Abstract
Embryonic stem cells (ESCs) hold great therapeutic potential due to their ability to differentiate into cells of the three primary germ layers, which can be used to repopulate disease-damaged tissues. In fact, two cell therapies using ESC derivatives are currently in phase I clinical trials. A main concern in using ESCs and their derivatives for cell transplantation is the ability of undifferentiated ESCs to generate tumors in the host. Positive selection steps are often included in protocols designed to generate particular cell types from ESCs; however, the transition from ESC to progenitor cell or terminally differentiated cell is not synchronous, and residual undifferentiated cells often remain. In our transplants of ESC-derived neural progenitors (ESNPs) into the adult mouse hippocampus, we have observed the formation of teratocarcinomas. We set out to reduce teratocarcinoma formation by enrichment of ESNPs using fluorescence-activated cell sorting (FACS) and have found that, although enrichment prior to transplant reduces the overall rate of teratocarcinoma formation, the tumorigenicity of cell batches can vary widely, even after FACS enrichment to as much as 95% ESNPs. Our data suggest that this variability may be due to the percentage of residual ESCs remaining in the transplant cell population and to the presence of pluripotent epiblast-like cells, not previously identified in transplant batches. Our data emphasize the need for stringent characterization of transplant cell populations that will be used for cell replacement therapies in order to reduce the risk of tumor formation.
Introduction
Cell transplantation therapies using embryonic stem cell (ESC) derivatives are on the horizon and have been proposed for various neurodegenerative diseases that are often resistant to drug treatment. Many studies have focused on the efficient derivation of specific subtypes of neurons or glial support cells from either mouse or human ESCs [reviewed in Cai and Grabel (5)]. Several reports have also demonstrated varied levels of efficacy of these cells in relieving disease symptoms when transplanted into animal models of neurodegenerative diseases (12,18,20,25,27,28,36). This success in animal models has recently led to the first FDA-approved clinical trials for ESC-based therapy in humans: one in which hESC-derived oligodendrocyte precursors are used to treat spinal cord injury (1) (ClinicalTrials.gov Identifier NCT01217008; now halted) and another in which hESC-derived retinal pigment epithelial cells are used to treat Stargardt's disease and age-related macular degeneration (34) (ClinicalTrials.gov Identifier NCT01345006).
As medicine moves in the direction of ESC-based therapies, we must be wary of the ability of residual undifferentiated cells to give rise to teratocarcinomas following transplantation. The ability of ESCs to proliferate and generate derivatives of all three primary germ layers makes them attractive candidates for cell-based therapies, but the retention of pluripotent cells could lead to the generation of teratocarcinomas in transplant patients. Stringent protocols to eliminate undifferentiated ESCs from the transplant population must be utilized.
In animal studies involving transplantation of a variety of ESC-derived cell types, there are mixed reports of graft overgrowth and teratoma formation (3,13,35). In addition, several studies have investigated the minimum number of contaminating pluripotent cells required for teratocarcinoma formation in different target tissues (16,23,24,31). These studies suggest a wide range of numbers of residual ESCs for tumor formation: from 2 to 100,000 cells, varying with transplantation site. For example, one study observed tumors only after injection of at least 50,000 undifferentiated mouse ESCs into the mouse heart, with the rate of tumor formation increasing with dosage of ESCs (31). On the other hand, as few as 400 mESCs generated tumors when transplanted into the adult mouse cerebral cortex (16).
Various methods exist for enriching the desired cell type for cell replacement [reviewed in Hentze et al. (19)], including fluorescence-activated cell sorting (FACS) using cell lines expressing constructs encoding cell type-specific fluorescent reporters or antibodies directed against cell surface proteins specific to the cell types of interest. Other methods aim to inhibit the survival and proliferation of undifferentiated ESCs within the transplant, for example, by chemically induced apoptosis in ESCs (2) or by inhibition of ESC self-renewal (30). Over the course of our work on transplanting ESC-derived neural progenitors (ESNPs) into the mouse hippocampus, we have observed the formation of transplant-derived teratocarcinomas (7). In an effort to reduce teratocarcinoma formation, we set out to enrich the transplant population by FACS based on the expression of the neuroectoderm-specific transcription factor sex-determining region Y-box 1 (sox1). We observe here that, although this step reduces the overall rate of teratocarcinoma formation, tumors may still form, depending on the makeup of the starting cell population. Although positive selection for Sox1-expressing cells increases the percentage of ESNPs in the transplant population, the number of undifferentiated ESCs comprising the Sox1-negative population can vary with cell batch. In addition to stage-specific embryonic antigen 1 (SSEA-1)-positive undifferentiated ESCs, we also observe cells expressing fibroblast growth factor-5 (FGF-5), which may represent an epiblast-like population. Epiblast, which is segregated from extraembryonic endoderm in the inner cell mass of the late blastocyst stage embryo, is a pluripotent cell capable of generating derivatives of all three primary germ layers of the mammalian embryo but no longer able to generate extraembryonic endoderm (15). We have previously shown that during in vitro neural differentiation, our ESCs transition through an epiblast intermediate, before becoming neuroectoderm (6). These epiblast-like cells may also contribute to the teratocarcinoma formation we observe in our ESNP transplants.
Materials and Methods
Culture of ESCs and In Vitro Neural Differentiation
Sox1-green fluorescent protein (GFP) (46C) mouse ESCs (gift of Dr. Austin Smith, University of Cambridge) were routinely cultured and differentiated into ES-derived neural precursor cells (ESNPs) as described (6). After 7 days of culture in N2B27 differentiation medium, neural cultures were either frozen, passaged (replated), or subjected to FACS 2 days prior to transplantation. For replating, cells were dissociated using trypsin, plated at 3.5–5.0 × 104 cells/cm2 on laminin-coated tissue culture dishes and further cultured in N2B27 for 2 days before transplantation. Frozen aliquots of day 7 differentiation cultures were thawed onto laminin-coated tissue culture dishes in N2B27 medium at 5.0 × 104 cells/cm2 and were either replated or subjected to FACS prior to transplantation.
Generation of the Sox1-GFP/Ubi-RFP Mouse ESC Line
Sox1-GFP (46C) ESCs were transduced with a lentiviral vector containing the mCherry fluorescent reporter under the control of the ubiquitin promoter (gift of Dr. Alex Lichtler, University of Connecticut Health Center). Clonal ESC lines were generated by serial dilution and expansion of red fluorescent protein (RFP)-positive clones.
Fluorescence Activated Cell Sorting (FACS)
Neural differentiation cultures were trypsinized and resuspended in phosphate buffered saline (PBS) supplemented with 2% fetal calf serum (FCS) and 20 mM glucose. Cells were then filtered through 45-μm mesh and treated with propidium iodide to eliminate dead and dying cells from the sort. Undifferentiated Sox1-GFP and Sox1-GFP/Ubi-RFP ESCs were used as negative gating controls. Cells were sorted and analyzed using a FACSVantage flow cytometer with FACS Diva software (BD Biosciences). FACS sorted Sox1-GFP/Ubi-RFP double- positive cells were collected in PBS supplemented with 10% FCS and 20 mM glucose and kept on ice until plating. Following FACS, cells were pelleted, resuspended in N2B27 media supplemented with epidermal growth factor (EGF) (10 ng/ml) and fibroblast growth factor-2 (FGF-2) (10 ng/ml), and plated onto laminin coated tissue culture dishes at 0.5 − 1.0 × 106 cells/ml.
Transplantation of ESNPs Into Mouse Hippocampus
Transplant procedures were done following approved Wesleyan University Animal Care and Use Committee protocols and in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. Two days following FACS or replating of ESNPs, cells were trypsinized and resuspended in N2B27 media. Approximately 50,000 cells in 1 μl of N2B27 were stereotaxically injected into the hippocampus of 6- to 8-week-old male 129SvEv (Taconic) or B6.CB17-Prkdc scid/SzJ (Jackson Laboratories) mice as described (7) with the following modifications: Anesthesia was induced via inhalation of isofluorane gas and monitored by observing breathing rate and responsiveness to toe-pinch test. The stereotaxic coordinates used for transplantation were −2.0 mm posterior from bregma, −1.9 5 mm lateral to the midline, and −1.9 mm ventral to the pial surface. Four weeks following transplantation, animals were sacrificed by a lethal dose of sodium pentobarbital (Nembutal; Abbott Labs, North Chicago, IL) and transcardially perfused with heparinized phosphate buffer (pH 7.4) followed by 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (pH 7.4). Brains were cryoprotected by sucrose gradient followed by embedding and freezing in tissue freezing medium. Care was taken when removing brains to collect tumors intact and still attached to the brain.
Immunocytochemistry and Immunohistochemistry
Cultured cells were fixed with 3.7% formaldehyde in PBS for 10 min followed by permeabilization with 0.5% PBS-Triton x 100. Cells were then blocked for 1 h at room temperature in 2% bovine serum albumin (BSA)/5% serum/0.1% PBS-Triton x 100 before incubation with primary antibodies in blocking solution overnight at 4°C. Secondary antibody incubations were done for 1 h at room temperature in blocking solution. The following antibodies were used: βIII tubulin (TUJ-1; Covance, mouse monoclonal, 1:500), Nestin (Chemicon, mouse monoclonal, 1:1000), NeuN (Chemicon, mouse monoclonal, 1:400), octamer-binding transcription factor 3/4 (Oct3/4; Santa Cruz, rabbit polyclonal, 1:1,000), smooth muscle actin (SMA; Sigma, mouse monoclonal, 1:1,000), SSEA-1 (Developmental Studies Hybridoma Bank, mouse monoclonal, 1:1,000), Troma-1 (Developmental Studies Hybridoma Bank, rat monoclonal, 1:10), RFP (Rockland, rabbit polyclonal, 1:1,000), GFP (Invitrogen, rabbit polyclonal, AlexaFluor 488 conjugated, 1:1,000), and FGF-5 (Santa Cruz, FL-268, rabbit polyclonal, 1:100). All fluorescent secondary antibodies (Alexa Fluorconjugated, Invitrogen) were used at 1:1,000. Cells were counterstained with either Hoechst 33342 or SYTOX green dye for 10 min prior to coverslipping with Gelvatol. For immunohistochemistry, brain/tumor sections were processed similarly to cultured cells, following rehydration in PBS and permeabilization with 0.5% PBS-Triton x 100. Slides were imaged using a Nikon Eclipse Ti with NIS Elements software.
Measurement of Teratocarcinoma Volume
Cryopreserved mouse brains were sectioned at 12 μm. Every 10th section throughout the tumor was collected beginning at the first observation of RFP-positive transplanted cells and ending when RFP-positive cells were no longer found. The first and last slides containing sections with RFP-positive tumors, as well as every fifth slide in between, were processed for hematoxylin and eosin (H&E) staining (below). H&E-stained slides were imaged using a Nikon Eclipse E400 microscope with NIS Elements software. The area of all tumor-containing sections was acquired by using the area measurement tool in the Nikon NIS Elements software. Total length of individual tumors was calculated and multiplied by the average area of the tumor sections to approximate individual tumor volumes.
Hematoxylin and Eosin Staining
Slides were stained with hematoxylin and eosin using standard protocols. In brief, slides were air dried, rinsed in water, soaked for 1 min in Gill's hematoxylin, and differentiated in tap water and acidic ethanol. After a rinse in 70% ethanol, they were stained for 3 min in eosin, then dehydrated in EtOH followed by xylenes, and finally coverslipped in Permount.
Statistical Analysis
The chi-square test for independence was used to compare tumor formation between transplants with FACS-enriched ESNPs or unsorted ESNPs. The difference in average tumor volume between conditions was tested using the Student's t test. Differences were considered to be statistically significant when p < 0.05.
Results
We routinely transplant ESNPs into the hippocampal dentate gyrus (DG) using mouse models of temporal lobe epilepsy (TLE) and have demonstrated that transplanted cells incorporate into the DG where they mature into dentate granule neurons (7,17). Teratocarcinoma formation by residual undifferentiated cells is a major concern in the transplantation of ESC derivatives. In our previous studies, we occasionally observed both teratocarcinoma and neural tumor formation (7).
We set out to reduce teratocarcinoma formation by transplanting populations of cells that had been enriched for ESNPs (up to 95% pure) by FACS for Sox1-GFP expression (Fig. 1). The Sox1-GFP (46C) mouse ESC line was used to derive ESNPs because GFP reporter expression under the control of the early neuroectoderm marker sox1 allows for the identification and isolation of live ESNPs as they arise in culture. We modified this cell line to constitutively express RFP (Fig. 1C, E) in order to trace transplanted cells in the host brain at all stages of differentiation and after long incubation periods of up to 2 months.
FACS-enrichment of Sox1-GFP/Ubi-RFP ESNPs. Sex-determining region Y-box 1–green fluorescent protein-positive embryonic stem cell-derived neural progenitors (Sox1-GFP-positive ESNPs) are enriched by fluorescence-activated cell sorting (FACS) and express the neural stem cell marker nestin. (A) Day 7 ESNPs isolated using FACS yield a 95% Sox1-GFP-positive population. (B) Sox1-GFP-positive ESNPs in culture 2 days following FACS. (C, E) All ESNPs express red fluorescent protein (RFP) under the control of an ubiquitin promoter. (D) Sox1-GFP-positive ESNPs 2 days following FACS express nestin. Scale bars: 100 μm (in B–E and insets in C and E).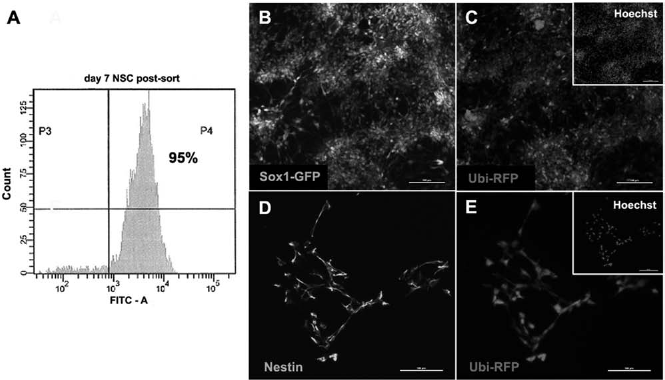
Immunocytochemical characterization of representative batches of FACS-enriched and nonenriched transplant cell populations examined on the day of transplantation (Fig. 2) showed that, in addition to Sox1- and nestin-positive ESNPs, batches contained up to 11% TUJ-1-positive neurons and 3% Troma-positive extraembryonic endoderm cells. We examined representative cultures for the presence of residual pluripotent cells by immunostaining for octamer-binding transcription factor 4 (Oct4) as well as the conventional ESC marker SSEA-1. Oct4-positive cells were observed at a frequency of about 5% of total cells (Fig. 2B). SSEA-1-positive cells were also observed, and their percentage was reduced by FACS for sox1 from about 7% in an unsorted batch to 3% following FACS (Fig. 2A, C). We chose to focus on SSEA-1 as an indicator of pluripotent ESCs in our cell batches as others have shown that Oct4 protein expression can be detected in transitioning progenitors of different cell lineages (10,33). We did not, however, observe any Sox1-GFP-positive cells that coexpressed either Oct4 or SSEA-1 in our transplant cell batches (Fig. 2F–H). In addition to SSEA-1- and Oct4-positive cells, differentiated cultures contained cells expressing the epiblast marker FGF-5 (Fig. 2B). Interestingly, both SSEA-1-positive and FGF-5-positive cells were predominantly found in compact clusters rather than dispersed throughout the cultures (Fig. 2D, E). Others have demonstrated that FGF-5-positive epiblast stem cells (EpiSCs) are pluripotent and can generate cells of all three germ lineages in teratoma assays (4,32). At early stages of neural differentiation, we observe a large population of FGF5-positive cells, which we believe represents a similar transition stage between ESC and neuroectoderm (6). Emerging Sox1-GFP-positive cells coexpress FGF-5 before maturing into FGF-5-negative/Sox1-GFP-positive ESNPs (Fig. 2I–K). It is possible that FACS enrichment for Sox1-GFP alone may include some of these FGF-5 coexpressing cells in the transplant population. We propose that this epiblast-like population in our transplant cell batches is an additional source of tumor-forming cells.
Characterization of representative ESNP transplant cell populations. Quantification of percentage of cells expressing markers of ESNPs (Nestin, Sox1), immature neurons (βIII tubulin [TUJ-1]), extraembryonic endoderm (Troma), and pluripotent cells (stage specific embryonic antigen 1 [SSEA-1], octamer-binding transcription factor 4 [Oct4], fibroblast growth factor-5 [FGF-5]) from two separate unsorted populations (A,B) and one FACS-enriched transplant population (C) at the time of transplantation. Note that in a FACS-enriched ESNP population with 95% Sox1-GFP (C) up to 2% of cells remain SSEA-1-positive. FGF-5-positive “epiblast-like” cells were observed as 11% of total cells on average (B). Error bars represent standard deviation. (D) FGF-5-positive cells are observed in compact clusters amidst neural rosettes containing nestin-positive ESNPs. (E) Residual undifferentiated ESCs expressing SSEA-1 are found in clusters. Sox1-GFP-positive cells in early neural differentiation cultures (F) and at the time of transplantation (G,H) do not coexpress Oct4 or SSEA-1. (I–K) Emerging Sox1-GFP-positive cells at early stages of neural differentiation coexpress FGF-5. Scale bars: 50 μm (D, F, and I–K) and 100 μm (E, G, and H). Insets in (D) and (E) are magnified at 2.5x.
We transplanted several batches of unsorted or FACS-enriched ESNPs, with varying percentages of Sox1-GFP-positive cells, into the adult mouse hippocampus. As we have previously shown, some transplants incorporated into the upper blade of the hippocampal DG and matured into neurons expressing the marker neuronal nuclei (NeuN) (Fig. 3A). In a number of cases, brains contained large heterogeneous masses of transplant-derived cells, identified as teratocarcinomas, based on expression of markers of the three primary germ layers as well as retention of SSEA-1-positive undifferentiated ESCs (Fig. 3). Overall, the rate of teratocarcinoma formation in animals receiving transplants of FACS-enriched ESNPs was reduced when compared to those receiving non-FACS sorted cells (20% and 87.5%, respectively; p = 3×10−6 by chi-square test for independence) (Table 1). We also quantified the total tumor volume per mouse to compare whether the overall tumor mass varied between FACS-enriched and nonenriched transplant cell batches. With or without FACS enrichment, the volume of tumors varied greatly with no significant difference between conditions (p = 0.50 by Student's t test) (Fig. 3F).
Transplantation of ESNP populations that contain undifferentiated ES cells can result in teratocarcinoma formation in the mouse brain. (A) Representative Sox1-GFP/Ubi-RFP ESNP transplant in which no tumor was observed but the graft incorporated into the upper blade of the hippocampal DG. Transplant-derived neural cells at varying stages of maturity are found within the graft including NeuN-positive mature neurons (inset). Scale bar: 100 μm. (B) Teratocarcinoma derived from Sox1-GFP/Ubi-RFP cells in the DG of mouse hippocampus. Scale bar: 500 μm. Nuclei are counterstained (A and B) by SYTOX Green. (C) Cells of the endodermal lineage present in the tumor express Troma-1. (D) βIII-tubulin expression in the tumor indicates the presence of cells of the ectodermal lineage. (E) SSEA-1-expressing cells can be found in tumors suggesting the presence of undifferentiated ES cells. (F) Smooth muscle actin (SMA)-expressing cells indicate the formation of mesodermal tissue. Nuclei are counterstained (C–F) by labeling with Hoechst 33342. Scale bars: 100 μm (C–F). (G) The average volume of tumors found in animals receiving transplants of FACS-enriched (sorted) ESNPs versus unsorted ESNP populations varies within each treatment group and is not significantly different between groups. Error bars represent standard error of the mean (SEM). Statistical significance determined by Student's t test, p = 0.50. Summary of Number of Tumors Observed Following Transplantation of Fluorescence-Activated Cell Sorting (FACS)-Enriched Embryonic Stem Cell-Derived Neural Progenitors (ESNPs) or Unsorted ESNPs The difference in the rate of tumor formation between FACs-enriched and unsorted ESNP transplants is statistically significant, p = 3×10−6, by chi-square test of independence.

While FACS enrichment results in an overall reduction in tumor formation, surprisingly, we do not observe a correlation between the relative enrichment of GFP-positive cells and a reduction in teratocarcinoma formation. In a set of transplants in which enrichment for Sox1-GFP-positive cells was approximately 95%, 6 of 10 transplants resulted in teratocarcinoma formation, whereas in a separate set with 90% enrichment, only 1 of 10 transplants resulted in tumor (Fig. 4). This suggests that, although ESNPs were derived by the same protocol each round, there is variability in the tumorigenicity of each transplant cell batch. Analysis of representative transplant cell populations revealed that, while the number of SSEA-1-positive cells is reduced following FACS enrichment, as little as 2% of the transplant population expressing SSEA-1 can result in teratocarcinomas. Furthermore, the presence of FGF-5-positive epiblast-like cells, which can persist even in mature neural cultures and after multiple passages, may also lead to tumor formation.
Tumorigenicity following ESNP transplants varies by ESNP cell batch. The rate of tumor formation varies in individual rounds of transplants with FACS-enriched ESNPs. The percentage of Sox1-GFP-positive cells in each transplant cell batch is indicated above bars. There does not appear to be a correlation between increased percentage of Sox1-GFP-positive cells and lowered rate of tumor formation. * indicates transplants with ESNPs from a common frozen batch. # indicates transplants into immunodeficient (SCID) mice.
Discussion
ESC derivatives are an attractive source for cell-based therapies because large amounts of material can be generated readily via defined differentiation protocols. Enrichment steps that increase the population of the desired cell type for transplant are included in many of these protocols. The exclusion of residual undifferentiated cells is also key to avoiding teratocarcinoma formation. Our data support the idea that enrichment of the desired cell type for transplantation is critical but that careful characterization of the total transplant population, with specific attention to all pluripotent cells, is imperative to reduce tumor formation.
Two previous studies, using ESNPs generated by a stromal-derived inducing activity (SDIA) protocol or a five-step embryoid body based protocol, transplanted FACS sorted Sox1-positive or the residual Sox1-negative cells into the mouse striatum (9,14). These studies did not directly compare tumorigenesis before and after FACS enrichment. In the first study (14), tumors were observed in 6 of 11 transplants with Sox1-negative cells, whereas 9 of 10 cases resulted in tumors from Sox1-negative transplants in the second study (9). Tumors were not found in any of the Sox1-positive transplants. The rates of tumor formation in these Sox1-negative cell transplants, although high, are very different from each other. Our data suggest that this may be due to different percentages of undifferentiated cells composing their individual Sox1-negative populations. These studies claimed that the lack of tumors in Sox1-positive transplants was due to 100% or 93.9% purity of the Sox1-positive postsort population. However, our data (Fig. 4) suggest that tumors can form after transplantation of 95% Sox1-positive cells and that tumorigenicity varies even among batches with the same percentage of Sox1-positive cells.
The absence of tumors observed in these studies may also be a result of more mature cells present at the time of transplant than those used in our study. For example, Fukuda et al. (14) report just over 20% of the population as TUJ-1 (βIII-tubulin)-positive at the time of FACS versus 10% and under TUJ-1-positive in our study. In addition, the ESNPs transplanted in our study were derived by monolayer differentiation, a method very different from those used in the two studies described, and were harvested for transplantation at an early stage, after just 7 days in neural differentiation medium. We and others have observed that transplantation of more mature neural cultures results in lessened tumorigenesis (3,21,22,26); however, depending on the goal of the transplant, more mature cells may not be an ideal option. Mature neural progenitors and immature neurons are more easily damaged during transplantation and may be at a survival disadvantage in vivo. In addition, some neurodegenerative diseases involve recurring neural damage, which may affect the survival of mature ESC-derived cells introduced by transplantation. For example, a loss of hippocampal inhibitory interneurons is associated with TLE (11,29), and there is evidence that the recurring seizures characterizing the disease further reorganize the neural circuitry and create an environment of ongoing neurodegeneration (8). ESC-based therapies for these types of diseases may necessitate a more immature progenitor capable of self-renewal as well as differentiation, so as to supply a continuing source of replacement cells.
We have shown that positive selection for a desired cell type can significantly increase the percentage of the selected cells for ESC-based cell transplantation therapies, but the composition of the remaining population can vary with respect to residual undifferentiated cells with tumor-forming capacity. These observations strongly suggest a need for stringent characterization of the total transplant cell population and exclusion of all pluripotent cells, be they SSEA-1- or Oct4-positive ESCs or FGF-5-positive epiblast-like cells.
Footnotes
Acknowledgments
We would like to thank Dr. Austin Smith at the University of Cambridge for providing the Sox-GFP (46C) mouse ESCs. We would also like to thank Dr. Alex Lichtler at the University of Connecticut Health Center for providing the ubiquitin-mCherry vector. Funding for L. Grabel was from Connecticut Stem Cell Initiative. The authors declare no conflicts of interest.
