Abstract
Canine mesenchymal stem cells (cMSCs) derived from umbilical cord blood represent a potentially useful source of stem cells for therapy. The aim of this study was to compare the effects of different transplantation times of cMSCs after spinal cord injury (SCI). A total of 21 dogs were subjected to SCI by balloon-induced compression of the first lumbar vertebrae for 12 h. Of the 21 dogs, 12 were divided into four groups of three according to the time of stem cell (1 × 106) transplantation at the injury site: control no treatment, 12 h, 1 week, and 2 weeks. The remaining 9 animals were negative harvest (HA) time controls for each treatment group (n = 3). Olby and Tarlov scores were used to evaluate functional recovery of the hindlimbs. Markers for neuronal regeneration (Tuj-1, nestin, MAP2, and NF-M), astrogliosis (GALC, GFAP, and pSTAT3), signal molecules for actin cytoskeleton (RhoA, Cdc42, and Rac1), inflammation (COX-2), and neurotrophins (NT-3) were evaluated by Western blot analysis. Scores of the 1-week transplantation group showed significant improvement compared to controls. Hematoxylin and eosin (H&E) staining revealed less fibrosis at the injury site in the 1-week transplantation group compared to other groups and immunohistochemistry showed increased expression of neuronal markers. Furthermore, in both 1-week and 2-week transplantation groups, Tuj-1, nestin, MAP2, NF-M, NT-3, and GFAP increased, but pSTAT3, GALC, and COX2 decreased. RhoA decreased and Rac1 and Cdc42 increased in the 1-week transplantation group. In conclusion, transplantation of cMSCs 1 week after SCI was more effective in improving clinical signs and neuronal regeneration and reducing fibrosis formation compared to the other transplantation times evaluated. Subsequently, these data may contribute to the optimization of timing for MSC transplantation used as a therapeutic modality.
Introduction
Responses to spinal cord injury (SCI) include disruption of the spinal cord vasculature, ischemia, glutamatergic excitotoxicity, oxidative cell stress, lipid peroxidation, inflammation, and scar formation (30,51). Inflammation or scarring as a result of SCI has been found to be detrimental to nerve regeneration (5,6,42). At the site of inflammation in the spinal cord, cyclooxygenase-2 (COX2) expression is markedly increased (21,40) and glial cells interact with inflammatory cells and injured axons (6). The formation of mechanically obstructive glial scars composed of connective tissue elements and glial cells such as astrocytes and oligodendrocytes were thought to explain the failure of central nervous system (CNS) regeneration after injury (6). Oligodendrocytes provide myelin sheaths for enhanced axonal transmission (6), and astrocytes are important for neurotransmitter regulation and ion homeostasis (6,47). But after injury they are the major cell type responsible for walling off areas of damage to protect normal tissue from further erosion. In addition, astrocytes respond to multiple extracellular signaling molecules through a complex assortment of intracellular signal transduction pathways that activate cyclic adenosine monophosphate (cAMP), mitogen-activated protein kinases (MAPK), NF-κB, Janus kinase-signal transducer and activator of transcriptions (Jak-STATs), and others, implicated as potential regulators of astrogliosis (11,16,33). For example, STAT3 is expressed by astrocytes, neurons, and other cell types in the CNS (2), and activation of STAT3 by phosphorylation increases markedly following CNS insults (17,43,50).
Targeting individual components might not be the most efficient approach to overcome inhibition by CNS myelin and glial scarring. Instead, identifying intracellular pathways common to multiple sources of inhibition may offer greater prospects for promoting axon regeneration. So far, the best characterized pathway involves small guanosine triphosphate hydrolases (GTPases) of the Rho family such as RhoA, Rac1, and Cdc42, which are known regulators of the actin cytoskeleton (25). RhoA has been shown to correlate with signals that induce growth, cone collapse, and axon guidance repulsion (7). Cdc42 is required for axon generation (9), while Rac1 is important for axon guidance rather than growth (3). It was also reported that neurotrophin expression was induced by human adult mesenchymal stem cell (MSC) transplantation (36). Neurotrophins (NTs) are a family of proteins including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), NT-3, and NT4/5 that induce the survival, development, and function of neurons (10,38). NT-3 helps to support the survival and differentiation of existing neurons and encourages growth of new neurons from neuronal stem cells (13,20,26,27).
These factors and how they interact with each other must be taken into consideration during the development of treatment strategies. However, to date there have been no reports detailing overall injured spinal cord regeneration with regard to MSC transplantation time. Furthermore, most previous studies employed parenchymal injections of MSCs directly into the lesion site 1 week after SCI to evaluate the effect of stem cells (4,18,22,23,39). Differential effects of transplantation times on human embryonic stem cells between 7 days and 10 months post-SCI have been reported (19). However, no studies have compared regenerative results inclusive of signal pathways and physical interactions between stem cells and microenvironments in relation to postinjury transplantation times. Therefore, in this study, we transplanted canine umbilical cord blood-derived mesenchymal stem cells (cUCB-MSCs) to the injured spinal cords at different times (12 h, 1 week, 2 weeks) after injury in an experimentally induced SCI dog model. We also evaluated the different transplantation times with respect to neuronal regeneration, the promotion of antiastrogliosis, neuronal extension, and anti-inflammation.
Materials and Methods
Animals
A total of 21 2–3-year-old dogs weighing an average of 5.85 ± 0.62 kg (12 female, 9 male) were used for the present study. All of the dogs were clinically judged to be in good health, neurologically normal, and had their own admission number from the Institute of Laboratory Animal Resources, Seoul National University (SNU-081006-2). During the experiment, all dogs were cared for in accordance with the Animal Care and Use Guidelines (Institute of Laboratory Animal Resources, Seoul National University, Korea). Body weights of the dogs were recorded every morning during the experimental period. The dogs were assigned to groups of three as follows: control with no treatment, transplantation (TP) of cUCB-MSCs at 12 h, 1 week, and 2 weeks after SCI, and negative control untreated harvest (HA) groups for each of the three different transplantation times. Spinal cords were harvested 8 weeks after MSC transplantation, 8 weeks after SCI for the control group, and 12 h, 1 week, and 2 weeks for the HA groups (Fig. 1).
Study design for the in vivo experiment. SCI, spinal cord injury.
Induction of Spinal Cord Injury
SCI was induced using a balloon compression method previously described (23). Briefly, the dogs were medicated and anesthetized with IV tramadol (4 mg/kg) (Toranzin; Sam Sung Pharm. Ind. Co., Korea), propofol (6 mg/kg) (Anepol; Ha Na Pharm., Korea), and subcutaneous atropine sulfate (0.05 mg/kg) (Atropine; Je Il Pharm., Korea). Anesthesia was maintained with 2% isoflurane inhalation (Aerane; Ilisung, Korea). A Datex-Ohmeda (Microvtec Display, UK) was used to monitor physiological measures including rectal temperature, oxygen saturation, and pulse rate during anesthesia. The dogs were placed in a ventral recumbent position and a hemilaminectomy was performed through a left paramedian approach at the forth lumbar segment (L4). A 3–5-mm hole was made in the left vertebral arch at L4 using a high-speed pneumatic burr and a 3-French embolectomy catheter (SORIN Biomedica, Italy) inserted. The balloon was advanced under fluoroscopic guidance until the tip of the catheter was at the cranial margin of the first lumbar segment (L1) vertebral body. The balloon was then inflated with 50 μl/kg of a contrast agent (Omnipaque; Amersham Health, Ireland), diluted 50:50 with saline. According to a previous study (23), the SCI model used occludes more than 80% of the spinal canal as confirmed by computed tomography. Following SCI, the soft tissues and skin were closed per standard methods. The balloon was fixed with a Chinese finger-type suture, and removed after 12 h. After the operation, the dogs were monitored in an intensive care unit, and if needed, manual bladder expression was performed at least three times daily until voluntary urination was established.
Isolation and Culture of cUCB-MSCs
The experimental materials consisted of cUCB-MSCs (from the Laboratory of Stem Cell and Tumor Biology, Department of Veterinary Public Health, Seoul National University). The culture procedure for cUCB-MSCs was previously described (15,23,41). Briefly, the low-density mononuclear cells were isolated using Ficoll-Paque Plus (Amersham Biosciences, Sweden). Next, the cells were cultured in Dulbecco's modified Eagle media-low glucose with 10% fetal bovine serum (Gibco-BRL, USA), 2 mmol/L L-glutamine (Gibco-BRL, USA), and 0.3% penicillin-streptomycin (Gibco-BRL, USA) at 37°C in 5% CO2 (26). The UCB-MSCs were cultured and the mononucleated cells that proliferated from the cord blood were characterized by fluorescence activated cell sorting (FACS) analysis (12,23,41).
Transplantation of cUCB-MSCs
Transplantation of cUCB-MSCs was performed at 12 h, 1 week, and 2 weeks after SCI. The dogs were anesthetized using the same methods described for induction of the injury. The control and untreated groups did not have any cells or medium transplanted after injury. In the transplant groups, a laminectomy was performed, and 1 × 106 cells suspended in 150 μl of PBS were transplanted into the injured region of the L1 spinal cord parenchyma with intact dura using a 30-gauge needle. All incisions were closed routinely.
Evaluations
Behavioral Assessment
Behavioral assessments were performed before the operation and weekly after the operation in order to evaluate functional recovery of the hindlimbs. Each dog was videotaped for a minimum of 10 steps from both sides and behind when walking on the floor. Dogs that could not bear weight on their hindlimbs were also videotaped while supported by holding the base of their tail. Using a 15-point scoring system (Table 1) (35) as well as revised and modified Tarlov scales (37), the dogs' gait was independently scored from the videotapes by two individuals unaware of the experimental conditions. A mean score was calculated every week after the SCI until end of experiment.
List of Olby Scores

Histopathological and Immunohistochemical Assessment
The control dogs were euthanized 8 weeks after SCI. The dogs in HA untreated groups were euthanized at each transplantation time (12 h, 1 week, and 2 weeks), and the dogs in TP group were euthanized 8 weeks after cUCB-MSC transplantation. The spinal cords from the 11th thoracic segment (T11) to the third lumbar segment (L3) of all dogs were extracted by dissection. The longitudinal half of each section was immediate fixed in 10% formalin at 4°C for 24 h. Each section was then placed in 10% sucrose/PBS at 4°C for 12 h and subsequently immersed in 20% sucrose solution overnight at 4°C. The other half of each section was immediate frozen with liquid nitrogen for Western blot analysis. The dura was removed by scissors, embedded in O.C.T compound (Tissue-Tek®), frozen, and longitudinally sectioned at 10 μm with a cryomicrotome. These sections were mounted on silane-coated slide glass and stained with hematoxylin and eosin (H&E) to detect vacuolar formation and fibrosis. The volume of fibrosis for each slide was calculated using an image analyzer program (Image J, version 1.38; National Institute of Health, USA) according to a previously described report (23). The area of the spinal cord was calculated from images of the longitudinal section at the epicenter of the injury. The area of fibrosis formation was calculated from the same image. Subsequently, the percentage of the fibrosis formation area in the longitudinal section of the epicenter of the injured spinal cord was analyzed. Primary antibodies were used against mature astrocytes [glial fibrillary acid protein (GFAP)], immature neurons [neuron-specific class III β-tubulin (Tuj-1)], neurofilament (NF-M), neural stem cells (nestin), and microtubules [microtubule-associated protein 2 (MAP2)] (Santa Cruz, CA, USA) for immunofluorescence determination. Tissues were fixed and permeabilized for 10 min with 0.1% (v/v) Triton X-100 and washed then preincubated with 1% bovine serum albumin (BSA; Sigma-Aldrich, St. Louis, MO, USA) in PBS for 30 min to decrease nonspecific antibody binding, and incubated with the primary antibodies for 24 h at 4°C. Secondary antibodies, anti-mouse IgM fluorescein isothiocyanate (FITC) were used against the primary antibodies. Propidium iodide (PI) (Invitrogen, USA) was added to identify nuclei. Tissues were mounted with aqueous mounting medium (DakoCytomation, USA) and kept in the dark at 4°C until analysis. Fluorescence images were visualized with a Fluoview 300 fluorescence microscope (Olympus, Tokyo, Japan).
Western Blot Analysis
The frozen half of each spinal cord section 0.25 cm from the epicenter of the injured region was used for Western blot analysis (each group, n = 3). The tissue was washed twice with PBS and lysed with buffer (20 mM Tris at pH 7.5, 1 mM EDTA, 1 mM EGTA, 1% Triton X-100, 1 mg/ml aprotinin, 1 mM phenylmethyl-sulfonylfluoride, 0.5 mM sodium orthovanadate) for 30 min on ice. Lysates were cleared by centrifugation (10 min at 15,000 rpm, 4°C) and protein concentrations were determined using the Bradford method (1). Equal amounts of protein (20 μg) were resolved by electrophoresis on 10% SDS-PAGE gels and transferred to polyvinylidene fluoride membranes. Membrane blots were washed with TBST (10 mM Tris-HCl, pH 7.6, 150 mM NaCl, 0.05% Tween-20), blocked with 5% skim milk for 1 h, and incubated with appropriate primary antibodies at recommended dilutions. Antibodies used include: anti-actin (Sigma), anti-Tuj1 (Abcam, UK), anti-nestin (Abcam, UK), anti-MAP2, anti-NF-M, anti-GFAP (Chemicon, Millipore), anti-NT-3, anti-Cox2, anti-GALC (galactosylceramidase; an oligodendrocyte marker), anti-pSTAT3 (a hallmark of astrogliosis), anti-RhoA, anti-Rac1, and anti-CDC42 (a neuronal extension marker, Santa Cruz Biotechnology, Santa Cruz, CA, USA). The membrane was washed and the primary antibodies were detected with goat anti-rabbit IgG or goat anti-mouse IgG conjugated to horseradish peroxidase. Bands were visualized by enhanced chemiluminescence (Amersham Pharmacia Biotech, England, UK).
Statistical Analysis
Data are presented as median and quartiles. Statistical analyses were performed using SPSS (version 17.0 SPSS Inc., Chicago, IL, USA). In all experiments, repeated measures of Kruskal-Wallis tests followed by Bonferroni corrected Mann-Whitney U-tests were used to compare four groups. A value of p < 0.05 was considered to be significant.
Results
Behavioral Analysis
All injured dogs showed complete pelvic limb paralysis after SCI, with the Olby score prior to SCI being 14, and 0 afterwards. However, at transplantation time, the scores of the 12 h, 1 week, and 2 weeks TP groups were 0, 2, and 2, respectively (Fig. 2A). Increases in the Olby scores were first observed 2 weeks after cUCB-MSCs transplantation, except for in the 2-week TP group. In all groups, the scores gradually increased during the study period, but the rate of increase decreased after 3 weeks. The Olby scores in the control, 12-h, 1-week, and 2-week TP groups increased to 2(2,2), 2(2,2), 4(2,7), and 2(2,2) at 3 weeks and 3(3,4), 5(4,5), 7(5,9), and 5(2,5) at 8 weeks after cMSCs transplantation, respectively. There was a significant difference between the 1-week TP group and the other groups from 3 to 5 weeks after cMSCs transplantation (p < 0.05). Although no significant differences were observed among TP groups from 6 weeks after cMSCs transplantation, there was a significant difference between the 1-week TP group and control group (p < 0.05). In addition, using revised and modified Tarlov scales, motor function in the 1-week TP group recovered significantly compared to the control group (p < 0.05) (Fig. 2B, Table 2 and 3).
Revised Tarlov Scores

p-Value based on Bonferroni corrected Mann-Whitney U-test.
Modified Tarlov Scores

p-Value based on Bonferroni corrected Mann-Whitney U-test.

Behavioral analysis following canine mesenchymal stem cell (cMSC) transplantation. Olby scores of control, 12-h, 1-week, and 2-week transplantation (TP) groups were 14 before SCI. Post-cMSC transplantation, the Olby score of the 1-week TP group was significantly increased compared to other groups 3–5 weeks after cMSC transplantation (A). ∗p < 0.05 versus control, 12-h, and 2-week groups. #p < 0.05 versus control. Motor function outcome 8 weeks after cMSC transplantation recorded by two grading systems (B, Revised Tarlov scores; C, Modified Tarlov scores). x = mean, + = median.
Histopathological and Immunohistochemical Assessments
Myelomalacic changes were observed in the 12-h HA group (data not shown). At 8 weeks after cMSC transplantation, atrophic and fibrotic changes in the injured region were detected in control and TP groups (Fig. 3). Sizes of the injured region in control, 12-h, 1-week, and 2-week groups were 1(0.6, 2.4), 0.8(0.4, 1.2), 0.5(0.2, 0.6), and 0.5(0.7, 1.2) cm, respectively. However, there was not a significant difference in lesion sizes among the four groups as determined by the Kruskal-Wallis test (p = 0.297). Histopathological analysis with H&E staining revealed severe parenchymal fibrosis in control and TP groups (Fig. 4). Most of the dogs in the control and TP groups had damaged tissues and vacuolar formation. However, fibrosis formation was decreased in the TP groups compared to the control group (Table 4). Fibrosis in the control group was observed cranially and caudally over the lesion (Fig. 4A). For the 12-h and 2-week TP groups, fibrosis was localized to the injured lesions (Fig. 4B, D) and there was less fibrosis in injured lesions in the 1-week TP group than in the other groups (Fig. 4C). Immunohistochemistry revealed neuronal markers in injured lesion (Fig. 5). Neuronal markers observed in the 12-h TP group were weak; however, these markers did not show up in controls. In the 2-week TP group, neuronal markers were more localized and encircled compared to the 1-week TP group.

Gross findings 8 weeks after SCI. Injured spinal cord lesions in all groups show atrophic and fibrotic changes. Scale bar: 1 cm.

Histopathological evaluation. Representative longitudinal sections of the lesions show fibrous tissue in the control (A), 12-h TP (B), 1-week TP (C), and 2-week TP (D) groups. (A) Diffused fibrosis formation was detected throughout the SCI lesion including the cranial and caudal regions. (B) Fibrosis and vacuolar cavity formation were decreased compared to controls and localized through the SCI lesion. (C) Vacuolar formation was detected around the center of the SCI lesion, but fibrosis formation was decreased compared with other TP groups. (D) Compared to the 12-h TP group, fibrosis and vacuolar cavity formation were localized in the epicenter of the SCI lesion. The red box represents magnified lesion and 1-cm section. Scale bar: 1 cm.

Immunofluorescence staining at different time points after transplantation. β-III tubulin (Tuj-1), glial fibrillary acidic protein (GFAP), microtubule associated protein (MAP2), neurofilament M (NF-M). Scale bar: 50 μm.
Percentage of Fibrosis Formation Area in a Longitudinal Section of the Epicenter of Injured Spinal Cord

All values are mean ± SD.
Area was calculated using an image analyzer program.
p < 0.05, transplantation versus control.
Inflammation and Astrogliosis
In HA groups, COX2 and GALC expressions were increased significant at 1 week after SCI, but decreased at 2 weeks (p < 0.05) (Fig. 6A, B). Expression of GFAP increased gradually until 2 weeks after SCI. The significant increase of pSTAT3 was observed in 1-week and 2-week HA groups compared to the 12-h HA group (Fig. 6B). We next examined whether cMSC transplantations were associated with anti-inflammation and antiastrogliosis. In TP groups, the level of COX2 was significantly decreased in all TP groups compared to the control group (Fig. 6A). The COX2 levels of 1-week and 2-week TP groups decreased more than in the 12-h TP group and 1-week and 2-week HA groups. However, the COX2 level in the 12-h TP group was not different from that in the 12-h HA group. In addition, expression of GALC in the 2-week TP group was decreased significantly compared to the control and other TP groups. The level of GALC in the 12-h TP group was significantly higher than in the 12-h HA group. GFAP levels in the 12-h and 1-week TP groups were increased significantly compared to control and HA groups.
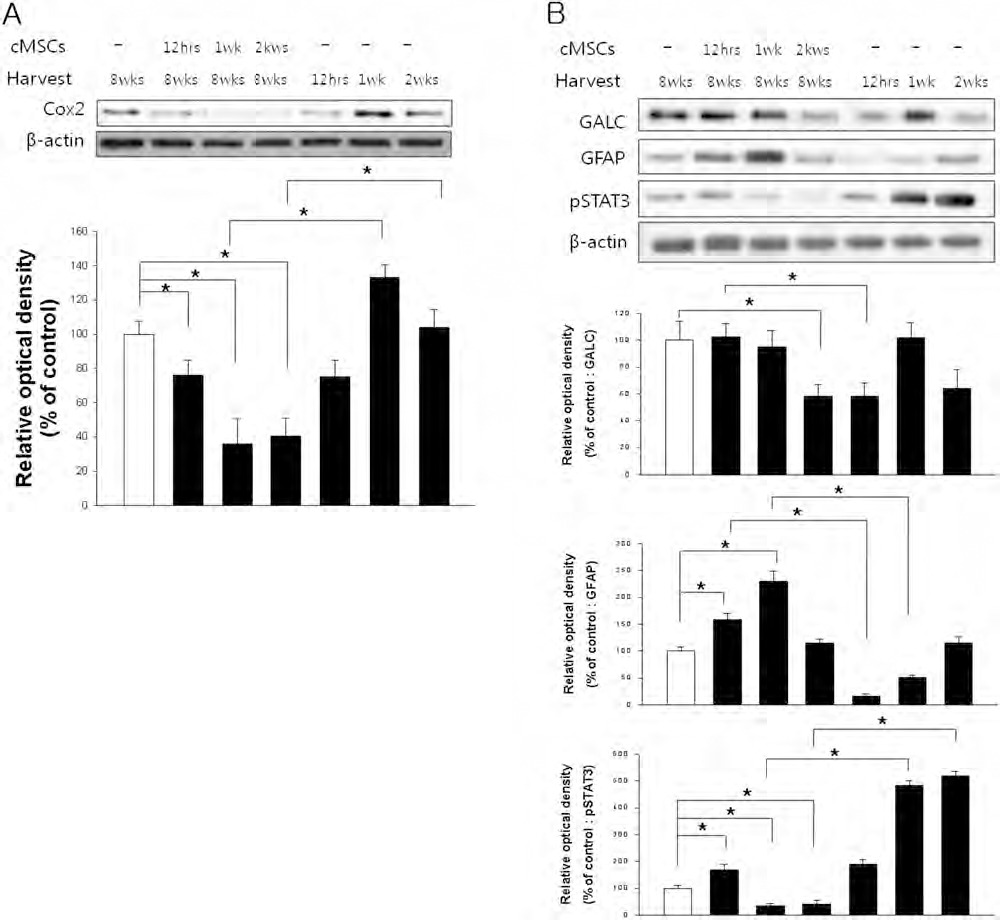
Inflammation and astrogliosis. (A) The expression of cyclooxygenase 2 (COX2) was significantly decreased in all TP groups compared to control. In 1-week and 2-week TP groups, the COX2 level was significantly decreased compared to all transplantation times. (B) In comparison to control, the level of galactosylceramidase (GALC) was significantly decreased in the 2-week TP group, and the level of glial fibrillary acidic protein (GFAP) was significantly increased in the 12-h and 1-week TP groups. Expression of phosphorylated signal transducer and activator of transcription 3 (pSTAT3) was significantly increased in the 12-h TP group compared to control, but decreased in 1-week and 2-week TP groups compared to control. The graph and bars denote means ± SE for each condition as determined by densitometry relative to β-actin. An ∗ and bar indicates that there is a significant difference (p < 0.05).
Expression of pSTAT3 in 1-week and 2-week TP groups was significantly decreased compared to the control group, 12-h TP group, and each HA group (Fig. 6B).
Intracellular Pathways for Regulators of Actin Cytoskeleton
RhoA, Rac1, and Cdc42 were examined to confirm the expression of intracellular pathways for actin cytoskeleton regulators at each transplantation time. In HA groups, RhoA levels were not significantly different. Rac1 levels gradually increased until 2 weeks. The Cdc42 level in 1-week and 2-week groups increased significantly compared to the 12-h group (Fig. 7). The expressions of RhoA in all TP groups were significantly decreased compared to the control group and all HA groups and the level in the 1-week group was lowest (p < 0.05). Levels of Rac1 and Cdc42 in the 1-week and 2-week groups were significantly increased compared to control, 12-h TP, and all HA groups (Fig. 7).

Intracellular pathways for regulators of actin cytoskeleton. The expression of Rho A was significantly decreased in all TP groups compared to control and each transplantation time. The levels of Rac1 and Cdc42 were significantly increased in 1-week and 2-week groups compared to control. The graph and bars denote means ± SE for each condition, as determined by densitometry relative to β-actin. An ∗ and bar indicates that there is a significant difference (p < 0.05).
Neurotrophin-3 and Neuronal Regeneration
In the present study, we investigated NT-3, Tuj-1, MAP2, NF-M, and nestin levels associated with various cMSC transplantation times. In HA groups, NT-3 and MAP2 expression were increased significantly at 1 week, but decreased at 2 weeks (p < 0.05). The expression of NF-M gradually decreased until 2 weeks. Expression of Tuj-1 and nestin were not significantly different among HA groups (Fig. 8). We next examined whether transplantation of cMSCs was associated with NT-3 production and neuronal regeneration. NT-3 and nestin levels in the 1-week and 2-week TP groups were significantly increased compared to control and all HA groups. The Tuj-1 level of all TP groups was significantly increased compared to control and all HA group. The transplantation of cMSCs significantly increased the MAP2 levels in 12-h and 1-week groups compared to the control and 2-week TP group. Although the MAP2 level of the 2-week TP group was significantly increased compared to the 2-week HA group, there was no significant difference compared to control. In addition, NF-M expression in 12-h and 1-week TP groups was significantly increased compared to control, and an increase in NF-M expression in the 1-week TP group was also observed when compared to each HA group.

Neurotrophin-3 and neuronal regeneration. The expressions of NT-3 and nestin were significantly increased in 1-week and 2-week groups compared to control, and the expressions of MAP2 and NF-M were significantly increased in 12-h and 1-week TP group compared to control. The level of Tuj-1 significantly increased in all TP groups compared to control and each transplantation time. The graph and bars denote means ± SE for each condition, as determined by densitometry relative to β-actin. An ∗ and bar indicates that there is a significant difference (p < 0.05).
Discussion
In the present study, transplantations of cMSCs 1 week after SCI significantly improved clinical signs evaluated by Olby (34,46) and Tarlov scales (37). The 12-h TP group showed no significant difference in Olby scores when compared to the control group in the early period. Therefore, these results suggest that immediate transplantation of cMSCs into the spinal cord after SCI delayed the improvement of clinical signs during the early treatment process, and the peak inflammation period should be avoided when transplanting cells. We also confirmed that cMSC transplantation 1 week after SCI reduced atrophic and fibrotic morphologic changes and increased expression of neuronal markers.
Identifying specific markers and intracellular pathways that are common to multiple sources of inhibition could offer greater prospects for evaluating injured spinal cord regeneration. Neuronal regeneration after SCI is influenced by inflammation, astrogliosis, neurotrophic factors, and the production of both growth promoting and inhibitory extracellular molecules (6). Inflammation within the spinal cord has been considered a potential source of cytokines and other signaling molecules that can lead to upregulation of inhibitory molecules after injury.
COX2 expressed within neurons and glia of the CNS plays an essential role in the development of inflammation (45). However, the ability of MSCs to create a tolerogenic niche by direct interaction with immune cells and secretion of regulatory molecules makes them attractive therapeutic candidates in the regulation of inflammatory response to injury (14,48). In the present study, cMSCs decreased COX2 expression in 1-week and 2-week transplantation groups. The glial cells of the CNS act as both structural and physiological support, and also respond to injury or disease. The formation of a mechanically obstructive glial scar composed of astrocytes, oligodendrocytes, and connective tissue elements was at one time thought to explain the failure of regeneration within the spinal cord after injury (6). In addition, phosphorylated STAT3 is a critical regulator of certain aspects of reactive astrogliosis and provides evidence that scar-forming astrocytes restrict the spread of inflammatory cells after SCI (11). This signaling mechanism is important because it determines not only the degree to which reactive astrocytes maintain or suppress function, but also whether reactive astrocytes initiate new injury-induced activities. Our results demonstrated that transplanted cMSCs affect immunomodulation, and modulate intracellular signaling which downregulates astrogliosis.
In order to further investigate molecular interactions associated with cMSC transplantation, we checked for expression of growth promoting and inhibitory extracellular molecules. This study showed that transplanted cMSCs modulated the activity of Rho GTPases (Rac1, Rho, and Cdc42), which play important roles in regulating the actin cytoskeleton and subsequent morphological changes in various cellular functions (8,24,49). In addition, constitutive activation of RhoA downregulates Rac1 activation via a Rho-associated kinase-dependent pathway, and activation of RhoA blocks neurite formation by inhibition of Rac1, suggesting an antagonistic effect for Rho and Rac in the control of neuronal morphology (44,49). Our results suggest that transplanted cMSCs promote the formation of neuronal network through regulating the intracellular pathways of the actin cytoskeleton to overcome repulsive forces which result from scar formation and severance of axon.
To further verify the role of cMSC transplantation in the regeneration of injured spinal cords, NT-3 was investigated as a neurotrophic factor. Exogenous cMSCs provided a supportive environment for neuronal repair and regeneration through trophic effects (4,29,31,32). Neurotrophic factors secreted by transplanted stem cells may have a more prominent therapeutic effect than differentiation of transplanted stem cells and establishment of functional connections (4). In the present study, levels of NT-3 and neuronal markers were increased in 1-week and 2-week TP groups. These results demonstrate that increases of GFAP or neuronal marker levels were consistent with NT-3 production, which was similar to that reported by a previous study (28).
Our results indicate that cMSCs transplanted to the site of SCI improved clinical signs by anti-inflammatory effect, regulating signal molecules related to astrogliosis, neuronal extension, and NT-3. These collective effects have been more enhanced in 1 wk transplantation after SCI than in 2-week transplantation after SCI. The optimal time for cMSC transplantation as a therapeutic modality may not be immediately after SCI or once fibrosis has progressed.
Footnotes
Acknowledgments
This work was supported by the Research Institute for Veterinary Science at Seoul National University, the BK21 program for Veterinary Science, NRF (MEST No. 2009-0081395), and the Seoul R&BD program (10548). The authors declare no conflicts of interest.
