Abstract
Chronic systemic immunosuppression in cell replacement therapy restricts its clinical application. This study sought to explore the potential of cell-based immune modulation as an alternative to immunosuppressive drug therapy in the context of pancreatic islet transplantation. Human amniotic epithelial cells (AEC) possess innate anti-inflammatory and immunosuppressive properties that were utilized to create localized immune privilege in an in vitro islet cell culture system. Cellular constructs composed of human islets and AEC (islet/AEC) were bioengineered under defined rotational cell culture conditions. Insulin secretory capacity was validated by glucose challenge and immunomodulatory potential characterized using a peripheral blood lymphocyte (PBL) proliferation assay. Results were compared to control constructs composed of islets or AEC cultured alone. Studies employing AEC-conditioned medium examined the role of soluble factors, and fluorescence immunocytochemistry was used to identify putative mediators of the immunosuppressive response in isolated AEC monocultures. Sustained, physiologically appropriate insulin secretion was observed in both islets and islet/AEC constructs. Activation of resting PBL proliferation occurred on exposure to human islets alone but this response was significantly (p < 0.05) attenuated by the presence of AEC and AEC-conditioned medium. Mitogen (phytohaemagglutinin, 5 μg/ml)-induced PBL proliferation was sustained on contact with isolated islets but abrogated by AEC, conditioned medium, and the islet/AEC constructs. Immunocytochemical analysis of AEC monocultures identified a subpopulation of cells that expressed the proapoptosis protein Fas ligand. This study demonstrates that human islet/AEC constructs exhibit localized immunosuppressive properties with no impairment of β-cell function. The data suggest that transplanted islets may benefit from the immune privilege status conferred on them as a consequence of their close proximity to human AEC. Such an approach may reduce the need for chronic systemic immunosuppression, thus making islet transplantation a more attractive treatment option for the management of insulin-dependent diabetes.
Keywords
Introduction
Islet transplantation offers a more physiological approach to the restoration of glucose homeostasis than exogenous insulin therapy (8,30), but its use is restricted to a discrete population of individuals with type 1 diabetes who experience frequent and unpredictable episodes of hypoglycemia. More widespread application of islet transplantation awaits solution of technical limitations, in particular the requirement for chronic systemic immunosuppression, which poses risks both to the islet graft and its recipient (4,11,17,32).
Development of a safe, biocompatible method of islet immune isolation that circumvents the need for systemic immunosuppression has been the subject of extensive research. Macro- and microencapsulation devices have been the preferred option, resulting in limited clinical application (6,39), but loss of capsule integrity and impaired gaseous and nutrient exchange undermine long-term β-cell function. Nanocapsule devices, formed from layers of biocompatible polymer applied to the islet surface, address some of these limitations, but the process is technically involved and incurs significant loss of β-cell mass (35,40). Hence, a clinical role for encapsulated islet transplants must await improvements in capsule composition and biocompatibility.
A more physiological approach to cellular immune evasion exploits the properties of cells with innate immunomodulatory capabilities involved in creating anatomical sites of immune privilege. Sertoli cells (SC) induce a state of immune neutrality in the testis to support resident germ cells and prevent rejection of allogeneic and xenogeneic intratesticular islet grafts. Furthermore, SCs may also confer immune privilege at anatomical sites that would otherwise be unable to sustain graft survival without systemic immunosuppression. When preengrafted to the renal capsule of chemically induced diabetic mice, SCs enhance subsequent islet allograft survival (20); examples of xenograft protection at ectopic sites in large mammals have also been documented (16). Intriguingly, such studies suggest that complete encapsulation of islets by SC is not a prerequisite to prevent rejection; nonetheless, obtaining sufficient numbers of SC for use in human transplantation would pose logistical challenges.
Human fetal membranes may provide an alternative source of immunoregulatory cells, readily obtainable in large numbers without ethical constraints. Amniotic membrane possesses anti-inflammatory and immune-suppressing properties that underlie its clinical use in the treatment of wounds, burns, and in ophthalmic surgery where it is grafted without rejection (2,9,14,33). Human amniotic epithelial cells (AEC) isolated from the membrane suppress T-cell activation in both mixed and mitogen-induced lymphocyte proliferation assays (24,41): AEC are amenable to both allogeneic and xenogeneic engraftment in immune-competent recipients (1,22). The expression of potential mediators of immune suppression, including HLA-G, Fas ligand and TGF-β have been identified in human AEC (13,23,24), which may serve to inhibit immune cell functions to create a microenvironment conducive to allogeneic graft survival.
In the present study we examined the potential of human AEC to modify the immune response to isolated human islets. Specifically, we sought to test the hypothesis that the presence of AEC in close proximity to human islets alters the immediate microenvironment sufficiently to induce a localized immunosuppressive response on invading peripheral blood lymphocytes. A rotational cell culture system (RCCS) (26,27) was employed to bioengineer novel cellular constructs composed of islets and AEC (islet/AEC), the functional and immunological characteristics of which were then investigated under in vitro conditions.
Materials and Methods
Human Islet Isolation
Pancreases from multiorgan donors were supplied by the UK Human Tissue Bank (De Montfort University, Leicester, UK) with the appropriate consent and local Research Ethics Committee approval. A total of seven pancreases (five female, two male; mean age 47.1 ± 3.3 years) were used, with an average cold ischemic time of 10.04 ± 1.25 h. Organs were dissociated by a combination of enzymatic digestion (Liberase HI, Roche Diagnostics, West Sussex, UK) and mechanical agitation, and islets separated from the resulting pancreatic digest using density gradient centrifugation on Ficoll columns as previously described (26,27). Staining with dithizone (500 μg/ml, Sigma Aldrich Ltd, Dorset, UK) was used to assist islet counting and conversion to islet equivalents (IEQ) (28), while trypan blue (0.4% v/v) exclusion confirmed islet cell viability. The islet preparations were seeded at a density of 750–1000 IEQ/ml in Medium 199 containing 100 U/ml penicillin, 100 μg/ml streptomycin, 10 μg/ml amphotericin B (Sigma Aldrich Ltd) supplemented with 10% fetal calf serum (First Link Ltd, Birmingham, UK), and maintained in nonadherent culture (27) for a period of 24 h to allow acclimatization.
Human Amniotic Epithelial Cell (AEC) Isolation
Human amniotic membrane was obtained according to ethically approved protocol and with informed consent from 17 women (mean age, 32.5 ± 1.6 years) undergoing elective Caesarean section. Samples (10 × 10 cm) of amniotic tissue were separated from the chorion layer by blunt dissection (avoiding areas overlying the placenta). The tissue was rinsed three times in phosphate-buffered saline (PBS, Sigma Aldrich Ltd) containing 200 U/ml penicillin, 200 μg/ml streptomycin, and 20 μg/ml amphotericin B, and reduced to small pieces for digestion in 0.25% (w/v) trypsin in Hanks balanced salt solution (HBSS, Sigma-Aldrich Ltd) for 20 min at 37°C. The resulting tissue suspension was passed through a 500-μm mesh to retain larger pieces of amnion, which were subjected to three further incubation cycles with trypsin to liberate all available epithelial cells. Pooled fractions of cell suspension thus obtained were centrifuged at 400 × g for 5 min and the pellets resuspended in RPMI-1640 supplemented with 10% fetal bovine serum (FBS, Sigma-Aldrich Ltd), 100 U/ml penicillin, 100 μg/ml streptomycin, and 10 μg amphotericin B. AEC were seeded at high density in T-75 flasks and cultured at 37°C, 5% CO2, 95% O2 in a humidified atmosphere for 48–72 h, to form a flattened confluent monolayer. In some instances cultures of AEC harvested at confluence by mild trypsinization (0.025% trypsin-EDTA in PBS, Sigma Aldrich Ltd) were resuspended in supplemented RPMI containing 10% DMSO and cryopreserved at −80°C for later analysis of immunomodulatory potential.
Peripheral Blood Lymphocyte (PBL) Isolation
Peripheral blood lymphocytes (PBL) were isolated from buffy coats obtained from nine healthy volunteers through the National Blood Service (NBS, Birmingham, UK) with local research ethics approval. Briefly, buffy coat fractions were resuspended in an equal volume of HBSS and layered onto 12 ml of Histopaque-177 (Sigma-Aldrich Ltd), centrifuged at 700 × g for 30 min (with no brake), and the resulting leucocyte layer harvested using a sterile pastette. The isolated PBLs were washed three times in HBSS, centrifuged at 500 × g for 10 min, resuspended in RPMI-1640 (supplemented as described above), and cultured in uncoated plastic petri dishes at 37°C, 5% CO2, 95% O2 overnight. A portion of the isolated PBLs was incubated with the mitogen phytohemagglutinin (PHA, 5 μg/ml, Sigma Aldrich Ltd) for 24 h prior to use in proliferation assays.
Islet/AEC Coculture: Conventional Static Culture Versus a Rotational Cell Culture System (RCCS)
For coculture studies islet suspensions were adjusted to a density of 500–1000 IEQ/ml and placed under either conventional static culture (CSC) conditions in 90-mm culture plates (NHS Logistics, Alfreton, UK) or in a rotational cell culture system (RCCS) in high aspect ratio vessels (HARVs, Cellon Ltd, Bereldange, Luxembourg) as previously described (27). The cultures were maintained at 30°C in a humidified atmosphere of 95% O2/5% CO2. Once confluent the AEC monolayers were disrupted by mild enzymatic digestion (0.025% trypsin-EDTA in PBS, Sigma Aldrich Ltd) and the resulting cell suspension washed in PBS and introduced to the islet cultures (both CSC and RCCS) at a final density of 1 × 105 cells/ml. The islet/AEC cocultures were maintained under conditions as described above for 72 h. Control cultures consisted of islets seeded at equal density (CSC and RCCS) in the absence of AEC.
Morphological Analysis of AEC Monocultures and Islet/AEC Cocultures Using Fluorescence Immunocytochemistry
For immunocytochemistry isolated AEC and islet/AEC cocultures maintained for 72 h either under CSC conditions or within the RCCS were anchored to glass coverslips and fixed with 4% paraformaldehyde for 30 min at room temperature (RT). Three 10-min washes in PBS were followed by antigen retrieval (0.3% Triton X-100, Sigma Aldrich Ltd) and blocking (10% normal goat serum in PBS for CK19, vimentin, FasL; 10% normal rabbit serum in PBS for insulin, Vector Laboratories Ltd, Peterborough, UK). The AEC or islet/AEC constructs were then incubated with primary antibodies, anti-human cytokeratin 19 (CK19), anti-human vimentin (Dako UK Ltd, Cambridgeshire, UK; 1:100), anti-human insulin (AbD Serotec, Oxford, UK; 1:10), or anti-human Fas Ligand (FasL, CD95L, Sigma Aldrich Ltd, 1:10) for 1 h at RT and at 4°C overnight. Secondary antibody (goat anti-mouse IgG-FITC for CK19, vimentin, and FasL, goat anti-rabbit IgG-TRITC for insulin; Cambridge Biosciences, Cambridge, UK, 1:100) was applied for 3 h at RT. The coverslips were rinsed and mounted in fluorescence mounting medium (Dako UK Ltd) before cell imaging using a Zeiss Axioskop 40 fluorescence microscope equipped with an AxioCam MRc color camera and incorporating Axiovision imaging software (Carl Zeiss, Hertfordshire, UK). Controls involved omission of the relevant primary antibody.
Insulin Secretory Capacity: Static Glucose Challenge
Cultures of islets or cocultures consisting of islets and AEC maintained either under CSC conditions or within the RCCS as described above were assessed for preserved glucose responsiveness. The impact of culture condition on islet function was determined by measuring insulin release in response to glucose under basal conditions viz. in the presence of 1.67 mmol/L glucose in modified HEPES-buffered HBSS comprised of (mmol/L): HEPES (9.9); NaCl (113.2); NaHCO3 (4.1); Na2HPO4 (0.33); KCl (5.36); CaCl2 (0.95); MgSO4.7H2O (0.8); KH2PO4 (0.44), containing 0.2% BSA, pH 7.4, at 37°C, and subsequent to stimulation with high glucose (16.7mmol/L) or a combination of 16.7 mmol/L glucose and 10 mmol/L theophylline according to methods previously described (26,27). The secretory capacity demonstrated by islets maintained under CSC conditions was compared with that seen in islets held within the RCCS and to islets in coculture with AEC under both culture conditions. Response to glucose stimulation was quantified by measurement of insulin in the incubation medium using a commercial ELISA (Diagenics Ltd, Milton Keynes, UK) and expressed as a ratio of insulin secretion under basal conditions (stimulation index, SI). The islets were assessed for insulin secretory capacity at 24 h postisolation and at 72 h after the initiation of the islet/AEC cocultures (viz. 5–7 days postisolation).
Immunomodulation: PBL Proliferation Assay
AEC Monocultures. Confluent monolayers of AEC were dispersed and transferred to 24-well plates at a final density of 5 × 104 cells/well in supplemented RPMI as described above. The cells were allowed to attach and flatten prior to the initiation of PBL proliferation assays. Resting or PHA-activated PBLs were added to each well at equal density (5 × 104/well) for coincubation at 37°C, 5% CO2, 95% O2. Activated PBLs in contact with AEC continued to be cultured in the presence of 5 μg/ml PHA. After 72 h the PBLs were harvested, solubilized (VialightPlus, cell lysis reagent, Lonza Ltd, Wokingham, UK), and analyzed for ATP content using a commercial chemiluminescence assay (Lonza Ltd) according to the manufacturer's instructions. Concentration of ATP per well, measured as relative light units (RLU), is directly proportional to cell number and thus indicative of the proliferative activity of PBLs in culture (34). Results were expressed as a percentage of control (i.e., resting PBLs incubated in the absence of AEC). In selected experiments cryopreserved AEC were rapidly thawed, rinsed in PBS, and seeded as described for the fresh AEC prior to their use in PBL assays as detailed above.
AEC-Conditioned Medium
In a separate set of experiments confluent monolayers of AEC were dispersed and replated in T75 flasks in supplemented RPMI medium as described above. The flasks were left for 72 h without a medium change to allow concentration of putative soluble factors released by the AEC. The resulting AEC-conditioned medium was harvested and centrifuged at 1300 × g to ensure removal of all cells and cellular debris prior to use in PBL proliferation assays. Conditioned medium (0.5 ml) was dispensed to the appropriate wells of a 24-well plate and 5 × 104 resting or PHA-activated PBLs were added, adjusting the total volume to 1.0 ml using standard RPMI medium. Plates were incubated at 37°C, 5% CO2, 95% O2. After 72 h the PBLs were harvested and processed as described above. Results were expressed as a percentage of control (i.e., resting PBLs incubated in the absence of AEC-conditioned medium).
Islet/AEC Cocultures
As the islets and islet/AEC cultures maintained within the RCCS demonstrated superior viability both with regard to morphology (islet/AEC integration) and insulin secretory capacity compared to those held under CSC conditions, these cultures were subjected to PBL proliferation studies. Cells were transferred from the HARVs to 24-well plates (50–100 IEQ or 50–100 islet/AEC aggregates/well). Following a 48-h period to allow attachment the islet or islet/AEC cultures were exposed to either resting or PHA-activated PBLs (5 × 104 cell per well) for a period of 72 h, after which time the PBLs were harvested and analyzed for ATP content as described above.
Statistical Analysis
Statistical differences between the culture conditions in response to insulin secretagogues were assessed by one-way analysis of variance (ANOVA) using islet monocultures maintained under CSC conditions as the control group. Significant differences in PBL proliferation in response to AEC, conditioned medium, islets or islet/AECs were determined using Mann-Whitney U and Tukey's multiple comparison tests (by Rank), with the response of resting PBLs serving as the control. In all comparisons a value of p < 0.05 was considered to be statistically significant. Statistical analysis was performed using SigmaStat software version 3.5 (Systat Software Inc, Chicago, IL, USA).
Results
Morphological and Immunocytochemical Assessment of Human Islet and Amniotic Epithelial Cells Postisolation
The human islet isolation protocol employed in the present study resulted in the harvest of structurally intact islets, which were well cleaved from the surrounding exocrine tissue, as previously reported (26,27). The purity of the islet suspension following Ficoll-assisted separation ranged from 70% to 85%, with islets mostly sized between 100 and 500 μm. Trypan blue exclusion served as an indicator of preserved islet structural integrity.
AEC plated at high density in T75 flasks readily attached and flattened to form a monolayer (Fig. 1A); the vast majority of these cells stained positive for the epithelial cell marker cytokeratin 19 (Fig. 1B). A discreet subpopulation of cells also stained positive for the intermediate filament marker vimentin (Fig. 1C) and a significant number (~30%) expressed Fas ligand (Fig. 1D). Islets held in coculture with AEC under CSC conditions demonstrated a degree of cell association: AEC were overlying islets in some instances (Fig. 2) and more robust, tightly formed cellular constructs exhibiting good integration of the two cell types were achieved by 72-h coculture of islets and AEC within the RCCS (Fig. 3). The vast majority of islets within the RCCS became associated with AEC although, in most instances, the AEC did not form a complete layer.

Morphological characteristics of human amniotic epithelial cells (AEC) in confluent monolayer culture. Phase contrast visualization of isolated human AEC (A). Immunocytochemical localisation of cytokeratin 19 (CK-19) (B), vimentin (C), and Fas Ligand (FasL) (D). Scale bar: 100 μm.
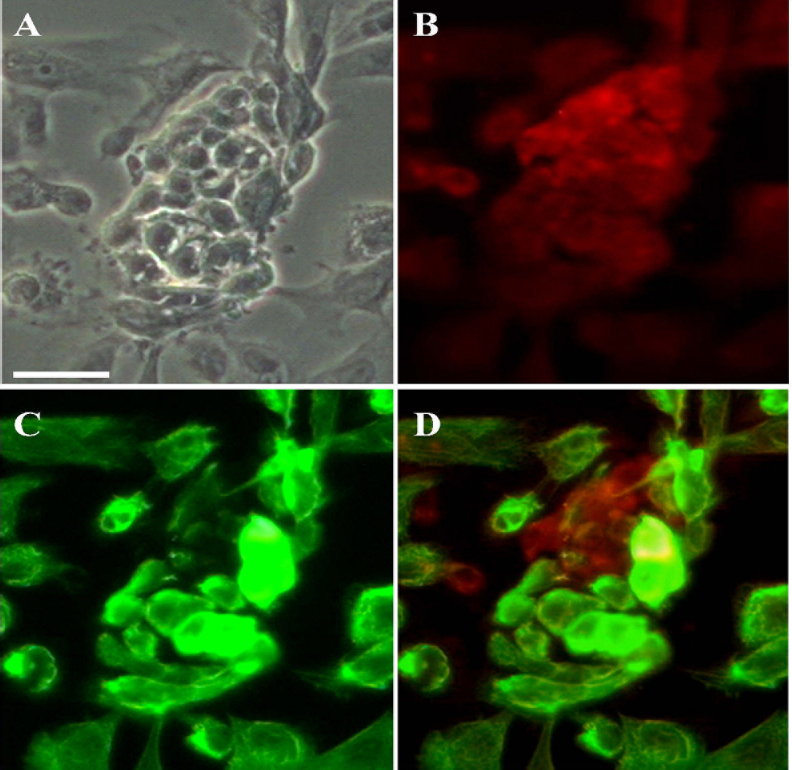
Morphological characteristics of human islet/AEC constructs formed by coculture under conventional static culture (CSC) conditions for 72 h. Phase contrast image of typical cell construct (A). Immunocytochemical localization of insulin (TRITC) (B) and CK19 (FITC) (C). Overlay image showing the spatial interaction of the two cell types (D). Scale bar: 50 μm.
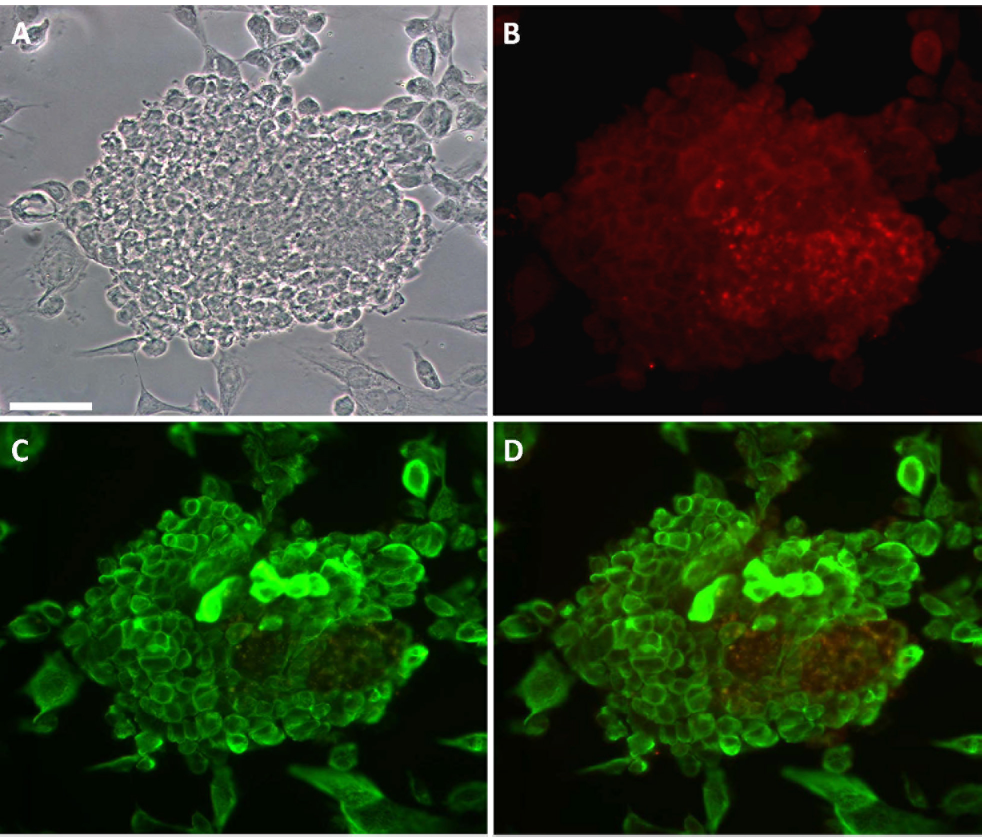
Morphological characteristics of human islet/AEC constructs formed in rotational cell culture (RCCS) over 72 h. Phase contrast image of typical cell construct (A). Immunocytochemical localisation of insulin (TRITC) (B) and CK19 (FITC) (C). Overlay image showing the spatial interaction of the two cell types (D). Scale bar: 100 μm.
Islet Secretory Function at 24 h Postisolation
Islets maintained under CSC conditions for a period of 24 h postisolation demonstrated functional viability as indicated by their response to a glucose challenge. Insulin secretion was consistently increased by 16.7 mM glucose (SI 2.63 ± 0.21) compared with basal release. This was further enhanced by the presence of 10 mM theophylline (SI 3.67 ± 0.34) (Fig. 4).
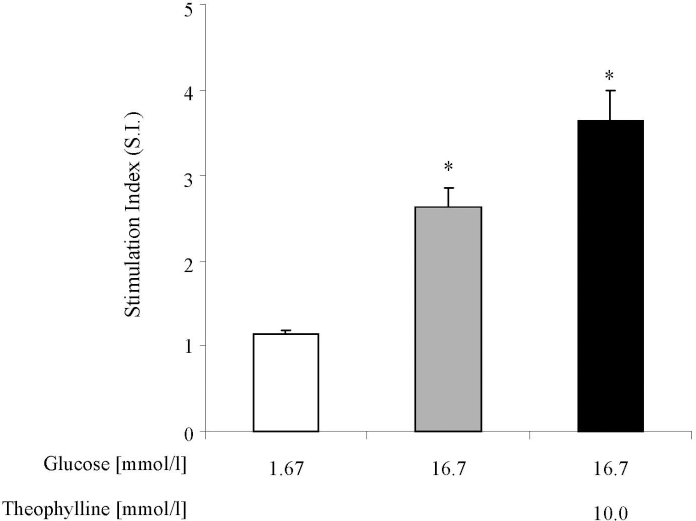
Insulin secretion from isolated human islets in response to nutrient stimulation during static challenge experiments performed 24 h postisolation. Islets were maintained under CSC conditions prior to assessment of secretory function. Insulin release was measured in response to 1.67 mmol/L glucose (basal release), 16.7 mmol/L glucose, and 16.7 mmol/L glucose plus 10 mmol/L theophylline. Results are expressed as the mean ± SEM fold increase in insulin release in response to nutrient stimulation relative to release under basal conditions. n = 7 independent islet preparations. The absolute mean value for insulin secretion under basal conditions was 86.5 ± 17.2 μUml −1 [20 islets]−1 h−1. *p < 0.05 versus basal conditions.
Impact of islet/AEC Coculture Condition on β-Cell Function
Following a 72-h period of culture islet and islet/AEC constructs were subjected to further glucose challenge studies. Preliminary experiments indicated that isolated AEC do not secrete insulin when maintained in either static or rotational culture (data not shown) and were therefore not assessed during this investigation. In the presence of elevated (16.7 mM) glucose, control islets held under CSC conditions throughout the period of the investigation responded minimally in terms of insulin secretion (SI 1.24 ± 0.07), as previously noted (26, 27), although a combination of 16.7 mM glucose and 10 mM theophylline elicited more marked (p < 0.05) insulin secretion (SI 1.53 ± 0.1) (Fig. 5). By contrast, maintenance of islets within the RCCS preserved glucose responsiveness with significant insulin secretion occurring in response to 16.7 mM glucose both in the absence (SI 1.59 ± 0.08; p < 0.05) and the presence (SI 2.49 ± 0.28; p < 0.01) of the potentiator. Coculture of islets with AEC under both CSC conditions or within the RCCS had an apparently beneficial effect on β-cell function, with islets continuing to respond to glucose stimulation (SI 1.65 ± 0.12 and 2.89 ± 0.34 for islets under CSC condition in response to 16.7 mM glucose alone and 16.7 mM glucose plus 10 mM theophylline, respectively; SI 1.83 ± 0.11 and 3.15 ± 0.32 for islets maintained in the RCCS in response to 16.7 mM glucose alone and 16.7 mM glucose plus 10 mM theophylline, respectively) (Fig. 5).

Glucose-stimulated insulin release from human islets (HI) maintained under conventional static culture (CSC) conditions or within the rotational cell culture system (RCCS) either in the presence or absence of human amniotic epithelial cells (AEC) for 72 h. Insulin release was measured in response to 1.67 mmol/L glucose (open bars), 16.7 mmol/L glucose (gray bars), and 16.7 mmol/L glucose plus 10 mmol/L theophylline (filled bars). Results are expressed as the ratio of stimulated insulin release compared to basal, mean ± SEM. n = 4. *p < 0.05, **p < 0.01 stimulated insulin secretion compared to basal release.
PBL Proliferation: Influence of AEC and AEC-Conditioned Medium
PBLs taken from healthy volunteers demonstrated a six- to ninefold stimulation in the presence of 5 μg/ml PHA for a period of 72 h (Fig. 6A, B). Resting PBLs failed to respond on contact with an equal number of AEC or on exposure to AEC-conditioned medium over the same time period. The proliferation of PHA-activated lymphocytes was abrogated by coculture with AEC (Fig. 6A). A similar inhibition to PHA-mediated PBL proliferation was seen in AEC subjected to a period of cryopreservation. Furthermore, AEC-conditioned medium had comparable immunosuppressive activity on PHA-activated PBLs (Fig. 6B).

Modulation of peripheral blood lymphocyte (PBL) proliferation by (A) the presence of human amniotic epithelial cells (AEC), and (B) exposure to AEC-conditioned medium (CM). Resting (r; open bars) or PHA-activated (s, filled bars) human PBLs were maintained in 24-well plates either alone, in the presence of an equal number of human amniotic epithelial cells, or 0.5 ml of AEC-conditioned medium for a period of 72 h. The rate of PBL proliferation following this period was measured using an ATP chemiluminescence assay. Data show the percentage increase above control (resting PBLs) from six individual AEC preps and represents the typical observation in fresh and cryopreserved AEC. *p < 0.05, **p < 0.01 compared to control. †p < 0.01 for PHA-activated PBL proliferation in the presence or absence of AEC/AEC-conditioned medium.
PBL Proliferation: Islets Versus Islet/AEC
Exposure of resting PBL to unmodified human islets that were maintained within the RCCS elicited a marked (p < 0.05) proliferative response (Fig. 7A). By contrast, the presence of AEC attenuated resting PBL proliferation. PHA-stimulated PBL proliferation was sustained on contact with isolated islets, but was significantly (p < 0.01) suppressed when islets were in coculture with AEC (Fig. 7B).

Modulation of peripheral blood lymphocyte (PBL) proliferation by exposure to human islets and human islet/AEC constructs. Resting (A) or PHA-activated (B) human PBLs were maintained in 24-well plates either alone or in the presence of human islets (HI) or islet/AEC constructs for a period of 72 h. The rate of PBL proliferation following this period was measured using an ATP chemiluminescence assay. Data depict the response from four individual human islet and AEC preps and represents the typical observation. *p < 0.01 compared to resting levels. †p < 0.01 for PHA-activated PBL proliferation in the presence or absence of human islet/AEC constructs.
Discussion
This investigation has demonstrated that human amniotic epithelial cells possess innate immunoregulatory properties in vitro as indicated by their ability to suppress mitogen-induced lymphocyte proliferation, thus confirming previous studies (1,24,41). In addition, the outcome of the coculture studies suggests, for the first time, that the immunosuppressive properties of AEC may confer a state of immune privilege in otherwise immunogenic cells. These novel observations are relevant to the potential use of human AEC as an adjunct to cell replacement therapies, such as islet transplantation. Conceivably, the creation of a localized region of immunosuppression might reduce or obviate the obligatory requirement for chronic immunosuppressive therapy.
Notwithstanding their disparate origins, the coculture of human islets and AEC under either conventional static or rotational cell culture conditions resulted in successful physical interaction between the two cell types. As previously reported (27), the RCCS provided a more conducive environment for cellular aggregation, with the formation of robust constructs exhibiting frequent spatial association of the insulin- and CK19-expressing cells and a preserved islet-like morphology. The high aspect ratio vessels (HARVs) are designed to create a microgravity environment with low shear forces permitting a greater degree of cell–cell interaction (38), which may underlie the efficient formation of stable islet/AEC constructs observed in the present study.
The close proximity of AEC to the human islets had no adverse effect on β-cell function. Indeed, the insulin secretion data indicate preservation of glucose sensitivity in human islets maintained in coculture with AEC. This may be compared with islets held alone under CSC conditions, which showed a diminution of glucose responsiveness. In previous studies we have demonstrated a beneficial impact of ductal epithelial cell coculture in preserving islet function (26), apparently due to their ability to provide trophic support to neighboring β-cells (29). AEC are also reported to synthesize and secrete a range of growth factors that may have relevance for the sustained functional viability of islets seen in the coculture model. Of note, mRNA expression of TGF-β, EGF, and KGF, known mediators of β-cell replication (7,12,25) has been reported in intact human amniotic membrane and isolated amniotic epithelial cells (21). Furthermore, dissociated AEC secrete biologically active neurotrophins including BDNF (18), which have been linked to β-cell development and survival (31). It is thus likely that the close association of AEC to islets in this coculture model permits the paracrine release of soluble mediators able to support insulin secretory capacity in the postisolation period with beneficial consequences for long-term β-cell function.
The immunomodulatory capabilities of human amniotic membrane have been studied extensively (15,22,36). Our findings that isolated AEC abrogate mitogen-induced PBL proliferation confirm the results of previously published studies using comparable amnion-derived epithelial cell populations (24,41). Hence, our extrapolation that the immunosuppressive properties of isolated AEC could be manipulated to confer a state of immune privilege on other cells capable of provoking an immune response. The findings of the conditioned medium studies support those of others (15,24,37), and further suggest that AEC secrete immunomodulatory factors at concentrations sufficient to create a region of localized immunosuppression, with the potential to alter the immunogenicity of other cells in their immediate vicinity. Thus, in our mixed islet/lymphocyte reaction sustained proliferation of resting PBL was demonstrated in the presence of unmodified islets, yet those that were closely associated (cocultured) with AEC failed to elicit an allogeneic response. This effect was not dependent on complete encapsulation of the islets by the AEC, further indicative of a role for soluble immunoregulatory factors. Also, the immunomodulatory response to activated (PHA-stimulated) T cells was as robust in the islet/AEC cocultures as in AEC monocultures. Combined, these data suggest that AEC exhibit a potent and generalized immunosuppressive capability, inducing an anti-proliferative response in T cells subjected both to specific and nonspecific antigen challenge.
Studies to identify the soluble factors involved in AEC-mediated immunosuppression and to characterize their T-cell targets are ongoing, yet initial immunocytochemical evidence indicates the potential involvement of Fas ligand (FasL), an immunomodulatory factor associated with naturally occurring T-cell evasion in the testis, eye, and brain (10). Localization of FasL within the placenta and amnio-chorionic membranes is implicated in maternal tolerance developed to the fetus during pregnancy (13,19). Thus, FasL in the AEC cultures raises the possibility of activated, FasL-mediated T-cell apoptosis. It is unlikely that a single mediator is responsible for immune adaptation and, indeed, other soluble factors have been identified within the AEC population, including TGF-β, HLA-G, and IL-10 (23,24), all of which have the potential to affect localized immunosuppression. A number of these mediators could potentially operate in concert to produce a microenvironment capable of sustaining allogeneic tissue by diminution of the T-cell response.
The extrapolation of the immune-neutralizing properties of one cell type to modify the immunogenicity of a cotransplanted cell population has been demonstrated elsewhere. Notably, in the context of islet transplantation the use of Sertoli cells (SC) to create a local milieu conducive to long-term allograft and xenograft survival has been demonstrated experimentally and clinically (16,20,39) and more recently the use of mesenchymal stem cells (MSC) to regulate the immunogenicity of islet allografts has also been reported (5). We propose that immunoprotection could be achieved by the use of AEC, effectively bioengineering a state of immune privilege within the graft tissue promoting the localized release of soluble immunoregulatory mediators. While the widespread clinical use of human SC and MSC would pose certain technical challenges, human amnion is readily available and not subject to the same ethical constraints. Additionally, the present studies suggest that amnion provides an expandable pool of immunomodulatory cells, which are amenable to cryopreservation, readily integrate with isolated human islets, and do so without causing adverse alterations to β-cell viability, cellular transplant volumes, or nutrient exchange. Furthermore, the experimental and clinical use of amniotic membrane is well established (3,9,14,33) and successful engraftment of human AEC without evidence of tumorigenesis has been reported (2). Future work will seek to demonstrate that the immune-evasive properties of islet/AEC constructs are sustained in vivo and define how such a bioengineered approach to immune suppression could be adapted for clinical use.
Footnotes
Acknowledgments
We gratefully acknowledge the financial support of The Sir Halley Stewart Trust, The James Tudor Foundation, The Rowlands Trust, The Eveson Charitable Trust, The South Warwickshire Diabetes UK Voluntary Group, and Worcestershire Acute Hospitals NHS Trust R&D.
