Abstract
Previous rodent studies employing monotherapy or combined immunosuppressive regimens have demonstrated a variable degree of spinal xenograft survival in several spinal neurodegenerative models including spinal ischemia, trauma, or amyotrophic lateral sclerosis (ALS). Accordingly, the characterization of optimal immunosuppressive protocols for the specific neurodegenerative model is critical to ensure reliable assessment of potential long-term therapeutic effects associated with cell replacement. In the present study we characterized the survival of human spinal stem cells when grafted into the lumbar spinal cords of a rodent model of ALS, SOD1 (G93A) male and female rats (60–67 days old). Four different immunosuppressive protocols were studied: i) FK506 (q12h); ii) FK506 (qd) + mycophenolate (PO; q12h, up to 7 days postop); iii) FK506 (qd) + mycophenolate (IP; q12h, up to 7 days postop); and iv) FK506 (qd) + mycophenolate (IP; qd, up to 7 days postop). Three weeks after cell grafting the number of surviving human cells was then systematically assessed. The highest density of grafted cells was seen in animals treated with FK506 (qd) and mycophenolate (IP; qd; an average 915 ± 95 grafted cells per spinal cord section). The majority of hNUMA-positive cells colocalized with doublecortin (DCX) immunoreactivity. DCX-positive neurons showed extensive axodendritic sprouting toward surrounding host neurons. In addition, migrating grafted cells were identified up to 500 μm from the graft. In animals treated with FK506 (q12h), FK506 (qd) + mycophenolate (PO; q12h) or FK506 (qd) + mycophenolate (IP; q12h), 11.8 ± 3.4%, 61.2 ± 7.8%, and 99.4 ± 8.9% [expressed as percent of the FK506 (qd) and mycophenolate (IP; qd)] cell survival was seen, respectively. In contrast to animals treated with a combination of FK506 + mycophenolate, robust CD4/8 immunoreactivity was identified in the vicinity of the injection tract in animals treated with FK506 only. These data suggest that a combined, systemically delivered immunosuppression regimen including FK506 and mycophenolate can significantly improve survival of human spinal stem cells after intraspinal transplantation in SOD1 (G93A) rats.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a lethal neuromuscular disease characterized by progressive degeneration of upper and lower motor neurons and corresponding motor and respiratory dysfunction. Approximately 10% of patients have an inherited form of the disease, and of these 20% carry a mutation in the superoxide dismutase 1 (SOD1) gene. Creation of the G93A SOD1 transgenic mouse and rat has provided experimental rodent models of ALS that mimic human disease with comparable progressive motor dysfunction and respiratory failure (17,23).
Despite the discovery of several putative mechanisms underlying the onset and progression of ALS, there are no therapeutic strategies that successfully modify disease progression or outcome (33). Recently, cell-based therapies have emerged as potential treatments for several neurological disorders including ALS (18,28). For example, Xu et al. (34) reported that spinal grafting of human spinal stem cells (hSSCs) to the lumbar spinal cord of G93A SOD1 rats delayed the onset and progression of disease, while also extending the life span by more than 10 days. Glial-derived neurotrophic factor (GDNF)-producing human cortical neural precursors spinally grafted to G93A rats significantly improved motoneuron survival (30). However, cell graft survival appears to be inconsistent in the G93A SOD1 spinal cord and seems to greatly depend on the immunosuppression regimen. Yan et al. (35) reported that FK506 monotherapy after hSSCs grafting to the spinal cord of G93A SOD1 mice was inferior compared to combination therapy with rapamycin. Similarly, Suzuki et al. (30) and Klein et al. (19) both reported poor graft survival with cyclosporin A immunosuppression after spinal grafting of human neural progenitors in the G93A rat. Other studies, while reporting positive functional outcomes after spinal cell grafting to rodent ALS spinal cord, did not provide systematic data on cell survival (9,10,34).
In contrast to rodent G93A SOD1 models, spinal grafting of human neuronal or neural precursors in models of spinal trauma or spinal ischemia show much more consistent survival and maturation using similar immunosuppressive protocols (4,5,24,31). The nature of this differential response to identical immunosuppressive therapies is not clear but may reflect several possible mechanisms including distinct types of acute and chronic inflammatory responses. Experimental and clinical data demonstrate a complexity of pathological and resulting inflammatory changes in ALS rodent models and human patients [see review (25)]. Thus, it is possible that the increasingly hostile environment (e.g., inflammatory mileau resulting from cellular degeneration and vascular leakage) present in the spinal cords of rodent ALS models might attenuate graft survival and that a more aggressive immunosuppressive therapy than routinely required in other spinal cord injury models would be needed to achieve consistent graft survival. Accordingly, the current study was designed to optimize an immunosuppressive protocol for the spinal transplantation of hSSCs to presymptomatic G93A SOD1 rats.
Materials and Methods
Spinal Cord Implantation of hSSCs
All animal studies were approved by the UCSD institutional animal care and use committee. SOD1 (G93A) ALS male and female rats (UCSD colony, Dr. D. W. Cleveland; 60–67 days old) were anesthetized with isoflurane (1.5–2% maintenance; in room air), placed into a spinal unit apparatus (Stoelting, Wood Dale, IL, USA), and a partial Th12–L1 laminectomy was performed using a dental drill (exposing the dorsal surface of L2–L5 segments). Using a glass capillary (tip diameter 80–100 μm) connected to a microinjector (Kopf Instruments, Tujunga, CA, USa), rats were injected with 0.5 μl (10,000 cells per injection) of the hSSCs in hibernation buffer. The duration of each injection was 60 s followed by a 30-s pause before capillary withdrawal. The center of the injection was targeted into the base of the ventral horn (distance from the dorsal surface of the spinal cord at L3 level: 1.1–1.2 mm) (16). Five injections (~800 μm rostrocaudally apart) were made on each side of the lumbar spinal cord. After injections, the incision was cleaned with penicillin-streptomycin solution and sutured in two layers.
Derivation of the Spinal hSSCs
Human SSCs were prepared from the cervical–upper thoracic region of spinal cord tissue obtained from a single 8-week human fetus after an elective abortion. The fetal tissue was donated by the mother in a manner fully compliant with the guidelines of NIH and FDA and approved by an outside independent review board. The spinal cord tissue was removed of meninges and dorsal root ganglia and dissociated into a single cell suspension by mechanical trituration in serum-free, modified N2 media. The modified N2 media was composed of: 100 mg/L human plasma apo-transferrin, 25 mg/L recombinant human insulin, 1.56 g/L glucose, 20 nM progesterone, 100 μM putrescine, and 30 nM sodium selenite in DMEM/F12. For growth of the hSSCs, 10 ng/ml basic fibroblast growth factor (bFGF) as the sole mitogen was added to the modified N2 media (growth media). The initial culture was serially expanded as a monolayer culture in precoated flasks (T-175) or plates (15). Briefly, the precoated vessels were prepared by incubating them for 1 h at room temperature with 100 μg/ml poly-D-lysine in 10 mM HEPES buffer at 0.165 ml/cm2. The vessels were washed three times with water and allowed to completely dry aseptically in the hood. They were then further incubated with 100 μg/ml fibronectin/PBS for 5 min or alternatively 25 μg/ml fibronectin/PBS for 1 h. The fibronectin solution was aspirated and the vessels were used immediately without drying. Approximately 6.1 × 106 total cells were obtained upon the initial dissociation of the spinal cord tissue. All of the cells were plated onto one 150-mm plate in 20 ml of the growth media.
The growth medium was changed every other day and in the alternate days, 10 ng/ml of bFGF was directly added to the culture. The first passage was conducted 16 days after plating. At this point, the culture was composed mostly of postmitotic neurons and mitotic hSSCs. The mitotic cells were harvested by brief treatment with trypsin (0.05% in 0.53 mM EDTA). Trypsin was stopped by addition of soybean trypsin inhibitor to 0.05% final concentration. The cell suspension was triturated with a pipette to obtain a single cell suspension and centrifuged at 1400 rpm for 5 min. The cell pellet was resuspended in growth media and the cells were replated in new precoated plates at 1.2 × 106 cells in 20 ml of growth media per 150-mm plate. The cells were harvested at approximately 75% confluence, which occurred in 5–6 days. This process was repeated for 20 passages. At various passages, the cells were frozen in the growth medium plus 10% dimethyl sulfoxide (DMSO) at 5 × 106–10 × 106 cells/ml using a programmable freezer. The frozen cells were stored in liquid nitrogen. Upon thawing, the overall viability and recovery was typically 80–95%. The resulting cell line, which was produced by epigenetic means only, using bFGF as the sole mitogen, was named “566RSC.” A cell bank of passage 16 cells was prepared and used for this study.
Preparation of hSSCs for Implantation
One day prior to each surgery day, one cryopreserved vial of the previously prepared passage 16 cell bank was thawed, washed, concentrated in a hibernation buffer, and shipped from the cell preparation site (Neuralstem, Inc., Rockville, MD, USA) to the surgery site (UCSD, San Diego, CA, USA) at 2–8°C by overnight delivery. Upon receipt the following day, the cells were used directly for implantation without further manipulation. Before and after implantation the viability of cells was measured with trypan blue (0.4%; Sigma). On average a 75–85% viability rate was recorded.
Experimental Groups: Immunosuppression Protocols
Before cell grafting animals were randomly divided into four experimental groups and received immunosuppressive treatment with Prograf (FK506; Astellas Pharma, Deerfield, IL, USA) alone or in temporary combination with Cellcept (mycophenolate mofetil; Roche Pharmaceuticals, Nutley, NJ, USA) as described in Table 1. These groups allowed us to examine the effect of adding oral or intraperitoneal mycophenolate to the FK506 regimen, and for practical reasons, whether once-a-day treatment would be effective.
Experimental Immunosuppression Protocols

Mycophenolate was always halted 7 days postop. FK506 was always subcutaneous and lowered to 1 mg/kg 14 days postop. qd: once daily; q12h: every 12 h; PO: oral gavage; IP: intraperitoneal.
Immunohistochemistry
Three weeks after cell grafting, rats were deeply anesthetized with pentobarbital and phenytoin and transcardially perfused with 200 ml of heparinized saline followed by 250 ml of 4% paraformaldehyde in PBS. The spinal cords were dissected and postfixed in 4% formaldehyde in PBS overnight at 4°C and then cryoprotected in 30% sucrose PBS until transverse sections (30 μm thick) were cut on a cryostat and stored in PBS. Sections were immunostained overnight at 4°C with primary human-specific (h) or nonspecific antibodies made in PBS with 0.2% Triton X-100: mouse anti-nuclear matrix protein/h-nuc (hNUMA; 1:100; Millipore, Temecula, CA, USA); goat anti-doublecortin (DCX; 1:1000; Millipore). Mouse anti-CD4 and anti-CD8 antibodies (1:500; AbD Serotec, Raleigh, NC, USA) were used for identification of T-lymphocyte infiltration. After incubation with primary antibodies, sections were washed three times in PBS and incubated with fluorescent-conjugated secondary donkey anti-mouse, or donkey anti-goat antibodies (Alexa 488, 546; 1:250; Invitrogen Corp., Carlsbad, CA, USA) and DAPI for general nuclear staining. Sections were then mounted on slides, dried at room temperature, and covered with Prolong anti-fade kit (Invitrogen Corp., Carlsbad, CA, USA).
Quantification of Grafted Cells
Fluorescent images were captured using a Leica DMLB microscope with a Zeiss Axiocam MRm monochrome camera. For quantification, four sections spanning two injection sites were taken from each animal and stained with hNUMA antibody. Quantification was performed using Image-Pro Plus (v.6.2.0.424; Media Cybernetics Inc., Bethesda, MD, USA), counting only hNUMA-positive nuclei and eliminating cells with less than 30% of the maximum staining intensity. Sections that included an obvious injection tract were excluded from analysis because the tract scar introduced significant artifact in the automated quantification. Sections within two to three sections (60–90 μm) were used. Any postprocessing was done with Adobe CS3 (Adobe Systems, Inc., San Jose, CA, USA) with equal changes to any images being compared.
Statistical Analysis
Multiple comparisons between individual grafting groups were performed using one-way analysis of variance (ANOVA) followed by Student-Newman-Keuls test. All results are shown as mean ± SEM. A value of p < 0.05 was considered to be statistically significant.
Results
Tolerability of Immunosuppressive Treatment
Preliminary experiments were performed to determine tolerable drug doses for both FK506 and mycophenolate in naive animals. We initially tested four mycophenolate doses: 30, 50, 70, and 100 mg/kg/day. After 2 days of treatment, doses of 50 mg/kg or higher resulted in severe diarrhea in 100% of animals with dramatic weight loss (up to 10% per day) and development of lethargic behavior. A dose of 30 mg4/kg/day resulted in mild diarrhea in all animals by 3–5 days of treatment but only mild weight loss (<5% per day) and negligible lethargic behavior. FK506 doses of 1 and 3 mg/kg/day were initially tested in naive animals. Using 3 mg/kg/day resulted in continued body weight decline until treatment was halted.
Using data from the dose tolerance study, it was determined the most suitable drug doses were 30 mg/kg/day mycophenolate and 3 mg/kg/day FK506. The FK506 dose was lowered to 1 mg/kg/day at 14 days postop due to animal health issues noted in earlier studies using higher doses of FK506 in long-term treatment. At the doses described here, the animals tolerated each immunosuppression regimen well, with the worse side effects being mild diarrhea, which subsided once the mycophenolate was stopped (7 days postop). Long-term FK506 treatment appeared to have no side effects (at 1 mg/kg/day).
Survival of Grafted Cells in all Experimental Groups
In the first group (group A) treated with FK506 monotherapy, only sporadic survival of hSSCs was noted in one animal (from five total); detection in other animals was unsuccessful (Fig. 1). In that one animal, the identified hNUMA-immunoreactive (IR) cells appeared near the injection tract, typically within the graft core (Fig. 1A) and represented only 11.8 + 3.4% (Fig. 1E) of the largest population detected in the FK506 (SC, qd) + mycophenolate (IP, qd) groups (group D). In all other animals the injection tracts were readily identifiable but were negative for hNUMA.

Immunohistochemical staining and quantification of human nuclei (hNUMA) in animals treated with each immunosuppression protocol. Presence of hNUMA-IR cells was inconsistent or not present in animals treated with FK506 (FK) monotherapy (A), while the addition of mycophenolate (Myc) significantly improved cell survival (B–D), particularly with intraperitoneal mycophenolate administration (D). These results are summarized by expressing the number of hNUMA-IR cells present as a percentage of the maximum number found in any of the groups (E). Scale bar: 200 μm. Dashed line indicates an injection tract. *p < 0.05 when compared to FK(qd) + Myc(ip; q12h) and FK (qd) + Myc(ip; qd). **p < 0.05 when compared to all three FK + Myc groups.
In group B addition of mycophenolate to the immunosuppressive regimen [FK506 (SC, bid) + mycophenolate (PO, bid)] revealed more consistent graft survival (4/5 animals had persisting grafts), with much higher cell density, and greater cell migration from the injection site (Fig. 1B). Cell counting showed cell survival at 3 weeks after grafting to be 61.2 ± 7.8% of the maximum detected in group D (Fig. 1E).
In groups C and D changing the mycophenolate route of administration to IP administered once or twice daily (versus PO) produced the greatest and most consistent graft survival (all animals had persisting grafts), as well as extensive cell migration away from the injection tract (Fig. 1C). The average number of hNUMA-IR cells per section in group C was 852 ± 87 and 915?95 in group D (representing the best survival and for the purpose of this study, designated 100%). While not quantitatively assessed, hNUMA-IR cells were often observed more than 500 μm away from the injection tract in the plane of the transverse section, as well as in sections several hundred microns rostral or caudal from the injection site. Administering mycophenolate once or as a divided dose twice daily had no statistically significant effect on cell survival (Fig. 1D).
Double staining of human cell grafts with hNUMA-IR and doublecortin (DCX) antibody revealed a strong overlap in grafted regions. Intense DCX staining was noted in all persisting cell grafts with virtually all DCX-IR cells also hNUMA-IR (Fig. 2A–D). However, in sharp contrast, animals treated with FK506 monotherapy lacked DCX and hNUMA staining in the injection tracts and putative graft core (Fig. 2E–H). In the one animal (FK506 only) that had sparse hNUMA staining in the injection tract, weak and disorganized DCX-IR structures were sometimes found in the injection sites (Fig. 2I–L).

Immunofluorescence staining of human grafted cells (hNUMA green) and doublecortin (DCX red). In FK506-treated rats, hNUMA and DCX were either absent completely (A–D) or only sparsely found (E–H) within the injection site. Large grafts of human origin were easily detected in animals treated with FK506 and mycophenolate (I). A majority of hNUMA-IR nuclei were found within DCX-positive structures, indicating a neuronal phenotype (J–L, and inset). Dashed line indicates an injection tract. Scale bar: 100 μm, and 25 μm for the inset (L).
CD4/8 immunohistochemistry was used to assess T-lymphocyte infiltration into the spinal cord after cell transplantation. In group A (FK506 only) injection tracts that were typically void of hNUMA-IR contained large populations of CD4/8-IR cells and no DCX staining (Fig. 3A–C). In sharp contrast, no or minimal CD4/8-IR cells were noted in the other three experimental groups with combined immunosuppression (Fig. 3D). In those latter three groups the injection tracts frequently void of CD4/8-IR consistently were strongly reactive for DCX (Fig. 3D–F).
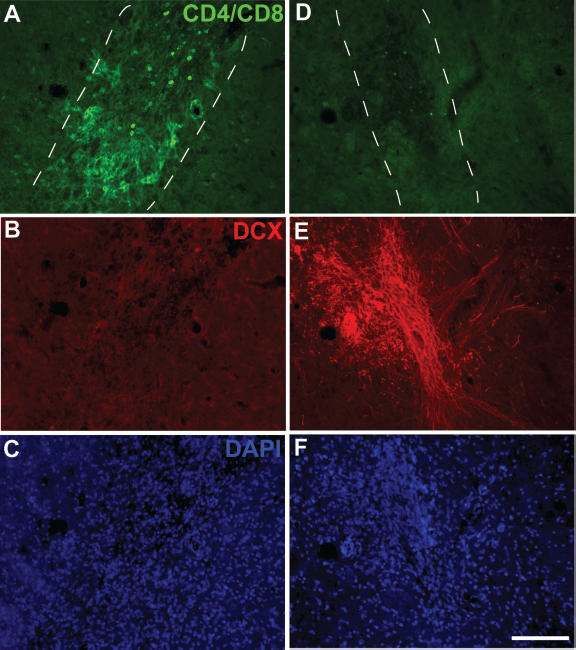
Immunohistochemical staining of T cells (CD4/8) and doublecortin (DCX). In FK506 monotherapy-treated animals, strong CD4/8 staining was noted in the spinal cord near and around all identified injection tracts and these sites were negative for DCX-IR (A–C). However, with combined immunosuppression (FK506 + mycophenolate) extensive DCX-IR was noted throughout the injection sites with elaborate DCX-positive fibers extending from the graft site (E) but were virtually absent of CD4/8-IR (D). Dashed line indicates an injection tract. Scale bar: 100 μm.
Discussion
Cell-based therapies are emerging as possible treatment options for numerous neurological disorders including spinal cord injury, spinal ischemia, stroke, Parkinson's disease, and ALS. However, similar to organ transplantation, graft rejection by the host immune system remains an obstacle in achieving long-term survival and maturation of grafted cells. Given the time frame required for the maturation of grafted human neural precursors to fully functional neurons with well-developed synaptic networks (2–3 months), optimal immunosuppressive protocols are required for each disease process to ensure a reliable and accurate assessment of the potential beneficial effects expected after spinal cell grafting.
Our current study was designed to optimize an immunosuppression protocol for spinal grafting of hSSCs to the lumbar spinal cord of a rat model of ALS. We used two drugs, as monotherapy or in combination: FK506 (tacrolimus) and mycophenolate. FK506 is a macrolide and acts by binding to the immunophilin FKBP12 (FK506 binding protein); this new complex interacts with and inhibits calcineurin, thus inhibiting both T-lymphocyte signal transduction and IL-2 transcription (22). Mycophenolic acid (active form of mycophenolate mofetil after hepatic metabolism) inhibits inosine monophosphate dehydrogenase, the rate-limiting enzyme for the synthesis of guanine monophosphate in the de novo pathway of purine synthesis used in B- and T-lymphocyte proliferation. In the current study, we found extensive CD4/8 staining near the injection sites in animals that received FK506 only and concurrently no grafted cells were noted at 3 weeks after grafting. While the complexity of xenograft rejection by a host has yet to be fully understood, T lymphocytes appear to play a key role [see review (14)]. The importance of T cells in rejection of neural grafts is exemplified by the long-term survival of transplanted human cells in athymic rats (13,36). The strong CD4/8 staining and lack of hNUMA-IR cells at injection sites in animals treated with FK506 monotherapy suggests that despite the actions of FK506, it is insufficient as a single immunosuppressive agent in ALS rats at the dosing regimen employed in the present study. Addition of mycophenolate dramatically improved hSCC graft survival and concurrently decreased detectable CD4/8-IR. No significant difference in graft survival was noted when the drugs were administered once a day or divided twice daily.
It is interesting that while FK506 has been successfully used as monotherapy for spinal transplantation of human cells in our previous spinal ischemia studies (4,24), it failed in the present and earlier studies involving G93A SOD1 animal models of ALS (35). While not assessed directly in the current study, a possible determining factor of the effectiveness of a given immunosuppressive treatment is the degree and duration of inflammation in the vicinity of grafted cells. In G93A SOD1 models of ALS, there is a progressive inflammatory response characterized by the presence of activated microglia and macrophages. Indeed, active inflammation is present from the presymptomatic to the end stages of the disease process (12). Additionally, graft survival is likely affected by infiltration of peripheral immune cells through the compromised blood–brain barrier in ALS-diseased rodents. Recent studies have demonstrated progressive disruption of the blood–brain barrier with vascular leakage and extracellular edema in G93A SOD1 mice (7,8). Zhong et al. (37) also reported progressive blood–spinal cord barrier disruption in SOD1 mice, resulting in microhemorrhages, release of neurotoxic hemoglobin-derived products, and reduced microcirculation. Similarly, infiltration of several vascular high molecular weight components (such as albumin, IgG, and C3 complement) into the cerebrospinal fluid in ALS patients was reported (1,20).
In contrast to ALS where spinal inflammation continues, likely worsening, until animal end stage, the traumatic-injured spinal cord is typically characterized by an initial acute inflammatory phase (hours to days) followed by a progressive loss of most inflammatory markers from the core of the injury over the following days to months (6). For example, there is at least partial restoration of blood–spinal cord barrier functionality 14–28 days after spinal trauma, as evidenced by a lack of extravasation of large and low molecular weight tracers (26). Interleukin-1β mRNA peaked by 12 h, and returned to levels found in sham-operated mice by 28 days after spinal cord contusion injury (27). Thus, it is possible that a differing time course and degree of spinal inflammation noted between mechanical spinal cord injury and experimental/clinical ALS influences cell graft survival and the efficacy of a given immunosuppressive regimen used to prevent cell graft rejection.
Additionally, it is possible that the plasma levels for both drugs have different plasma/clearance profiles and can directly contribute to the failure of the FK506 as monotherapy even if administered twice daily. Previous kinetic studies in rats show that an IV bolus of FK506 (1 mg/kg, bid) resulted in trough concentrations below 10 ng/ml by 6–12 h after injection (29). Therapeutic doses of FK506 have been reported to be 10–20 ng/ml, while doses of 20 ng/ml and higher are associated with neurotoxicity in rats and humans (2,29). Thus, it is possible that FK506 was not effective because the targeted plasma level was only transiently achieved after each injection. Mycophenolate has a significantly longer half-life than FK506, 16–18 h compared to 11 h, respectively (21,32); thus, it is possible that animals receiving mycophenolate benefited mostly from its longer half-life rather than its mechanism of action.
In animals that received combined immunosuppression, sizable human cell grafts were identified that were also intensely immunoreactive for DCX. DCX is a microtubule-associated protein and with few exceptions is exclusively expressed by immature neurons from about embryonic day 10 to about 2–3 weeks after mitosis is complete (3,11). This would suggest that the majority of grafted cells developed a neuronal phenotype by 3 weeks after grafting.
Combined immunosuppression also nearly completely prevented CD4/8 staining in grafted spinal cords. Given that FK506 and mycophenolate have relatively different mechanisms of action, and that the latter can also affect B cell activity, it is likely that these actions complement each other and result in a synergistic interaction. Our results are supported by a previous study by Yan and colleagues (35) that used the same cell line as the current report, and demonstrated that combination therapy of FK506 and rapamycin significantly improved cell graft survival and animal behavior in the G93A mouse model of ALS. Inconsistent cell graft survival using immunosuppression monotherpy has been reported previously. Cyclosporine monotherapy was used by Suzuki et al. after transplanting human-derived cortical neural precursors to the spinal cord of G93A rats and grafts were found in only 3/6 animals at 2 weeks postgrafting and 4/6 animals by 6 weeks. In a similar study the same group described random variability of cell survival while grafts were identified in only 8 of 14 rats (19). In several other cell replacement studies that employ rodent ALS models and report positive results in functional outcome (e.g., improved survival and neurological outcome) no systematic data on the graft survival in individual animals and correlative behavior were provided (9,10,34).
Overall, we demonstrated that the immunosuppression provided by FK506 alone is insufficient to maintain a low population of active T cells after cell transplantation in the G93A ALS rat model. Addition of mycophenolate seemed to supplement the inhibition of T-cell proliferation and leads to a robust graft survival when analyzed at 3 weeks after grafting.
Footnotes
Acknowledgments
This work was supported by the A. Alfred Taubman Foundation and California Institute for Regenerative Medicine (RC1-00131-1).
