Abstract
A key limitation to the success of islet transplantation is islet cell exhaustion and cell death during islet isolation and following transplantation. Endoplasmic reticulum (ER) stress has been identified as an important mechanism in the development of β-cell dysfunction, cell death, and diabetes. This study investigated the role of ER stress in islet loss during human islet isolation and posttransplantation in a diabetic athymic mouse model. Islets were isolated from human organ donor pancreata using intraductal enzymatic dissociation and continuous density gradient purification. ER stress mediators were assessed by Western blot and by RT-PCR. Caspase-3 activity was quantified by a bioluminescent peptide cleavage assay. Normal and streptozotocin-treated diabetic nude mice were transplanted with 2,000 IEQ of human islets under the kidney capsule and the grafts were harvested 3 or 28 days after transplantation. The grafts were analyzed for the presence for ER stress signals by immunohistochemistry. Isolated islets demonstrated higher levels of ER chaperone Bip, ER stress mediators eIF2α, ATF, spliced XBP-1, and CHOP, and also ER stress-associated apoptotic signals like JNK, caspase-3/7, and cleaved PARP. Donor pancreatic tissue did not show expression of any of these ER stress mediators. After transplantation, low expression of only protective ER stress mediators was evident in the grafts from the normal recipients. In contrast, both protective and apoptotic ER stress mediators were highly expressed in the grafts of hyperglycemic mice. ER stress mediators were induced during islet isolation and may contribute to islet apoptosis and cell death. Islet isolation activates ER stress and apoptotic pathways in isolated islets. Hyperglycemia may prolong this ER stress signal in engrafted islets, converting the protective aspects of the ER stress response to a proapoptotic response and thus contribute to deterioration of β-cell function and survival.
Introduction
Transplantation of pancreatic islets offers a potentially effective therapy for persons with type 1 diabetes mellitus (T1D) but is currently constrained by poor long-term success rates, with only 10% of recipients remaining insulin independent 5 years following transplantation, despite encouraging early results (46,47). The cause of this graft failure is not well understood but probably involves both immune and nonimmune mechanisms. Significant attention has been focused on the immunological factors (innate, acquired, and autoimmune), which may limit graft survival, but less is known about the role of the graft stress response in affecting graft survival. Islet cells face a number of stresses during isolation and after transplantation that result in apoptosis and a decline in functional cell mass (1,3—6, 36,38,43). Principal among these, hypoxia incurred during islet processing and nutrient deprivation during engraftment claim a significant proportion of the transplanted cells (9,14). The insufficient β-cell mass that survives is thereafter exposed to metabolic stress, resulting in erosion of the surviving graft and the development of a vicious cycle leading to graft failure (40). The concept of “β-cell exhaustion” to explain this type of graft loss was proposed long before a molecular mechanism for the process was defined (41).
Current understanding of the process of β-cell failure in type 2 diabetes (T2D) can provide valuable insight. In T2D, insulin resistance and chronic β-cell glucotoxicity pose a high metabolic demand for insulin and result in oxidative stress through the generation of reactive oxygen species (20,25). Together these events disrupt endoplasmic reticulum (ER) homeostasis, promoting the misfolding of nascent proteins in the ER. The ER stress pathway is a quality control mechanism that senses the accumulation of unfolded or misfolded proteins and activates an adaptive response termed the unfolded protein response (UPR) (21,42,44). This comprises three signaling pathways: inositol requiring protein-1 (IRE1) (31,48, 49), protein kinase RNA (PKR)-like ER kinase (PERK) (7,16), and activating transcription factor-6 (ATF6) (17,18). The UPR, a series of signaling cascades, initially has a protective mechanism aimed at alleviating the load of misfolded proteins in the ER. Prolonged ER stress, however, can shift UPR signaling to a proapoptotic mechanism via activation of CCAAT-enhancer binding protein (c/EBP)-homologous protein 10 (CHOP), c-Jun N-terminal kinase (JNK) pathway, and the caspase cascade (32,45,49,52,53). Pancreatic β-cells extensively perform the function of insulin synthesis and secretion and therefore have highly developed endoplasmic reticulum and high levels of UPR transducers, as well as chaperones immunoglobulin heavy chain-binding protein (Bip) and 94 kDa glucose regulated protein (GRP94) (15,35). It makes them highly susceptible to ER stress and even a minimal disturbance in the homeostasis may result in accumulation of unfolded proteins and activation of ER stress pathway that might contribute to β-cell failure and apoptosis (34,35).
Whereas studies of islet transplantation, and most theories regarding transplant failure, have focused on immunologic donor—recipient incompatibility, the concept of metabolic incompatibility has not been examined. The short-term success of the Edmonton protocol, however, which included the avoidance of steroid-based immunosuppression, illustrated that recipient metabolic demand and graft mass are crucial to achieve insulin independence in the short term. We presume ER stress to be one of the signaling pathways activated due to isolation stress and may contribute for apoptosis and cell death in isolated islets. The present study focused mainly on the role of ER stress in the context of apoptosis of isolated human islets and islet transplantation grafts. In this study, we document the induction of UPR pathways following isolation, and their persistence after transplantation. These findings may indicate that such factors as ER stress and UPR activation need to be examined more carefully as potentially modifiable causes of islet graft failure.
Materials and Methods
Pancreas Procurement, Islet Isolation, and Culture
Human pancreata were procured from multiorgan cadaveric donors after appropriate consent was obtained by personnel from Quebec-Transplant, the local organ procurement organization. Prior to isolation, a wedge biopsy was obtained, embedded in Tissue-TEK OCT (Sakura Finetek, USA), snap frozen in liquid nitrogen, and stored at −80°C. Islet isolation was performed at the Human Islet Isolation Laboratory, McGill University Health Centre, according to the protocol previously described by Ricordi et al. (39). Briefly, a 30°C solution of 2 mg/ml Liberase® enzyme blend (Roche Molecular, Indianapolis, IN) in Hanks' balanced salt solution (HBSS) (Mediatech, Herndon, VA) supplemented with 0.2 mg/ml DNAse I (Hoffman-La Roche, Montreal, Que.) was infused into the main pancreatic duct using a syringe. The distended pancreas was placed in a sterilized aluminum digestion chamber (Bio-Rep, Miami, FL) through which HBSS supplemented with penicillin (100,000 U/L) and fungizone (2,500 μg/L) (Gibco, Burlington, Ontario) was recirculated at 37°C. Extent of tissue digestion was assessed by staining aliquots of digestate with dithizone (Sigma, St. Louis, MO), and visualizing the islets under an inverted light microscope (Nikon, Montreal, Quebec). Cooling the circuit to between 5°C and 10°C when the majority of the islets were seen to be free of surrounding acinar tissue terminated the digestion process. The digestate was collected, centrifuged (400 x g), and washed three times in HBSS/ fetal calf serum (FCS) solution and islets were purified on a discontinuous UW-Ficoll density gradient (UW-Belzer solution, Barr Laboratories; Ficoll®, Sigma, St. Louis, MO) using a COBE 2991 Cell Processor (COBE BCT, Denver, CO). Approximately, 2,500 islet equivalents (IEQ) of isolated islets were frozen in embedded in Tissue-TEK OCT and 5,000 IEQ cultured in CMRL-1066 (Invitrogen) supplemented with 0.1% bovine serum albumin (BSA), penicillin/streptomycin (100 μg/ ml) (Wisent), and fungizone (Invitrogen) at 37°C in a 5% CO2 incubator. Culture medium was changed every other day.
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Approximately 2,500 IEQ were washed with PBS and total RNA was extracted with RNeasy kit (Qiagen). A total of 2 μg RNA was reverse transcribed with oligodT and the Thermoscript RT system (Invitrogen) and amplified with PCR using Taq polymerase (Bioshop) with the primers specific for X-box binding protein 1 (XBP-1). The housekeeping gene, ribosomal protein S16, was used as a reference. The primer sequences are reported in Table 1.
Sequences of the Primers Used for RT-PCR
hXBP1, human X-box binding protein 1; hS16, human ribosomal protein S16.
Cell Lysis and Western Immunoblot
Samples of islets (5,000 IEQ per group) were centrifuged, washed twice in PBS at 4°C, and the pellet resuspended in a Nonidet-P40 lysis buffer (Nonidet-P40, PMSF, aprotinin, sodium orthovanadate; Sigma-Aldrich Canada, Oakville, Ont.). The cells were sonicated for 15 s and centrifuged at 14,000 rpm for 20 min. The supernatant was recovered, frozen, and stored at −20°C. Minor variations in sample size were corrected by performing a total protein assay on an aliquot of each whole cell lysate using Bradford protein dye (Bio-Rad, Mississauga, Ont.). For all Western blots, equivalent amounts of protein were loaded in each well. Between blots, this varied from 75 to 100 μg/well. The islet cell lysates were diluted 4:1 in 6x sample buffer [0.375 M Tris (pH 6.8), 0.416 M SDS, 30% (v/v) glycerol, 2.89 mM bromophenol blue, 12% (v/v) β-mercaptoethenol]. The samples were boiled for 5 min (100°C) and 15-μl aliquots were loaded into 12% (v/v) Tris-glycine gels (Helixx, Toronto, Ontario) for protein separation by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The electrophoresed proteins were transferred to nitrocellulose (Bio-Rad, Mississauga, Ont.) and the membranes blocked overnight at 4°C in blocking buffer [26.4 mM Tris, 0.15 mM NaCl, 0.05% (v/v) Tween-20, 1% (w/v) bovine serum albumin, 1% chicken albumin (Sigma-Aldrich, Oakville, Ontario)]. The blocked membranes were probed with PARP1 [poly(ADP-ribose) polymerase 1; Cell Signaling Tech], eIF2α (eukaryotic initiation factor 2α; Cell Signaling Tech.), and anti-JNK rabbit polyclonal antibodies (Santa Cruz, CA, USA), overnight at 4°C. To evaluate kinase activation, blocked membranes were also exposed to anti-phospho eIF2α (Ser51) (Cell Signaling Tech.) and anti-phospho JNK (Thr183/Tyr185) (New England Biolabs, Mississauga, Ontario) for 48 h at 4°C. Membranes were then washed [26.4 mM Tris, 0.15 mM NaCl, 0.05% (v/v) Tween-20] and incubated with horseradish peroxidase-conjugated polyclonal anti-rabbit IgG secondary antibody (Amersham, Oakville, Ont., Canada) (in washing buffer with 5% milk) for 1 h. Proteins were visualized using the enhanced chemiluminescence (ECL) system (Amersham, Oakville Ont.). The membranes were stripped for 30 min at 65°C in stripping buffer [0.7% (v/v) 2-mercaptoethanol, 2% (w/v) SDS, 62.6 mM Tris, pH 6.7], washed, and reblotted. All experiments were repeated at least three times, with representative blots selected for presentation.
Apoptosis Assays
Caspase-3 activity was measured with a homogeneous luminescent assay that measures caspase-3/7 activities (Caspase-Glo® 3/7 Assay, Promega); 1,500 IEQ per sample were used. Caspase-3 activity was measured against a standard curve prepared with recombinant human caspase-3 (R&D Systems) in 96-well plates. Upon caspase-3 activity measurement, total DNA was extracted to correct for sample-to-sample size variation. The amount of DNA was measured with Quant-iT PicoGreen dsDNA assay kit (Invitrogen) containing a dsDNA binding fluorescence dye.
Islet Transplantation
CD-1 nude male mice (Charles River) (7 weeks old) weighing 25—30 g were used as islet recipients. Graft recipients were either normal or diabetic. Mice were made diabetic by a single intraperitoneal dose (250 mg/ kg) of streptozotocin in NaH2PO4 buffer under sterile conditions. Following establishment of diabetes, animals were anesthetized and LinBit slow-release insulin pellets (LinShin, Toronto, Ontario) were implanted subcutaneously. Marginal size grafts of 1,500 IEQ were transplanted under the left kidney capsule of both normal and diabetic mice. LinBit pellets were removed during surgery. Blood glucose measurements and weighing were performed after transplantation, every 2 days. The left kidney was removed to confirm return of hyperglycemia. Islet grafts were retrieved and paraffin embedded after 3 or 28 days for analysis. Three to five mice were tested for each category. All animal procedures were approved by the McGill University Animal Care Committee.
Immunohistochemistry and Immunofluorescence
Frozen sections for pancreas and the islets were fixed with acetone for 10 min. Antigen retrieval was performed by microwave heating for paraffin sections. All the antibodies [insulin, glucagon, CHOP, XBP-1, Bip, ATF6α (Santa Cruz Biotech) and cleaved caspase-3 (Cell Signaling)] were used at 1:200 dilution and the sections were incubated at 4°C overnight. The Immunocruz staining system (Santa Cruz Biotech) was used for immunoperoxidase staining. For immunofluorescence, Texas-red conjugated anti-rabbit antibody was used and sections were mounted with Prolong-Antifade mounting medium (Invitrogen) and analyzed with Nikon inverted microscope. As a negative control, primary antibody was replaced with appropriate serum.
Statistical Analysis
Data was normalized to the control and statistical analysis was performed using a paired t-test and calculating one-tailed p-values. The p-values were further adjusted using Bonferroni correction by multiplying with number of tests performed. Values of p < 0.05 were considered significant. Results are expressed as the means and error bars represent SEM.
Results
Islet Isolation Stress Induces Apoptosis in Isolated Islets
The cleavage status of PARP1 and the level of caspase-3 activity were examined in isolated human islets at 0, 1, 3, and 5 days postisolation. PARP cleavage was detected by Western blotting and was apparent after 1 day in culture (Fig. 1A) and protein expression was consistent until day 5. Activation of caspase-3, measured using a luminescent assay, was apparent in isolated islets and was observed to increase fourfold until day 3 and then reduced thereafter (Fig. 1B). These results are consistent with previously published data (37), indicating that apoptotic pathways are induced in human isolated islets due to isolation stress and these pathways are mediated by caspases.

Apoptosis occurs in isolated human islets. Isolated human islets were cultured up to 5 days postisolation and samples were taken at indicated times postisolation. Day 0 samples were collected immediately after the isolation. (A) Poly(ADP-ribose) polymerase 1 (PARP) and cleaved PARP were detected by immunoblot. The blot shown is representative of three independent experiments. (B) Caspase-3 activity was measured using caspase-3/7-Glo assay at the time of sample collection. After the assay, DNA was extracted from cell pellet and DNA content was measured to standardize caspase-3 activity to total DNA. Data are means of four independent experiments and error bars represent SEM. The p-values were adjusted using Bonferroni correction. *p < 0.05 compared with day 0.
Activation of UPR in Isolated Islets
We assessed the expression of ER stress markers in cultured isolated islets at various time points. For the first series of experiments, changes in the phosphorylation of eIF2α and JNK were measured by Western blot analysis. As previously reported (2,37), strong activation of JNK1 was observed immediately after the isolation and the signal decreased over time (Fig. 2A). Phosphorylation of eIF2α also followed a similar pattern (Fig. 2A). Next, we investigated activation of the XBP-1 by semiquantitative RT-PCR. Appearance of the spliced form of XBP-1 mRNA immediately followed islet isolation, and then diminished with time (Fig. 2B).

Characterization of the unfolded protein response (UPR) in isolated human islets. Isolated human islets were cultured up to 5 days and samples were taken every other day. Day 0 samples were taken immediately after the isolation. (A) Total cell lysates were analyzed for eukaryotic initiation factor 2α (eIF2α) and c-Jun N-terminal kinase 1 (JNK1) activation by immunoblot using phospho-specific antibodies against eIF2α and JNK1. Data are means of four independent experiments and error bars represent SEM. (B) X-box binding protein 1 (XBP-1) mRNA splicing were measured by semiquantitative RT-PCR. Percent XBP-1 splicing was calculated as described in Materials and Methods. Data are means of three independent experiments and error bars represent SEM. The p-values were adjusted using Bonferroni correction. *p < 0.05 and **p < 0.001 compared with day 0.
Absence of UPR Signals in the Donor Pancreas
Frozen sections of donor pancreas and isolated islets showed strong staining for both insulin and glucagon (Fig. 3). Islets within intact pancreas stained negative for Bip, CHOP, and ATF-6, and weakly for XBP-1 (Fig. 4Aa—e). In contrast, isolated islets stained strongly positive for all mediators (Fig. 4Af—j). Islets within the intact pancreas stained negative for cleaved caspase-3 (p17 subunit) whereas isolated islets were strongly positive (Fig. 4B).

Isolated islets are functional and express hormones insulin and glucagon. Frozen sections of human pancreas and isolated islets were analyzed for the presence of insulin and glucagon by immunofluorescence. The pancreatic sections were positively stained with insulin and glucagon (b, c). Frozen sections of isolated islets were also positively stained for insulin and glucagon (e, f). (a, d) The negatively stained sections for pancreas and isolated islets, respectively. Only representative images are shown.

(A) Analysis of ER stress mediators Bip, CHOP, XBP-1, and ATF6α in donor pancreas and isolated islets. Expression of endoplasmic reticulum (ER) stress mediators. Binding immunoglobulin protein (Bip), c/EBP-homologous protein 10 (Chop), XBP-1, and activating transcription factor-6α (ATF6α) were examined in the frozen sections of human pancreas and isolated islets by immunohistochemistry. The pancreatic sections (b, c, d, e) did not stain for these markers whereas the frozen sections of isolated islets (g, h, i, j) were positively stained for them. (a, f) The negatively stained sections for pancreas and isolated islets, respectively. Only representative images are shown. (B) Analysis of proapoptotic signals CHOP and cleaved caspase-3 in the donor pancreas and isolated islets. Frozen sections of human pancreas and isolated islets were analyzed for the presence of apoptotic signal CHOP and cleaved caspase-3 by immunofluorescence. The pancreatic sections did not stain for CHOP and cleaved caspase-3 antibodies (b, c) whereas frozen sections of isolated islets showed strong staining for both CHOP and cleaved caspase-3 (e, f). (a, d) The negatively stained sections for pancreas and isolated islets, respectively. Only representative images are shown.
UPR in Transplanted Islets Relates to Metabolic Demand
To assess the effects of glycemic stress on UPR signals in transplanted islets, a marginal mass of isolated human islets was transplanted under kidney capsule of normal and diabetic athymic mice. In the normal group, mean blood glucose 28 days after transplantation was 5 ± 2 mmol/L, whereas in the hyperglycemic stress group it was 15 ± 2 mmol/L. Islet grafts from both groups, retrieved at 3 and 28 days after transplantation, all showed strong staining for the hormones, indicating graft viability in all cases (Fig. 5).

Expression of islet hormones, insulin and glucagon, in islet transplanted grafts from normal and diabetic mice. Hyperglycemia was induced in nude mice by injecting streptozotocin and isolated islets were transplanted into the kidney capsule of normal and diabetic mice. The grafts were retrieved 3 and 28 days after transplantation and were analyzed for expression of insulin and glucagon by immunofluorescence. Islet grafts from normal (A) and diabetic (B) mice expressed both insulin and glucagon at 3 days and 28 days. Only representative images are shown.
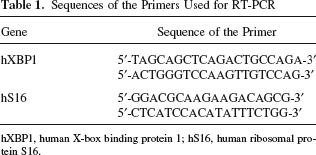
Graft sections were then analyzed for the UPR mediators Bip, XBP-1, and ATF6α by immunohistochemistry (Fig. 6). The grafts from the diabetic recipients, at both 3 days and 28 days after transplantation, revealed moderate or intense immunoreactivity for the UPR intermediates. On the other hand, the 3-day and 28-day grafts from normal recipients showed weak immunoreactivity for the UPR intermediates.
Hyperglycemia induces ER stress mediators in the isolated islet grafts. Hyperglycemia was induced in nude mice by injecting streptozotocin. The isolated Islets were transplanted into normal and diabetic mice both and the grafts were analyzed after 3 and 28 days for the presence of ER stress mediators Bip, XBP-1, and ATF6α by immunohistochemistry. The sections from the normal mice did not stain for these mediators (left panels). The sections from 3-day and 28-day grafts of diabetic mice were stained positively for all the markers (right panels). Only representative images are shown.
Caspase-3 Activation in the Islet Transplantation Grafts in Diabetic Mice
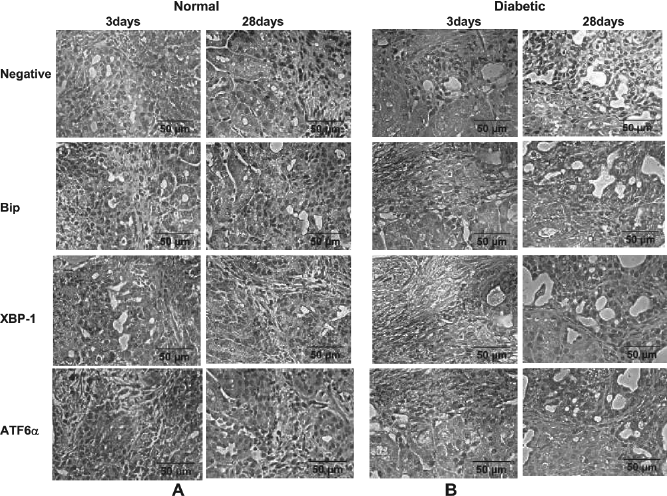
To establish the proapoptotic nature of the UPR, grafts were analyzed for CHOP and caspase-3 activation by immunofluorescence. A small number of cells were observed positive for CHOP and cleaved caspase-3 in the sections of the grafts from diabetic recipients whereas none or very few cells positively stained for CHOP and cleaved caspase-3 in the grafts from normal recipients (Fig. 7).
Hyperglycemia induces expression of ER stress-associated apoptotic mediators CHOP and cleaved caspase-3. The islet transplantation grafts were examined for ER-associated apoptotic marker CHOP and cleaved caspase-3 by immunofluorescence. No signals were observed for the grafts from the grafts of normal mice (left panels). A few cells positively stained for CHOP and cleaved caspase-3 in the grafts from diabetic mice (right panels). Apoptotic cells are marked with arrow. Only representative images are shown.
Discussion
Human islets are exposed to a variety of stresses during isolation, which may lead to cell death and graft failure (1,6,37,40). ER stress is known to be activated by mechanical or chemical stress, hypoxia, and other environmental or biochemical disturbances, many of which may occur during islet processing (8,14,23). It has also recently been found to be of great importance in β-cell loss and both type 1 and type 2 diabetes (11,24,34,35). In the latter, it may accelerate the process of cell loss, as increased metabolic demand is placed on a diminishing β-cell mass. The analogous situation in islet transplantation could see a marginal engrafted β-cell mass being affected by the disproportionately high metabolic demand of the recipient. While this has been hypothesized in the past, no mechanistic evidence has been provided for such a process to occur in the transplant context.
Isolated islets showed higher levels of activated caspase-3/7 and also the signs of PARP cleavage during the first 3 days in culture, indicating the activation of apoptotic signaling pathways. Caspase-12 is specifically involved in ER stress-related apoptosis in rodents (28, 32) and activates the downstream effector caspases-3, −6, and −7, followed by PARP cleavage (10,19). In humans, caspase-12 is inactivated by mutations in the majority of human populations (10,13,50,51) so the preliminary ER stress-related apoptotic effector is not known. Isolated islets also revealed elevated levels of ER stress transducers, such as phosphorylated eIF2α (P-eIF2α), spliced XBP-1, and phosphorylated JNK (P-JNK) immediately after isolation but these levels subsequently reduced in culture. The observation that the peak of UPR activation (P-eIF2α and sXBP-1) coincides with proapoptotic JNK activation, and these are followed by activation of apoptotic executors (caspase-3/7 and cleaved PARP). These results suggest that UPR signals activated during the islet isolation procedure are shifted to the apoptotic pathway by the third day of culture. The inference was further supported by the immunohistochemical studies of pancreatic and islet sections. Both the UPR and proapoptotic signaling molecules ATF6α, Bip, CHOP, and cleaved caspase-3 were not detected in the donor pancreatic tissue but were highly expressed in the isolated islets, implicating that UPR and apoptotic pathways must have been induced due to islet isolation stress. Another study has shown the isolation stress recruits JNK and NF-κB (nuclear factor of kappa light polypeptide gene enhancer in B-cells 1) pathway in islets, which are normalized in culture beyond 48 h (33). Therefore, ER stress is another signaling pathway activated during islet isolation procedure and could be responsible for loss of β-cells during islet isolation and engraftment.
Histological examination of islet grafts from normoglycemic or hyperglycemic nude mice showed that the grafts from all the recipients expressed insulin and glucagon. The islet grafts in normoglycemic mice showed either minimal or no expression of UPR mediators, similar to the situation in the native donor pancreas, whereas higher levels of UPR signals were observed even as early as 3 days after transplantation in hyperglycemic recipients and were maintained up to at least 28 days. Staining of sections of these grafts for the presence of apoptotic signals like CHOP and cleaved caspases-3 revealed the presence of scattered apoptotic cells in the grafts from diabetic mice only. These changes appeared to be consistent whether or not hyperglycemia was reversed in the recipient. Therefore, while the transplantation procedure is a potent stimulator of the ER stress response, potentially leading to transient cell loss by apoptosis, a continued metabolic demand placed on the graft may allow sustained activation of the UPR. This appears to be minimal in the grafts of normoglycemic mice, those in which the native endocrine pancreas function effectively suppressed or replaced that of the transplanted islets, and it did not potentiate apoptotic signaling pathways. In recipients that were hyperglycemic or perhaps marginally normoglycemic, where the transplanted islets were the only source of insulin, a more pronounced UPR was seen and this was also associated with histological evidence of continued apoptosis. Recent data suggest that the UPR serves only a protective function in islet grafts of normoglycemic mice (22). That study, however, did not specifically look at apoptotic activity in the grafts, nor did it compare recipients in which normoglycemia was maintained by native insulin secretion. The use of this control in the present study reveals important differences in UPR signaling and apoptotic activity when blood glucose is tightly controlled.
A large number of transplanted islets are lost during the early phase of transplantation due to nonspecific inflammatory responses (30). While these factors are certainly important in determining the fraction of transplanted islets surviving and engrafting within the liver, this study suggests that processes and pathways activated during this period may remain active and participate in long-term islet cell loss, even in an initially normoglycemic recipient. Activation of the UPR in islet grafts suggests that functional overload of islets may occur immediately following transplantation, at a time when the cells are recovering from the injury associated with isolation and implantation. At this time, cells may be viable yet damaged by inflammation and ischemia, and thus more susceptible to the deleterious effects of ER stress. Even mild hyperglycemia, under these conditions, could stimulate UPR-dependent apoptotic pathways. Earlier studies have shown that hyperglycemia induces ER stress and apoptosis through activation of the JNK pathway and caspase-1 in β-cells (12,26,27). Therefore, maintaining strict normoglycemia with exogenous insulin in the early phase following clinical islet transplantation may be crucial for long-term graft survival (29).
This study is an attempt to assess the role of ER stress in association with cell death during islet isolation and transplantation. The most important observations of this study are the induction of the UPR and ER stress-associated apoptosis in isolated human islets contributing to the significant islet losses pretransplantation. The exposure of these distressed islets to other kinds of stresses during transplantation or posttransplantation shifts UPR to apoptotic pathways, contributing to further β-cell loss. This continuous cell death process results in a vicious cycle of reduced, and metabolically challenged, β-cell mass and could be responsible for early or delayed graft failure. There is an ongoing need to understand the complex network of stress signaling pathways triggered by the isolation process and design appropriate strategies for interfering with them in the initial phase.
Footnotes
Acknowledgment
The authors declare no conflict of interest.
