, continuous color scheme.
Real-Time PCR
RNA (200 ng) per sample was reverse transcribed using avian myeloblastosis virus (AMV) reverse transcriptase (Promega, Madison, WI, USA). Samples were incubated at 37°C for 1 h and 95°C for 10 min. cDNA was diluted 1/10 in RNAse free water. Real-time PCR reactions were prepared in a final volume of 20 μl using cDNA derived from 200 ng of input RNA and performed using AmpliTaq Gold® (Applied Biosystems, Foster City, CA, USA). For each gene TaqMan® (Applied Biosystems) primer probes were used. The probes contain a 6-carboxy-fluorescein phosphoramidite (FAM dye) label at the 5′ end of the gene and a minor groove binder and nonfluorescent quencher at the 3′ end and are designed to hybridize across exon junctions. Thermal cycling conditions comprised polymerase activation at 95°C for 8 min, then 45 cycles of denaturation at 95°C for 10 s, and annealing and extension at 60°C for 60 s. Reactions were performed in a Rotor-gene RG-3000 real-time PCR machine using Rotor-gene 6 analysis software (Qiagen). Each measurement was performed in duplicate and the threshold cycle (Ct), the fractional cycle number at which the amount of amplified target reached a fixed threshold, was determined. ACt, the difference between the threshold cycle of a given target cDNA and the internal standard gene 18S rRNA subunit cDNA (55), was calculated for each sample.
Applied Biosystems TaqMan® Primer/Probe Sets Used for Real-Time PCR
TaqMan® primer probe kits used are the following. Eukaryotic 18S rRNA (Hs99999901_s1), insulin (Hs00 356618_m1), B-cell CLL/lymphoma 2 (BCL2) (Hs0023 6808_s1), BCL2–like 1 (BCL-xL) (Hs00236329_m1), myeloid cell leukemia-1 (Mcl-1) (Hs03043899_m1), BCL-2 antagonist/killer 1 (BAK) (Hs00832876_g1), BCL2-associated × protein (BAX) (Hs00180269_m1), BCL2 antagonist of cell death (BAD) (Hs001889 30_m1), BH3 interacting domain death agonist (BID) (Hs00609630_m1), BCL2-interacting killer (BIK) (Hs00 609635_m1), BCL2-like 11 (BIM) (Hs00375807_m1), BCL2-modifyng factor (BMF) (Hs00372938_m1), Bcl-2/E1B-19K interacting protein 3 (BNIP3) (Hs0096 9291_m1), Bcl-2/E1B-19K interacting protein 3-like protein (BNIP3L) (Hs01087963_m1), Beclin-1 (Hs00 186838_m1), Harakiri (HRK) (Hs00705213_s1), inducible nitric oxde synthetase (iNOS) (Hs00167257_m1), myeloid cell leukemia-1 (Mcl-1) (Hs00766187_m1), monoocyte chemoattractant protein 1 (Mcp-1) (Hs00 234140_m1), NOXA (Hs00560402_m1), BCL2-binding component (Hs00248075_m1), and tissue factor (Hs00 175225_m1).
Immunoblotting
Human islet or acinar protein lysates were made within 1 h of isolation. Mouse thymus, splenocytes and brain, islets from transgenic mice expressing Bcl-2 under control of the rat insulin promoter (1), and HeLa cells were used as positive control tissues. To show cleaved Bid, mouse splenocytes or human islets were treated with FasL for 48 h before lysates were made. Cells were washed three times in PBS and lysed in 10 [0.l of lysis buffer (20 mM Tris/HCl, pH 7.4, 135 mM NaCl, 1.5 mM MgCl2, 1 mM EGTA, 1% Triton X-100, 1× protease inhibitor cocktail (Sigma Aldrich, St. Louis, MO, USA), and 1x phosphatase inhibitor cocktail (Sigma Aldrich). Samples were snap frozen and thawed three times, before being centrifuged at 10,000 times; g for 5 min. Protein was quantified using the bicinchoninic acid (BCA) protein assay kit (Thermo Fisher Scientific, Rockford, IL, USA). Twenty micrograms of protein per sample was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE; Biorad Laboratories, Hercules, CA, USA) and transferred to nitrocellulose membrane (GE Healthcare Biosciences, Piscataway, NJ, USA) using standard procedures. Membranes were blocked in 5% skim milk in PBS-Tween 20 overnight at 4°C, incubated with the appropriate primary antibody, and followed by secondary antibodies made in 5% skim milk in PBS-Tween 20. Immunoreactive bands were visualized with the ECL or ECL Plus detection systems (GE Healthcare Biosciences). Primary antibodies used in this study were: anti-human Bax (clone AC2) 1:2000, anti-rat Bid (clone AE6) 1:1000, anti-mouse Bim (clone 3C5) 1:1000, anti-Mcl-1 (clone AA2), anti-Bcl-2 (all gifts from Professor Andreas Strasser, Walter and Eliza Hall Institute, Melbourne, Australia), Bcl-xl (Cell Signalling, Danvers, MA, USA) 1:2000, BNIP3 (Sigma-Aldrich) 1:300, rabbit anti-beta actin (Abcam, Cambridge, MA, USA) 1:1000. Anti-rabbit or rat HRP-conjugated secondary antibodies were purchased from Dako (Glostrup, Denmark).
Statistical and Image Analysis
Microarray analysis was performed using GenePat tern version 3.2.3 (52) hosted at the Peter Wills Bioinformatics Centre (http://pwbc.garvan.unsw.edu.au/gp). Data analysis was performed using GraphPad Prism (GraphPad Prism Software, San Diego, CA). Data are represented as mean ± SEM and significance by Student's t-test. Western blot films were scanned using Canon CanoScan Toolbox Version 4.1 and contrast uniformly lightened using Adobe Photoshop Elements Version 6.
Results
Gene Expression Profiles of Isolated Human Islets Are Consistent with Hypoxia and Lack of Nutrients
Microarray analysis of eight transplant quality islet preparations using Affymetrix whole genome arrays was performed (Fig. 1). Analysis of Bcl-2 family molecules revealed that Mcl-1 was the most abundant prosurvival transcript, with levels very consistent between islet preparations and approaching expression levels of insulin. Bcl-xL was also expressed at high levels when compared to the other prosurvival molecule Bcl-2. The hypoxia-induced cell death molecules BNIP3, BNIP3L, and Beclin-1 were highly expressed in all islet preparations. BH3-only molecules Bim and Bid were the most abundant proapoptotic transcripts, although their expression levels were generally lower than those of the prosurvival members of the family.

Expression of the pro- and antiapoptotic molecules of the Bcl-2 family in isolated human islets. Gene expression was analyzed in eight independent islet donor samples by Affymetrix HG-U133+2 whole human genome microarrays. A heat map of the B-cell CLL/lymphoma 2 (Bcl-2) family genes ranked from highest to lowest expression is shown. The average expression level is given, with average >6 being scored as detected above the level of background noise. See Materials and Methods for gene abbreviations.
Real-time PCR using RNA from independently isolated human islets from 20 pancreas donors broadly confirmed the transcripts found in the microarray studies (Fig. 2). Insulin levels were measured in all preparations, and its expression did not vary widely between donors. Expression levels of individual pro- and antiapoptotic genes did not vary greatly between islet preparations. The most highly expressed BH3-only molecules by RT-PCR were Bmf, Bid, Puma, and Noxa. Others, including Bad, Bik, and Bim, were expressed at lower levels, just on the limits of detection in this assay. Although a recent report has suggested Hrk may be involved in cytokine-induced death of islet cells (23), we did not find evidence of its expression in human islets by RT-PCR or gene array, which agrees with studies using a β-galactosidase reporter that found expression only in neural tissue (13). While normally considered a prosurvival molecule, Mcl-1 has a short, proapoptotic domain (Mcl-1s) that we detected at very low levels by RT-PCR. Also highly expressed were the multidomain effector Bax, and the proapoptotic BNIP3, BNIP3L, and Beclin-1, confirming the microarray results.

Human islet cells express high levels of BNIP3, BNIP3L, Beclin-1, Bax, Mcl-1, and Bcl-xl. Quantitative RT-PCR using TaqMan primer probe sets for each gene was run in duplicate in each of three independent experiments, and the ΔCt was calculated by normalizing the gene of interest to the 18S housekeeping gene. The data represent the mean ΔCt for between 2 and 20 different donors for each gene. The numbers on each bar represent the number of different donors analyzed for each experiment. (A) Prosurvival molecules, (B) proapoptotic molecules, and (C) proinflammatory molecules.
Tissue factor and monocyte chemoattractant protein 1 (Mcp-1) are thought to be important in the inflammatory response in the recipient after transplantation. They were already highly expressed at the mRNA level after isolation (Fig. 2C), and without contacting blood, confirming previous reports (4,27,39,44). Inducible NO synthase was expressed at very low levels in all islet preparations tested.
Human Islets Express Bcl-2 Family Members at the Protein Level
Prosurvival molecules Bcl-xl and Mcl-1 were expressed in human islets and acinar cells after isolation. A band of 30 kDa was seen in all islet samples probed with an antibody recognizing Bcl-xl (Fig. 3A). Mcl-1 was detected in all islet samples as indicated by a 42-kDa band in Figure 3A. A weaker band at 30 kDa was also detected in some preparations corresponding to the alternatively spliced proapoptotic Mcl-1s. When probed with an antibody recognizing Bcl-2, a band of 26 kDa was detected in islets from transgenic mice expressing human Bcl-2 in their β-cells (1) (Fig. 3B). Bcl-2 protein was not detected in islets from several human donors (Fig. 3B), consistent with its low level of mRNA.
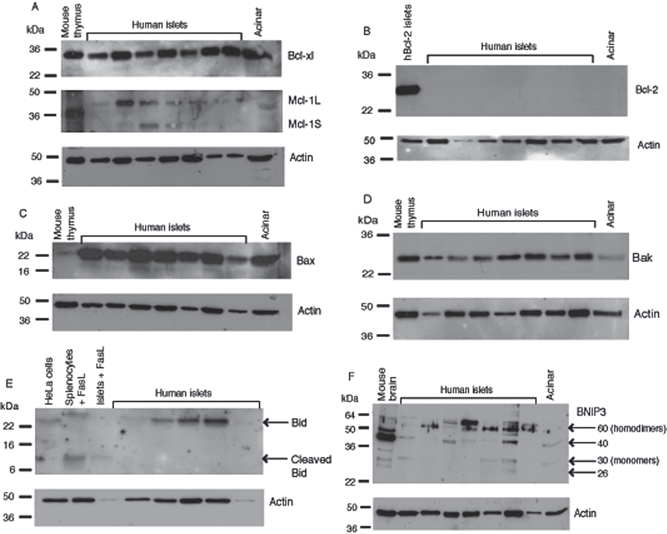
Human islet cells express Bcl-xl, Mcl-1, Bax, Bid, and BNIP3 after isolation. Western blotting was performed with antibodies to Bcl-xl (A), Mcl-1 (A), Bcl-2 (B), Bax (C), Bak (D), Bid (E), BNIP3 (F), and β-actin as a loading control. Molecular weight markers are shown in kDa. The results are representative of three independent experiments performed with five to seven different islet preparations.
Bax protein expression was observed at high levels in isolated islet preparations and acinar cells from all donors examined (Fig. 3C). Bak protein was also expressed, but at lower levels compared to Bax (Fig. 3D). Bid was expressed in isolated islets, but its lower molecular weight cleaved form was not detected. This suggests that islets are not exposed to stimuli that activate Bid during isolation such as FasL, granzyme B, and proinflammatory cytokines. Cleaved Bid was detected in human islets after treatment with recombinant FasL, indicating the islets are susceptible to proapoptotic stimuli, and that like in mouse islets, Bid is activated by the Fas-FasL pathway (Fig. 3E).
Other groups have reported the proapoptotic protein BNIP3 to exist in both monomeric and dimeric forms, and that the dimer is stable even under reducing conditions (11). We detected a 60-kDa dimer in human islets probed with an antibody to BNIP3 (Fig. 3F). The apparent molecular weight of native BNIP3 has been reported to be 21.5 kDa (11). In human islets from several different donors we observed no bands at 21.5 kDa, suggesting the unmodified protein monomer is not present. Slower migrating, posttranslationally modified forms of the native BNIP3 protein have been reported in cancer cell lines (38). We observed bands at 26 and 30 kDa, representing BNIP3 monomers, in some of the human islet donors. We also detected anti-BNIP3 reactive bands at around 40 kDa. Other studies using the same antibody in cancer cell lines also demonstrated BNIP3 bands between 30 and 50 kDa that may represent unphosphorylated forms or degradation products of the BNIP3 homodimer. We did not find an antibody recognizing BNIP3L that was able to detect protein in human islets by Western blotting.
Expression of Bcl-2 Family Molecules Does Not Correlate with Quality of Islet Preparations
Quality analysis of human islet preparations used for RT-PCR and Western blot analysis were performed (Table 1). The average percentage of viable islets as determined by fluorescence microscopy was 77 ± 12% (n = 9 donors). An average of 78 ± 12% (n = 20 donors) monodispersed islet cells were live cells as indicated by absence of 7-AAD staining. Of those live cells an average of 37 ± 11% cells in the islet preparations were NPG positive (indicating β-cells) (n = 20 donors). Of these β-cells, viability was 72 ± 20% (n = 20 donors), as measured by NPG-positive cells with high levels of TMRE staining. The average β-cell viability index of the islet cells, calculated by multiplying the proportion of NPG-positive cells by the proportion of viable cells, was 0.260 ± 0.102 (n = 20 donors). The average stimulation index (SI) from low (3 mM) to high (20 mM) glucose of islets that performed well when grafted into mice was 4.35 ± 2.3 (Table 2). This was significantly more than that of the islets that performed poorly in mice (mean SI 2.3 ± 0.47, p < 0.0001 unpaired t-test). This association between islet function in vitro and in vivo has been noted anecdotally by others.
Islet Quality Control Information for Pancreas Donors Used for This Study
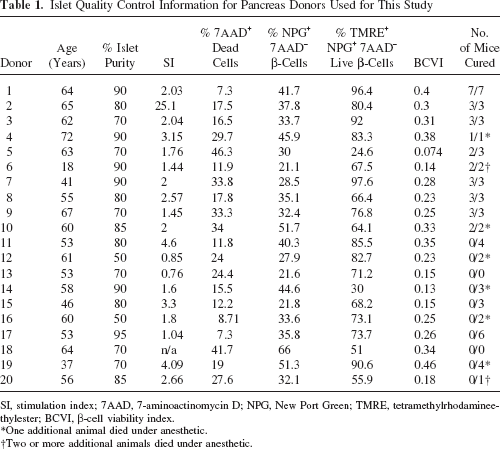
SI, stimulation index; 7AAD, 7-aminoactinomycin D; NPG, New Port Green; TMRE, tetramethylrhodamineethylester; BCVI, β-cell viability index.
One additional animal died under anesthetic.
Two or more additional animals died under anesthetic.
Summary of Expression Studies for the Pro- and Antiapoptotic Members of the Bcl-2 Family in Human Islets
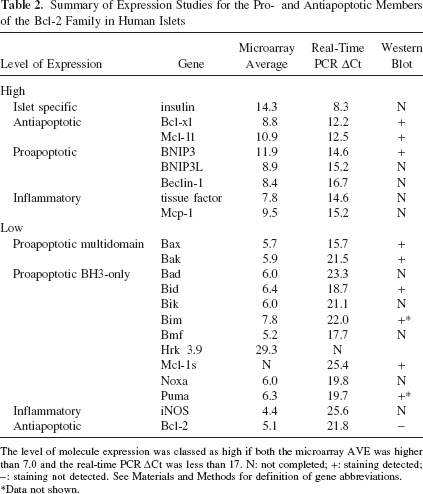
The level of molecule expression was classed as high if both the microarray AVE was higher than 7.0 and the real-time PCR ΔCt was less than 17. N: not completed; +: staining detected; -: staining not detected. See Materials and Methods for definition of gene abbreviations.
Data not shown.
Although the islet preparations varied a great deal in their function and viability, we did not observe a correlation of gene or protein expression with quality control data suggesting the expression of Bcl-2 family genes in isolated islets is similar regardless of in vitro function or whether they were capable of correcting blood glucose levels in diabetic mice.
Discussion
We examined the expression of the Bcl-2 family of pro- and antiapoptotic molecules in human islets directly after isolation. The prosurvival molecules Bcl-xl and Mcl-1 were highly expressed in isolated human islets as was the multidomain proapoptotic effector Bax. The proapoptotic molecules BNIP3, BNIP3L, and Beclin-1 were all highly expressed, indicating exposure of islets to oxygen and nutrient deprivation during isolation. While we observed expression of most genes in this family, we did not observe a correlation of expression of pro- or antiapoptotic molecules with transplantation outcome or other quality control criteria, suggesting all human islet preparations may be exposed to similar stresses.
Limited previous information about the expression and function of Bcl2 family members is available in human islets. Expression of Bcl-xl, Mcl-1, Bax, and Bad has been demonstrated by immunohistochemistry in human pancreas cells (20,29,30,61). Expression of Bcl-2, Bcl-xl, Bad, Bik, and Bid in isolated human islets has also been shown by RT-PCR (21,61). The proapoptotic molecules BNIP3 and BNIP3L have been shown by microarray analysis to be upregulated in isolated human islets (34).
The Bcl-2 family members that are more highly expressed may provide a clue to the stresses experienced in islet isolation. Higher levels of Bcl-xl and Mcl-1 mRNA and protein that were observed suggest they may be more important than Bcl-2 in prosurvival responses in islets. Physiological expression of Bcl-x may protect β-cells from apoptosis. Mouse β-cells lacking Bcl-xl are abnormally sensitive to apoptotic stimuli including inducers of ER stress, gamma irradiation, and proinflammatory cytokines (10). Mcl-1 mRNA has a short half-life, and the protein is rapidly degraded by proteasomes, suggesting it plays a role in control of apoptosis in response to rapidly changing environmental stimuli (63). We observed the short proapoptotic Mcl-1s in isolated human islets, consistent with islet exposure to a cytotoxic environment during isolation. Overexpression of Bcl-2 provides protection from only a limited set of apoptotic signals including staurosporine (1), thapsigargin to a lesser extent (10), and minimal protection from cytokine-mediated cell death (51), suggesting a minor role for Bcl-2 in islet antiapoptotic responses.
Several BH3-only proteins, including Bid, Bad, and Bmf, are relatively easy to detect in healthy cells and require posttranslational modification to induce proapoptotic activity (48). Bid and Bad have both been implicated in the extrinsic apoptosis pathway (death receptors and proinflammatory cytokines) in islets. Bid was one of the more highly expressed transcripts in human islets and was also detected at the protein level. We did not observe cleaved Bid (tBid) immediately after islet isolation, but it was detectable after incubation of islets with FasL. These data suggest that, like in mouse islets, Bid is activated by death receptors but that the islets had not been exposed to death receptor activation during isolation. Bad was shown to be dephosphorylated by calcineurin in rodent, but not human β-cells in response to proinflammatory cytokines (15). We observed low levels of Bad in human islets in RT-PCR and microarray studies. Bmf is normally sequestered into the cytoskeletal structure and is released in response to stress stimuli such as detachment of cells or anoikis (49). Cell detachment is a stress likely to occur in islet isolation. It remains to be determined if Bmf is activated in islet isolation.
Other BH3-only molecules, including Puma, Bim, and Noxa, are regulated by transcription factors such as p53 (50) or CCAAT/enhancer binding protein (CHOP) (47). Bim and Puma are required for glucose- or ribose-induced apoptosis in mouse islets (37), while a role for Noxa in islets remains to be determined. In our study Bim, Puma, and Noxa were expressed at low levels. Without similar expression data from laser-dissected islets prior to isolation, we cannot conclude that these transcripts have altered during islet isolation. However, our data confirm the expression in human islets of molecules that are known to be activated by apoptotic stimuli in mouse islets.
Bax and Bak are multidomain proapoptotic effector molecules downstream of the BH3-only molecules. Bax was highly expressed in all isolated human islet preparations, whereas Bak was expressed at low levels. Previous data from our laboratory demonstrated that Bax, but not Bak, is transcriptionally upregulated in islets by high concentrations of glucose or ribose (37). The difference in expression of these molecules may reflect the apparent bias towards requirement for Bax over Bak in islets, although both Bax and Bak were required for death receptor-induced apoptosis of mouse islets (36).
BNIP3 and BNIP3L are activated by oxygen deprivation or changing nutrient conditions, and together with Beclin-1 are implicated in autophagy (14,33). Comparison of gene expression in isolated human islets relative to laser dissected, β-cell-rich areas from a pancreas biopsy of the same donor found increased expression of hypoxia- and oxidative stress-induced genes, including BNIP3, in isolated islets (34). High levels of BNIP3, BNIP3L, and Beclin-1 suggest that islets are exposed to lack of oxygen and nutrients during isolation, even though temperatures are kept low to minimize cellular metabolism. A correlation between reduced oxygen supply, upregulation of the hypoxia-inducible factor HIF-1α, and the occurrence of apoptosis has been shown in isolated human and rat islets (41).
Further work is required to compare gene expression in isolated islets with that in biopsies of the pancreas taken without cessation of blood supply or prior to islet isolation to confirm whether isolation stresses have led to increased expression of these molecules. It remains possible that expression of these molecules is activated in the donors before pancreas retrieval, as brain death is associated with damage that can lead to activation of apoptotic pathways (2,12,46).
Inhibition of proapoptotic Bcl-2 family molecules during islet isolation may reduce islet cell death and slow or prevent initial graft loss after transplantation. Treatment of islets with antioxidants (8,35,45) or anti-inflammatory agents (22,53,64) promotes islet survival and this may be in part due to prevention of activation of proapoptotic signaling in β-cells. The iron chelator and antioxidant desferoxamine has been used during islet isolation and may promote vascularization of grafts (32); however, it also targets HIF-1α and thus may induce hypoxic stress in islets (9). Targeting apoptosis with synthetic caspase inhibitors such as zVAD-FMK (17,19,40,42), or overexpression of the X-linked inhibitor of apoptosis (XIAP) (18,24) has also been beneficial in preventing posttransplant islet loss. Understanding the expression of apoptotic genes and their targets is therefore critical for guiding strategies to improve islet quality after isolation.
Footnotes
Acknowledgments
This study was supported by The Australian Federal Department of Health and Ageing and the Juvenile Diabetes Research Foundation International. We thank the Tom Mandel Islet Transplant Program and The Centre for Transplant and Renal Research, Westmead Millennium Institute for Medical Research for isolated human islets, Stacey Fynch and Gaurang Jhala for excellent technical assistance, and Professor Andreas Strasser and Dr. Lorraine O'Reilly from the Walter and Eliza Hall Institute of Medical Research for antibodies. We thank Dr. Helen Speirs from the Ramaciotti Centre for technical assistance and advice regarding microarrays. The authors declare no conflicts of interest.
