Abstract
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is one of the consolidation modalities for adult patients with T-cell lymphoblastic lymphoma (T-LBL). However, the optimal conditioning regimen needs to be explored. In the present study, 40 patients with T-LBL undergoing allo-HSCT were retrospectively analyzed, including 23/40 (57.5%) with total body irradiation (TBI)–based conditioning regimen and 17/40 (42.5%) with busulfan (BU)-based regimen. TBI–based regimen significantly increased the cumulative incidence (CI) of grade II to IV acute graft-versus-host disease (aGvHD) as compared with BU-based regimen (13.0% vs 0%, P = 0.000). The relapse risk was significantly lowered in TBI-based group with a 2-year CI of relapse (CIR) of 9.1% as compared with that of 49.6% in BU-based group (P = 0.008). The 1-year and 2-year non-relapse mortalities (NRMs) for all patients were 5.0% and 10.3%, respectively. The 1-year and 2-year NRMs were 8.9% and 16.0% in TBI-based group, and 0.00% and 0.00% in BU-based group (P = 0.140). The 2-year probabilities of overall survival (OS) and relapse-free survival (RFS) were 83.0% [95% confidence interval, 63.4%–100%] and 74.0% (95% confidence interval, 54.4%–93.6%) in TBI-based group, which were higher than that of 35.0% (95% confidence interval, 0.0%–72.2%) and 50.0% (95% confidence interval, 24.5%–75.4%) in BU-based group, respectively (P = 0.020 for OS and P = 0.081 for RFS). In multivariate analysis, TBI-based regimen significantly reduced the risk of relapse [subdistribution hazard ratio (SHR) = 0.030, 95% CI, 0.002–0.040, P = 0.000] and improved the OS [hazard ratio (HR) 0.121, 95% CI, 0.021–0.683, P = 0.017] as an independent prognostic factor. These results suggested that TBI-based regimen might be an optimal choice for adult patients with T-LBL undergoing allo-HSCT.
Keywords
Introduction
T-cell lymphoblastic lymphoma (T-LBL) is a relatively rare type of hematological malignancies originating from T-cell precursors, which accounts for about 90% of all lymphoblastic lymphoma and occurs mostly in children and adolescents 1 . Although the complete remission (CR) rates could be achieved for more than 90% of patients and the survival of adult T-LBL has been substantially improved by adopting pediatric-inspired chemotherapy regimens for B-cell acute lymphoblastic leukemia (ALL), the prognosis of adult T-LBL was inferior to pediatric patients with 40% to 50% of cumulative incidences of relapse (CIRs) and only 50% to 55% of long-term disease-free survival2–4.
The optimal consolidation treatment modality for adult T-LBL has not been defined. Autologous hematopoietic stem cell transplantation (auto-HSCT) has been used as a consolidation modality to reduce the risk of disease recurrence, but the risk remains to be higher with 5-year relapse rates of 26.5% to 56.0%5–8. Liu et al 5 reported that tandem auto-HSCT could decrease the progression/relapse rate as compared with chemotherapy alone or single auto-HSCT, the 3-year progression/relapse rate remained to be higher of 26.5%. Allogeneic HSCT (allo-HSCT) as a consolidation modality for T-LBL could decrease the relapse rate, especially for high-risk patients, but might increase non-relapse mortality (NRM) as compared with auto-HSCT 8 . Levine et al 8 reported that the CIRs of T-LBL at 1 and 5 years after transplantation were significantly lower in allo-HSCT than that in auto-HSCT (32% vs 46%, P = 0.05; 34% vs 56%, P = 0.004, respectively), while the NRM in allo-HSCT was higher than that in auto-HSCT at 6 months after transplantation (18% vs 3%, P = 0.002). Along with the optimized prophylaxis regimens for graft-versus-host disease (GvHD)9,10, the effective prevention and treatment modalities for infection, and so on, NRM was significantly decreased over the last decades11,12. The optimal conditioning regimen for allo-HSCT needs to be explored to decrease the relapse risk and improve the survival of adult patients with T-LBL.
In the present study, 40 patients with T-LBL undergoing allo-HSCT in our center were retrospectively analyzed and two types of conditioning regimens of total body irradiation (TBI)–based and busulfan (BU)-based were compared to determine the optimal conditioning regimen. The results suggested that TBI-based regimen could decrease the risk of relapse and improve the survival of adult patients with high-risk T-LBL. To our knowledge, this is the first report about evaluating the effects of different conditioning regimens for adult T-LBL.
Patients and Methods
Patients, Donors, and Grafts
Fifty patients were diagnosed with T-LBL in our center from May 1, 2017, to December 31, 2020. Forty of them underwent allogeneic peripheral blood stem cell transplantation (allo-PBSCT) and were enrolled in the retrospective study. T-cell lymphoblastic lymphoma was diagnosed according to World Health Organization (WHO) 2016 criteria 1 . Donors were from human leukocyte antigen (HLA)-matched related and unrelated donors and haploidentical family donors. HLA-matched was defined according to ≥8/10 matched loci at HLA A, B, C, DRB1, DQB1 loci between donor and recipient, while HLA haploidentical according to >3/10 mismatched loci between recipient and family donor. Grafts were from mobilized peripheral blood stem cells (PBSCs) with granulocyte–colony stimulating factor (G-CSF). This study had ethical approval from the local ethical committees and was conducted in accordance with the Declaration of Helsinki. All patient data originated from clinical trials with mandatory written informed consent.
Conditioning Regimens
Myeloablative conditioning (MAC) regimens including TBI-based or BU-based were prescribed to all patients. Total body irradiation–based regimen contained fractioned TBI (fTBI) 10 Gy at 2.0 Gy for five times, etoposide (VP-16) 15 mg/kg/d for 2 days, cyclophosphamide (CY) 50 to 60 mg/kg/d for 2 days. Busulfan-based regimen was composed of intravenous BU 3.2 mg/kg/d for 4 days, CY 50 to 60 mg/kg/d for 2 days, and VP-16 15 mg/kg/d for 2 days. Total body irradiation–based regimen was preferred to patients in partial remission (PR) status, while for patients in CR status, TBI- or BU-based regimen was chosen according to patient’s choice and physician’s decision.
GvHD Prophylaxis
Rabbit anti-human thymocyte globulin (ATG, 10 mg/kg, Thymoglobin®, Genzyme Polyclonals S.A.S., LYON, AUVERGNE RHONE ALPES, France)–based regimen or low-dose ATG (2.5 mg/kg/d on days −2 to −1) plus low-dose post-transplant cyclophosphamide (50 mg/kg/d on day 3) (low-dose ATG/PTCy)–based regimen were used for prevention of GvHD after haploidentical transplantation, while cyclosporine (CsA) combined with mycophenolate mofetil (MMF) and short-course methotrexate (MTX) were provided for prophylaxis of GvHD after HLA-matched sibling donor transplantation and unrelated donor transplantation with additional ATG 5 mg/kg9,10,13. Cyclosporine was prescribed at 2 mg/kg/d as a continuous infusion, and its dose was modified to obtain nadir serum levels between 200 and 300 ng/ml. Mycophenolate mofetil was administered for 30 days at 15 mg/kg oral three times per day (maximum dose 3 g per day) and then stopped if no GvHD.
Infection Prophylaxis
All patients received prophylactic posaconazole and acyclovir from the beginning of conditioning to at least 6 months after transplantation. Compound sulfamethoxazole tablets were prescribed to all patients from the engraftment to at least 1 year for preventing Pneumocystis jiroveci pneumonia. Quantitative real-time polymerase chain reaction (PCR) assays for cytomegalovirus (CMV) DNA in serum and Epstein–Barr (EBV) DNA in whole blood were performed once or twice per week. Preemptive therapies with ganciclovir for CMV and Rituximab for EBV reactivation were described in reference 10 .
Definition
Evaluation of treatment response included computed tomography (CT) scanning and/or PET-CT scan and bone marrow examination. Complete remission was defined as the complete disappearance of all clinical, radiological, and histological/immunophenotypic evidence of the disease. Partial remission was defined as a 50% or greater reduction in the area of tumor based on bidimensional measurements 14 . Neutrophil engraftment was defined as achieving an absolute neutrophil count greater than 0.5×109/l on the first day of 3 consecutive days without G-CSF, while platelet engraftment was defined as a platelet count greater than 20×109/l on the first day of 7 consecutive days without platelet infusion 15 . Chimerism analysis was performed on bone marrow aspiration using short tandem repeat (STR)–PCR in patients with a sex-matched donor, and fluorescence in situ hybridization (FISH) analysis of X and Y chromosome in sex-mismatched patients. Complete chimerism was defined by the detection of ≥95% of donor cells using STR-PCR and FISH16–18. Acute GvHD (aGvHD) was graded according to the modified Glucksberg criteria 19 and chronic GvHD (cGvHD) according to the revised Seattle criteria 20 .
Statistical Analysis
All statistics and Kaplan–Meier estimates were performed using IBM SPSS version 23.0. The characteristics of patients in the two different conditioning regimen groups were compared using the χ2 test. The probabilities of overall survival (OS), relapse-free survival (RFS), and GvHD-free relapse-free survival (GRFS) were estimated using the Kaplan–Meier method with log-rank test. Cumulative incidence of relapse, NRM, relapse-related mortality (RRM), and grade II to IV aGvHD were estimated using the Gray method. Cumulative incidence functions were used to estimate RRM and NRM in a competing risk setting 21 , because NRM and relapse are competing events. To estimate the cumulative incidence of aGvHD, we considered relapse and NRM as competing events. Multivariate analysis was performed using the Cox regression hazard model for OS, RFS, and GRFS. Factors with a P value < 0.20 and those that may affect survival were included in the multivariate analysis. The incidences of opportunistic infections were compared using the χ2 test. All the P values were two-sided, and statistical significance was set at P < 0.05.
Overall survival was defined as the time from HSCT to death, regardless of the cause. Relapse-free survival was defined as the time from HSCT to disease relapse/progression, or death from any cause. The GRFS was defined as being alive with neither grade III to IV aGvHD nor severe cGvHD nor disease relapse at any time point 22 . Relapse was defined as the reappearance of the underlying disease. Death without evidence of relapse or progression was defined as NRM.
Results
Patient Characteristics
Ten out of 50 patients diagnosed as T-LBL in our center did not receive allogeneic transplantation. Four of 10 patients achieved CR with first-line chemotherapy and subsequently relapsed. Four developed disease progression and two achieved PR after first-line therapy. The two patients with PR refused allogeneic transplantation. All the 10 patients did not respond to salvage chemotherapy and died from disease progression with a median OS of 8 (range, 6–14) months. Forty adult patients with T-LBL undergoing allo-PBSCT were enrolled into the retrospective study. All patients were in stage III/IV at diagnosis. The majority of patients (31/40, 77.5%) were male with a median age of 28 years old (range, 18–50) at transplantation. A total of five patients received local irradiation therapy for mediastinum residual mass, of which three (two in BU-based group and one in TBI-based group) at 1 month pre-transplant, and two in BU-based group at 3 months post-transplant. Most patients (26/40, 65.0%) received transplantation from haploidentical family donors, while 14 (14/60, 35.0%) from HLA-matched donors (related or unrelated). In total, 23 patients (57.5%) received TBI-based conditioning regimen (TBI-based group) and 17 (42.5%) received BU-based regimen (BU-based group). The other characteristics including B symptoms, mediastinal mass, lactate dehydrogenase (LDH) level at diagnosis, and so on between two groups were similar as shown in Table 1.
Patient and Donor Characteristics.

Additional ATG (5 mg/kg) for matched related family donor GvHD prophylaxis. ATG: anti-thymocyte globin; BU: busulfan; CR: complete remission; CSA: cyclosporine A; GvHD: Graft-versus-host disease; HCT-CI: hematopoietic cell transplantation-specific comorbidity index; LDH: lactate dehydrogenase; MMF: mycophenolate mofetil; MNC: mononucleated cells; MTX: methotrexate; PBSC: peripheral blood stem cell; PR: partial remission; TBI: total body irradiation; ECOG: Eastern Cooperative Oncology Group; ABO: blood types A, B, O, and AB; HLA: human leukocyte antigen.
Engraftment
All patients engrafted successfully and achieved full donor chimerism at 30 days after transplantation. One patient in TBI-based group suffered from secondary graft failure at 46 days post-transplant. For all patients in the cohort, the median time for neutrophil engraftment was 11 (range, 9–21) days, while for platelet engraftment was 12 (range, 10–40) days. The median time for neutrophil engraftment was 12 (range, 9–21) days in TBI-based group and 11 (range, 10–18) days in BU-based group, respectively (P = 0.283), while for platelet engraftment was 12 (range, 10–40) days and 11.5 (range, 10–13) days, respectively (P = 0.368).
GvHD and Infectious Complications
Among all the patients, nine developed any grade of aGvHD with a CI of 22.8% within 180 days after transplantation and three developed grade II to IV aGvHD with a CI of 7.7%. No patient developed grade IV aGvHD. The CIs of any grade aGvHD within 180 days after transplantation had no significant difference between TBI-based and BU-based groups (21.7% vs 5.9%, P = 0.222), while the CI of grade II to IV aGvHD was significantly higher in TBI-based group than that in BU-based group (13.0% vs 0%, P = 0.000). Only one patient in BU-based group developed mild cGvHD at 7 months after transplantation.
Among all 40 patients, 6 patients developed CMV reactivation (2 in BU-based and 4 in TBI-based group, P = 0.622) and 2 suffered from CMV pneumonia (both in TBI-based group, P = 0.499) within 180 days after transplantation. Both the patients recovered from CMV pneumonia with antiviral therapy. Seven patients experienced EBV reactivation (three in BU-based and four in TBI-based group, P = 0.983) and two developed EBV-associated post-transplant lymphoproliferative disorder (PTLD, both in TBI-based group, P = 0.499). One patient developed PTLD of high-grade B-cell lymphoma at 1.5 months after transplantation and recovered from Rituximab 375 mg/m2 once a week for four times, while the other developed peripheral T-cell lymphoma (non-specific subtype) at 3 months post-transplant and recovered from four cycles of chemotherapy with hyper-CVAD regimen (fractionated CY, vincristine, doxorubicin, and dexamethasone).
Relapse, RRM, and NRM
All the six patients in PR status at transplantation achieved CR after allo-PBSCT. In the whole cohort, 10 patients (2 in TBI-based group and 8 in BU-based group) relapsed with a median relapse time of 5 months (range, 2–16). In the TBI-based group, one patient relapsed at 5 months post-transplant and was still alive till the end of follow-up after second allo-HSCT, while the other patient relapsed at 8 months and lost follow-up at 14 months post-transplant. Compared with patients in TBI-based group, all eight relapsed patients died of disease progression in BU-based group. One out of two relapsed patients in TBI-based group and four of eight in BU-based group were in non-remission status at transplantation. For CR patients at transplantation, the 1-year and 2-year CIRs were 5.0% and 5.0% in TBI-based group, while 38.46% and 38.46% in BU-based group (P = 0.020) (Fig. 1A). For total patients, the 1-year and 2-year CIRs were 9.1% and 9.1% in TBI-based group, which were significantly lower than that of 41.2% and 49.6% in BU-based group (P = 0.008) (Fig. 1B). The 1-year and 2-year RRMs were 0% and 7.0% in TBI-based group, which were significantly lower than that of 41.8% and 50.1% in BU-based group (P = 0.011) (Fig. 1C).

(A) The cumulative incidences of relapse (CIRs) for CR patients at transplantation. (B) The CIRs for all patients by conditioning regimens. (C) Non-relapse mortalities (NRMs, P = 0.140) and relapse-related mortalities (P = 0.011) for all patients by conditioning regimens. BU: busulfan; TBI: total body irradiation.
Three patients died of non-relapse causes, and all were from TBI-based group. One died from secondary graft failure at 2 months, one from poor graft function at 1 month, and another from veno-occlusive disease/sinusoidal obstruction syndrome (VOD/SOS) at 3 months after transplantation. No patients died of GvHD and infection. The 1-year and 2-year NRMs for all patients were 5.0% and 10.3%, respectively. The 1-year and 2-year NRMs were 8.9% and 16.0% in TBI-based group, which had no significant differences with that of 0.00% and 0.00% in BU-based group (P = 0.140) (Fig. 1C).
Survival
For all patients, the median follow-up time was 15.5 (range, 1.0–45.0) months. The 1-year and 2-year probabilities of OS (Fig. 2A), RFS (Fig. 2B), and GRFS (Fig. 2C) were 76.0% [95% confidence interval, 62.2%–89.7%] and 65.0% (95% confidence interval, 45.4%–84.6%), 72.0% (95% confidence interval, 58.2%–85.7%) and 64.0% (95% confidence interval, 46.3%–81.6%), and 70.0% (95% confidence interval, 56.2%–83.7%) and 62.0% (95% confidence interval, 46.3%–77.2%), respectively. The 1-year and 2-year probabilities of OS of 91.0% (95% confidence interval, 79.2%–100%) and 83.0% (95% confidence interval, 63.4%–100%) in TBI-based group were significantly higher than that of 58.0% (95% confidence interval, 34.4%–81.5%) and 35.0% (95% confidence interval, 0.0%–72.2%) in BU-based group (P = 0.020) (Fig. 3A). The 1-year and 2-year probabilities of RFS in TBI-based group were 82.0% (95% confidence interval, 66.3%–97.6%) and 74.0% (95% confidence interval, 54.4%–93.6%), which were higher than that of 59.0% (95% confidence interval, 35.4%–82.5%) and 50.0% (95% confidence interval, 24.5%–75.4%) in BU-based group, respectively (P = 0.081) (Fig. 3B). The 1-year and 2-year probabilities of GRFS were similar between TBI- and BU-based groups [78.0% (95% confidence interval, 60.3%–95.6%) and 71.0% (95% confidence interval, 51.4%–90.6%) vs 59.0% (95% confidence interval, 35.4%–82.5%) and 50.0% (95% confidence interval, 24.5%–75.4%), P = 0.146] (Fig. 3C).

(A) Overall survival (OS). (B) Relapse-free survival (RFS). (C) Graft-versus-host disease/relapse-free survival (GRFS).
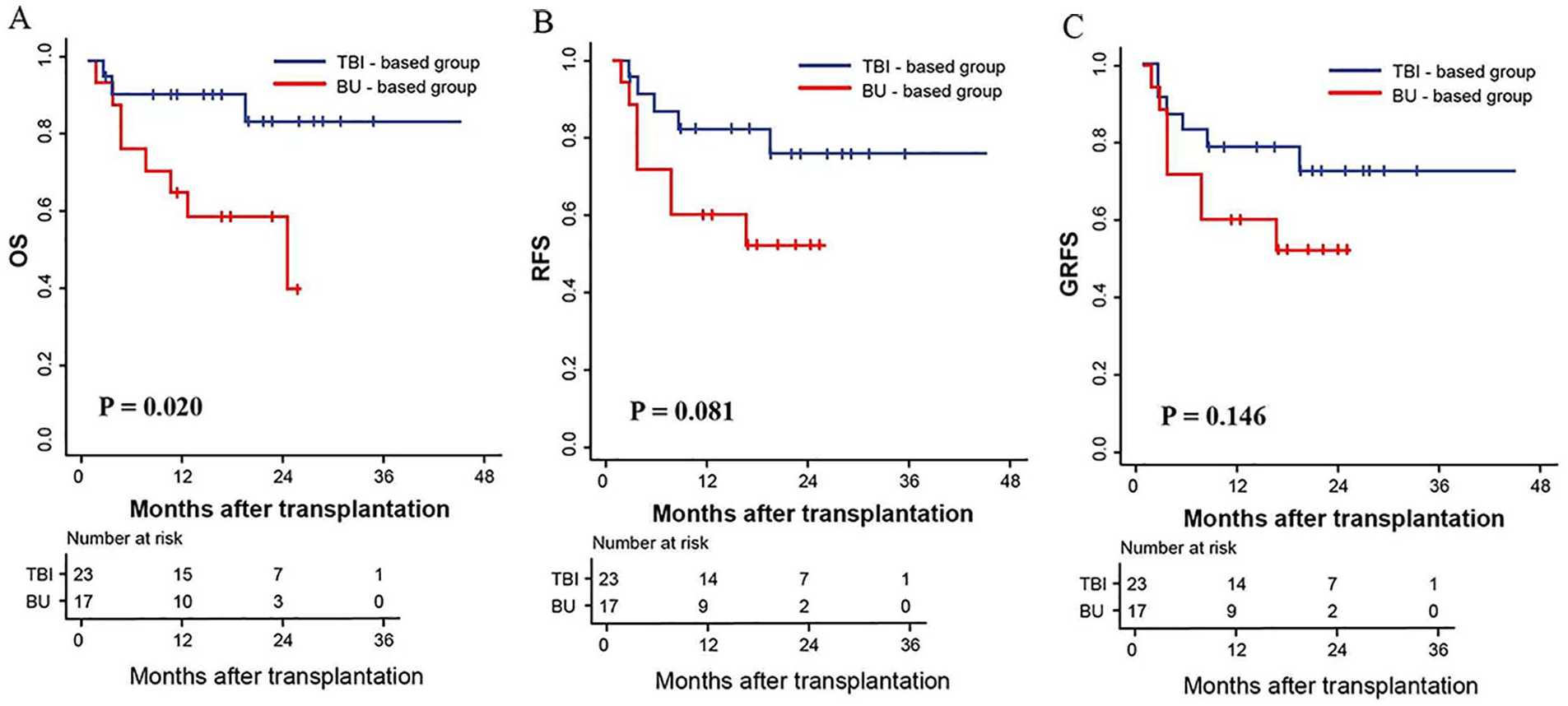
(A) Overall survival (OS) by conditioning regimens. (B) Relapse-free survival (RFS) by conditioning regimens. (C) Graft-versus-host disease/relapse-free survival (GRFS) by conditioning regimens. BU: busulfan; TBI: total body irradiation.
Univariate and Multivariate Analysis
The results of univariate and multivariate analysis are shown in Table S1 in the Supplemental Material and Table 2, respectively. In multivariate analysis, conditioning regimen as an independent prognostic factor significantly affected the OS, RFS, and the risk of relapse. Total body irradiation–based conditioning regimen increased the OS [hazard ratio (HR), 0.121, 95% CI, 0.021–0.683, P = 0.017] and RFS with a strong trend (HR, 0.302, 95% CI, 0.078–1.165, P = 0.082), while BU-based regimen increased the risk of relapse (HR, 33.32, 95% CI, 6.62–166.679, P = 0.000). Elevated LDH level negatively affected the OS (HR, 9.145, 95% CI, 2.062–40.565, P = 0.004), RFS (HR, 6.375, 95% CI, 1.829–22.217, P = 0.004) and GRFS (HR, 4.567, 95% CI, 1.447–14.418, P = 0.010). Partial remission status at transplantation significantly decreased the RFS and GRFS as an independent risk factor. The presence of B symptoms at diagnosis significantly increased the risk of relapse [subdistribution hazard ratio (SHR), 7.662, 95% CI, 1.056–55.593, P = 0.040], while TBI-based regimen reduced the relapse risk (SHR, 0.030, 95% CI, 0.002–0.040, P = 0.000). Partial remission status at transplant had a strong trend toward relapse (SHR, 5.178, 95% CI, 0.923–29.055, P = 0.062).
Multivariate Analysis for OS, RFS, GRFS, and Relapse.

The risk factors for OS, RFS, and GRFS were estimated using Cox regression and were described as HR. Relapse were estimated using the competing regression and were described as SHR. BU: busulfan; CI: cumulative incidence; CR: complete remission; GRFS: graft-versus-host disease/relapse-free survival; HR: hazard ratio; LDH: lactate dehydrogenase; OS: over survival; PR: partial remission; RFS: relapse-free survival; SHR: subdistribution hazard ratio; TBI: total body irradiation; HLA: human leukocyte antigen.
Discussion
In the present study, the results showed that TBI-based conditioning regimen could significantly reduce the CIR and improve the OS and RFS, which suggested that TBI-based MAC regimen might be an optimal conditioning regimen for adult T-LBL patients receiving allo-HSCT.
Total body irradiation as a conditioning regimen was first reported in the 1970s 23 and then widely used for its efficiency. The optimal TBI dosage and fractionation remain to be undefined24,25. Initial experience using TBI was based on a single dose of 10 Gy 26 . Dose-fractionated strategy could increase the total dose of TBI to 12 Gy or above and lower the risks of early acute toxicities, but the late effects of TBI remain to be notable, which include growth and endocrine dysfunction 27 , organ-specific damages like as cardiovascular and pulmonary28,29, ocular complications and second malignancies 30 , and so on. Vitale’s study compared the safety and efficacy of a 10 Gy single-dose TBI (sTBI) with a 10 Gy fTBI (3.3 Gy on 3 consecutive days) and suggested that fTBI was the only significant factor associated with a lower incidence of interstitial pneumonia (IP) (P = 0.002), a lower mortality from GvHD and IP (P = 0.03 and P = 0.001, respectively), and a better 100-day survival (P = 0.03) 31 . A 12 Gy dose fTBI still resulted in a higher risk of developing cataract that eventually required surgery 32 and central nervous system (CNS) complications 33 . As patients enrolled in our study were mostly adolescent and young adult patients, a 10 Gy fTBI was chosen to maintain efficacy and lower acute and late toxicities.
As an alternative to TBI, non-TBI regimens such as BU substituting for TBI have been investigated34,35. Total body irradiation–based and BU-based regimens were the most common used MAC regimens for patients with ALL36–39. For pediatric patients, TBI-based regimen was superior to chemotherapy-based regimen with a lower CIR and a better OS 40 . While for adult patients, the benefit of lowering the relapse frequencies from TBI-based conditioning regimen was offset by its higher NRM as compared with BU-based regimen, resulting in a similar RFS and OS41–43. Kalaycio et al 44 comparatively analyzed the data of adult ALL patients with BU- and TBI-based regimens, which showed the incidence of initial relapse in extramedullary sites following BU was 50%, but only 9% following TBI (P = 0.005), but no survival advantage in either event-free survival (EFS) (P = 0.11) or OS (P = 0.20) for TBI-based regimen. An European Society for Blood and Marrow Transplantation (EBMT) study suggested that TBI-based regimen could not bring any survival benefit for T-cell ALL patients of ≥35 years old, owing to a higher risk of NRM as compared with BU-based regimen (38% vs 9%, P = 0.01) 45 . On the contrary, a study by Shigematsu et al 46 showed the results of a medium-dose VP-16 combined with CY and fTBI (12 Gy) conditioning regimen for adult ALL patients and found this regimen resulted in a good disease control without an increase of NRM (1-year NRM 14.0%). In our study, NRMs had no significant difference between the two conditioning regimens (P = 0.140), although the NRM was higher in TBI-based group than that in BU-based group (15.95% vs 0%). The NRMs were relatively lower not only for all patients in the study, but also for patients with TBI-based regimen compared with that in previous studies4,7,8,11,38. The risk of NRM after transplantation mainly correlated with GvHD and infection. The incidence of grade II to IV aGvHD was only 13.04% in TBI-based group, while no patient in BU-based group in our study, which was relatively lower compared with that of 40% to 47%43,47,48 in previous studies. The lower incidences of aGvHD might come from the effective prevention of GvHD9,10,13. A lower incidence of cGvHD was present in the whole cohort, which might be related to the lower incidence of aGvHD, the early death of some relapsed patients in the BU-based group, the shorter period of the follow-up, and the small size of sample. All the patients suffering from CMV and EBV infection recovered, and no patient died from infection. Older age is an important risk factor for NRM, especially in patients receiving MAC regimens 49 . In a study by Marks et al 50 evaluating the effects of different fTBI-based MAC regimens (12 Gy < TBI <14.5 Gy) for ALL patients, over 60% of patients were younger than 30 years old in each group and the treatment-related mortalities (TRMs) at 3 year were 9% to 27%. In our study, the majority (70%) of patients were younger than 30 years old, which might be one of the causes for a lower NRM.
As a result of the lower risk of disease recurrence and NRM, patients in the TBI-based group had a better OS than the BU-based group. The 1-year OS and RFS of 91.0% and 82% in the TBI-based group were higher than that of 58.0% and 59% in the BU-based group (P = 0.020 and P = 0.081, respectively). The lower number of patients developed severe aGvHD (only one patient with grade III aGvHD in TBI group), and no patient experienced severe cGvHD might be related with the similar incidences of GRFS between two groups.
The prognostic factors for adult T-LBL with chemotherapy have been largely studied51–53, but fewer studies concerned the prognostic factors for patients with allo-HSCT. In GRAALL-LYSALL03 study, multivariate analysis showed that LDH level (normal or elevated) was an independent prognostic indicator for EFS (HR, 2.8, 95% CI, 1.3–5.9; P = 0.010) and OS (HR, 3.4, 95% CI, 1.3–8.7; P = 0.012) 54 , while other prognostic factors like as CNS involvement 55 , bone marrow involvement 56 , and disease status at transplantation 8 were also reported. Hu et al 47 analyzed the prognostic factors for T-LBL patients, of which 22 patients (45.8%) received allogeneic transplantation and found that a higher Ki-67 score, evaluated LDH, and pleural effusion at diagnosis significantly affect the prognosis of patients. Our study found that elevated LDH level at diagnosis as an independent prognostic factor negatively affected the OS, RFS, and GRFS. Partial remission status at transplantation leaded to a higher risk of relapse, a poorer RFS, and GRFS. Total body irradiation–based conditioning regimen as an independent prognostic factor significantly reduced the risk of disease recurrence, increased the OS and RFS. Interestingly, a trend toward haploidentical transplantation decreased the incidence of relapse as a prognostic factor, which indicated that a haploidentical donor should be preferred to a HLA-matched donor for higher risk T-LBL patients.
In conclusion, our data showed that adult T-LBL patients achieving CR at transplantation might have an excellent prognosis with TBI-based conditioning regimen. For patients in PR status, allo-HSCT might not be an optimal choice due to the high risk of relapse, but it could be operated after achieving CR with alternative strategies like as CD7 chimeric antigen T-cell therapy 57 , new drugs like venetoclax combined with chemotherapy 58 . The results suggested that TBI-based conditioning regimen might be an optimal regimen for adult T-LBL patients receiving allo-HSCT.
Our study also had some limitations for a retrospective study with a relative small number of sample, different donor sources and GvHD prophylaxis strategies, and a relative shorter follow-up. Thus, a well-designed prospective study with a larger sample size and a longer follow-up is required.
Supplemental Material
sj-docx-1-cll-10.1177_09636897221108890 – Supplemental material for Total Body Irradiation–Based Conditioning Regimen Improved the Survival of Adult Patients With T-Cell Lymphoblastic Lymphoma After Allogeneic Peripheral Blood Stem Cell Transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897221108890 for Total Body Irradiation–Based Conditioning Regimen Improved the Survival of Adult Patients With T-Cell Lymphoblastic Lymphoma After Allogeneic Peripheral Blood Stem Cell Transplantation by Jiahua Niu, Zhixiao Chen, Jie Gao, Huiying Qiu, Liping Wan, Ying Wang, Wenwei Wang, Yin Tong, Chongmei Huang, Yu Cai, Xiaowei Xu, Kun Zhou, Ying Zhang, Xinxin Xia, Chang Shen, Yu Wei, Tingfeng Chen, Xianmin Song and Jun Yang in Cell Transplantation
Footnotes
Author Contributions
Conception and design: Xianmin Song and Jun Yang
Provision of study materials or patients: All authors
Collection and assembly of data: Jiahua Niu, Zhixiao Chen
Analysis and interpretation: Jiahua Niu, Zhixiao Chen and Jie Gao
Manuscript writing: Jiahua Niu
Final approval of manuscript: All authors
Accountable for all aspects of the work: All authors
Ethical Approval
This study was approved by the Ethics Committee of the coordinating institution of Shanghai General Hospital (Code: 2022KY023).
Statement of Human and Animal Rights
This study was approved by the Ethics Committee of the coordinating institution of Shanghai General hospital (Code: 2022KY023).
Statement of Informed Consent
Informed consent was waived because we used datasets provided by Shanghai General Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Three-year development project from Shanghai Shen Kang Hospital Development Center (SHDC2020CR1012B, 16CR1010A for Xianmin Song, SHDC12018X09 for Jun Yang); Clinical Research Plan of SHDC (SHDC2020CR3028B for Chun Wang); Clinical Research Innovation Plan of Shanghai General Hospital (CTCCR-2018BP03 for Jun Yang, CTCCR-2018B02 for Chun Wang); Medical Guidance Project of Science and Technology Commission of Shanghai Municipality (18411968400 for Jun Yang); Clinical Research Special General Project of Shanghai Municipal Health and Family Planning Commission (201840043 for Jun Yang); and National Clinical Research Center for Hematologic Disease (2020ZKPC02 for Xianmin Song).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
