Abstract
Spinal cord injury (SCI) is a common clinical disease that places a heavy burden on families and society. Cellular therapy provides a method of giving a supplement of cells lost in the injury and promoting functional recovery after SCI. Neural stem cells (NSCs) and olfactory ensheathing cells (OECs) are two most promising cell types. NSCs have the potential of differentiating into neurons and glial cells, and OECs could help the axons of neurons pass through the glial scar to promote functional recovery. NSCs were isolated from the cortices of fetal rats on days 12–14 of embryonic development and OECs were isolated from the olfactory bulbs of adult rats. In vitro coculture studies demonstrated OECs could promote NSCs to differentiate into neurons. Four groups of rats that had been 3/4 spinal cord transectioned at T9 were injected with DMEM/F12 solution, NSCs, OECs, and NSCs + OECs, respectively, 7 days post-SCI. Twelve weeks postoperation, the hindlimb locomotor function of rats in the cotransplantation group was significantly improved compared with that in the other three groups. Histological observation and immunohistochemical staining of NF-200 both showed new nerve fibers across the injured region. Cotransplantation of NSCs and OECs might have a synergistic effect on promoting neural regeneration and improving the recovery of locomotion function. Cotransplantation of NSCs and OECs was better than a single graft of either NSCs or OECs. These findings have provided a new way of thinking in the treatment of SCI.
Introduction
Spinal cord injury (SCI) is one of the most serious clinical diseases and the incidence has increased in recent years. Traffic accidents and trauma often cause SCI, resulting in life-long paralysis or even death, which places a heavy burden on families and society. Paraplegia caused by SCI is a constant challenge in clinical medicine, and facilitating recovery of function after SCI is still an important and difficult problem to neuroscientists. It was previously thought that the axons of adult mammals regenerate poorly after central nervous system (CNS) injury, but studies in the last two decades have shown that CNS neurons and axons still have the regenerative capacity in a suitable microenvironment (9,25,32,45,49). In view of the current research, the disadvantages of the microenvironment within the injured spinal cord for axon regeneration and extension toward the target site are as follows: the formation of glial scar in the lesion area, a process that produces inhibitory chondroitin sulfate proteoglycans (CSPGs) (35,40); secondary apoptosis and demyelination after SCI (15,30); inadequate endogenous neurotrophic factors (NTFs) and stem cells (46,50); and the secretion of axon growth inhibitors by oligodendrocytes (13,34,41).
Cellular therapy provides a method of giving a supplement of cells lost in the injury and promoting functional recovery after SCI. Embryonic stem cells (ESCs) (12), bone marrow stromal cells (BMSCs) (28), Schwann cells (49), olfactory ensheathing cells (OECs) (25,36), and neural stem cells (NSCs) (27,45) have been tried in the treatment of SCI. Among them, NSCs and OECs are the two most promising cell types. NSCs are undifferentiated populations of cells residing in the tissue lining the ventricular system of both the embryonic and adult mammalian CNS. NSCs have the ability to self-renew and the potential of differentiating into any one of the principal CNS cell types (neurons, astrocytes, and oligodendrocytes) according to the local environment. These properties make NSCs a good source of cells for cell replacement therapy after CNS injury. Also, implanted NSCs may proliferate, migrate, differentiate, and constitutively secrete NTFs that support host axonal regeneration after SCI (27,42). However, implantation of NSCs alone does not produce a significant restorative effect because the majority of NSCs transplanted into the spinal cord differentiate into astrocytes (14). Although astrocytes can secrete NTFs and limit the pervasion of the inflammatory reaction, the formation of glial scar blocks axon growth. How to maximize the differentiation of NSCs into neurons and guide the axonal regeneration through the astroglial scar is the key problem in the treatment of CNS injury.
In recent years, the role of OECs in SCI repair has attracted the extensive attention of researchers. The OECs reside in the outer layer of the olfactory bulb, termed the olfactory nerve layer, and also ensheath the olfactory nerve as it travels from the olfactory epithelium to the olfactory bulb (11,48). Based on antigenic criteria and morphology, there are two types of OECs: one has Schwann cell-like properties with a spindle-like morphology and the other type has astrocyte-like properties with a flattened fried egg morphology. Unlike oligodendrocytes, which are found only in the CNS and Schwann cells that are found only in peripheral nervous system (PNS), OECs are a unique type of glial cell that arise from the olfactory placode and occur both peripherally and centrally along the olfactory nerve (37). After being transplanted into injured spinal cord, OECs have been demonstrated to stimulate the regeneration of severed descending axons and improve functional recovery (25,48). Although the precise molecular mechanism of OECs supporting axonal growth is not fully understood, OECs express many molecules that are beneficial for such a task, including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial cell-line derived neurotrophic factor (GDNF). Moreover, they secrete extracellular matrix (ECM) such as laminin, fibronectin, and type IV collagen, which promote axonal growth (11). The primary advantage of using OECs may be that they migrate into and through the glial scar and therefore facilitate axonal regrowth through an injury barrier. Clinical study has also shown that OECs transplantation promotes partial functional recovery in patients after SCI (16).
Cellular replacement and axon guidance are both necessary for the repair of SCI. In consideration of the complementary biological properties of NSCs and OECs, according to our previous hypothesis (1), in the present study we separately cultured NSCs and OECs and cocultured NSCs with OECs in vitro to investigate the interaction between the two kinds of cells. Then, for the first time, we cotransplanted NSCs and OECs into the injured area of 3/4 spinal cord-transected rats to investigate their synergistic effect on the repair of adult rat SCI.
Materials and Methods
Isolation, Purification, and Culture of NSCs
NSCs were isolated from the cortices of fetal rats on days 12–14 of embryonic development. In brief, the pregnant rats were anesthetized by intraperitoneally injecting 5% chloral hydrate (8 ml/kg body weight). The embryos were isolated from the uterus, and the cortices were dissected under a stereomicroscope in a sterile condition. After removing the meninges and blood vessels under a stereomicroscope, the tissue was cut into 1-mm3 pieces and incubated with 0.25% trypsin (Gibco, USA) at 37°C for 15 min. Trypsinization was stopped by adding Dulbecco's modified Eagle medium/nutrient mixture F12 (1:1) (DMEM/F12; Gibco, USA) with 10% fetal bovine serum (FBS; Hyclone, USA). Then the cell suspension was centrifuged at 1000 rpm for 5 min, and the tissue was resuspended in DMEM/F12 medium with 2% B-27 supplement (Gibco, USA) and basic fibroblast growth factor (bFGF; 20 ng/ml; Sigma, USA). After filtration, cells with a density of 1 × 105/ml were cultured in a humidified incubator at 37°C with 5% CO2.
After 1 day of culture, fibroblast cells showed a quick attachment on the culture dish, and the suspended NSCs with medium were transferred to a new culture dish. The medium was changed every 2 days.
Isolation, Purification, and Culture of OECs
OECs were isolated from the outer two layers of olfactory bulbs. Briefly, adult male Sprague-Dawley (SD) rats (2 months old; purchased from the Animal Center of Haidian, Beijing) were killed by decapitation, and the olfactory bulbs were placed in ice-cold Hank's balanced salt solution (HBSS) in a sterile condition. The same as stated above, after centrifugation, the cell suspension was centrifuged at 1000 rpm for 5 min, resuspended in DMEM/F12 medium with 10% FBS. The tissue was triturated with a fire-polished Pasteur pipette (15–20 times) and filtered through a cell strainer (200 mesh) to obtain a single cell suspension. Cells were then plated in a culture dish at a density of 1 × 105 /ml and maintained at 37°C with 5% CO2.
According to the differential rates of attachment of fibroblast cells and OECs (33), after 12 h of culture, suspended cells were centrifuged and subsequently expanded in DMEM/F12 medium with 20 μmol/L forskolin (Sigma, USA) and 20 μg/L bovine pituitary extract (BPE; Invritrogen, USA). Twelve hours later, the supernatant and suspended cells were transferred to another new culture dish coated with poly-l-Lysine (Sigma, USA). After 2 days of culture, cells were treated with 1 × 10-5 mol/L cytosine-barabinoside hydrochloride (Ara-C; Sigma, USA) for 24 h, then washed with PBS and subsequently expanded in DMEM/F12 medium with 20 μmol/L forskolin and 20 μg/L BPE. The medium was changed every 2 days.
Coculture of NSCs and OECs In Vitro
In order to investigate the differentiation of NSCs and the effect of OECs on NSCs differentiation, NSCs were cultured alone and cocultured with OECs in DMEM/F12 medium with 10% FBS. After 5 days, the coverslips with cultured NSCs grown in the absence or presence of OECs were removed from the culture dishes and processed for immunocytochemistry.
Immunocytochemistry
NSCs were immunostained with nestin, which is widely used in characterization of NSCs, and OECs displayed positive immunoreactivity to S100β and P75 (low-affinity nerve growth factor receptor). So the characterization of NSCs and OECs was performed by using rabbit anti-nestin antibody (1:150; Sigma, USA) and rabbit anti-S100β antibody (1:200; Sigma, USA), respectively. The differentiation of NSCs was tested by using monoclonal mouse anti-β-tubulin III antibody (1: 100; Chemicon, USA) and polyclonal rabbit anti-glial fibrillary acidic protein (GFAP) antibody (1:1000; Chemicon, USA). Cells cultured on coverslips were fixed with 4% paraformaldehyde at room temperature for 30 min, washed several times in phosphate buffered saline (PBS, pH 7.4), and rinsed in PBS with 0.1% Triton X-100 (PBST) for 30 min. Then cells were incubated with a blocking solution including 5% goat serum for 1 h. Coverslips were then incubated at room temperature for 2 h in PBST containing the diluted primary antibodies. After washing, cells were incubated for 1 h in PBST containing the secondary antibodies conjugated to fluorescein isothiocyanate (FITC) or tetraethyl rhodamine isothiocyanate (TRITC). Nuclear staining was performed by incubating the coverslips in a solution PBST with 4′,6-diamidino-2-phenylindole (DAPI; 1:1000; Dojindo, Japan) for 10 min. Finally, after washing several times, the preparations were visualized by using a confocal laser scanning microscope (Olympus, Japan).
Establishment of Thoracic SCI Rat Model and Cell Transplantation
Twenty-four male SD rats (200–250 g; purchased from the Animal Center of Haidian, Beijing) divided into four groups (control group, NSCs group, OECs group, and NSCs + OECs group) were anesthetized by intraperitoneally injecting 5% chloral hydrate (8 ml/kg body weight). Under an operation microscope, laminectomy was performed at the level from the eighth to tenth thoracic vertebrae, followed by a right lateral 3/4 incision at the T9 level. After local hemostasis, muscle and skin were sutured. If the rat had died of excessive loss of blood during the operation, we performed a supplemental operation to ensure there was the same number of samples in the four experimental groups. Seven days after SCI, the four groups of rats were injected with 10 μl DMEM/F12 medium, NSCs suspension, OECs suspension, and NSCs + OECs suspension, respectively, by a microinjector. Cell suspension at a density of 1 × 109/ ml was obtained for transplantation. For cotransplantation, NSCs and OECs were mixed in equal number to make a homogenous suspension. The 5-μl suspension was injected into two sites rostral and caudal (about 0.5 mm) to the lesion at a depth of 2 mm with the microinjector retained for 5 min. An immunosuppressant was not used in our study (31,44). The rats were returned to their cages with easily accessible water and food at room temperature during the recovery period of up to 12 weeks. The bladder was massaged twice daily after SCI until normal bladder function was restored.
All the animal studies were performed in line with the US National Institute of Health Guide for the Care and Use of Laboratory Animals and approved by the Beijing Administration Committee of Experimental Animals.
Behavioral Assessment
Once prior to the operation, 1 day after, and then once every week after the operation, two observers blind to the surgery carried out the Basso-Beattie-Bresnahan (BBB) test in an open field to evaluate the recovery of locomotor function after SCI (2).
Immunohistochemistry
Twelve weeks post-SCI, the rats from each group were deeply anesthetized by intraperitoneally injecting 5% chloral hydrate (8 ml/kg body weight) and perfused transcardially with 0.1 M PBS (pH 7.4), followed by 4% paraformaldehyde in PBS for fixation of tissue. The brain and spinal cord were carefully excised and fixed in the same fixative at 4°C for 24 h and stored in 30% sucrose in PBS overnight. Serial tissue sections of 10 μm thickness were cut in cryostat microtome (Leica CM 1900, Germany). Monocolonal rabbit anti-neurofilament 200 (NF-200) antibody (1:100; Sigma, USA), and polyclonal mouse anti-S100 antibody (1:150; Chemicon, USA) were used. These sections were incubated for 2 h in primary antibody. The sections were then washed three times with 0.1 M PBS (pH 7.4) and incubated with secondary antibodies conjugated to FITC or TRITC for 1 h. Finally, the sections were coverslipped and then visualized by a confocal laser scanning microscope.
Semithin Section with Toluidine Blue Staining
After the perfusion described above, the middle part of the injured spinal cord was transected into small segments of 2 mm in thickness, and then the segments were washed three times with PBS, 5 min each time, followed by postfixation in 1% osmium tetraoxide solution at 4°C for 2 h. After that, the segments were washed three times with PBS and dehydrated in upward gradient alcohol and acetone, then embedded in Epon resin. Semithin sections (0.5 μm) were cross cut using an ultramicrotome, stained in 1% toluidine blue solution for 5 min, washed with distilled water, dehydrated in absolute alcohol, transparentized with xylene, and coverslipped immediately with mounting media. Then the sections were observed under a light microscope equipped with a digital camera system (DP-71; Olympus, Japan).
Statistical Analysis
Data were presented as mean ± SD. Statistical comparisons were performed using ANOVA followed by Bonferroni correction to determine where the significant differences occur, and differences of p < 0.05 were considered statistically significant.
Results
Purification and Characterization of NSCs
Because fibroblast cells showed a quick attachment on the culture dish, after 24-h culture NSCs were not adherent and transferred with medium to a new culture dish. As shown in Figure 1, in vitro NSCs were small and round with little morphological evidence of differentiation after 1 day of incubation (Fig. 1A). Then the cells were expanded as floating aggregates called neurospheres during subsequent culture. After 5 days of incubation, the neurospheres increased both in size and amount in serum-free medium containing 20 ng/ml bFGF, which is a key factor of promoting NSC proliferation and maintaining the undifferentiated status of NSCs (Fig. 1B). In order to identify the purity of NSCs, we detected the expression of nestin, an acknowledged protein marker for NSCs. The results showed that regardless of the form (single cells or neurospheres) NSCs showed positive immunoreactivity to nestin (Fig. 1C, D).

Primary culture and characterization of NSCs. (A) Phase-contrast image of NSCs cultured 1 day in serum-free medium. (B) Phase-contrast image of NSCs cultured 5 days in serum-free medium. (C, D) Immunostaining of NSCs with nestin antibody and DAPI. Scale bars: (A, B) 100 μm, (C, D) 20 μm.
Purification and Characterization of OECs
Observation by microscope showed that OECs were initially stubby and became spindly later. Because the meninges and blood vessels could not be removed completely, there was a certain amount of flattened fibroblasts with poor refractivity (Fig. 2A). After purification, the majority of mature OECs were bipolar or multipolar Schwann cell like with a spindle morphology, and astrocyte-like OECs with a fried egg morphology were few (Fig. 2B). Most OECs were appulsive with good refraction. Immunocytochemical staining of OECs showed positive immunoreactivity to S100β as green stain (Fig. 2C), while fibroblasts showed negative immunoreactivity to S100β. The number of S100β-positive cells of 10 random images was counted by using the Image-Pro Plus software. The purity of OECs increased to 92.3 ± 6.8% compared with that of unpurified OECs (Fig. 2D).

Primary culture and characterization of OECs. (A) Phase-contrast image of OECs cultured 5 days before purification; arrows indicate OECs. (B) Phase-contrast image of OECs cultured 5 days after purification. (C) Immunostaining of OECs with S100β antibody and DAPI. (D) The purity of OECs before and after purification (*p < 0.05). Scale bars: 100 μm.
Differentiation of NSCs In Vitro
The multipotentiality of NSCs is demonstrated by their capacity to differentiate into neurons, astrocytes, and oligodendrocytes. When cultured in serum-free medium with bFGF, NSCs kept the state of proliferating and undifferentiating for a long time, as shown above. What was the destiny of NSCs cultured in medium with FBS? The floating neurospheres were isolated by centrifugation, after being mechanically dissociated to single-cell suspension, and then the cultures were plated in DMEM/F12 medium with 10% FBS. After 1 day of culture, most single NSCs attached on the dish. Observation under light microscope revealed that single cells adhering onto the culture dish showed changed cell morphology; they were no longer round, but exhibited short processes (data not shown).
After 5 days of incubation, the majority of cells (>90%) displayed positive immunoreactivity to astrocytic marker GFAP (Fig. 3B), but there was almost no expression of early neuronal marker β-tubulin III (Fig. 3C). Simultaneously we observed fibrous astrocytes and protoplasmic astrocytes (Fig. 3B). These findings demonstrated that NSCs were inclined to differentiate into astrocytes rather than neurons or oligodendrocytes when cultured alone in the medium with FBS.

In vitro differentiation of NSCs cultured alone and cocultured with OECs 5 days in DMEM/F12 medium with 10% FBS. (A, E) Image of DAPI immunostaining. (B, F) Image of GFAP immunostaining. (C, G) Image of β-tubulin III immunostaining. (D, H) Overlay of images. Scale bars: 20 μm. (I) The ratio of NSCs differentiated into neurons (*p < 0.05).
Previous research has indicated that the behavior of NSCs is regulated by the intrinsic programs and extrinsic signals from the culture medium (5,10,19,29,39). With the presence of NGF or BDNF, more NSCs differentiate into neurons (4,5,8). What is the in vitro behavior of NSCs in response to the NTFs secreted by OECs? For coculture, NSCs and OECs were mixed in equal number to make a homogenous suspension in DMEM/F12 medium with 10% FBS. As shown in Figure 3, after 5 days of coculture, while observing the expression of GFAP (Fig. 3F), we also detected a certain quantity of β-tubulin III expression (Fig. 3G). The ratio of NSCs differentiated into neurons increased to 24.6 ± 3.8% (Fig. 3I) compared with NSCs cultured alone according to an analysis of 10 random images. The presence of OECs resulted in a significant increase in the ratio of NSCs differentiated into neurons.
Recovery of Locomotor Function
In 1995, Basso, Beattie, and Bresnahan established a widely used statistical method for evaluating the behavioral consequences of SCI to the rats (2). The rats were rated on a scale of 0 to 21, 0 being no function and 21 being normal. The method involved the following parameters: joint movements, the ability for weight support, limb coordination, foot placement, and gait stability. Figure 4 shows the BBB scale score of the four experimental groups. For all four groups, 6 weeks after the operation, the average BBB scale score became stable. For the NSCs + OECs group, the BBB scale score reached 15.6 on average and the rats displayed consistent plantar stepping, consistent forelimb–hindlimb coordination, and occasional toe clearance during forward limb advancement. Twelve weeks later, the average BBB scale score of the cotransplantation group reached 17 and the animals showed frequent toe clearance with the predominant paw position parallel at initial contact and lift off. In contrast, in the other three groups, the average BBB scale scores were 9.1 (control group), 13.2 (NSCs group), and 14.6 (OECs group), significantly lower than that of the NSCs + OECs group (p < 0.01). The NSCs group and the OECs group also showed significant differences in BBB scale scores compared with the control group (p < 0.05). Although the BBB score of the OECs group was a little higher than that of the NSCs group, there was no significant statistical difference between them (p > 0.05).
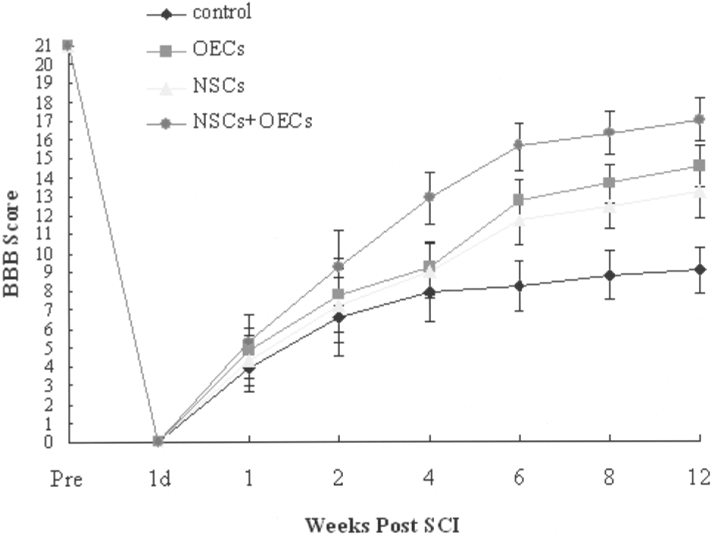
BBB open-field walking scores of four experimental groups. Twelve weeks after SCI, the average BBB scale score of the cotransplantation group reached 17 and was significantly higher than that of the other three groups.
Axon Regeneration
After perfusion and obtaining the samples as described in the Materials and Methods section, in gross observation (Fig. 5A) the region that was about 2 mm from the epicenter of the injury site in the rostral or in the caudal end was a transition zone between the denatured and normal spinal cord (Fig. 5B). Analysis of this region might illuminate our results better. Immunohistochemistry of the caudal part (2 mm from the epicenter) of the injured spinal cord stained with NF-200 antibody (green stain) showed that few NF-200-positive nerve fibers were seen in the control group (Fig. 6A). There were also a few NF-200-positive nerve fibers observed in the OECs group (Fig. 6B) and in the NSCs group (Fig. 6C), but a lot of regenerated NF-200-positive nerve fibers passed through the lesion area to the caudal end of the injured spinal cord in the NSCs + OECs group (Fig. 6D). After SCI, hemorrhage, degeneration, and necrosis occurred, followed with cystic cavity formation. As well, many small cavities were observed in the transition zone (Fig. 6). In our experiment, the mean cystic cavity size in the injured area showed a significant reduction in the cotransplantation group compared with the other three groups (data not shown). Figure 6E shows the density of regenerated nerve fibers in the four experimental groups. The percentage of area of NF-200-positive nerve fibers was calculated by analyzing five random images with Image-Pro Plus software. As a result, the percentage of the NSCs + OECs group (11.2 ± 1.03%) was significantly higher than that of the other three groups (p < 0.05).

(A) Dorsal view of spinal cord in the four experimental groups 12 weeks after the operation. Scale bar: 5 mm. (B) A schematic illustration of spinal cord: the asterisk indicates the epicenter of the injured spinal cord where the right 3/4 was cross cut; the triangle indicates the caudal part 2 mm from the epicenter where immunohistochemistry staining with NF-200 and S100 was applied, analyzed within the four groups in Figure 6.
Semithin transections of the lesion region of the cotransplantation group stained with toluidine blue showed there were few regenerated axons in the control group (Fig. 7A). In the OECs group (Fig. 7B) or in the NSCs group (Fig. 7C), there were only a small number of regenerated axons. In contrast, there were a number of myelinated axons in the scar tissue (Fig. 7D). Five random visual fields of the oil-immersed lens were selected for each specimen. The myelinated axon density (number of axons/mm2) was calculated. Statistical analysis (Fig. 7E) demonstrated that the density of myelinated axons of the NSCs + OECs group (14,359 ± 903) was significantly higher than that of control group (4,259 ± 1,112), OECs group (9,217 ± 868), and NSCs group (6,852 ± 751). The quantification result of myelinated axon density was consistent with the result showed in Figure 6E. Longitudinal tissue sections of the lesion area in the injured spinal cord of the NSCs + OECs group stained with NF-200 antibody showed that regenerated nerve fibers, which were NF-200 positive, existed throughout the lesion region (Fig. 8).

Immunohistochemistry of the caudal part (2 mm from the epicenter) of the injured spinal cord stained with NF-200 antibody (green) and S100 antibody (red). (A) Control group. (B) OECs group. (C) NSCs group. (D) NSCs + OECs group. Scale bars: 30 μm. (E) The density of NF-200-positive nerve fibers in the four groups (*p < 0.05).

Semithin transections stained with toluidine blue showed regenerated nerve fibers in the middle part of the injured spinal cord in the four groups. Scale bars: 20 μm. (A) Ccontrol group. (B) OECs group. (C) NSCs group. (D) NSCs + OECs group. (E) The density of myelinated axons in the four groups (*p < 0.05).
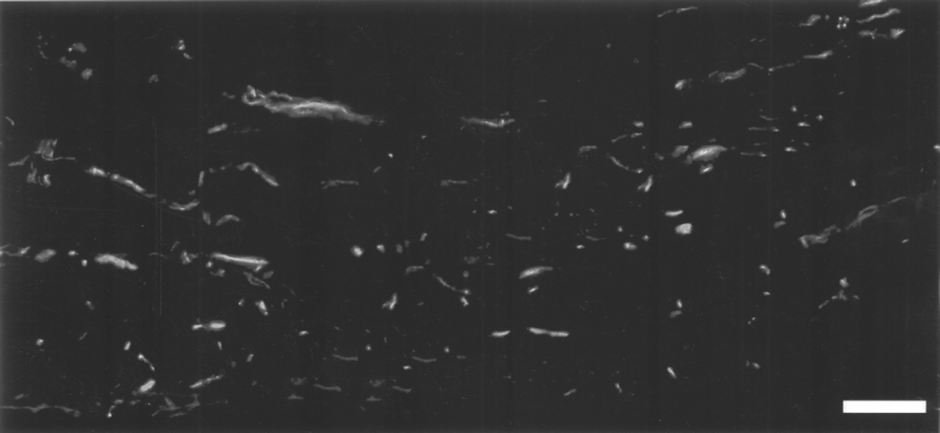
Regenerated nerve fibers in the middle part of the injured spinal cord in the cotransplantation group. A longitudinal section stained with NF-200 antibody. Scale bar: 30 μm.
Discussion
The aim of our study was to determine the effect of cotransplantation of NSCs + OECs after SCI by taking advantage of the complementary biological properties of the two types of cells. Our results indicated that cotransplantation of NSCs + OECs into the injured spinal cord 1 week after SCI significantly improved the recovery of locomotor function, reduced spinal cord damage, and resulted in many regenerated nerve fibers traversing the lesion area.
The capacity of axon regeneration of adult mammalian CNS is limited, and how to form supplemental cells to make up for those lost in the injury and how to activate the function of endogenous cells is a key problem. SCI leads to locomotor dysfunction below the level of injury due to interruption of the corticospinal tract and accumulative death of neurons, astrocytes, and oligodendrocytes in and around the lesion site (3). After the initial injury and following necrosis, a fluid-filled cavity develops, resulting in the loss of gray and white matter. The cavity can expand to additional spinal cord segments around the injury site, causing additional cell death and increased loss of function (47). The multipotential differentiation capability of NSCs makes them a possible cell type in the treatment of SCI. However, the majority of surviving transplanted NSCs differentiate into astrocytes in the lesioned spinal cord (6,14). Identification of effects that control the behavior of NSCs may allow for eventual manipulation of NSCs in the CNS therapies to replace cells (38). NSCs exhibited different cell behaviors in response to extrinsic signals (5,10,19,29,39). The promotion of NSCs differentiation into more neurons becomes one of the critical factors in the utilization of NSCs in neural restoration.
In our in vitro study, NSCs could maintain an undifferentiated status for a long time when cultured in the serum-free medium with bFGF (Fig. 1). But when NSCs were cultured alone in the medium with 10% FBS, through the agency of FBS, the undifferentiated condition was broken quickly. After 5 days of culture, immunocytochemistry of NSCs double stained with GFAP (green stain) and β-tubulin III (red stain) antibodies showed that over 90% of NSCs differentiated into astrocytes and almost no NSCs differentiated into neurons (Fig. 3). This result was consistent with the previous reports (4,5,8,43). In vivo, the activation of glial cells forms a glial scar that inhibits the regeneration of axons and repair of SCI. The astrocytes are densely packed and secrete inhibitory molecules (CSPGs) into the ECM, making it difficult for injured neural fibers to regenerate across the injury site (46). This means that the effect of repairing SCI by transplantation of NSCs alone is finite.
The presence of OECs has changed such circumstances. OECs are a unique type of glial cells that are distributed in olfactory bulbs or olfactory mucosa and can still grow in the maturation phase. Much research has confirmed that transplantation of OECs can help the axon pass through the glial scar and is effective for repairing nerve fiber injuries in the spinal cord (11,20,24–26,36,37). But primary culture of OECs is often contaminated with fibroblast cells because of the incomplete removal of meninges and blood vessels. In our study, if the purification procedure was not carried out, there were a lot of fibroblast cells in the culture dish (Fig. 2A). With the continuation of the culture, the fibroblasts showed a much faster proliferation than OECs, with the proportion of fibroblast cells increasing quickly, and eventually led to a useless result in the OECs culture. The method based on differential rates of attachment of different cells is easy to carry out and economic, so it is often used in purification of OECs (32). In this study, we obtained a highly purified culture of OECs (Fig. 2B) according to the above method to lay the foundation of later transplantation. The isolation of OECs was carried out in adult rat olfactory bulbs in our experiment, while it requires an intracranial surgery when getting to the human olfactory bulb in clinical use. The clinical potential of OECs may seem difficult due to the inaccessibility of the human olfactory bulb, but it has been shown that OECs can be obtained from human olfactory mucosa (7,11,17). So the nasal cavity is an alternative accessible source of OECs and intracranial surgery is not necessary.
OECs secrete NTFs such as NGF, BDNF, and GDNF, which not only promote axonal regeneration but also provide neurotrophic support to injured host cells (11). These NTFs have an evident effect on NSCs differentiation (4,5,8,10,19,29,39). These NTFs may be related to enhancing the survival of NSCs differentiated into neurons rather than directly promoting undifferentiated NSC differentiation into neurons. In this study, we cocultured NSCs with OECs in the medium with 10% FBS in vitro; after 5 days of culture, NSCs differentiated into more neurons compared with NSCs cultured alone in the medium with 10% FBS (Fig. 3). The effect of OECs on differentiation of NSCs was obvious. The presence of OECs made NSCs differentiate into a more useful condition for repair of SCI in the medium with serum, which is also contained in vivo. It seems that cotransplantation of NSCs and OECs is preferable in the treatment of SCI.
In order to confirm our presumption, a thoracic SCI rat model was established. In the four groups of rats, the spinal cord was 3/4 transected on the right side at the T9 level. The subsequent secondary injury triggered by the initial acute mechanical injury is a degenerative process that includes edema, ischemia, inflammation, ionic imbalance, excitotoxicity, and apoptosis (21). Previous studies and clinical research (16,21) have indicated that the subsequent secondary injury mentioned above gradually disappeared about 1 week after SCI. In order to minimize the adverse impact of secondary injury on transplanted cells, the four groups of rats were injected with DMEM/F12 medium, NSCs, OECs, and a mixture of the two types of cells, respectively, 1 week post-SCI. The 3/4 transection of spinal cord caused the loss of sensory, motor, and autonomic function; as a result, the BBB scale score soon dropped to the bottom line after 1 day in all four experimental groups (Fig. 4). After 1 week, there were no significant differences in BBB scale score between the four groups (p > 0.05). One week after the cell transplantation (2 weeks post-SCI), the average BBB score of the cotransplantation group was slightly higher than that of the other three groups, and 4 weeks after operation the NSCs + OECs group had an obvious increase in BBB score compared with the other three groups. At the end of the BBB open field test (12 weeks post-SCI), the average BBB scale score of the cotransplantation group was significantly higher than that of the other three groups (p < 0.01). From 6 to 12 weeks after operation, the degree of increase of the BBB scale score in the four groups was not large, and the result indicated that the initial 4–5 weeks after cell transplantation was a critical recovery period of locomotion function. Previous studies have showed that both NSC transplantation alone (6,14,19,27,45) and OEC transplantation alone (20,24–26,36) have a positive effect on repair of SCI. Compared to these studies, our results of NSCs or OECs transplantation alone had the similar effect to a certain extent.
The immunohistochemistry of the caudal part of the injured spinal cord stained with NF-200 antibody showed that many regenerated nerve fibers existed in the cotransplantation group (Fig. 6D) and the number was significantly higher than that of the other three groups (Fig. 6A, B, C), especially than that of the control group (Fig. 6A). Also, we observed cavities (Fig. 6), while the formation of the cystic cavity generated a physical barrier to spontaneous regeneration. Cell therapy can help overcome this barrier through a variety of methods. Such strategies have demonstrated that reduction of the cavity size correlates with an increase in functional recovery. The longitudinal and transverse tissue sections of the middle part of the injured spinal cord in the NSCs + OECs group also showed that regenerated myelinated nerve fibers traversed the lesion area (Figs. 7 and 8). These fibers probably played a role in establishing functional connection with the host spinal cord, leading to a partial recovery of sensory and locomotor functions (18), which also confirmed the result of the BBB behavioral test. But it is still difficult to prove whether the recovery was the result of transplanted cells or the activation of endogenous cells. The precise molecular mechanism of NSCs and OECs in vivo is still not fully understood, and 5-bromodeoxyuridine (BrdU) labeling may be a helpful method in understanding the in vivo behavior of transplanted cells. But owing to its bromine side group, BrdU is toxic to experimental cells and animals and negative effects of BrdU on the survival of labeled cells have been reported (22,23). As we first cotransplanted NSCs and OECs into the injured spinal cord in the treatment of SCI, further experiments are necessary to analyze the more precise mechanism.
Conclusion
In summary, OECs could promote NSCs differentiation into neurons. Cotransplantation of NSCs and OECs might have a synergistic effect on promoting neural regeneration and improving the recovery of locomotion function after SCI. Cotransplantation was better than a single graft either by NSCs or OECs.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (grant No. 30670528), the National Basic Research Program (also called 973 Program) of China (grant No. 2005CB623905), the Tsinghua Yue-Yuen Medical Science Fund, and the Wu-Shun-De Foundation of Tsinghua University.
