Abstract
Femoral head osteonecrosis (ON) is a serious complication of steroid administration. We examined whether implantation of autologous bone marrow-derived endothelial progenitor cells (EPCs) can augment neovascularization and bone regeneration in steroid-induced osteonecrosis of the femoral head. Forty 12-week-old male New-Zealand white rabbits were divided into group I (left untreated, n = 12), group II (core decompression, n = 12), and group III (core decompression + autologous EPCs implantation, n = 16) after receiving an established inductive protocol for inducing steroid-associated ON. Four weeks later, these rabbits were euthanized, bilateral femora were dissected for Micro-CT-based microangiography to assess vascularization, and then the osteonecrotic changes and repair processes were examined histopathologically. Quantitative analysis showed that new vessel formation in group III was significantly greater compared with other groups at 4 weeks after treatment. The histologic and histomorphometric analyses revealed that the new bone volume was significantly higher in group III than in groups I and II 4 weeks after treatment. A combination of EPCs and core decompression enhances the neovascularization and bone regeneration in rabbit steroid-induced femoral head ON. Local implantation of EPCs may provide a novel and effective therapeutic option for early corticosteroid-induced ON.
Introduction
Corticosteroid-induced osteonecrosis of the femoral head (ONFH) is one of the most serious complications in patients who have received corticosteroids as treatment for underlying diseases, such as systemic lupus erythematosus, nephrotic syndrome, and renal transplantation (30). Once the femoral head collapses, the hip joint tends to be destroyed as ONFH is generally localized to the weight-bearing area in younger adults. The precise pathomechanism of ONFH still remains unclear; ischemia is the assumed cause (3). Various surgical treatments aimed at hip preservation have attempted to induce revascularization or transposition of the necrotic area (5,25,31). However, these treatments were limited in their ability to support subchondral bone and were not necessarily successful.
Many studies suggest that implantation of endothelial progenitor cells into ischemic tissue can promote angiogenesis in peripheral artery disease such as myocardial infarction, cerebral and critical limb ischemia (4,8,12, 16,20,23,33). Recently, in bone fracture or defect model, local endothelial progenitor cell (EPC) implantation not only improved vascularization but also enhanced bone regeneration (26,40,46). However, the effect of EPCs on vascularization and bone regeneration in ONFH has not been studied.
Therefore, in the present study, we investigated whether local EPC implantation plays a beneficial role in vascularization and bone regeneration of femoral head in a steroid-induced rabbit model 4 weeks after EPC transplantation.
Materials and Methods
All experimental procedures adhered to the recommendations of the US Department of Health for the care and use of laboratory animals, and were approved by the Ethics Committee of Shanghai Jiao Tong University.
EPC Culture and Characterization
In vitro culture and expansion of EPCs was performed as previously described (46). Briefly, bone marrow (6 ml from each rabbit) was aspirated from the bilateral iliac crest of healthy New Zealand white rabbits (12 weeks old, 2–2.5 kg) with a 20-ml syringe containing 1000 units of heparin. Mononuclear cells were separated by density gradient centrifugation with Histopaque-1077 (Sigma). Isolated cells were seeded on culture plates and maintained in endothelial cell-basal medium supplemented with EGM-2MV (Clonectics). After 4 days in culture, nonadherent cells were removed by washing with phosphate-buffered saline (PBS) and new medium was applied thereafter. The medium was subsequently changed every 3 days for further cultivation. Cell colonies were selected and passaged when necessary. Third passage cells were used for all subsequent in vivo and in vitro studies. As a functional test, cultured EPCs were seeded onto 35-mm dishes (Becton Dickinson, USA). Twenty-four hours after seeding, the EGM-2 medium was replaced with fresh medium containing 10 mg/ml DiI complex and acetylated low-density lipoprotein (DiI-ac-LDL) (Molecular Probes, USA) according to the manufacturer's directions. After another 4 h of culturing, the cells were washed several times with PBS, and then fixed for 10 min with 2% paraformaldehyde. Cells were washed three times with PBS, incubated in EMG-2 medium containing 10 mg/ml ulex europaeus agglutinin (UEA) (Vector, USA) for I h, washed three times with PBS, and then observed under a Nikon (Tokyo, Japan) fluorescent microscope. DiI-ac-LDL and UEA were not used in the controls.
Confirmation of EPCs was performed by immunofluorescent staining using antibodies against endothelial cell markers such as von Willebrand factor (vWF, Antibody Corporation), CD31 (Antibody Corporation). Autologous rabbit EPCs were labeled with the green fluorescent marker PKH2-GL (Sigma) and then implanted, in order to examine whether implanted autologous rabbit EPCs survive at the implanted site.
Animal Osteonecrosis (ON) Model and Treatment Protocol
Forty 12-week-old male New Zealand white rabbits were housed at the Experimental Animal Center of the investigators' hospital and received a standard laboratory diet and water ad libitum. An ON model of the femoral head was established according to previous inductive protocols (40). In brief, one injection of 10 μg/kg body weight of lipopolysaccharide (LPS; Sigma) was given intravenously, and then three injections of 20 mg/kg body weight of methylprednisolone (MPS; Pfizer, USA) were given intramuscularly, at a time interval of 24 h. It was reported that ON gradually developed 6 weeks after injection of MPS, which was similar to stage II of ON clinically (Ficat and Arlet classification system). No rabbits died of the inductive protocol throughout the experiment period.
Six weeks after the last injection of MPS, the rabbits were randomly divided into three groups. Group I (n = 12) served as controls and did not receive any therapy. Group II (n = 12) only underwent bilateral core decompression of the femoral head. Group III (n = 16) underwent bilateral core decompression of the femoral head and autologous EPC transplantation. For the rabbits in group III, a total of 1 × 107 EPCs/each animal were resuspended in 1 ml PBS. Half of the composite solution (0.5 ml) was slowly injected into the right femoral head through the tunnel made by drill; another 0.5 ml was injected into the left side. The hole was sealed by an absorbable collagen sponge plug.
Then 4 weeks later, the femoral heads of all rabbits were assessed by microangiography and histological examination. All of the treated animals lost weight and several rabbits had to be killed or died precociously. Thus, only 10 of the 12 animals (20 femoral heads) in group II and 12 of the 16 animals (24 femoral heads) in group III were available for study.
Surgical Procedure
For the core decompression, the animals were anesthetized with intravenous pentobarbital and operated according to the modified previous technique (50). Normal saline was infused intravenously through an ear vein during the procedure. A standard lateral approach to expose the lateral aspect of the femur just distal to the greater trochanter was made under aseptic conditions. A drill with an outer diameter of 1 mm was inserted at the flare of the greater trochanter and into the femoral neck and head. The location was confirmed radiographically. Both sides of femoral head were performed. The wound was then closed layer by layer.
Assessment of Femoral Head Vascularization
Femoral head blood vascularization was measured using a new technique: the microCT technique. Five rabbits of each group were assessed at 4 weeks after treatment.
Perfusion and Decalcification.
Under general anesthesia with 3% sodium pentobarbital (1 ml/kg), the abdomen cavity of the animals was opened (Fig. 1A), and a scurf needle with 25-mm syringe was inserted in the abdominal aorta distal to the heart with ligation of that proximal to the heart. The vasculature was flushed with 50 U/ml heparinized normal saline at 37°C and at a flow speed of 20 mm/min via a syringe. As soon as the outflow from an incision of the abdominal vein was limpidness, 10% neutral buffered formalin (37°C) was pumped into the vasculature to fix the nourished skeletal specimen. The formalin was then flushed from the vasculature using heparinized normal saline, and the vasculature was injected with Microfil, a lead chromate-containing confected radiopaque silicone rubber compound based on the manufacturer's protocol (Microfil MV-122, Flow Tech; Carver, MA, USA) (Fig. 1B). Animals were then euthanized with an overdose of sodium pentobarbital and stored at 4°C for 1 h to ensure polymerization of the contrast agent before microangiography. Bilateral femoral samples were then harvested and fixed in paraformaldehyde (4%) for decalcification with ethylenediaminetetraacetic acid (EDTA, 10%, pH 7.4). Success of decalcification was confirmed by anteroposterior view radiographs taken using a cabinet X-ray system (Specimen Radiography System, Faxitron 43855C, Fraxitron X-ray Corporation, Wheeling, IL, USA) under an exposure condition of 40 kV/30 s. Then both proximal 1/3 of bilateral femoral samples of each group were obtained for evaluations.

Surgical technique for microfil infusion and visualization of vessel networks. (A) Exposure of the abdominal aorta (arrow) for cannulation and injection of heparin. (B) Direct visualization of microfil contrast agent within the intestine or other arteries.
Microangiography.
With the help of one experienced microCT application specialist, the proximal parts of the bilateral femoral samples of each rabbit were placed into a polymethylmethacrylat (PMMA) sample tube, respectively. The femoral shaft was fixed in the tube with its long axis perpendicular to the bottom of the tube for microCT scanning using microCT (GE, USA). The scan was performed at a resolution of 36 μm per voxel with 1024 × 1024 pixel image matrix. For segmentation of blood vessels from background, noise was removed using a low pass Gaussian filter (Sigma = 1.2, Support = 2) and blood vessels were then defined at a threshold of 85. In order to reconstruct the three-dimensional (3D) architecture of vasculature in the proximal femur, the blood vessels filled with microfil were included with semiautomatically drawn contour at each two-dimensional (2D) section by built-in “Contouring Program” for automatic reconstruction of 3D image of vasculature in the decalcified sample. In addition, axial slices through the samples were sequentially visualized and the number of vessels (main trunk only) penetrating the bone vessels was collated. All voxels counted in the specified microfil range represented the vessel volume (VV). After microangiography, the decalcified samples were embedded in paraffin and cut into 5-μm-thick sections along the coronal plane for the proximal parts. Sections were stained with hematoxylin-eosin (H&E) for evaluation of ON and repair process.
Tissue Preparation and Assessment of Bone Regeneration
Bone regeneration at the implanted sites was assessed by the density of new bone. Five rabbits in each experimental group were sacrificed at 4 weeks, and bilateral proximal one-third femurs were harvested. The bone specimens were decalcified in 10% EDTA (pH 7.4) and embedded in paraffin. Sagittal plane sections of the femoral head (thickness 5 μm) were prepared and stained with H&E before being examined with a light microscope. Five random fields of each section were pictured; new bone was measured with image-analysis software (NIH image) and new bone density was defined as the ratio of new bone area to total implant area × 100. Osteoid tissue was excluded from new bone calculations, because the border between the new bone and osteoid tissue is unclear, and inclusion of osteoid tissue leads to overestimation.
Statistical Analysis
Data are expressed as mean ± SD. Statistical analysis of the data was performed using ANOVA with a Bonferroni post hoc analysis for multiple analysis and Pearson correlation coefficients were calculated using GraphPad Instat Software (GraphPad Software, Inc., San Diego, CA, USA). The results were taken to be significant at a probability level of p < 0.05.
Results
Characterization of EPCs
Freshly isolated EPCs appeared round and were of various sizes. Typical cell colony-forming units were found 6 days after culture. The adherent cells presented a typical spindle shape (Fig. 2). The clusters came into contact with each other by 7–8 days, and cell confluence was achieved by 10–12 days. At 17–21 days after culture, 1.14 ± 0.12 × 107 cells were harvested. The third passage of EPCs showed the typical cobblestone appearance characteristic of an endothelial monolayer (Fig. 3A). These cell colonies also ingested Ac-LDL and bound UEA-1 (Fig. 3B, C). These cells expressed the typical endothelial cell phenotypic markers, vWF (Fig. 4A) and CD31 (Fig. 4B).

After 6 days, the primary passage of EPCs to form typical cell colony-forming units. The adherent cells presented a typical spindle shape. Scale bar: 100 μm.

The characteristics of the third passage of EPCs. (A) The typical cobblestone appearance characteristic of endothelial cells. (B) EPCs labeled with UEA-1. (C) EPCs took up Dil-Ac-LDL. Scale bars: 10 μm (A), 20 μm (B,C).
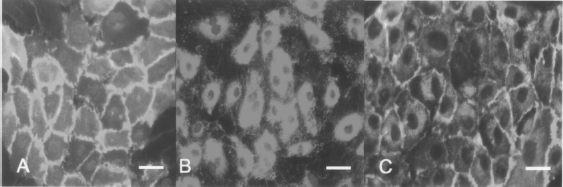
Immunofluorescent staining of EPCs. (A) Immunofluorescent staining by anti-vWF. (B) Immunofluorescent staining by anti-CD31. (C) Dual positive vWF and CD31. The cells were counterstained by DAPI. Scale bars: 20 μm.
Assessment of Femoral Head Neovascularization
We examined whether transplanted bone marrow-derived EPCs survived at the implanted site in steroid-induced osteonecrotic femoral head in rabbits. Before transplantation, third passage EPCs were isolated and labeled with the green fluorescent marker PKH2-GL according to the manufacturer's instruction (Sigma). On the day of surgery, rabbits in group III (n = 16) underwent bilateral core decompression of the femoral head and autologous green fluorescence-labeled bone marrow-derived EPCs (1 × 107 cells/animal) in the ischemic osteonecrotic femoral head. At 4 weeks after cell implantation, frozen tissue sections were prepared from the ischemic tissues. Fluorescence microscopic examination revealed that labeled EPCs were distributed and survived in the femoral head at the implant site 4 weeks after implantation (Fig. 5). The implanted EPCs contributed to the formation of new vessels.
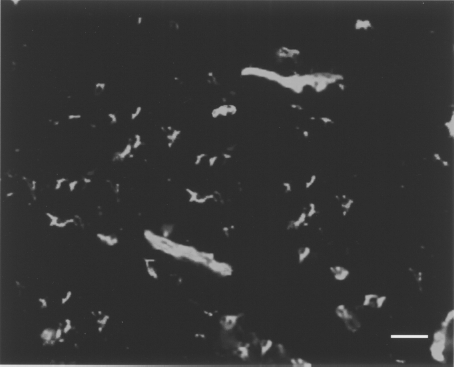
Detection of fluorescently labeled EPCs 4 weeks after implantation. Examination with fluorescence microscopy revealed that cells labeled with a fluorescent dye prior to implantation were still present in implant sites in the group of EPCs implantation. Scale bar: 20 μm.
Blood vessel microarchitecture of each group was reconstructed in three dimensions for presentation. The samples of group I showed that these vasculatures were not visible in and around the necrotic lesion of femoral head, the samples of group II showed lightly increasing capillary vessels, and the samples of group III showed intensive vascular architecture (Fig. 6). Quantitatively, Figure 7A shows the average number of vessels penetrating the proximal femur at 4 weeks after treatment. It was 8.2 ± 0.51 in group I, 16.1 ± 0.74 in group II, and 41.3 ± 3.32 in group III. Penetrating capillary vessels number between group III and group II showed apparent difference. Capillary vessel number in group III was significantly larger than that of group II and group I (p < 0.001). Capillary vessel number in group II was also significantly large compared with group I (p < 0.01). Figure 7B shows similar results.

Representative images of microCT reconstructed 3D microangiography of proximal femur from group I (A), group II (B), and group III (C) rabbits. Compared with other groups, the density of vessels in the femoral head of group III was obviously increased. Scale bars: 5 mm.

MicroCT quantification of new vessel formation in the femoral head. Mean number of blood vessels (A) and vessel volume (B) of group III were significantly higher than that of other groups (∗p < 0.05).
Formation of New Bone Surrounding the Necrotic Zone
Subchondral bone of the femoral heads in group I showed sparser trabecular bone with empty lacunae or pyknotic nuclei of osteocytes, and the medulla was full of fatty cells. In the rabbits of group II, empty lacunae and pyknotic nuclei of osteocytes within the bone trabeculae were also observed, but some of the marrow fat cells decreased in size compared to that of group I, and capillary formation, hemorrhage in the medulla, and osteoblasts were observed (Fig. 8A, B), but new bone formation was not prominent.

Histological findings of the femoral head 4 weeks after treatment. (A) Representative photomicrograph from group I showed massive lamellar trabeculae had become empty, surrounded by more marrow fat cells with increased size dominantly occupying marrow space. (B) In group II, limited repair appositional bone formation with lining cells around the necrotic bone and hemorrhage in the medullary cavity was observed. (C) Photomicrographs showing development of new bone in group III, 4 weeks after treatment of EPCs. Scale bars: 20 μm.
The histologic observations suggested that EPCs had potential to regenerate bone and repair the ON. In group III, the reparative response resulted in progressive development of a reactive margin, or interface, between the dead zone and adjacent viable tissues (Fig. 8C). During the repair process of ON, primitive mesenchymal cells and capillaries proliferated and invaded dead trabecular bone, differentiated into osteoblasts, and laid down new living bone surrounding dead trabecular bone, which was later remodeled. Histomorphometric analysis revealed that the new bone volume was significantly higher in group III than in groups I and II at 4 weeks (Table 1).
Comparisons of New Bone Volume Among Three Groups After 4 Weeks

p < 0.001 compared to group I.
p < 0.01 compared to group II.
Discussion
ONFH is a painful disorder of the hip that often leads, in its final stage, to femoral head collapse, osteoarthritis, and the need for total hip replacement (39). Steroid use is among the most widely recognized risk factors for osteonecrosis (18,29). The pathophysiology of ONFH is uncertain for most cases, with speculation of vascular impairment and changes in cell biology (38,49). To date, the best approach for precollapse steroid-induced ONFH remains unanswered. Vascularized fibular grafts have demonstrated the highest rates of success in treating early stage ONFH by establishing a new source of circulating blood, but it is a complex procedure with a higher rate of morbidity (1,52). In recent years, there has been a growing interest in the implantation of bone marrow mononuclear cells for ONFH, and its clinical effectiveness has been demonstrated by several studies. Some researchers thought that the efficacy of such implantation was due to a supply of EPCs included in bone-marrow mononuclear cells (2,10,15,17,47,51). More recently, Séguin et al. found that nontraumatic (mainly steroid-induced) ON may well be associated with regional endothelial dysfunction. Endothelial cell activation, apoptosis, or injury appear to be possible explanations for the development of microcirculatory thrombus at the level of microvascular environment of bone and then ischemic death of the osteocytes, suggesting that ON might be a disease of endothelial cells (43).
There have been some experimental studies of implantation aimed at promoting vascularization in patients with ischemic disease using EPCs (12,21,35,36). However, there has been no previous research on EPC implantation aimed at improving neovascularization and bone regeneration in the ONFH. Therefore, we investigated whether EPCs could induce neovascularization and subsequent bone regeneration in rabbit steroid induced femoral head ON.
Our study showed that local implantation of EPCs could increase vascularization and promote bone regeneration in steroid-induced ON of the femoral head in rabbit model.
The current study applied a novel technique, using microCT imaging, to visualize and quantify new blood vessel formation and vascularization in rabbit femoral head. Traditionally, approaches to measure vessel formation and structure have relied on 2D qualitative strategies. In recent years, contrast-enhanced microCT has been utilized to assess tissue microvasculature networks in many fields. This technique is quantitative and effective for assessing vascularization (6,13,24,27,32,48). Here, we quantified vessel formation within the femoral head by applying this technique. This study demonstrated a significant increase in blood vessel volume and penetrating vessel number in group III compared to groups I and II. This result was similar to Tan et al.'s report (46). In their study, bone marrow-derived endothelial cells significantly promoted neovascularization in the rabbit bone defect model. In addition, endothelial cells derived from bone marrow, peripheral and umbilical blood have been demonstrated to greatly promote new vessel formation in ischemic tissues as well as local blood flow (4,12, 20,21,33). We believe that there are several reasons to account for the improved vascularization. First, the implanted EPCs could survive and contribute to blood vessels formation (9,22, 41,42,53). Second, multiple angiogenic growth factor (such as VEGF) secreted from implanted endothelial cells could enhance angiogenesis (14,28,45,54). Bone marrow-derived endothelial cells could be a promising cell source for vascularization. The use of EPCs as the cell source is advantageous; EPCs can be harvested easily and exhibit strong proliferation potential. Additionally, harvesting bone marrow cells is generally considered to be less invasive than the harvest of vascular cells from autologous blood vessels (7,22).
The bone regeneration in group III was more rapid and of greater integrity than that of the control group. One likely explanation for this finding is the promotion of neovascularization. New blood vessels are intimately involved in ON because blood provides oxygen, nutrients, and growth factors and removes waste products. A study has shown a direct correlation between angiogenesis and bone repair in models of bone damage (44). Adding VEGF also leads to a significant increase in bone formation, which indicates that induced angiogenesis could enhance tissue regeneration (11). Another reason is synergic effects on bone formation between endothelial cells and osteoblasts (19).
Chronic pain while walking and functional limitation are some of the clinical manifestations of ON in most patients. The goal of management of the patient with ON is control of pain and improvement in function and quality of life, so evaluation of pain plays important role in the treatment of ON. In animal experiments, behavior assessment is crucial to the process of evaluation of pain and discomfort. Because behavioral responses to pain are influenced by many factors including species, breed, age, sex, source of pain, and coexisting disease, many systems for measuring pain have been developed and used for different species and location. Ochiai et al. applied behavior test to indicate that extracorporeal shock wave therapy is a useful treatment for knee osteoarthritis of rat (37). Neugebauer et al. suggested that behavioral measurements of knee joint pain associated with arthritis in animal models include indirect (weight bearing, gait analysis, spontaneous mobility, and sensitivity of the paw to von Frey filaments or heat) and more direct measurements (probing the sensitivity of the knee, knee extension angle, and vocalizations evoked by stimulation of the knee) (34). However, as so far there are no validated behavioral systems for measuring pain from hip ON in rabbits, we could not accurately evaluate the effect of local EPC transplantation on pain of the hip in rabbits. Considering behavioral assessment of models of ON is important in testing treatments, we will focus on this aspect in the next study.
Indeed, untreated ON of the femoral head generally results in a progressive course of subchondral fracture, collapse, and painful disabling arthritis. Many patients with AVN present later, often times with large lesion and secondary collapse. The treatment of late-stage ON has been a challenge to orthopedic surgeons. In the present, the application of femoral head-preserving procedures, such as core decompression, electrical stimulation, transtrochanteric rotational osteotomy, and bone marrow cell implantation mainly focus on early stage, small lesions, and precollapse cases. Among them, the use of bone marrow cells was initiated in an effort to enhance revascularization and to arrest the progression of necrosis. Many authors have reported good results with the technique for early stage ON (Ficat stage I–II). In the present study, because the bone marrow-derived EPC transplantation could be considered to be one of bone marrow cell implantation, we also selected early stage ON as our study objective. The present study strongly suggests that a combination of autologous EPC transplantation and core decompression is associated with better histological and radiographic results than is core decompression in precollapse hips, particularly those with Steinberg Stage-II disease in an animal model. The technique of local implantation of EPCs may provide a novel and effective therapeutic option for early corticosteroid-induced ON in a clinical situation. For chronic AVN, especially postcollapse patients, we cannot comment as to whether this technique iss appropriate for them.
Conclusions
Implantation of EPCs could increase vascularization and bone regeneration in the early stages of ON of the femoral head in a rabbit model. Local implantation of EPCs into a necrotic lesion of the femoral head may provide a novel and effective therapeutic option for early corticosteroid-induced ON.
Footnotes
Acknowledgment
This study was supported by the Key Scientific Foundation of Health Ministry, P. R. China (200802029).
