Abstract
In islet transplantation, in addition to immunologic and ischemic factors, the diabetic/hyperglycemic state of the recipient has been proposed, although not yet validated, as a possible cause of islet toxicity, contributing to islet loss during the engraftment period. Using a miniature swine model of islet transplantation, we have now assessed the effect of a persistent state of hyperglycemia on islet engraftment and subsequent function. An islet–kidney (IK) model previously described by our laboratory was utilized. Three experimental donor animals underwent total pancreatectomy and autologous islet transplantation underneath the renal capsule to prepare an IK at a load of ≤1,000 islet equivalents (IE)/kg donor weight, leading to a chronic diabetic state during the engraftment period (fasting blood glucose >250 mg/dL). Three control donor animals underwent partial pancreatectomy (sufficient to maintain normoglycemia during islet engraftment period) and IK preparation. As in vivo functional readout for islet engraftment, the IKs were transplanted across an immunologic minor or class I mismatch barrier into diabetic, nephrectomized recipients at an islet load of ∼4,500 IE/kg recipient weight. A 12-d course of cyclosporine was administered for tolerance induction. All experimental donors became diabetic and showed signs of end organ injury, while control donors maintained normoglycemia. All recipients of IK from both experimental and control donors achieved glycemic control over long-term follow-up, with reversal of diabetic nephropathy and with similar glucose tolerance tests. In this preclinical, large animal model, neither islet engraftment nor subsequent long-term islet function after transplantation appear to be affected by the diabetic state.
Introduction
The diabetic condition renders a chronic state of hyperglycemia with relative or absolute insulin deficiency that leads to severe systemic and organ-specific effects. 1 –9 Allogeneic islet transplantation may be an alternative treatment modality in select type 1 diabetic patients. 10 –17 In islet transplant recipients, it has been proposed that the hyperglycemic state in the diabetic recipient may be an important factor contributing to islet toxicity during the period of islet engraftment. 18 This factor may be especially relevant in the peritransplant setting, as the nonvascularized islets may be in double jeopardy, with oxidative injury resulting from both ischemia–reperfusion and hyperglycemic conditions. In addition, hyperglycemia may indirectly affect islet engraftment by causing systemic metabolic derangements. In view of these potentially deleterious effects, islet transplant centers typically maintain their patients on exogenous insulin therapy in the peritransplant period to mitigate damage from hyperglycemia. 12,13
We sought to test whether a persistent state of hyperglycemia can independently affect islet engraftment or cause long-term islet dysfunction in a preclinical, large animal model of islet–kidney (IK) transplantation in miniature swine. Although several studies performed in vitro and in mouse models have suggested glucose-mediated islet toxicity and apoptosis, 3,19 –21 to our knowledge, no one has evaluated this in a large animal model in the in vivo setting. Our study utilized a model of vascularized islet transplantation in a miniature swine model that was established previously in our laboratory. 22 –24
Materials and Methods
Animals
All animals were housed at the Transplantation Biology Research Center, Massachusetts General Hospital. Experiments were performed in compliance with institutional guidelines and were approved by the subcommittee on research animal care. Minor and class I antigen mismatched donor and recipient pairs were selected from our herd of partially inbred miniature swine. 25,26 Typically, the IK transplant donors were >16 mo of age and the recipient animals were 2 to 3 mo of age. All IK transplants, except for one, were performed across an immunologic minor mismatched barrier (Table 1). One animal in the experimental arm donated an IK across a class I mismatched barrier due to the unavailability of a minor mismatch recipient.
Islet–Kidney (IK) Donor–Recipient Pairs.

Note. IK donor was >16 mo old, and the IK recipient was 2 to 4 mo old. All the transplants were performed across immunologic minor mismatch barrier, except for a single immunologic class I mismatch transplant (*).
IK Transplantation Model
An overview of the IK transplantation model is shown in Fig. 1. In the experimental arm, after total pancreatectomy, autologous IK were prepared at <1,000 IE/kg of donor weight. The control arm donor animals underwent a partial pancreatectomy and autologous IK preparation. Islets were allowed to engraft over a 4- to 8-wk period. IK were then transplanted into diabetic, nephrectomized, recipient animals at ∼4,500 IE/kg of recipient weight, along with the administration of intravenous cyclosporine for 12 d for tolerance induction.

Experimental design using the porcine islet–kidney (IK) model is shown. Control arm: donor animal underwent partial pancreatectomy + autologous IK preparation, with an engraftment period of 4 to 8 wk. Experimental arm: donor animal underwent total pancreatectomy + autologous IK preparation at a decreased islet load (≤1,000 IE/kg), with an islet engraftment period of 4 to 8 wk. As an in vivo functional readout, the IK was then transplanted across an immunologic minor or class I mismatch barrier into a pancreatectomized, nephrectomized recipient at an islet load of ∼4,500 IE/kg. IK recipient received a 12-d course of cyclosporine for tolerance induction.
Partial Pancreatectomy
Distal pancreatectomy (60%) with splenectomy was performed in the control donor animal through a midline laparotomy as previously described. 22,23 The distal pancreas was transected at the junction of the splenic vein—superior mesenteric vein confluence, distended on the back table, and transported for islet isolation.
Total Pancreatectomy in the Experimental Donor
The distal pancreas and spleen were dissected as in the control donor animal. Next, the duodenal and proximal splenic lobes of the pancreas were dissected free. The pancreatic duct was doubly ligated. The whole organ was distended and transported to Joslin Diabetes Center, (Boston, MA, USA) for islet isolation. 27
Islet Transplantation (IK Preparation)
Isolated islets were cultured overnight at 25°C, 5% CO2, and prepared the next day for transplantation. The autologous islet suspension was injected underneath the renal capsule to prepare an IK in the donor as previously described.
Donor Animal Care
The experimental arm donor animals received a high-protein diet supplemented with pancreatic enzymes (pancreazyme) daily, while the control donors received normal supplementation. Blood sampling was done to check complete blood count, fasting blood glucose (FBG), blood urea nitrogen (BUN) and creatinine, amylase, lipase, liver function tests, and lipid profiles. All animals were observed at least twice daily.
Total Pancreatectomy in the IK Recipient
Through a left paramedian incision, the pancreas was removed. The pancreatic duct was doubly ligated. The animal also underwent bilateral external jugular catheter placement for drug and fluid administration and blood chemistry sampling.
IK Transplantation
In the donor animal, the composite left IK graft was removed with the renal vessels and the ureter through a left paramedian incision. The organ was perfused with cold Euro-Collins solution on the back table. In the recipient, through a right paramedian incision, a bilateral nephrectomy was performed. The IK was placed in the right abdomen, and the following anastomoses were performed: renal vein to inferior vena cava, renal artery to aorta, and an ureterocystostomy.
Recipient Animal Care
Blood was sampled to check complete blood count, BUN, creatinine, and electrolytes, as warranted. Morning FBG was checked daily. The animals received a special diet containing pancreatic enzymes for exocrine insufficiency. Intravenous cyclosporine was administered for 12 d for tolerance induction (cyclosporine blood trough 300–450 ng/mL for minor mismatch and 500–700 ng/mL for class I mismatch). 28 Animals were observed at least twice daily. Insulin therapy was instituted for all FBG > 250 mg/dL for at least 2 consecutive days.
Intravenous Glucose Tolerance Test (IVGTT)
After an overnight fast, the animal was sedated with an intramuscular ketamine injection at 5 mg/kg. After checking the FBG, dextrose was infused intravenously at 0.5 g/kg. Blood glucose was checked at the following time points (minutes) after infusion or stopped earlier if blood glucose returned to baseline: 2, 5, 10, 15, 30, 45, 60, 90, 120, 150, and 180.
IK Biopsy
The animal was placed in a lateral decubitus position, and a right flank incision was made to access the IK. With a #15 blade, a 1 × 1 cm biopsy of the allograft was obtained and hemostasis achieved with electrocautery.
Graftectomy
The IK in the recipient was accessed through a flank incision and freed from attachments. The composite graft was removed en bloc upon ligation of the renal vessels and the alloureter.
Histology
Tangential sections were taken to include islet and renal tissue. Formaldehyde-processed specimens were stained with hematoxylin and eosin (H&E) and insulin-specific antibodies. Insulin staining was performed on 4.0-µm sections. Sections were incubated at room temperature in a humidified chamber for 20 min with 10% normal goat serum in phosphate buffered saline (PBS; pH 7.4) and then for 60 min with guinea pig antiporcine insulin antibody (Dako, Carpinteria, CA, USA) diluted 1:10 in PBS. They were next incubated with a 1:200 dilution of biotinylated goat antiguinea pig secondary antibody (Vector Laboratories, Burlingame, CA, USA) for 60 min. The tissue-bound primary antibodies were detected by an avidin–biotin–peroxidase complex (Dako) that was visualized by staining with 0.02% hydrogen peroxide containing 0.3 mg/mL 3,3′-diaminobenzidine (DAB) in 0.05 mol/L Tris buffer. Sections were counterstained with Gill’s single-strength hematoxylin.
Results
IK Donors
Glycemic control
The 3 donor animals on the control arm maintained normoglycemia during the islet engraftment period (Fig. 2A).
The remnant pancreas plus the transplanted islets were apparently sufficient to maintain normal glucose homeostasis. In contrast, the 3 experimental donors were severely diabetic (FBG > 400 mg/dL) after the total pancreatectomy (Fig. 2A). On postoperative day (POD) 1 after autologous islet transplantation, in all 3 animals the hyperglycemia was corrected to FBG < 120 mg/dL. The following day the FBG started to increase, and by POD 3 it was >250 mg/dL, and it persistently remained at this level. Thus, throughout the islet engraftment period, the experimental donors were in a persistent diabetic state.

The islet–kidney (IK) donors on the control and experimental arms showed very different glucose regulation. (A) Fasting blood glucose (FBG) curves in the control animals showed normoglycemia. Experimental animals showed a chronic hyperglycemic state with FBG consistently around 250 mg/dL. (B) Control animals showed normal glucose tolerance on intravenous glucose tolerance test. Experimental animals showed severe glucose intolerance. Time points 2- and 5-min blood glucose values that read as “high” on the glucometer are represented as 600 mg/dL on the graphs. (C) Control donors showed normal renal function during the islet engraftment period. In contrast, the experimental donors showed mild renal insufficiency during the follow-up. Serum creatinine values for control (D1–D3) versus experimental (D4–D6) arm donor animals are shown. (D) IK donors on the experimental arm showed histological signs of diabetic nephropathy. Renal biopsies were performed on the day of autologous islet transplantation and at the time of IK donation. Representative histology from animal D5 is shown.
IVGTTs were performed in the donor animals in both groups. The 3 control animals showed near-normal responses to a glucose challenge, with blood glucose levels returning to baseline values within 45 min postinfusion (Fig. 2B). In contrast, the experimental donors showed severe glucose intolerance on IVGTT, requiring about 3 h to clear the intravenous glucose load (Fig. 2B). Thus, the autologous islets in the experimental arm donors were in a decompensated state under severe hyperglycemic stress during the engraftment period.
Clinical picture
The control donors showed a clinical behavior very similar to naive animals. They maintained normal activity, appetite, and weight. They showed no signs of infection and maintained normal hematopoietic profiles (data not shown). The clinical picture of the experimental donor animals was very similar to poorly controlled diabetic patients. All 3 animals developed central venous catheter-related infections, requiring broad-spectrum antibiotic coverage and early removal of central venous lines. The animals were also more prone to developing pressure ulcers and local infections of the distal extremities. Wound healing in these animals (including the incision sites) was significantly delayed, with an increased risk of stitch abscess formation and wound dehiscence. The animals showed polyuria, polydipsia, and a 10% weight loss, despite aggressive supplementation and increased care.
Although the experimental donors showed clinical effects of poorly controlled diabetes, they maintained the hyperglycemic state without any ketoacidosis or hyperosmolar hyperglycemic nonketosis. They did not show overt pancreatic exocrine insufficiency (no steatorrhea or lipid positive stools). They also developed elevations in liver enzyme values with normal total bilirubin values and mild hypertriglyceridemia that gradually resolved by the time of IK donation (data not shown). The animals developed renal insufficiency with elevations in BUN and creatinine values that persisted until the time of IK donation but did not significantly worsen over the follow-up (Fig. 2C). All 3 animals also had persistent glucosuria without ketonuria or proteinuria.
IK donor histology
For histological confirmation of diabetic changes in the experimental animals, biopsies of the liver and the kidney were obtained. On histology, all 3 animals showed progressive diabetic nephropathy. There were severe vacuolar changes in the renal tubules, consistent with diabetic tubular injury. No histological evidence of glomerular injury was noted (Fig. 2D).
IK Recipients
Fasting glycemic control
All 3 recipient animals in the control arm showed a high FBG after total pancreatectomy that was immediately corrected after IK transplantation (Fig. 3A–C). Two to three days after IK transplantation, there was transient decompensation in glucose regulation likely due to the diabetogenic stress of cyclosporine. The FBG normalized spontaneously after cessation of the cyclosporine and overall, the IK maintained normoglycemia in the animals long-term. To confirm that glucose regulation was strictly due to the IK, graftectomy was performed. After graftectomy, the FBG dramatically increased to >600 mg/dL (Fig. 3B). One animal died on POD 50 from intestinal volvulus, thought to be unrelated to the function of the IK (Fig. 3C).

Islet–kidney (IK) recipients on the experimental arm showed better fasting glycemic control than the control arm animals. Blue lines represent fasting blood glucose (FBG) readings, and orange bars represent exogenous insulin requirements. (A–C) Control animals showed immediate correction of the hyperglycemic state upon IK transplantation. Overall, the animals showed normal FBG during the follow-up. But during periods of diabetogenic stress, such as immunosuppression with cyclosporine and catheter-related infection, the animals showed transient hyperglycemia. A: R1, B: R2, C: R3. (D–F) The experimental animals also showed immediate correction of the diabetic state upon IK transplantation. In addition, they maintained normoglycemia during the cyclosporine period. D: R4, E: R5, F: R6. TPx, total pancreatectomy; IK Tx, islet–kidney transplant; Gx, graftectomy; IV, death secondary to intestinal volvulus.
In the experimental arm, similar to the controls, all 3 recipients showed high FBG after total pancreatectomy that was immediately corrected after IK transplantation (Fig. 3D–F). Unlike the control recipients, the fasting glucose remained in the normal range in these animals during the cyclosporine course. In fact, 1 animal received a higher dose of cyclosporine compared to the rest of the IK recipients (due to a class I mismatch), but it still showed no glycemic decompensation (Fig. 3F). This animal died acutely on POD 11 during the period of cyclosporine infusion, presumably from drug toxicity.
IVGTTs
The response to glucose challenge was similar in the 3 control animals over follow-up up to day 95 (Fig. 4A–C). IVGTTs performed after day 120 showed worsening of the response compared to earlier time points in both control IK recipients. IVGTT response was slightly delayed compared to control donor animals, with blood glucose levels returning to baseline values between 60 and 90 min postinfusion. IVGTTs in the experimental arm animals were similar to the controls (Fig. 4D, E) if not slightly improved. In both animals, glucose levels returned to baseline within 60 min. Especially at day 126 IVGTT, the experimental arm IK recipient showed an improved response compared to the control IK recipient at a similar time point.
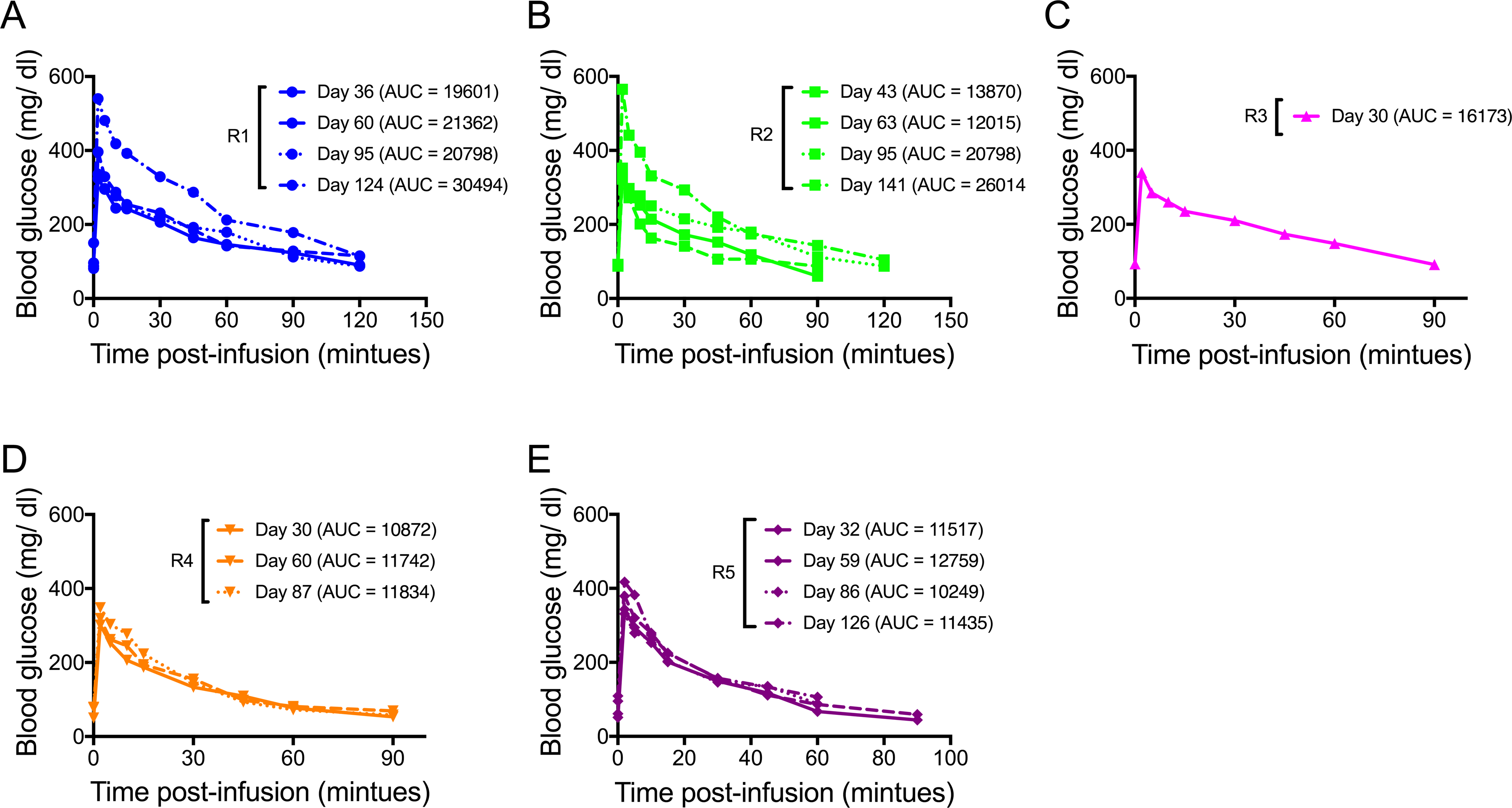
Islet–kidney (IK) recipients on the experimental arm showed better glucose tolerance on intravenous glucose tolerance test (IVGTT) than the control arm animals. (A–C) Control animals showed mild glucose intolerance on IVGTT, with baseline blood glucose values returning within 90 min postinfusion. A: R1, B: R2, C: R3. (D, E) Experimental IK recipients showed slightly better IVGTT response than the controls. The IVGTT response did not improve or worsen during the follow-up. D: R4, E: R5.
Renal function
All 3 recipients in the control arm showed normal renal function throughout the follow-up, suggesting that there was no clinical rejection of the IK (Fig. 5A). In contrast, the experimental arm recipients showed some renal dysfunction, especially in the early posttransplant period (Fig. 5B). The renal function did eventually improve and stabilized over time, but at a level higher than the baseline. Since the transplanted IKs had histological evidence of diabetic nephropathy, serial biopsies of these kidneys were obtained to monitor for changes upon correction of the diabetic state. IK transplantation showed reversal of diabetic nephropathy by POD 35 as evidenced in biopsy (Fig. 5C).

Renal function in the islet–kidney (IK) recipients. (A) Control animals showed normal blood urea nitrogen (BUN) and creatinine values before and after IK transplantation, suggesting that the IK maintained renal function very similar to the native kidneys. (B) Experimental animals showed renal dysfunction upon IK transplantation, indicating that the renal portion of the IK was not functioning as well as the native kidneys. Initially, the BUN and creatinine values worsened during the cyclosporine period, possibly secondary to drug-related toxicity to an organ that was already undergoing diabetic injury. The renal function improved after the cyclosporine course but settled at levels above the baseline. (C) Representative histology of the renal portion of the IK at the time of transplantation into animal R4 (animal 17238) showed severe vascular changes in the renal tubules consistent with diabetic nephropathy. Renal biopsy from postop day 35 after IK transplantation in recipient R4 showed reversal of diabetic nephropathy.
IK histology
Histology in the control animals showed normal renal parenchyma (data not shown). The IKs showed viable islet clusters underneath the renal capsule without leukocytic infiltration. Representative histology of the islets from an experimental arm IK recipient is shown (Fig. 6).

Islet–kidney (IK) histology for IK recipient R6 is shown. Islet graft section was stained with insulin (brown) and then counterstained with hematoxylin. Viable islet clusters containing insulin were noted without leukocytic infiltration.
Discussion
Our experimental model to study transplantation parameters in the diabetic state was achieved through autologous islet transplantation at an intentionally lower islet load (<1,000 IE/kg). The animals were persistently hyperglycemic during the engraftment period without suffering from any major acute diabetic complications, such as diabetic ketoacidosis or hyperosmolar hyperglycemic nonketosis. At the organ-specific and systemic levels, the animals showed effects of the diabetic state. Therefore, we believe that we now have a reliable and reproducible large animal model of the chronic diabetic state that does not require any exogenous insulin therapy or excessive medical care. The use of this model in partially inbred miniature swine can allow us to thoroughly study important clinical conditions such as diabetic nephropathy, cardiomyopathy, neuropathy, retinopathy, or other metabolic dysfunctions in a preclinical large animal model across controlled genetic backgrounds and at a given hyperglycemic state. In fact, histological analysis of the IK donors and recipients in this study showed progression to diabetic nephropathy and its reversal. Thus, the model may also have utility in understanding the role of therapeutic interventions in reversing or slowing diabetic organ dysfunction at a particular hyperglycemic state. The fact that these manipulations can be done in large animals should enable routine biopsies and blood chemistries over a long-term follow-up and have greater clinical relevance.
Using the IK model, we studied whether a persistent diabetic state may adversely affect the islet engraftment process and the functional capacity of the transplanted islets. We hypothesized that given the evidence for glucose toxicity, and possibly the ensuing organ-specific and islet injury secondary to the diabetic state, we would see a functional or morphologic reduction in β cell mass manifested as poor glucose homeostasis in the experimental IK recipients. The results of this study refute this hypothesis. We conclude that hyperglycemia, with an exposure time between 30 and 60 d, does not negatively affect the islet implantation/revascularization process, nor does it cause long-term islet dysfunction.
The study suggests that transplanted islets can be quite resilient under hyperglycemic conditions (at least up to 55 d of hyperglycemia). Even though the transplantation site (kidney) may undergo severe changes due to hyperglycemia, the islets still maintained the ability to implant, revascularize, and function. In the experimental donors, upon autologous islet transplantation, there was initial correction of the diabetic state and then decompensation. Possibly the transplanted islets were able to maintain normoglycemia for about a 24-h period, but the stress for the given islet mass was too much, and the islets decompensated. Obviously, this decompensation did not affect the engraftment process any more than in the controls. Even though the animals were diabetic and showed severe glucose intolerance, the level of hyperglycemia did not worsen in these animals, suggesting that the transplanted islets were insufficient to correct the diabetic state, but successfully prevented it from worsening over the follow-up.
If anything, the IK recipients in the experimental arm seemed to maintain slightly better glycemic control than the control animals. This was further supported by the results of the IVGTTs, and the relatively lower exogenous insulin requirements seen in the IK recipients in the experimental arm (Fig. 3). Although this may suggest that an effective and largely functional β cell mass was transplanted in the experimental arm, the slightly improved glycemic control in these IK recipients may be due to decreased renal function, as this would lead to decreased insulin clearance by the transplanted kidney. In the future, it would be worth investigating this finding by utilizing this model but performing IK transplants into recipients at a lower islet load, such as 2,500 to 3,000 IE/kg, with preservation of 1 of the 2 native kidneys in order to control the potential differences in insulin clearance associated with altered renal function in the experimental arm IK recipients.
Lastly, we also note that the transplantation site of the renal capsule in our model may provide a very different environment for islet engraftment than the usual clinical setting of islet infusion into the liver via the portal venous system. The renal subcapsular space may provide a more immunoprotective and anti-inflammatory environment compared to the liver. Therefore, the negative effect of glucose toxicity may be mitigated. Future investigations in our laboratory will look further into the relationship between islet engraftment and function as well as the choice of islet transplantation site in a particular glycemic environment.
Footnotes
Acknowledgments
The author would like to thank Aseda Tena, Jessica Sayre, and Meghan Cochrane for their help with animal care.
Ethical Approval
This study was reviewed and approved by the Harvard Medical School Institutional animal care and use committee (IACUC).
Statement of Human and Animal Rights
All experiments were performed in compliance with the institutional guidelines.
Statement of Informed Consent
Statement of Informed Consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: First author’s work was supported by the following grants: American College of Surgeons Resident Research Scholarship, American Society of Transplant Surgeons—Roche Laboratories Scientist Scholarship, and Ruth L. Kirschstein National Research Service Award.
