Abstract
The advent of Highly Active Antiretroviral Therapy (HAART) has radically changed incidence characteristics and prognosis of HIV-positive patients affected by lymphomas. At this time there is consensus in the literature that, in first line, HIV-positive patients should always be treated with curative intent preferentially following the same approach used in the HIV-negative counterpart. On the contrary, an approach of salvage therapy in HIV-positive lymphomas is still a matter of debate given that for a wide range of relapsed or resistant HIV-negative Hodgkin's disease (HD) and non-Hodgkin lymphoma (NHL) patients, autologous peripheral or allogeneic stem cell transplantation are among the established options. In the pre-HAART era, therapeutic options derived from pioneering experiences gave only anecdotal success, either when transplantation was used to cure lymphomas or to improve HIV infection itself. Concerns relating to the entity, quality, and kinetics of early and late immune reconstitutions and the possible worsening of underlying viroimmunological conditions were additional obstacles. Currently, around 100 relapsed or resistant HIV-positive lymphomas have been treated with an autologous peripheral stem cell transplantation (APSCT) in the HAART era. Published data compared favorably with any previous salvage attempt showing a percentage of complete remission ranging from 48% to 90%, and overall survival ranging from 36% to 85% at median follow-up approaching 3 years. However, experiences are still limited and have given somewhat confounding indications, especially concerning timing and patients' selection for APSCT and feasibility and outcome for allogeneic stem cell transplant. Moreover, little data exist on the kinetics of immunological reconstitution after APSCT or relevant to the outcome of HIV infection. The aim of this review is to discuss current knowledge of the role of allogeneic and autologous stem cell transplantation as a modality in the cure of HIV and hemopoietic cancer patients. Several topics dealing with practical aspects concerning the management of APSCT in HIV-positive patients, including patient selection, timing of transplant, conditioning regimen, and relapse or nonrelapse mortality, are discussed. Data relating to the effects of mobilization and transplantation on virological parameters and pre- and posttransplant immune reconstitution are reviewed. Finally, in this review, we examine several ethical and legal issues relative to banking infected or potentially infected peripheral blood stem cells and we describe our experience and strategies to protect positive and negative donors/recipients and the health of caretakers.
Introduction
HIV-positive subjects have an increased risk of developing lymphomas when compared with the general population (39,129). Aggressive lymphomas with a poor clinical outcome account for the vast majority of the cases (71,87,120). The advent of Highly Active Antiretroviral Therapy (HAART) has profoundly modified the characteristics, management, and prognosis of HIV-associated lymphomas, making aggressive therapy, including autologous or allogeneic stem cell transplantation, a fascinating tool to treat patients (71,86). In fact, in NHL-HIV (non-Hodgkin lymphomas-HIV) with a favorable viroimmunological profile, it has already been demonstrated that standard or infusional chemotherapy regimens such as CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), CDE (cyclophosphamide, doxorubicin, etoposide), or EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) can be safely and concomitantly administered with HAART. Outcomes in terms of hematological and extrahematological toxicity, percentage of complete response (CR), event free survival (EFS), and overall survival (OS), closely resembled those of HIV-negative NHL cases (86, 101,116,120,122,132). Similar results have been achieved using ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) or Stanford V associated with HAART in HIV-positive Hodgkin's disease (HD) (16,121,126). Thus, the majority of authors agree that fit, not profoundly immunodeficient (i.e., with more than 100 CD4/mm3) HIV-positive lymphoma patients, assuming an effective HAART, should be treated in first line with curative intent similar to the HIV-negative counterpart; aggressive chemotherapy should be applied whenever it is appropriate according to histology, stage, and prognostic risk factors (16,83,86,88,116).
Additional concerns still impact combining anti-CD20 monoclonal antibody (Rituximab) with chemotherapy given the potential overlapping of side effects in the immunological asset (120,123,124). Persistent neutropenia, development of deep hypogammaglobulinemia, higher risk of death due to early or late bacterial and/or opportunistic infections, including the reactivation of viral infections such as hepatitis C or B, herpes simplex or zoster, parvovirus or JC virus, are still matters of debate. Benefit in terms of response has been shown by Spina and colleagues in adding rituximab to CDE (122), by Wilson (heading the US NCI Lymphoma Group) and Levine et al. in experimenting anti-CD20 associated with EPOCH (72,73), and, more recently, by Ribera and colleagues in associating rituximab to CHOP (101). Implementation of vigilance during chemotherapy and follow-up, amelioration of antifungal and bacterial prophylaxis, and prompt diagnosis of infection may improve the efficacy of rituximab, at least in the outcome of HIV-positive NHL showing more than 50 CD4/mm3 (124).
Despite dramatic improvements obtained in first line chemotherapy, patients with a relapsed or resistant HIV-positive lymphomas still have little chance of survival (85,134). Before experimental introduction of autologous peripheral stem cell transplantation (APSCT) in the HIV-positive setting, studies focusing on salvage therapy in the HAART era were rare and disappointing (17,125). The most representative of these studies compared the efficacy of two high-dose cytosine-arabinoside and cisplatin-based regimens: ESHAP (etoposide, cisplatin, high-dose aracytine, methylprednisolone) and DHAP (dexamethasone, high-dose cytarabine, cisplatin), in 26 refractory or relapsed NHLs in HIV-positive patients (17). Partial response (PR) or CR were obtained in 1/13 and 7/13 cases treated with DHAP and ESHAP, respectively. Unfortunately, the median OS with the best treatment was only 7 months.
Clinical Aspects of Apsct
In the HIV-negative population, APSCT is an established salvage therapy for a wide range of relapsed or resistant HD and NHL (108,114). In the HIV-positive population, reservations were raised from unsuccessful application of such an aggressive therapy in the pioneering experience of the pre-HAART era, especially when bone marrow rescue was used (41). Poor results were also registered in the allogeneic setting (6,27,46,54,130). With the introduction of HAART in 1996, the overall survival of patients with an advanced HIV infection dramatically improved (31–34). Hence, in the late 1990s, enthusiasm returned for reexploration of more aggressive salvage therapy in HIV-positive cases.
From 2000 to 2008, several authors investigated the feasibility and utility of APSCT in HIV patients with HD or NHL, stating that it represents a successful salvage therapy even in HIV-positive lymphomas (43,66, 97,113). Published data compared favorably with any previous salvage attempt, but experience is still limited, especially when APSCT has been explored in small and highly selected series of HIV-positive lymphomas, ranging from 14 to 68 cases. Presently, the majority of the data have been obtained from retrospective studies documenting mixed HD and NHL histology. In these studies the timing of APSCT (CR vs. first or subsequent relapse or resistance), exclusion and inclusion criteria to enroll patients (CD4 < or >100/mm3, presence of OI, viral control, organ dysfunctions), and conditioning regimens are heterogeneous (32,66,82,97,113,127). Table 1 reports the most important data obtained from the six largest studies available on APSCT in HIV positive lymphomas.
Summary of Papers Evaluating Autologous Peripheral Stem Cells Transplantation (APSCT) in HIV-Related Lymphoma
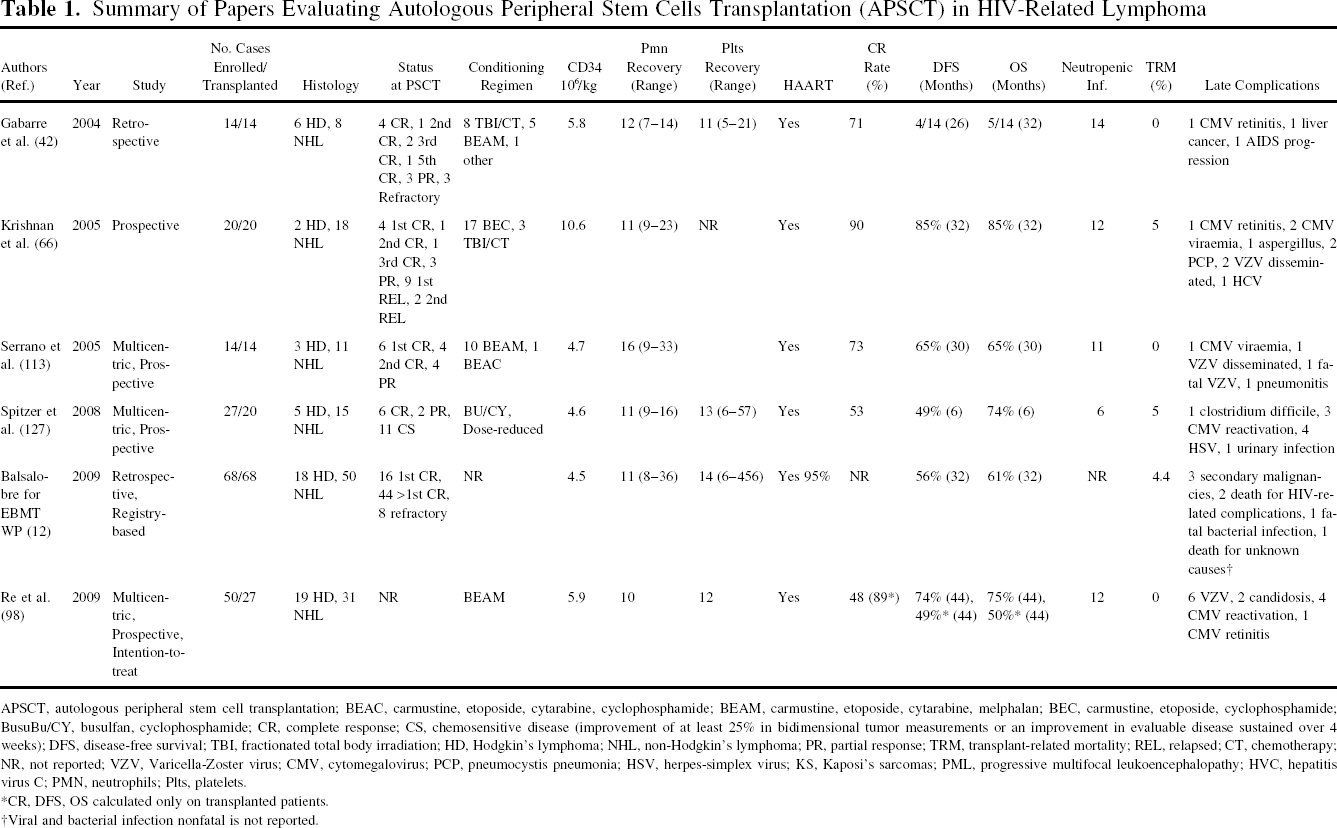
APSCT, autologous peripheral stem cell transplantation; BEAC, carmustine, etoposide, cytarabine, cyclophosphamide; BEAM, carmustine, etoposide, cytarabine, melphalan; BEC, carmustine, etoposide, cyclophosphamide; BusuBu/CY, busulfan, cyclophosphamide; CR, complete response; CS, chemosensitive disease (improvement of at least 25% in bidimensional tumor measurements or an improvement in evaluable disease sustained over 4 weeks); DFS, disease-free survival; TBI, fractionated total body irradiation; HD, Hodgkin's lymphoma; NHL, non-Hodgkin's lymphoma; PR, partial response; TRM, transplant-related mortality; REL, relapsed; CT, chemotherapy; NR, not reported; VZV, Varicella-Zoster virus; CMV, cytomegalovirus; PCP, pneumocystis pneumonia; HSV, herpes-simplex virus; KS, Kaposi's sarcomas; PML, progressive multifocal leukoencephalopathy; HVC, hepatitis virus C; PMN, neutrophils; Plts, platelets.
CR, DFS, OS calculated only on transplanted patients.
Viral and bacterial infection nonfatal is not reported.
APSCT Timing, Outcome, and Patient Selection
Compared to standard salvage therapy, a remarkably high percentage of success in obtaining CR, ranging from 53% to 90%, was uniformly observed in all six large series. Conversely, long-term OS and DFS had a higher variability, ranging from 32% to 85% (Table 1).
As in HIV-negative lymphomas, long-term outcomes were strictly linked to lymphoma characteristics: best outcomes were observed in the Krishnan series in which enrolment was limited to patients with well-controlled HIV infection. In fact, patients were transplanted if they failed to enter a CR after a standard dose regimen or had a high-risk NHL in first CR or had a chemosensitive relapse, while no cases with heavy pretreatments were enrolled (66).
Histology was also relevant in determining outcome. Poorer DFS caused substantially by a high percentage of relapse mortality was reported by Gabarre et al., who enrolled patients with more advanced and multiple relapsed disease and also included cases with highly aggressive histology such as Burkitt's and primary effusion lymphomas (PEL) (42). In addition, the impact of histology and disease status on outcome was emphasized in the EBMT (European Bone Marrow Transplantation) retrospective analysis performed by Balsalobre et al. showing that cumulative incidence (CI) of relapse was dependent on an NHL histology other than diffuse large B cell lymphoma, although this was also significant to the use of >2 pre-APSCT chemotherapy lines and to not being in CR at transplantation (12). Patient selection is an additional important factor in determining outcome, as suggested by the data from Spitzer et al., who reported a 53% rate of CR by transplanting only 20 of the 27 enrolled cases (127). Similar conclusions can be drawn from an update of the Italian Cooperative Group on AIDS and Tumors (GICAT) and from our experience at the National Cancer Center of Aviano, showing that the median OS of the entire population enrolled was 33 and only 7 months, respectively, (82,96). Nevertheless, in both studies, OS in the population that actually received transplant was 75% and 61%, respectively, at a median follow-up of more than 40 months.
The GICAT study prospectively enrolled 50 HIV-positive lymphoma patients on an intention-to-treat study with selection based on performance status (PS), CD4 count, presence of active or recent opportunistic infections (OI), no organ dysfunction or active central nervous system (CNS) disease. In this series, transplant was performed only in chemosensitive lymphomas defined at the end of the chemotherapy debulking program (i.e., in 27/50 patients) (96,97). Unlike the former, we enrolled and treated, aiming to transplant, all cases referred to the Aviano Institute for a salvage treatment excluding only cases with Burkitt histology or active CNS disease. No case was excluded from treatment and analysis based on poor risk defined by poor viroimmunological assets (less than 50 CD4/mm3), presence of concomitant illnesses leading to organ dysfunction, or history of recent/active OIs. This poor risk population that did not meet predesigned criteria to enter the Italian GICAT protocol was approximately 2/3 of the entire population of HIV-positive lymphomas that came in our Institute for salvage therapy from 2001 to 2007. This population substantially was responsible for the low median OS (7 months) observed in the entire population in contrast to an OS of 61% at a median follow-up of 44 months observed in the population (24/50 cases) that actually reached transplant (82).
In summarizing information on the outcomes currently reported, it could be stated that APSCT should certainly be considered the most successful salvage therapy in HIV-positive lymphomas showing relapse or having achieved only a partial response. This approach could be safety applied to approximately 50% of HIV-positive lymphomas with relapsed or refractory disease. A different strategy including tailored chemotherapy regimens monoclonal antibody or new drugs should be evaluated for the remaining substantial fraction of patients unable to sustain an aggressive treatment.
As documented by Krishnan et al., a low transplant-related mortality and a high probability of success may be obtained by considering transplantation as early as possible after the demonstration of limited chemosensitivity (66).
Inclusion of APSCT in first line therapy of poor risk NHL still remains a question of debate. Nevertheless, it must be emphasized once more that long-term survival and low mortality observed in cases selected for their favorable viroimmunological profile and shorter period of pretreatment chemotherapy are certainly very exciting and encouraging in open, new controlled trials exploring this matter.
Conditioning Regimens and Toxicity
A few studies, including our experience, considered more than 10 cases of transplanted patients and were therefore analyzed in this review for toxic events and conditioning regimens (12,32,42,66,82,98,113,127). Among these studies, the EBMT Lymphoma Working Party performed the largest registry-based analysis summarizing the European experience on APSCT in HIV-positive lymphomas, including 68 patients treated in 20 European institutions (12). Remarkable data were reported by Diez-Martin et al., who described the first comparative analysis between HIV-positive lymphomas and a matched cohort of HIV-negative lymphoma patients. This retrospective EBMT study included 53 patients (66% non-Hodgkin and 34% Hodgkin lymphomas within each cohort) (32).
Table 1 reports the conditioning regimens described in the literature, which were mostly chemotherapy based.
A dose-adjustment protocol was applied in two studies; in the Aviano experience, a dose reduction from 25% to 50% was applied whenever an organ dysfunction was registered and in the Spitzer et al. study as an appropriate conditioning regimen (82,127). Two studies used a total-body irradiation regimen that was given to 3 and 8 cases, respectively (42,66). The EBMT registry reported a nonrelapse mortality of 7.6% that was mainly related to bacterial infections (4 /5 cases reported) (12).
In our experience, nonrelapse mortality in the first 100 days from transplant was slightly higher (4/24, 16.7%) and always occurred in the context of a preexisting organ dysfunction determining a Sorror's HCT-CI score >2 (Hemopoietic Cell Transplantation-specific Comorbidity Index) (82,119). Two anecdotal cases of toxicity related to treatment have been described. One case of transplant-related mortality (5%) regimen related was reported by Spitzer et al., who observed a fatal veno-occlusive disease despite using a dose-adjusted conditioning regimen based on BU/CY (busulfan/cyclophosphamide) (127). A second case of fatal toxicity due to conditioning regimen accounting a transplant related mortality of 5% was observed by Krishnan et al. in the oldest transplanted case: a 68-year-old patient died of a cardiomyopathy and renal failure at +22 days (66,127).
In summary, published studies show that, in not profoundly immunodeficient well-fit cases, fatal toxic events at 3 months did not exceed 5%, which is not substantially different compared to HIV-negative cases (12,66,97,113,127).
Peripheral Blood Stem Cell (PBSC) Harvest
Despite initial caution given the presence of HIV myelodysplasia, no relevant alerts were registered on hemopoietic mobilization and harvest procedures (12,66, 97,113,127). However, to avoid additional negative effect on myelopoiesis, a change in HAART therapy avoiding zidovudine when APSCT is planned may be necessary (66).
Because stem cell transplant is among the most advanced clinical applications in transfusion medicine, autologous peripheral blood stem cell (APBSC) collection, processing, and storage involves technical, legal, and ethical considerations proper to autologous blood donation/transfusion. In fact, in attempt to satisfy patients' requests, transfusion medicine centers may put other patients at risk of acquiring a blood-borne infection by processing and banking APBSC that may harbor serious infectious agents. We examined the ethical and legal issues related to the banking of infected and potentially infected APBSC, and describe our hypothesis on how transfusion services may deal with them.
Ethically, the decision that favors maintaining known infectious APBSC for transfusion into the autologous donor/patient is premised on strong protection of the patient's autonomy and self-determination—the right of each patient to choose the type of therapy that reflects that patient's preference and values (13). Hospitals that deny patients access to APBSC programs because the donated autologous units are infectious may also be subjected to public criticism for violating individual rights on the ethical principle of justice, namely that HDC programs should be available to all eligible patients, irrespective of their infectious state. However, whatever course of action a hospital transfusion service selects, it is prudent that the ethical, legal, and risk management consequences be thoroughly evaluated and that an appropriate protection for all patients and healthcare workers should be established.
In our institute, the study protocol obtained approval from the Ethics Committee of the Centro di Riferimento Oncologico-IRCCS Aviano and, accordingly, all patients sign an informed consent form.
In its 2004 directive, the European Commission enumerated deferral criteria for autologous donors that included history of HBV, HCV, HIV, and human T-lymphotropic virus 2 infections (26). However, member states were allowed to “establish specific provisions for autologous donations by such persons.” In Italy, current legislation requires that the labeling of autologous blood collections specify both that the donation was made autologously and that the unit will be used for autologous purposes only. Further to concerns for privacy, when an autologous unit has a test result reactive for the above-mentioned infectious agents, the transfusion service provides that an additional “Biohazard” label may be applied (instead of listing the agent for which the unit is reactive) (26). According to European and Italian laws concerning technical requirements for blood and blood components, we developed a specific policy regarding PBSC collection, processing, storage, and transfusion.
All procedures for HIV patients are routinely performed in separate rooms, with individual use instruments, and by using appropriate biosafety systems (masks, sterile white coats, and sterile gloves). Controlled cleaning procedures are applied for all apparatuses and environments used. PBSC are harvested by leukapheresis and a femoral venous access to provide high flow rates. A short-term femoral venous double-parallel-lumen catheter remains in situ for the days needed to reach the target number of CD34+ cells. For each procedure, 12 L of whole blood is processed. This large volume apheresis is due to the fact that HIV patients are CD34+ cell poor mobilizers. An anticoagulant ACD-A solution is used; when needed, intravenous calcium is administered to minimize citrate toxicity. Apheretic products from HIV patients are routinely processed using standard aseptic procedures in laminar flow cabinets placed in separate clean rooms.
A cryopreservant solution composed of 10% sterile apyrogen DMSO (dimethyl sulfoxide) and 0.8% albumin in physiologic solution is mixed with an equal volume of leukapheresis product to reach the concentration of 5% DMSO in the final freezing mixture. All procedures are performed by using adapted safety systems to protect both operators and products from contamination. Samples for sterility and vitality testing (quality control) are obtained after addition of the freezing mixture and before the products are divided into multiple bags.
Apheretic products are cryopreserved in single poly-(ethylene co-vinylacetate) plastic bags. Each sterile bag, upon double sealing, is placed into another Teflon bag. This second bag is then sealed and identified through application of a bar code label. The first inner bag keeps conditions sterile. Entry/exit luer-lock connectors are used to prepare samples, mix solutions, and to split apheretic products into cryocyte bags. Double bags containing PBSC at the appropriate cell concentration suspended in cryopreservant solution are placed in a controlled-rate freezing device. The different steps of the freezing program are controlled using a probe inserted into a bag containing a buffy coat sample from a normal donor mixed with the same cryopreservant solution. In order to induce crystallization, the apparatus operates with an automatic method and cools the chamber down to about −50°C by modulating different nitrogen quantities. This event forces the supercooling phase of the product and induces the peak of crystallization (69).
Distinct freezing and storage apparatuses are used for HIV patients. After controlled-rate freezing, the double bags are placed into a preformed, prechilled aluminum cassette in the storage freezer in which liquid nitrogen is contained in the outer jacket. This cryogenic system provides a minimal interior temperature gradient: from −188°C at the top to −195°C at the bottom of the storage area (1).
Before thawing, one bag at a time is double checked for integrity and patient data using Blood Bank Operational Software (regional network). Blood bar-coding identification systems are available to minimize the rate of mistransfusion.
The bags are then thawed using a specific dry device to reduce the risk of bags and access lines being bacterially contaminated, as well as of identification labels detaching. Immediately after thawing, the product is given to the Patient Care Unit for infusion. Our APBSC processing method, based on a double bag and storage freezer without liquid or gas phase nitrogen in a cryogenic chamber, noticeably reduces bag rupture rate, ranging from 1.06% to 9.6%, as reported in the literature (61,81), to 0.24% and, consequently, decreases biohazard. Furthermore, this processing method leads to more than 95% stem cell viability after thawing (a schematic flow-chart of collection, storage, and distribution of APBSC for HIV patients is summarized in Fig. 1). These results highly recommend the PBSC processing methods described in this article, to prevent the risk of infection and improve quality in hemopoietic stem cell transplantation (1).

Schematic flow-chart of collection, storage and distribution of APBSC: different pathways for HIV patients. The present flow-chart is a visual representation of the sequence of our processing/working map applied to noninfected or HIV-infected patients. A completed flow-chart shows our clinical and laboratory strategies into a plan from which we can work out the details of what each procedure will be applied in separated pathway.
PBSC Engraftment
No relevant alerts were registered as concerns the hemopoietic engraftment (12,66,85,97,113,127,134). Granulocytes promptly recovered in all cases at a median range of 10 to 16 days. Platelets rarely failed recovery and were self-supporting at a median range of 11 to 14 days: as observed in the HIV the negative cases, the lower the number of reinfused CD34, the longer the time to engraft (Table 1). Sporadic cases with a partial or very late platelet recovery were recorded in the majority of series (12,66,97,113,127).
In order to understand whether HIV infection may influence the time to recovery, Serrano et al. compared PBSC engraftment after APSCT in a small group of HIV-negative or HIV-positive cases treated in their institute (112). By matching HIV-positive cases and negative controls for lymphomas characteristics, they showed that HIV-positive patients had short but significant delay in neutrophil and platelet recovery, possibly resulting from heavier bone damage due to concomitant administration of HAART or to recurrent reactivation of viral infections, such as transient cytomegalovirus (CMV) viremias requiring myelo-toxic drugs (112).
A retrospective study of the EBMT Lymphoma Working Party reported the larger comparative analysis between HIV-positive lymphomas and a matched cohort of HIV-negative lymphoma patients (12). It included 53 patients (66% NHL and 34% HD) within each cohort. Both groups were comparable except for the higher proportion of males, mixed cellularity HD, patients receiving growth-colony stimulating factors (G-CSF) prior to engraftment, and the lower proportion of total-body irradiation-based conditioning within the HIV-positive lymphoma cohort. Incidence of relapse, overall and progression-free survival was similar in both cohorts. A higher nonrelapse mortality within the first year post-APSCT was observed in the HIV-positive lymphoma group (8% vs. 2%), mainly due to early bacterial infections, although it was not statistically significant and had no influence on survival. Although neutrophil engraftment was similar in both groups, G-CSF was more frequently used within the HIV-positive lymphoma cohort. Regarding platelet engraftment, a lower CI and a mild delay were observed within the HIV-positive lymphoma cohort, which might be related to different factors such as posttransplant G-CSF use, HAART combination, or bone marrow damage due to chronic HIV infection.
HAART Coadministration During APSCT
Despite initial caution in using HAART in association with several chemotherapeutic agents, such as ones contained in the CHOP regimen, due to a higher frequency of mucositis and neurotoxicity, no conclusive data are presently available on the potential toxicity of chemotherapeutic drugs used concomitantly with HAART, even in the conditioning regimen (131). Significant nausea, mucositis, and grade 1–3 (WHO scale) hepatic toxicity were often registered with frequencies similar to HIV-negative cases (12,66,97,113,127). Nevertheless, in HIV-positive patients, gastrointestinal toxicities or mucositis were the main problems causing dyscontinuation of HAART (66).
Secondary Malignancies
The EBMT Lymphoma Working Party analysis reported three posttransplant malignancies in two cases out of 68 transplanted patients. One patient had an in situ epithelioma at +9 months and a myelodysplasia at +4 years and a second patient had kidney adenocarcinoma at +3 years. No patients showing a secondary malignancy received total-body irradiation during the conditioning regimen (12). Gabarre et al. reported one case of death at +25 months from undifferentiated liver cancer in absence of serological markers of hepatitis B or C (42). Krishnan et al. provided additional data on HIV-positive transplanted patients at the 11th International Conference on Malignancies in AIDS and Other Acquired Immunodeficiencies, reporting the development of a treatment-related myelodisplasia in 1/32 transplanted cases (67).
APSCT in Pediatric Patients
Two cases were published on the application of APSCT in children with lymphomas and HIV infection (36,65). Fluri et al. successfully treated a 13-year-old patient with a refractory Burkitt's lymphoma at second relapse (36). Rituximab was included in the debulking chemotherapy program and in the BEAM (carmustine, etoposide, cytosine-arabinoside, melphalan) regimen. A positive/negative selection was applied to the graft. A severe Escherichia coli infection occurring on day 0 and a peri-oral herpes simplex were successfully treated with meropenem and acyclovir/valcyclovir. Monthly immunoglobulin administration was required in pre- and posttransplant phases. Hemopoietic engraftment was attained at +9 and +26 with >500/mm3 neutrophils and >20,000/mm3 self-supporting platelets, respectively. The clinical and radiological evaluation demonstrated a continuous complete remission at +26 months.
Allogeneic Hematopoietic Stem Cell Transplantation
Experience in the use of allogeneic hematopoietic stem cell transplantation is even more limited and no definitive conclusion can be established on feasibility and indications. In 2006, the Center for International Blood and Marrow Transplantation Research (IBMTR) reported 27 HIV-positive cases who underwent allogeneic transplant from 1986 to 2003 (48). The reasons for transplant included hemopoietic cancers and aplastic anemia but also attempts to ameliorate HIV infection itself. Stem cell donors were mainly HLA-identical siblings (70%), while 19% had a syngeneic donor and only three cases had an HLA-matched unrelated donor (MUD). The origin of CD34 was mainly bone marrow (85%) and only 15% PBSC. Only recently, one case transplanted with allogeneic stem cells obtained from cord blood has been reported (48). In the IBMTR registry, 21/27 (78%) cases had a transplant before 1996 and before the introduction of HAART. Global probability of survival at 2 years was 22%. Nevertheless, in the six cases transplanted after 1996, four survived compared to 2 out of 21 receiving transplant before the HAART era.
A number of successful treatments of cancers using allogeneic hemopoietic stem cell transplantation (HSCT) were described more recently as case reports (23,136). With a myeloablative regimen, Schlegel et al. (107) and Campbell et al. (23) successfully cured two cases of chronic myeloid leukemia. Sorà et al. (118) and Wolf et al. (136) were successful in treating, respectively, a case of acute myeloid leukemia and a case of severe aplastic anemia transplanted from a MUD. With a reduced intensity conditioning regimen and allogeneic stem cell rescue, Kang et al. (59) and Bryant et al. (22) treated three cases of HIV-positive lymphomas, obtaining two long-term survivals. Especially in the allogeneic setting, the management of HAART and consequent viroimmunological reconstitution is a crucial matter. As for APSCT, mucositis and vomiting or nausea appear as major problems that cause HAART discontinuation. In contrast with APSCT, where discontinuation of HAART mainly caused a transient and asymptomatic viral load increase, a febrile syndrome resembling an acute HIV infection was reported from Sorà et al. (118) and from Wolf et al. (136) at HAART discontinuation during allogeneic hematopoietic and lymphopoietic recovery. Febrile syndrome corresponded to viral load increase. No definitive data are now available on the benefit obtained in the viroimmunological asset after an allogeneic transplant.
Immunological and Virological Aspects of Apsct
Pretransplantation Immunity
At diagnosis and before transplantation, the immune system of HIV-positive patients with lymphoma (HIV-positive lymphomas) is already irreversibly damaged by HIV disease itself, by previous chemotherapeutic regimens, and, possibly, by additional ill-defined immunopathogenetic mechanisms related to the tumor itself (35,62,79,106). In fact, the preexisting HIV infection induces quantitative as well as qualitative alterations of the immune system by causing a depletion of CD4 T cells through complex mechanisms, involving exhaustion through immune activation, dysregulation of apoptosis, syncytia formation, and direct destruction of infected cells (35). A reactive expansion of the CD8 T-cell counterpart, alterations in the composition of T-cell subsets and a general reduction in the T-cell repertoire are other distinctive hallmarks of HIV infection (25). The majority of HIV-infected patients show a poor HIV-specific proliferative or cytotoxic response, although these abnormalities are partially reversible during HAART (51). This virus also affects innate immunity by reducing the number and functions of cellular subsets, such as peripheral blood dendritic cells and NK cells and by altering immune functions mediated by extracellular components (34,70). In addition, HIV causes a profound perturbation of the cytokine networks; in particular the virus impairs IL-2 (interleukin-2) and IL-12 secretion, while increasing the production of IL-4, IL-10, and of proinflammatory molecules such as IL-6 or TNF-α (tumor necrosis factor-α) (35,102,111). Chemokines are a heterogeneous family of chemoattractive cytokines playing an important role in the regulation of immune response and homeostasis (3). In the last few years, the unexpected correlation between the chemokine system and HIV has produced fundamental advances in the knowledge of AIDS pathogenesis (76). To initiate infection, HIV interacts with two types of cellular receptors: CD4 and a second chemokine receptor such as CCR5 or CXCR4 (76). The natural course of HIV disease is characterized by the emergence of CXCR4 or CCR5 tropic viral strains that target different cellular subsets expressing the respective co-receptor (47). Furthermore, HIV induces, in lymphoid organs and in the peripheral blood, a sustained increase in the production of chemokines such as RANTES, MIP1–a, and MIP1-β (76,140). Besides their known role as HIV-suppressive factors, the chemokine/chemokine receptor axis also mobilizes hematopoietic stem cells (HSC) more rapidly than with G-CSF (92); collectively, the present data suggest that pathological factors influencing the homeostatic regulation of the chemokine system may have important effects both on HIV disease and on stem cell transplantation procedures (93).
The lymphopenia observed in HIV-positive lymphomas before transplantation has peculiar characteristics. First of all, because CD4 T lymphocytes are depleted by the virus itself and/or by previous chemotherapeutic (CT) regimens, CD4 lymphopenia is of severe grade, with absolute numbers often <100 cells/mm3 (42,95,97). Although viral-dependent immunopathogenetic effects are probably the most important causes of lymphodepletion in HIV, it may be conceivable that antineoplastic therapies also may contribute significantly to immune derangement. In fact, several papers by Mackall et al. have demonstrated, in the HIV neoplastic population, that CD4 cells are particularly sensitive to CT-mediated destruction and that they recover slowly at the end of therapy (77–79), probably because, as suggested by Foulds et al. (37), of their reduced constitutive proliferative capacity compared to CD8 T cells. It is not clear at the moment whether the severe CD4 lymphopenia observed in these patients results from the sum of the two independent hits by CT and HIV, from a viral-dependent increase in the susceptibility of CD4 T cells to damage by cytotoxic agents, or from other unknown mechanisms. Obtaining this information is essential to plan appropriate therapeutic strategies enhancing CD4 T-cell numbers and functions.
While in routine clinical practice the absolute CD4 T-cell counts constitute an appropriate surrogate marker to monitor HIV disease, quantitative measurements do not reflect the heterogeneous nature of this population, which may have important consequences on the behavior of HIV disease and on the mechanisms driving immune reconstitution. Therefore, more sophisticated phenotypic and functional assays are needed to recognize the different CD 4 T-cell subsets having peculiar roles in the regulatory or effector phases of the immune response, such as naive, central memory, effector memory, or Th1/Th2 cells (104,137,142). Several recent papers suggested a role for different CD4 and CD8 T-cell subsets in the immune response and in disease progression in patients having HIV infection and/or lymphomas (63,80). Current data suggest that, before transplantation, naive CD4 cell numbers in HIV-positive lymphomas are very low (i.e., usually below 20–40 cells/mm3), while the reduction of the central and effector “memory” subsets is less severe, probably due to the chronic antigenic stimulation taking place during HIV infection that preferentially boosts their proliferation and expansion (14,95,99).
Compared to CD4 T cells, the CD8 T-cell subset has a completely different behavior. CD8 T-cell numbers are mostly increased in the HIV-positive lymphomas subjects, while in the HIV-negative population with lymphomas CD8 T-cell numbers are generally low or in the normal range (14,95,99). However, the quantitative differences in CD8 T cells between HIV-positive lymphomas and HIV-negative lymphoma patients are only a part of the picture, because CD8 T cells from HIV-positive lymphomas are phenotypically and functionally different from CD8 T cells in HIV patients with lymphoma. These differences are mostly due to the preexisting chronic viral infection that promotes, in CD8 T cells, the overexpression of several inhibitory or “aging” receptors (PD-1, CD57), the downregulation of membrane receptors involved in costimulation pathways (CD28), the altered expression of genes involved in cellular functions, and profound metabolic and bioenergetic deficiencies. These findings define a novel cellular subset, the “exhausted” T cells, that are considered different from anergic cells, but similar to the mechanism during physiological aging (8,90,91,135).
A reduction in thymic function is one of the major mechanisms responsible for the quantitative immunological abnormalities present in HIV-positive lymphoma patients. The development of lymphomas affects thymic function independently from the histopathological characteristics of the tumor and from the type of chemotherapeutic treatment and worsens the well-known HIV-dependent thymic defect (95). Recently, much has been learned on the role of the thymus in different disease states and there is general agreement in the literature that thymus function is impaired by HIV infection (53). These conclusions are based on the use of several different techniques measuring thymic function: number of naive cells measured by flow cytometry, T-cell receptor excision circles (TRECs) content measured by reverse transcriptase polymerase chain reaction (RT-PCR), thymic morphology by computed tomography scans (52); in particular, TRECs content has been widely used for this purpose, although the results obtained by this technique may be altered in HIV-positive subjects by the influence of ongoing HIV replication on the rates of cell division, increased apoptosis, and modifications of the life span of CD4 and CD8 subsets (52,53). Although these effects may be negligible under effective HAART, none of the methods currently used to assess thymic function can be considered completely reliable due to the complex interactions existing between HIV and the immune system. New tools are therefore required to more accurately measure thymic function in order to assess its role in maintaining immunological homeostasis in HIV disease and in other immunopathological conditions (52).
Although the detrimental effects of HIV are mostly directed against CD4 T lymphocytes, this virus also leads to impaired B cell function and antibody responses through mechanisms that remain poorly defined (84). Recently, the interplay between HIV and B cells has been changed by the use of the anti-CD20 monoclonal antibody rituximab in the therapy of NHL, including conditioning regimens for APSCT candidates (108). This antibody alters the immunological status of treated patients, causing a depletion of B lymphocytes and accelerating the rate of CD4 T-cell depletion and of HIV replication in the peripheral blood (30,120). Finally, NK cell functions become compromised in HIV-infected individuals in early stages of the infection; these compromised NK cell functions adversely affect several aspects of the host's antiviral adaptive immune responses (35,70).
Effects of Mobilization Procedures on Immunological and Virological Parameters
G-CSF is used for leukapheretic procedures in both HIV-positive and HIV-negative patients undergoing APSCT. Besides its capacity to induce stem cell mobilization, G-CSF has an important role in the regulation of T lymphocyte and accessory cell activity (50). Effects on the immune system are mediated via binding of the cytokine to its receptor, expressed on T lymphocytes and monocytes; these consist in the subsequent increase of the differentiation and proliferation of lymphocytes and in the dysregulation of the cytokine network (38). G-CSF administration may be particularly detrimental for HIV-infected subjects by inducing CD4 lymphocyte activation and promoting viral replication (60).
Because the majority of the information on the effects of G-CSF on lymphocyte functions and HIV life cycle were initially based on in vitro data, in vivo measurements were rapidly needed. Campbell et al. (24) suggested that in HIV-positive subjects undergoing stem cell mobilization, G-CSF produced transient, although measurable, increases of HIV RNA plasma levels consistent with its ability to promote in vitro active viral replication. On the contrary, HIV DNA levels remained stable, suggesting that the viral reservoir was not substantially modified after the administration of the cytokine. Based on these findings, it was hypothesized that patients who have increased HIV plasma levels after G-CSF administration possess a subset of infected cells expressing high levels of the cytokine receptor that are susceptible to stimulation by G-CSF. According to these authors, the interactions between this cytokine and its receptor promote viral replication in succession and the generation of new virions in infected cells, finally leading to infection of other susceptible cells and the worsening of viral-dependent immunopathogenetic effects. In addition, their findings provided evidence that the increased HIV-1 replication during G-CSF treatment was associated with activation of HIV-1 variants that, before treatment, were minor components of the plasma quasispecies. These data suffered from substantial limitations, because they were obtained in a small number of HIV-positive patients with unknown immunological characteristics, who were not enrolled in a transplantation program. Therefore, we thought that more reliable information could be obtained in patients with a well-defined immunological status as part of an APSCT procedure. For this reason, we started a monoinstitutional prospective study aiming at obtaining information on clinical and biological parameters, such as the kinetic measurement of immune and viral markers in vivo, in HIV-positive lymphoma patients undergoing stem cell mobilization procedures for transplantation. Our data showed that, during mobilization procedures, G-CSF administration rapidly induced a sharp increase of CD4 counts in HIV-positive patients, while the effects were less relevant in the HIV-negative control group (20). The reasons for this increase in CD4 cells are not clear at the moment, but it is reasonable to hypothesize that they may result from boosted cytokine-dependent proliferation or mobilization of CD4 cells from peripheral lymphoid organs to the peripheral blood. In these patients we measured selected virological parameters that provided information on HIV disease; we used HIV RNA levels as sensitive markers of ongoing viral replication and HIV DNA levels as indicators of the extent of the viral reservoir. Our data demonstrated that neither viral marker had substantially increased after G-CSF; therefore, it may be concluded that an effective response to HAART may guarantee the control of HIV replication even in the presence of a potent exogenous stimulation such as cytokine administration.
Immune Reconstitution After APSCT
Available data in the literature suggest that in HIV-positive individuals, after APSCT, there is a profound immunosuppression similar to what has been consistently observed in transplanted HIV-negative subjects (40,42,44,66,97). However, detailed biological and clinical data in the HIV-positive setting are still limited while the kinetics and the mechanisms driving immune reconstitution after APSCT have been extensively studied in the HIV-negative population (7,40,44). Briefly, the type (unmanipulated vs. selected CD34+ cells) and the amount of reinfused apheretic product influence immune reconstitution, while the importance of the genetic characteristics of the host and the histopathological classification of the tumor are quite limited (29,45). In addition, the timing of immunological recovery has been considered an important prognostic factor in patients treated with standard chemotherapeutic regimens and in patients transplanted for lymphomas (45).
In HIV-positive patients, the situation is less clearly defined and it is complicated by the presence of the additional, viral-dependent immunopathogenetic effects. HIV does not infect bone marrow stem cells, but infects bone marrow stromal cells, a condition that is sufficient to induce hematopoietic suppression in vivo (11). Major mechanisms driving this suppression are a reduced clonogenic capacity and a decrease in the number of primitive progenitor cells in the bone marrow. In addition, upregulation of Fas and Fas ligand contribute to the inhibition of hematopoiesis (58) by amplifying apoptotic pathways. Finally, a perturbation of cytokine pathways is an additional pathogenetic mechanism promoting immune dysregulation. Particularly in severe lymphopenic HIV-positive patients, the upregulation of IL-7 production by stromal cells has been suggested to be an important mechanism of immune reconstitution because this cytokine is able to increase CD4 counts (18,128). In accordance with this putative role, stromal cells from HIV-positive patients with very low CD4 counts spontaneously produce more IL-7 compared to HIV-positive patients having higher CD4 counts (57). On the contrary, other experimental and in vivo data suggest that the IL-7 increase may be ineffective in sustaining immunological reconstitution and in conditioning a better prognosis (2,15). The reasons for the discrepancies between high IL-7 levels and absence of activity are presently unknown, although it has been hypothesized that circulating IL-7 levels may increase as a result of diminished absorption by peripheral lymphoid cells, rather than from an increased production by stromal cells. In fact, the consistent activation associated with HIV-1 infection induces a downregulation of membrane IL-7 receptor, a decrease of the cellular binding and a consequent increase of free circulating cytokine levels. This hypothesis is reinforced by the observation that IL-7 receptor downregulation is negligible in the less pathogenic HIV-2 infection (2). According to these data, the use of IL-7 to treat HIV-1 infection may be ineffective.
A perturbation of other cytokine networks is also considered an important pathogenetic mechanism driving immunosuppression in infectious diseases and cancer (35,49). For example, a Th1 to Th2 switch has been considered one of the hallmarks of HIV-mediated immunodeficiency and has been implicated in the genesis of Burkitt's lymphoma (75). Recent studies have reported that, after APSCT in the HIV-negative population, Th2 cells predominate, although it is not clear at the moment whether this imbalance correlates with an increased susceptibility to infections or recurrence of the tumor. We planned to perform a detailed analysis of cytokine pattern in patients transplanted in our institute in order to assess the possible relationship between HIV infection, lymphomas, and stem cell transplantation. A preliminary analysis suggests that, in HIV-positive patients, plasma levels of several cytokines are usually altered before and after APSCT and both quantitative and qualitative differences exist compared to the HIV-negative APSCT patients. However, our data are still incomplete and further studies are required before definitive conclusions can be drawn (P. De Paoli, manuscript in preparation).
Despite their importance in the pathogenesis of HIV disease and in stem cell physiology, no data are currently available on the chemokine secretion pattern during APSCT procedures in HIV-positive Lymphomas. This may also represent an important issue to be investigated in the near future.
From an immunologic point of view, APSCT in HIV-positive lymphomas is characterized by a short period of absolute cytopenia. Data in the literature and our observations suggest that the duration and entity of this period are not substantially influenced by the association of the HIV infection (20,42,99). In HIV-negative lymphoma patients, absolute lymphocyte count measured 2 weeks after the end of CT is considered an important prognostic factor, independent from the histopathological classification of lymphoma and the clinical characteristics of the patients (94). No information pertaining to this issue is available in the HIV-positive lymphoma subjects, but it is conceivable that additional factors influencing lymphocyte counts in the HIV population (i.e., efficacy of antiretroviral therapy, presence of co-infections) may impede the use of absolute lymphocyte counts as reliable prognostic factors in these patients.
After the period of absolute lymphopenia, lymphocyte subsets recover. Enhanced bone marrow activity, increased thymopoiesis, or peripheral expansion of already existing lymphocytes are considered the most important mechanisms driving lymphocyte reconstitution after APSCT, but qualitative and quantitative aspects of immune reconstitution differ among distinctive lymphocyte subsets.
CD4 T cells recover slowly and reach pretransplantation levels at least 1 year after transplantation. A profound depression of the CD4 naive subset lasts for more than 6 months and, therefore, the majority of the CD4 cells are composed of memory cells (14,99). The grade of immunodeficiency may be less dramatic than expected: Resino et al. stated that, although the reconstitution of the memory subset was suboptimal, HIV-positive lymphoma patients showed an appropriate immune reconstitution after APSCT, close to that observed in the HIV-negative group (99). In a larger number of patients, we observed that patients under HAART after transplantation may partially recover some of their immunological parameters in 12 months (95), but because no cases of prolonged HAART failure have been reported so far, it is not known at the moment in what way resistance to antiretroviral treatment may eventually affect the pattern of immune reconstitution.
More detailed information regarding quantitative, phenotypic, and molecular tests on CD4 subsets have already been obtained, but the behavior of important functions (i.e., antigen specificity, proliferation, cytokine secretion, etc.) of CD4 T cells during immune reconstitution after APSCT is, as yet, partially unknown. It has been suggested that CD4 central memory T cells play a central role in the effective immune control of HIV infection and cancer (63,80). Future studies should take into consideration quantitative and qualitative regulation and the effects of different therapeutic strategies on CD4 central memory T cells during immune reconstitution.
While CD4 T-cell recovery after APSCT has been, at least partially, characterized, the qualitative behavior of the CD8 T-cell subset is still less defined. Many studies have not reported the kinetic measurements and a multiparametric phenotypic characterization of this subset. The available data on the phenotypic characteristics of recovering CD8 T cells in HIV-negative patients with lymphoma undergoing APSCT suggest the presence of an expansion of antigen-primed cells rather than naive cells (45). Many CD8 T cells are CD45R0+, CD57+, and CD28–, with a phenotype initially ascribed to anergic or terminally differentiated cells; more recently this condition has been more appropriately defined as cellular “exhaustion” occurring in situations of prolonged antigenic immune activation (135). Our preliminary kinetic observations in HIV-positive lymphoma patients, before and after transplantation, are in favor of this hypothesis. We also observed that, shortly after transplantation in HIV-positive lymphoma patients, there is a massive expansion of CD8 T cells (mean >1500 cells/mm3, 1 month after grafting) lasting for 1 to 3 months [(117), P. De Paoli, manuscript in preparation]. It is not clear at the moment whether this increase depends on a higher proliferation rate, the blocking of apoptosis, or migration of CD8 T cells from lymphoid organs to peripheral blood. Studies are ongoing in order to establish the functional specificity of expanded cells.
Although CD4 and CD8 T cells have been extensively characterized phenotypically, there is no clear correlation at the moment between defined subsets and the extent of a protective response against chronic infections and tumors. Traditionally, the fundamental characteristic describing the behavior of a T-cell response was considered its ‘magnitude’ (the quantitative measurement of defined subsets), but this characteristic is probably unable to entirely reflect the effector potential of T cells. Because a low frequency of high-quality T cells may be as protective as a high frequency of lesser quality T cells, Seder et al. recently proposed measuring a whole set of T-cell activities, rather than magnitude alone, by means of a multiparametric assessment aiming at the definition of “the quality of the T-cell response” (110). They proposed that this quality could be measured by simultaneous analysis using flow cytometry demonstrating the ability of single antigen-specific T cells to produce distinctive cytokine patterns, express surface molecules acting as receptors, and proliferate in response to well-defined stimulatory/costimulatory signals. Future studies may take into consideration the importance of analyzing the kinetics of the T-cell response quality to optimize effective therapeutic strategies against chronic infections and tumors.
Impaired T-cell-mediated immunity may also depend on reduced diversity of the T-cell repertoire (TCR). Benicchi et al. showed that the TCR in HIV-positive lymphomas is already impaired before transplantation (14); they suggested that this defect could be attributed to hematologic malignancy, the intrinsic nature of the immunological disorder, or the cytotoxic effects of CT. By prolonging their observation period, these authors demonstrated that, 1 year after transplantation, the pattern of TCR expression was not different from that of the pretransplant period and that there was a tendency toward a TCR diversification. Therefore, the long-term effects of APSCT in HIV-positive lymphoma patients on the TCR need to be clarified.
Very few data have been published on B lymphocyte repopulation after transplantation; this information gained particular importance after the introduction of rituximab in therapeutic protocols for lymphomas. In fact, the use of rituximab in HIV-negative patients induces a complete depletion of B lymphocytes for 6–9 months and the suppression of immunoglobulin production for 1 year after APSCT (115). Recent data suggest that rituximab also impairs differentiation of naive B cells into plasma cells, resulting in hypogammaglobulinemia and causing a delayed recovery of memory B cells with abnormal cell markers and functions (89). In addition, we observed a novel aspect related to the use of rituximab in the HIV-positive lymphoma patients transplanted in our institute: the expected profound depletion of B lymphocytes for 6 months after APSCT was followed by a robust, short lasting, polyclonal expansion of this subset (>500 B cells/mm3); presently, studies are ongoing to characterize the phenotype and function of these cells.
Although the innate immune response plays an essential role in controlling the very early phases of HIV infection, studies in HIV controllers suggested that innate immunity is also an essential part of the immune response during the chronic phase of infection (51,70). However, few data on the role of innate immunity in immune reconstitution after APSCT are available in the HIV-negative setting. It has been demonstrated in these patients that, after APSCT, NK cells constitute the major lymphocyte subset in the first days after transplantation (45). Whether NK cell numbers do have prognostic significance, as demonstrated in lymphoma patients after conventional CT, is not clear at the moment. In the peripheral blood of HIV-negative subjects, counts of myeloid dendritic cells become normal 3 months after transplantation, while counts of plasmacytoid cells remain low, but no clear correlations between biological and clinical parameters have been reported (45). No studies on innate immunity in transplanted HIV-positive patients have been reported so far.
Kinetics of Viral Infections After APSCT
A few years ago, the possibility of eradicating the HIV reservoir by a combination of antiretroviral therapies and CT was hypothesized as a feasible goal of ongoing research (56,139). A first approach studying the influence of HIV disease on transplantation procedures was realized by Gabarre et al., who demonstrated that large numbers of HIV-1 proviral copies (106 to 109 copies) are reinfused with the graft and therefore could reinfect the recipient, although the profound cytopenia induced by high-dose CT could in vivo limit de novo infection of target cells and subsequent viral replication (42). These authors suggested the possibility that transplantation could modify the relationship between recipients and the virus, but they did not measure in vivo the kinetics of HIV viremia and proviremia after the engraftment. Therefore, the biological and clinical effects of proviral reinfusion were not known. We performed time course measurements of HIV viremia and proviremia in the HIV-positive lymphoma patients included in the transplantation program in our institute to resolve this matter. We observed that, when immunological reconstitution takes place, the proviral load rises accordingly, suggesting that a measurable proportion of recently generated CD4 T cells and monocytes is infected.
It was recently demonstrated that in HIV-positive patients proviremia also persists after allogeneic bone marrow transplantation using an HIV-negative donor, confirming the hypothesis that even aggressive cytotoxic therapies are unable to eradicate viral reservoirs (10). Based on these considerations, the continuous use of HAART is mandatory to maintain HIV viremia below detection values, permit immune reconstitution, and the control of HIV disease. Occasionally viremia may become detectable after APSCT, because of noncompliance with HAART or due to the development of resistant viral strains (P. De Paoli, manuscript in preparation).
After transplantation, the majority of concomitant infections are caused by herpes viruses (28,31,74). Despite the profound T-cell depletion induced by HIV infection that can be an additional factor favoring the reactivation of herpes viral infections, this reactivation is not very frequent during the posttransplantation period. In our patients, we observed rare cases of symptomatic CMV infection, accompanied by the detection of CMV p65 antigen or CMV-DNA by PCR in the peripheral blood. Blips of Epstein-Barr virus (EBV) viremia have been demonstrated in a small proportion of HIV-positive lymphoma patients, but their diagnostic and prognostic significance is still not clearly defined (19,133). After transplantation, EBV is seldom detectable even in the peripheral blood of HIV-positive lymphoma patients and, usually, the infection is asymptomatic. Due to the limited experience in this field, we suggest that evaluating the role of EBV in the cancerogenesis of these patients will require a longer observation period [(33), P. De Paoli, personal observations].
The establishment of immune correlates of protection and their possible relationship with the presence of ongoing viral replication markers may be particularly important not only to understand which factors interfere with a protective immune response, but also to identify strategies that establish pathogen-specific immunopotentiation procedures.
The qualitative and quantitative aspects of the regeneration of antigen-specific cellular immunity have been studied in HIV-negative cases undergoing APSCT. Reusser et al. have demonstrated that cytomegalovirus-specific CD8 cytotoxic activity and CD4 T helper responses are restored in a large proportion of patients in the first 3 months posttransplant and that specific CD8 CTLs afford protection from CMV infection (9,100). At the moment, data from literature on this topic are very limited and more studies are needed to clarify this issue.
Human herpes virus 8 (HHV-8) is causally implicated in the development of HIV-associated cancers, such as Kaposi's sarcoma (KS) and lymphoproliferative disorders (109). Few studies have demonstrated in the mouse model that HHV-8 establishes a persistent infection in HSC (138), those that may represent a source of virally infected cells. An anecdotal case of KS triggered by endogenous HHV-8 reactivation after allogeneic hematopoietic transplantation has recently been described in the HIV-negative setting (21). Due to higher HHV-8 sero-prevalence in HIV-positive patients, it may be expected that the frequency of HHV-8-related diseases after APSCT may be much higher compared to the HIV-negative counterpart, but epidemiological or clinical data are presently not available. In our experience, only 1 out of 24 transplanted cases worsened his preexisting KS.
Co-infection with hepatitis C virus (HCV) or hepatitis B virus (HBV) is an important additional pathogenetic factor in HIV-positive patients. Information on the consequences of HBV or HBV co-infection on APSCT is limited. We recently reported that HCV infection did not prevent APSCT in one HIV-positive lymphoma patient and that HCV viremia does not increase during immunosuppression following transplantation (117).
Future Direction in the Use of the Hemopoietic Stem Cell
Although the demonstration of HSCT efficacy in the cure of HIV-positive cancers is still under scrutiny, encouraging data on its high efficacy despite a low frequency of life-threatening adverse events (at least in well-fit patients) have rapidly demonstrated that in the HAART era there are no rational reasons to exclude HIV-positive patients from aggressive curative programs including an autologous or allogeneic hematopoietic stem cell transplant (12,66,97,113,127). Moreover, growing positive data on the engraftment of stem cells in HIV-positive recipients affected by a hematopoietic cancer have opened several fascinating scenarios, including the development of genetic therapies aimed at inducing HIV infection resistance. Research on this approach has its rationale in the fact that the chance of long-term survival in HIV-infected patients with chemosensitive cancers ultimately depends on the control of HIV infection itself. Particularly in the setting of HIV and hemopoietic cancers, where a transplant is the main curative option in the HIV-negative counterpart, manipulation of HSC before reinfusion constitutes a very attractive opportunity. In fact, HSC represent an ideal target for a genetic approach to treat HIV infection, given that HIV does not infect bone marrow stem cells whereas HSC can produce CD4+ cells, dendritic cells, and macrophages that are all the cells involved in HIV pathogenesis: the introduction of genetic modifications able to confer HIV resistance to progeny may generate a self-renewing source of a spectrum of susceptible HIV targets (64,68,103,105,141). The regenerative potential of HSC may give a theoretical advantage over the expansion and replacement of gene-modified T cells (103). Theoretical disadvantages may be the long period that elapses between HSC infusion and T-cell regeneration and the relative low engraftment that engineered CD34+ cells have currently demonstrated when reinfused, in absence of a myeloablative regimen or in absence of a positive selective pressure given from a high HIV viral load (4,5,64). In the City of Hope National Medical Center, Krishnan et al. treated five HIV-positive lymphomas with APSCT. Recipients were reinfused with a mixture of unmanipulated HSC and with selected CD34+ cells transduced with retrovirus encoding ribozymes targeted to tat and rev. Compared to prior studies on HIV-infected volunteers who received transduced HSC in the absence of ablative chemotherapy regimens, HIV-positive lymphomas showed a higher level of genetically modified cells just after transplantation. Unfortunately, long-term durability and kinetics of the manipulated graft were still disappointing because the level of marked cells rapidly declined over the subsequent 6 months (68). Kang et al. transplanted one patient with acute myelogenous leukemia and one with HD and HIV infection with an allogeneic graft characterized by genetically modified, selected CD34+ cells transduced with either a transdominant rev or control vector. They used a nonmyeloablative conditioning regimen. After 2 years from engraftment, a small amount of gene-modified CD4+ cells was still detectable (59).
Despite disappointing preliminary data on long-term evidence of engineered cell proliferation, overall research demonstrating feasibility and low toxicity of reduced conditioning regimens for HSC transplant and data on autologous and allogeneic PBSC ability to engraft cells that could possibly confer HIV resistance appear fascinating. Very encouraging suggestions also emerged from research conducted by Hutter et al. (55), who reported on allogeneic HSCT of a 40-year-old man with HIV-1 infection since 1995 having had a relapse of acute acute myelogenous leukemia first diagnosed in 2006. Considering that about 1% of Europeans, and a greater number in northern Europe, inherit a CCR5 δ32 mutation from both parents, preventing the CCR5 receptor from appearing on the surface of cells, they engrafted the HIV-positive leukemic patient with peripheral stem cells from a donor selected from the Bone Marrow Donor Registry both for being identified as HLA identical and by being homozygous for the CCR5 δ32 mutation. With ongoing engraftment, CCR5-polymerase chain reaction patterns of PBSC were transformed into a homozygous δ32/δ32 genotype. In rectal biopsies, taken on day +145, macrophages showed an expression of CCR5, whereas a remarkable CCR5–expressing population could not be found on the mucosal CD4+ T cells. During the whole follow-up period, measurement of serum HIV-1 RNA remained negative and the semiquantitative proviral DNA assay was under the limit of detection since day +61. Although HAART had been discontinued for longer than 200 days, HIV-1 viral load could not be detected in the peripheral blood, bone marrow, and rectal mucosa (55).
Finally, especially in the HAART era, it must be emphasized that patients with HIV infection would be considered individuals affected by a chronic and degenerative disease. From this point of view, data on the patients' immune system showing signs of accelerated aging are representative. Capacity of pluripotent stem cells to repair tissues in which they reside holds an alternative great promise in the development of novel therapeutic opportunities for cell replacement, even in HIV-infected patients. Due to the great potential of stem cells and HSC in the development of new therapeutic options for cancers and degenerative diseases, data on their ability to engraft and regenerate hemopoietic and lymphoid tissue in HIV patients are mandatory bases for the development of future approaches in advanced therapies against HIV.
Conclusions
Autologous or allogeneic transplantation are considered powerful strategies to treat poor risk, resistant, or relapsed hemopoietic cancers in the HIV-negative population. Although the advent of HAART allowed a high rate of success in the first line treatment of HIV-positive lymphomas, no satisfactory treatment exists at present for relapsed or resistant cases when using only standard dose chemotherapy. In the HAART era, only a few HIV-negative cases underwent an allogeneic transplant showing remarkable success. About 100 relapsed or resistant HIV-positive lymphomas have already been treated with an APSCT. Autologous transplant compared favorably with any previous salvage attempt in each series reported at present. Continuous complete remission and OS ranged from 36% to more than 80% at median follow-up approaching 3 years. The success is as dramatic, compared to the results of a standard regimen that, at best, gave CR in 7/13 cases with a median OS of only 7 months. Uniformly positive data also emerged from the ability of CD34 stem cells to be mobilized in the peripheral blood and engraft HIV-positive recipients. Nonrelapse mortality generally did not exceed 5% and only increased in the presence of concomitant chronic disease, organ dysfunction, or old age. Death due to infection appeared slightly increased compared to HIV-negative controls. Few data exist on viroimmunological kinetics; many studies relied on quantitative data, but the functional evaluation and protective role of reconstituted immune cells has not yet been performed.
Despite the above-mentioned improvements, caution must be used when approaching transplant in the HIV-positive population. Overall applicability of APSCT in HIV-positive cohort with relapsed/resistant lymphomas is certainly lower than in HIV-negative population and approached 50%. Especially when HIV infection is associated to chemosensitive cancers and an aggressive therapy is needed, its presence requires appropriate screening, careful management, concomitant HAART administration, and attentive supportive care as well as surveillance. Similar to elderly cancer patients, relapsed or resistant HIV-positive lymphomas frequently shows concomitance with an organ dysfunction, a poor performance status, or a viroimmunological asset that may prevent transplant or may result in a significant increase in the risk of death. Nevertheless, in well-fit HIV-positive lymphomas with a good viroimmunological asset, transplantation should be considered the gold standard. In order to optimize benefit and perform the procedure with low transplant-related mortality, transplantation should be considered as early as possible at first relapse or soon after demonstration of scarce chemosensitivity.
In the end, due to the great potential of stem cells and, in particular, of the HSC in the development of new therapeutic options for cancers, degenerative disease, and HIV gene therapy, experimental data concerning their ability to engraft and regenerate hemopoietic and lymphoid tissue in HIV patients are mandatory for the development of future curative approaches.
