Abstract
Amyotrophic lateral sclerosis (ALS) is a fatal disease that involves the degeneration of cortical and spinal motor neurons. Mutant SOD1G93A rats constitute a good animal model for this pathological condition. We have previously demonstrated that transplantation of neonatal olfactory ensheathing cells (OECs) into the dorsal funiculus of the spinal cord of mutant SOD1G93A transgenic rats increases the survival of spinal motor neurons and remyelinates the impaired axons through the pyramidal tract. In the present study, we examine whether intracranial cell implantation could also exert a similar effect on cortical motor neurons and on the lower motor neurons in the spinal cord. We injected OECs from the bulb of 7-day-old GFP green rats into the corona radiata of adult SOD1 mutant rats stereotaxically to observe any changes of the upper motor neurons as well as the lower motor neurons. We found that more motor neurons at both the motor cortices and ventral horns of the spinal cord survived in grafted ALS rats than in control rats. Prolonged survival and behavioral tests including a screen test, hind limb extension, rotarod, and gait control showed that the treated animals were better than the control group. This manuscript is published as part of the International Association of Neurorestoratology (IANR) supplement issue of Cell Transplantation.
Introduction
Amyotrophic lateral sclerosis (ALS) is a progressive fatal neurodegenerative disease characterized by skeletal muscle wasting and paralysis (69). The majority of patients with ALS usually die within 5 years after clinical onset and diagnosis (57). Most ALS occurrence is sporadic, and only 10% is familial (fALS). About 20% of fALS cases arise from mutations in the Cu/Zn superoxide dismutase (SOD1) gene on chromosome 21 (56). Cistaro et al.'s study suggests that brain hypermetabolism is involved in ALS (13). The study groups consisted of patients with ALS of either bulbar or spinal onset and a control group, studied using [18F]fluorodeoxyglucose positron emission tomography (FDG PET). Researchers found that glucose metabolism was significantly reduced in the large frontal and parietal zones in bulbar onset patients compared with both controls and spinal onset patients. Recent studies relating to cell transplant treatment in the SOD1G93A animal model of ALS have achieved encouraging results in presymptomatic as well as symptomatic animals (12,24,36,46,67).
The pathological findings of ALS include the degeneration of the corticospinal tracts associated with loss of upper and/or lower motor neurons and demyelinated axons. The corticospinal tract degeneration is most evident in the lower parts of the spinal cord, but it can be traced up through the brain stem to the posterior limb of the internal capsule and corona radiata by means of fat stains, which show the macrophages that accumulate in response to myelin degeneration (57).
Olfactory ensheathing cells (OECs), first described by Golgi and Blanes at the end of the 19th century, coexist in the peripheral nervous system (PNS) and central nervous system (CNS) and share properties with glia from the PNS, Schwann cells, and astrocytes, a glial cell type from the CNS (19,53,68). The OEC can promote regeneration and remyelination of injured spinal pathways and enhance motor recovery in experimental animal models with spinal cord injury (42,54,55).
Our previous research showed that OEC transplantation could provide protection to motor neurons and enable remyelination in mutant SOD1G93A transgenic rats with ALS (43). Furthermore, translational studies elucidated that fetal OEC transplantation into the corona radiata in ALS patients under local anesthesia are also able to slow the rate of clinical progression of ALS with even lower risks relevant to the operative procedure (6,33).
In the present study, we focused on the possible mechanisms by which OEC transplantation into the corona radiata prolongs survival and improves the overall function of transgenic SOD1G93A rats with ALS.
Materials and Methods
The study and use of animals was approved by the research ethics committee of Beijing Hongtianji Neuroscience Academy.
Preparation of Rat Olfactory Ensheathing Cells
OECs were obtained from the olfactory bulb of approximately 7-day-old neonatal “green” male rats (Lewis-EGFP F463.5, stable-single insertion site, single-chromosome 5) ubiquitously expressing green fluorescent protein (GFP), purchased from the Rat Resource Research Center (Department of Veterinary Pathobiology, University of Missouri, Columbia, MO, USA) (61). The tissues were incubated for 40 min at 37°C in Dispase II (Roche, Mannheim, Germany), then transferred into a 0.25% collagenase type IA (Sigma-Aldrich, St. Louis, MO, USA) in Dulbecco's modified Eagle's medium/Ham's F12 (DMEM/F12) (Hyclone, Logan, UT, USA), and incubated for 30 min at 37°C. After centrifugation, the cell pellet was resuspended in the DMEM/F12 with 10% fetal bovine serum (FBS; Hyclone) and plated on poly-l-lysine (PLL)-coated dishes (Corning, Corning, NY, USA). At 80–90% confluence, the cells were harvested and passed with DMEM/F12 containing 1% insulin–transferrin–selenium supplement (ITS-X, Gibco, Gaithersburg, MD, USA) and 50 ng/ml neurotrophin 3 (NT3, PeproTech EC Ltd., London, UK) onto a PLL-coated dish. Cells were cultured in this medium and were harvested until full confluence.
Assessment of OEC Purity
To assess the purity of OECs before transplantation, small aliquots of cells of five generations were seeded into 24-well plate (Corning) for 24 h then fixed in 4% paraformaldehyde (Sigma-Aldrich) for p75 low-affinity neurotrophin receptor (p75 NTR; 1:500, Santa Cruz Inc., Dallas, TX, USA) immunocytochemical labeling (1,38). The number of positive p75 and GFP (by fluorescence) double-labeled cells was then quantified in five randomly selected fields at 400x under a fluorescence microscope and was divided by the total number of GFP-expressing OECs in that field to obtain the mean percentage of cells expressing p75.
Animals
Sprague–Dawley rats carrying the SOD1G93A mutation have been characterized previously by Herbik et al. (30) as an effective animal model of ALS. The SOD1 rats used in this study were obtained from the Taconic Company (Germantown, NY, USA). The colony was developed by crossing male founders with wild-type female rats, and the heterozygous SOD1G93A progeny was identified using genomic PCR of tail DNA with primers specific for mutated hSOD1. Sprague–Dawley pups were genotyped between 4 and 5 weeks old. A small piece of tail was cut and boiled in 300 μl of 0.02 M EDTA (Sigma-Aldrich) and 1 mM NaOH (Chemicalreagents, Beijing, China) for 1 h. The samples were then vortexed and centrifuged for 5 min. PCR was then performed using 4 μl of this solution. For each sample, a master mix was used that contained 2.5 μl 10 × PCR buffer, 5 μl 5 × Q solution, 0.2 μl Taq DNA Polymerase (Qiagen, Valencia, CA, USA), 2.5 μl 2.5 mM dNTP Mix (Invitrogen, Carlsbad, CA, USA), 0.05 μl primer (5′-CAG CAG TCA CAT TGC CCA GGT CTC CAA CAT G-3′), 1 μl human SOD1 primer (5′-CCA AGA TGC TTA ACT CTT GTA ATC AAT GGC-3′), and 9.75 μl ddH2O. Positive and negative controls were used in every PCR reaction (45). The rats' mean onset of clinical weakness was 122.9 ± 14.1 days, and the duration of the disease was 8.3 ± 0.7 days (52).
The study included male SOD1G93A rats transplanted with OECs, medium + SOD1G93A rats, and sham-operated transgenic rats, with the groups balanced with regard to date of birth and body weight. The wild-type male rats were used as normal control. The groups and numbers of rats used are listed in Table 1.
The Number of Rats Used for Each Part of the Study

WT, nontransgenic Sprague–Dawley rats; OECs, olfactory ensheathing cells; SOD1G93A, transgenic Sprague–Dawley rats expressing familial amyotrophic lateral sclerosis (ALS) mutant (G93A) of human superoxide dismutase 1 (SOD1).
OES Transplantation
Cell transplantation was performed with 90-day-old rats [wild-type (WT) and transgenic SOD1G93A] using stereotaxic coordinates. Under chloral hydrate (Sigma-Aldrich) anesthesia (4%, 12 ml/kg), each rat was fixed into the stereotaxic apparatus (Stoelting, Wood Dale, IL, USA) (3), and a hole was made about 0.5 mm in diameter into the skull at the site +2 mm to the anterior fontanelle, 1.5 mm on the right side of the midline. A glass micro pipette (Hamilton, Bonaduz, Switzerland) was then inserted into the corona radiata (2.75 mm depth from surface). A 5-μl cell suspension (5 × 105 OECs) or DMEM/F12 medium was injected slowly over a 10-min time span. All animals were administered benzylpenicillin (Shiyao Pharmaceutical Group, Shijiazhuang, China; 10 mg/kg/day, subcutaneous injection), continuing for 3 days after surgery without cyclosporine, that is, no immunosuppression in our study.
Behavior, Survival Time, and Quality Assessment
Behavior survival time and a quality assessment were performed everyday after the operation, while behavioral observations were carried out by a blind observer. Disease onset, survival time, body weight, and behavioral tests were measured throughout the study. Disease onset was determined when tremor or subtle weakness was identified during suspension by the tail and/or during voluntary walking. The endpoint was determined when the rats were not able to get up from a lying position (37). These signs of impending death or actual death provided the experimental endpoint for survival and histological analysis.
The behavioral test included a screen test, hind limb extension, a rotarod performance test, and assessment of gait abnormality.
Screen Test
The screen test could serve as an indicator of general muscle strength (14). We used a 29 × 30-cm screen that could be rotated from 0° (horizontal) to 90° (vertical). Grid size of the screen was 0.6 × 0.7 cm. The screen was situated such that, when in the vertical position, it was 70 cm above a foam rubber pad (7.5 cm thick). Each rat was placed on the horizontally positioned screen. We performed it with slight modifications of scores: 0 = grasping the screen with forepaws for more than 5 s; 1 = grasping the screen with forepaws within 5 s; 2 = grasping the screen with forepaws for more than 3 s; 3 = grasping the screen with forepaws within 3 s; 4 = falling off instantaneously.
Hind Limb Extension
The hind limb extension test was performed with slight modifications accordingly to the method reported by Garbuzova-Davis et al. (25). The extension reflex was evaluated holding the rat by their tails suspended. The scores were recorded as 0, 1, 2, 3, and 4 after evaluation of the reflex of lower limb according to published procedures.
Rotarod
Motor impairment was evaluated with a rotarod (Columbus Instruments, Columbus, OH, USA) (2). The test was performed everyday once the animals were 90 days old. Each rat was placed on a rod rotating at 10 rpm, and we recorded the time the rat stayed on the rod until it fell off. The evaluation scores were as follows: 0 = >90 s; 1 = 60–90 s; 2 = 30–60 s; 3 = 10–30 s; 4 = falling off the rod within 10 s.
Gait Abnormality
Gait abnormalities were scored according to a severity scale ranging from 0 to 4: 0 = normal gait; 1 = mild gait impairment; 2 = medium gait impairment; 3 = severe gait impairment; 4 = absence of gait (paralysis) (22). The gait impairment was revealed by spontaneous locomotion in the open field cage. Paralysis was defined as the complete loss of gait associated with the loss of the physiological muscle tone.
Preparation of Tissue Sections
Four rats from each group were evaluated for histological study at 125 days (5 weeks after transplantation) and 139 days (7 weeks after transplantation) (n = 6 for OECs + SOD1G93A group). All SOD1G93A rats in each group as well as the WT controls were deeply anesthetized by intraperitoneal injection of chloral hydrate (Chemicalreagents) and transcardially perfused with normal saline solution (Shiyao Pharmaceutical Group), followed by 4% paraformaldehyde in 0.1 M phosphate-buffered solution (PBS, Gibco). The cerebral and lumbar spinal cords (between the end of T13 and the beginning of L4, including the lumbar enlargement) were dissected and isolated, immersed in the same fixative for 1 h, and cryoprotected in a series of sucrose (Chemicalreagents) solutions: 10%, 15%, and 20% sucrose in PBS at 4°C for 2 days. The tissue was then embedded in Tissue-Tec (Sakura Finetek, Tokyo, Japan) and frozen in liquid nitrogen-cooled isopentane (Chemicalreagents). The tissue was cut in coronal 20-μm-thick serial sections. Sections were mounted on gelatinized glass slides and stored at −20°C. A set of sections included 15 sections in total (every fifth section) from each animal.
Nissl Staining and Motor Neuron Count
To observe the number of motor neurons at 5 and 7 weeks after OEC transplantation in each group, we used Nissl staining. The serial transverse sections of cerebral and lumbar spinal cord were stained with 1% cresyl violet (Sigma-Aldrich) for 3–5 min, rinsed in 95% ethanol, and dehydrated in 100% ethanol. Finally, sections were mounted with Eukitt (Kindler Gmbh & Co., Freiburg, Germany) and examined for quantification of motor neurons.
For quantitative analysis of healthy motor neurons, serial cross sections (20 μm thick) of the cerebral and lumbar spinal cords were made and processed for Nissl staining and choline acetyltransferase (ChAT). Motor neuron cells in the cerebral cortex and lumbar ventral gray matter were counted in every fifth section, and the results of at least 10 sections from four rats were averaged (45,66). Cell count and morphometry were performed using the NIH ImageJ software (Bethesda, MD, USA).
Immunohistochemistry
Selected sections were stained with immunostaining for ChAT. For immunohistochemical staining, cerebral cortex and spinal cord sections were permeabilized with 0.2% Triton X-100 (Sigma-Aldrich) and 4% paraformaldehyde and blocked with 1 mg/ml bovine serum albumin (BSA, Sigma-Aldrich) and 0.5% normal goat serum (Funakoshi, Tokyo, Japan) in PBS (46). The sections were incubated overnight at 4°C with rabbit anti-ChAT (Chemicon, Temecula, CA, USA; AB5042, 1:400). The sections were treated with a second antibody conjugated with fluorescent dye/goat anti-rabbit IgG Texas Red (Santa Cruz Inc.) in the dark for 2 h at room temperature. Sections were coverslipped (Menzel-Glaser, Braunschweig, Germany) with 30% glycerin (Chemicalreagents) and observed under confocal microscope digitalized microphotographs of immunofluorescent sections that were saved. The Texas Red was excited by a Dye Laser at 595–605 nm and was emitted at 620 nm. The EGFP was excited by an argon laser at 488 nm and was emitted at 530 nm.
Western Blot
Rats were anesthetized and killed with an overdose of pentobarbital (Sigma-Aldrich) (150 mg/kg), perfused with ice-cold PBS from the left ventricle. The cerebral cortex and lumbar spinal cord were removed, frozen, and stored at −80°C until protein extraction. Tissues were homogenized in ice-cold lysis buffer (0.32 mol/L sucrose, 1 mmol/L ethylenediaminetetraacetate, 5 mmol/L Tris–HCl, pH 7.4, 0.1 mmol/L phenylmethylsulfonyl fluoride, 10 μmol/L pepsin, 10 μmol/L pepstatin A, and 1 mmol/L β-mercaptoethanol; all Sigma-Aldrich). The protein content was determined by Bio-Rad protein assay (Bio-Rad, Hercules, CA, USA). Equal amounts of protein per lane (50 μg) were loaded onto an 8% polyacrylamide gel (Sigma-Aldrich) and separated by electrophoresis at 90 V for 30 min and then 120 V for 1.5–2.5 h. Proteins were then transferred to nitrocellulose (Millipore, Billerica, MA, USA) at 200 V for 2 h, and the membrane was blocked with 5% nonfat dry milk (Applgen Technologies Inc., Beijing, China), 0.5% Tween-20 in Trisbuffered saline (Gibco) for 2–2.5 h. The nitrocellulose was then incubated with rabbit anti-ChAT (Chemicon, AB5042, 1:800) overnight at 4°C. The membrane was treated with horseradish peroxidase-conjugated secondary antibody (1:1,000, Cell Signaling Technology, Danvers, MA, USA) for 60 min at 37°C and then exposed to X-ray film (Fujifilm, Tokyo, Japan). The X-ray films were scanned, and the optical density was determined by Bio-Rad image analysis. As an internal control, the same nitrocellulose was incubated with an antibody specifically for β-actin (Santa Cruz, 1:1,000) after being stripped.
Statistical Analysis
The quantitative data were expressed as mean ± standard error of the mean (SEM). Statistical analysis was performed with repeated measures of ANOVA using Statview (Abacus Concept, Piscataway, NJ, USA). Analysis of variance and the Student–Newman–Keuls test were used for behavioral data of the multiple comparisons. The statistical analysis of onset age and endpoint was performed with the Kaplan–Meier survival analysis from SPSS 11.5J for Windows (SPSS Inc., IBM, Armonk, NY, USA). A value of p < 0.05 was considered statistically significant.
Results
Identification of OECs
The OECs were observed after 3-day (Fig. 1A) and 7-day cultures with GFP (Fig. 1B) and P75 red fluorescence marker (Fig. 1C). An enrichment of OEC culture of up to five generations could be harvested depending on the proportion of P75 in the starting population of cells.

The morphological identification of olfactory ensheathing cells (OECs) before transplantation. (A) OEC observation at 3 days of culture. (B) The expression of green fluorescent protein (GFP) by transplanted enhanced GFP-expressing OECs (EGFP-OECs) at 7 days of culture. (C) The expression of P75 low-affinity neurotrophin receptor (P75 NTR) by transplanted EGFP-OECs at 7 days of culture. (D) Merged image of (B) and (C). Scale bar: 50 μm.
EGFP-labeled OEC groups were observed under confocal microscopy to evaluate the migration of transplanted OECs. Two weeks following transplantation, OECs showed a restricted pattern of cell dispersion (Fig. 2A) in comparison with that of the intact corona radiata; there was a diffuse distribution of GFP-expressing OECs in the corona radiata of SOD1G93A rats. Five weeks postgraft, proliferation of GFP-expressing OECs was found, which indicated that a net cell migration occurred rather than a pressure-induced spreading of cells (Fig. 2B). Seven weeks after transplantation, there was little distribution of GFP-expressing OECs in the corona radiata (Fig. 2C).
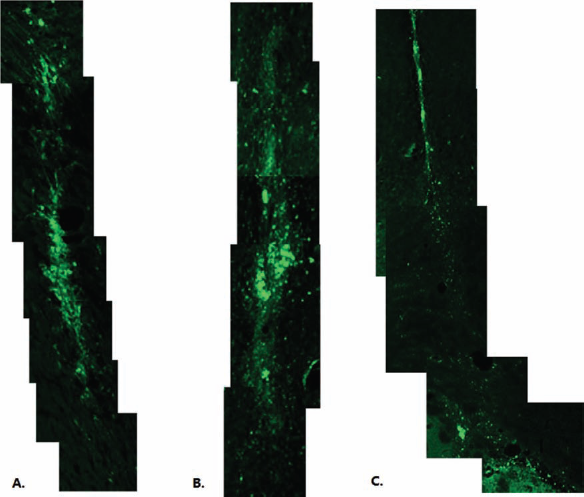
EGFP-OEC was traced at 2, 5, and 7 weeks after transplantation. (A) Two weeks postgrafting, there was a diffuse distribution of GFP-OECs in the corona radiata of Cu/Zn superoxide dismutase G93A mutated amyotrophic lateral sclerosis (ALS) (SOD1G93A) rats. (B) Five weeks posttransplantation, the image showed that there was a more distal dispersion of GFP-expressing OECs. (C) Seven weeks after transplantation, little distribution of GFP-expressing OECs was observed in the corona radiata.
Clinical Assessment and Behavioral Test
The procedure of the corona radiata transplantation was generally well tolerated. Within a few hours postoperatively, animals recovered and were able to stand on their hind limbs. No motor impairment was visible after surgery. Behavior survival time and quality assessment was carried out everyday after the cell implantation.
The analysis of disease onset and animal survival time is shown in Figure 3. OEC-treated SOD1G93A rats had a significantly prolonged survival time compared with SOD1G93A rats and medium-treated SOD1G93A rats, respectively (p < 0.01). There was no significant difference between the SOD1G93A rats and medium + SOD1G93A rats (p > 0.05). On average, the SOD1G93A rats survived for 137.72 days, whereas the OECs + SOD1G93A rats survived for 153.38 days. Medium + SOD1G93A rats survived for 137.90 days. Kaplan–Meier survival analysis revealed a significant increase (more than 15 days) in the survival of OECs + SOD1G93A rats versus SOD1G93A rats or medium + SOD1G93A rats (p < 0.01) (Fig. 3A). Also, there was a significant difference between OECs + SOD1G93A rats and SOD1G93A rats (or medium + SOD1G93A rats) for the day of onset (Fig. 3B).

Symptomatic effects and prolongation of the life span induced by OECs. (A) Kaplan–Meier survival analysis reveals a significant increase in survival of OEC + SOD1G93A rats versus SOD1G93A (p < 0.01). (B) The disease onset time of OEC-transplanted rats is delayed by about 10 days compared with nontreated SOD1G93A rats (p < 0.01). (C) The disease duration in OEC-transplanted rats is significantly longer than in SOD1G93A nontreated rats (p < 0.01).
Body Weight
The rats were observed after transplantation (90 days). The daily recorded body weights showed that there was no significant difference in weight between the disease onset and before 110 days among all groups (Fig. 4A). After approximately 110 days, the weight in the SOD1G93A and medium + SOD1G93A groups started to decline progressively, whereas the animals in the group of the OECs + SOD1G93A started to lose weight from day 120.

The effects of OEC transplantation on behavioral tests in SOD1G93A rats. (A) The body weight from the age of 90 days to death. (B) The total behavioral test included four independent tests (screen test, hind limb extension, rotarod performance test, and gait abnormality). A total score of 16 represents a complete loss of motor function. OEC graft improves the motor function of G93A rats as shown by behavioral tests. This improvement occurs in different behavioral tests aimed at exploring various features of motor activity. Graphs indicate various motor activities: (B1) screen test: indicates the time that the rat is able to hold the grid with its forelimbs before falling; (B2) hind limb extension: indicates the stronger of the hind limb following tail suspension; (B3) rotarod test: indicates the time the rat is able to stay on a rotating rod (rod speed = 10 rpm); (B4) gait abnormality: the rat physiological muscle tone during the disease. Results are shown as the mean ± SEM.
Behavioral Tests
A total score of 16 from four different tests represents a normal motor function, while a score of 0 means complete loss of motor function (Fig. 4B). Behavioral tests were performed and included a screen test (Fig. 4B1), a hind limb extension (Fig. 4B2), a rotarod performance test (Fig. 4B3), and a gait abnormality test (Fig. 4B4). All behavioral observations were recorded by a blind observer. A total score of 16 represented a normal motor function. OEC transplantation improves the motor function of SOD1G93A rats as shown by behavioral tests. This improvement occurs in different behavioral tests aimed at exploring various features of motor activity. Similar to the body weight, at about 120 days, the SOD1G93A and medium + SOD1G93A groups began to decline, but in the OEC transplantation group the impairment and abnormality were better than in the other two groups.
Histology and Western Blot Analysis
The Nissl staining (Fig. 5A1–G3) detection of the cerebral cortex was used to count the number of motor neurons. The count of the motor neuron result (Fig. 6A) showed a significant loss in motor neuron cells in SOD1G93A and medium + SOD1G93A rats in comparison with wild-type rats in the cerebral cortex (p < 0.001). SOD1G93A rats that received OEC transplantation showed a smaller neuronal loss area and collapse than WT SOD1G93A rats (Fig. 5B2–D2, E2–G2). At 7 weeks, Nissl staining of OEC transplants showed that nearly all the motor neurons suffered from some degree of cell loss (neuronal degeneration) in the SOD1G93A and medium + SOD1G93A rats (Fig. 5F2, F3, G2, G3). However, motor neuron cells were more clearly recognized in the figure, and the number of motor neuron cells was more than in the SOD1G93A or medium + SOD1G93A rats (Fig. 6A). The expression of ChAT with immunohistochemistry staining was similar to that with Nissl staining (Fig. 5A4–G4).
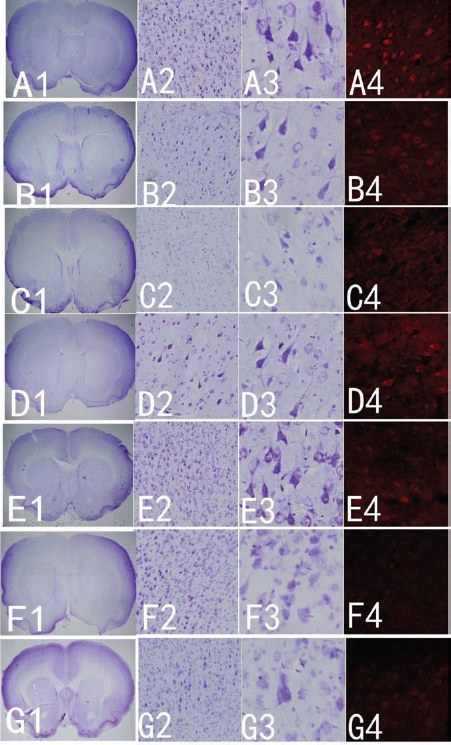
The histological evidence for motor neuronal protection in OEC + SOD1G93A rats. (A1–G3) The distribution and population of motor neurons in the cerebral cortex from hemisections of corona radiata in different groups by Nissl staining. (A1–G1) Location of the cerebral cortex. (A2–G2) Low-magnification image of Nissl staining of the cerebral cortex of wild-type (WT), SOD1G93A, medium + SOD1G93A, and OEC-treated rats demonstrating the features of motor neurons. (A3–G3) Higher magnification of Nisslstained cerebral cortex of WT, SOD1G93A rat, medium + SOD1G93A, and OECs treated showing the features of motor neurons. OEC graft protects the neurons in the cerebral cortex. (A) The normal group; (B) 5 weeks (125 days) after OEC transplantation, the motor neurons are less than in WT but are more than the medium transplanted (C) and SOD1G93A (D); after 7 weeks (139 days) of treatment, the motor neurons in the OEC transplantation (E) were more deficient than before, but in the medium + SOD1G93A (F) and SOD1G93A (G) groups, the motor neurons were close to death. (A4–G4) The expression of choline acetyl transferase (ChAT) immunohistochemistry in the cerebral cortex of the different groups.

Motor neuron quantification. (A) The number of motor neurons in cerebral cortex about 5 weeks after OEC transplantation. (B) The number of motor neurons in cerebral cortex about 7 weeks after OEC transplantation. (C) The number of motor neurons in the lumbar spinal cord about 5 weeks after OEC transplantation. (D) The number of motor neurons in the lumbar spinal cord about 7 weeks after OEC transplantation. There were significant differences between the OEC + SOD1G93A rats and SOD1G93A rats or medium + SOD1G93A rats (p < 0.01). Comparison between groups regarding cell number was made by using one-way ANOVA. #, compared to WT group; *, compared to SOD1G93A or medium transplantation group.
The result of the motor neuron cell count in the lumbar spinal cord through the Nissl staining showed that ALS rats transplanted with OECs had more motor neurons and ChAT protein levels than both the SOD1G93A and medium + SOD1G93A rats (Figs. 6 and 7).

OEC transplantation effect on lumbar spinal cord. (A1–G3) The distribution of motor neurons in hemisections of the lumbar spinal cord in the different groups by Nissl staining. (A1–D1) Nissl staining of lumbar spinal cord sections from different groups at 5 weeks after treatment. The images show the qualitative differences in cell density within the hemisection following OEC treatment in the SOD1G93A rats. (A2–G2) The low magnification of Nissl staining in the lumbar spinal cord. (A3–G3) High magnification of Nissl staining in the lumbar spinal cord. (A) The normal group; (B) 5 weeks after OEC transplantation; compared with group A, there are many motor neurons in the hemisection, but some of them were deformed (B3). Medium + SOD1G93A (C) and SOD1G93A rat (D) showed fewer cells at the age of 125 days (5 weeks). (A1–D1) Nissl staining of lumbar spinal cord sections from different groups at 5 weeks after treatment. Seven weeks (139 days) after OEC transplantation, the cells in the group of OEC treatment were rarely seen and were deformed (E), and no motor neurons were observed in the other groups (F, G). (A4–G4) The expression of ChAT immunohistochemistry in the lumbar spinal cord in the different groups.
Western blot quantification was conducted to evaluate the ChAT protein levels in the cerebral cortex and spinal cord between the OEC-transplanted and the nontransplanted SOD1G93A rats. The results show that the protein levels of ChAT in both SOD1G93A and medium + SOD1G93A are significantly decreased. The ChAT protein levels in the OECs + SOD1G93A rats were significantly higher than in the SOD1G93A and medium + SOD1G93A rats (p < 0.01) (Fig. 8).

The expression levels of ChAT protein in the cerebral cortex and lumbar spinal cord. The expression levels of ChAT protein in the cerebral cortex about 5 weeks (A) and 7 weeks (B) after OEC transplantation. The protein levels of ChAT were significantly decreased across all groups when compared with the wild-type (p < 0.01); however, the OEC + SOD1G93A rats were also significantly different compared to the SOD1G93A rats or medium + SOD1G93A rats (p < 0.01). The protein levels of ChAT in the lumbar spinal cord about 5 weeks (C) and 7 weeks (D) after OEC transplantation showed the protective effects of OEC treatment. #, compared to WT group; *, compared to SOD1G93A or medium transplantation group.
Discussion
Over the years, many therapeutic approaches have been attempted in mice and rat models, often with significant, albeit limited, benefits in disease onset, progression, and life span (15). A variety of cell transplantation experiments have been employed to treat animal models of ALS (9,31,40). The donor cells that showed benefit included human umbilical cord blood-derived cells (10,25,28), olfactory ensheathing cells (43), mesenchymal stem cells (28,51), Sertoli cells (29), and neural stem cells (16). Huang's group found that OEC transplantation resulted in some functional improvement for ALS patients, and there was no difference in the functional improvement between transplanting the cells into the spinal cord or the brain (6,8,33,34). Moreover, several clinical trials transplanting other kinds of cells, such as mesenchymal stem cells, hematopoietic stem cells, and neural stem cells, have shown safe and beneficial effects in patients with ALS (12,17,26,39,48–50).
OECs are a candidate cell for transplantation after genetic modification because they can be engineered to produce neurotrophins (58), they can be safely harvested, and they may be autologously transplanted in humans (21). It was discovered that OECs are neural crest cells and share a common developmental heritage with Schwann cells (4,23). Their mechanism for functional recovery includes a neuroprotective effect, supporting axonal regeneration, remyelination of demyelinated axons, neuroplasticity, neuromodulation, neurogenesis, angiogenesis, reducing scar and cavity formation, and/or strong phagocytic activity (27,31). Recently, therapies based on the use of OECs have been clinically studied in spinal cord injury or stroke, ALS, and other neurological lesions (7,11, 32,35,44,46).
The major finding in our study is that OECs from GFP-transgenic rats could migrate and remain alive at least 7 weeks without immunosuppression (Fig. 2). A previous study reported that astrocytes in the glial scar released tumor necrosis factor-α (TNF-α) (64), which may attract OECs to the site and possibly explain the extensive migration. From the fifth month of age, SOD1G93A mice displayed impaired exploratory activity and motor coordination (41), followed by asymmetric hind limb weakness, spasticity, and atrophy, exacerbated by high-intensity endurance exercise training in males (47). Male rats transplanted with OECs had a mean disease onset of 136 days of age and survived for 153.38 days (Fig. 3B, C) in our study, which was significantly delayed compared to nonvehicle or vehicle treatment. All parameters here considered, scoring of motor deficits, performance on the screen test, hind limb extension, rotarod performance test, and gait abnormality, were able to differentiate between treated and untreated male rats. OEC transplantation showed significant effects on motor behavior at the age here considered (Fig. 4).
OEC-transplanted ALS rats had a significantly higher number of motor neurons and ChAT protein levels than nontransplanted rats in the cerebral cortex (Figs. 5–8). Astoundingly, the number of motor neurons and the protein levels in the spinal cord in the OEC-transplanted group were also much higher than in the nontransplanted one, which resulted in longer survival in rats treated with OECs.
As we described in our clinical study paper (6), the key point for neural network restoration (KPNNR) in the brain represents a point at which numerous projection fibers, association fibers, and commissural fibers converge and pyramidal tract passes through. After being transplanted into this important point in the brain, the cells will initiate an extensive bidirectional remodeling in the entire neural network including the cerebrum, cerebellum, and spinal cord. The cells were transplanted at the KPNNR in this study, supporting our hypothesis about the existence of the KPNNR.
This raises the question as to why ALS rats with OECs transplanted into the corona radiata have a higher quantity of both upper and lower motor neurons and quality of life than control animals. First, this probably was a result of neuroprotection on upper motor neurons and remyelination on axonal demyelination by the OECs (18,43,59,60,63); second, there probably was neuromodulation or neurostimulation (5,62,65) as well as neuronutrition by axonal transport from upper motor neurons to lower motor neurons (20). We hypothesized that the transplanted OECs through secretion of growth factors such as nerve growth factor (NGF) and glial-derived neurotrophic factor (GDNF) would change the environment of the brain to protect upper motor neurons and remyelinate demyelinated axons, modulate or stimulate, and give more nutrition to lower motor neurons through the upper motor neurons and their axonal plasma transport.
Hardships and challenges still await patients with ALS in our quest to improve their neurological functions and quality of life. Even though it is impossible to clinically cure this disease in the near future, the scientific community should encourage any efforts to discover effective therapeutic strategies globally. Fortunately, present clinical studies have already shown that cell therapy could restore patients' neurological functions by neuroprotection or some other mechanisms to some degree and for a period of time (6,33).
In summary, our findings demonstrate that transplantation of OECs is feasible in transgenic ALS rats. Through cell injection before disease onset, at a time point when no major motor neuron loss had occurred, more motor neurons at both motor cortices and ventral horns of the spinal cord survived in grafted ALS rats than in control rats. Prolonged survival and motor skills including a screen test, hind limb extension, rotarod, and gait control showed that the treated animals were better than the control group. Further studies are warranted to confirm the exact mechanism by which upper and lower motor neurons survive after OEC transplantation.
Footnotes
Acknowledgment
The authors declare no conflict of interest.
