Abstract
Bone marrow and tissue precursor cells have been postulated to replenish grafts of transplanted islets. Several investigators have reported that bone marrow cells can promote the regeneration of injured islets. In this study, we investigated the potential of recipient-derived precursor cells to form new pancreatic endocrine cells in islet grafts transplanted under the kidney capsule. Mouse insulin promoter (MIP)-green fluorescence protein (GFP) mice, which express GFP only in β-cells, or β-actin GFP mice, which express GFP ubiquitously, were used to determine if the recipient-derived cells differentiate into β-cells or other types of endocrine cells. We transplanted MIP-GFP islets into wild-type mice, wild-type islets into MIP-GFP mice, β-actin GFP islets into wild-type mice, and wild-type islets into β-actin GFP mice. β-Actin GFP bone marrow cells were then injected into wild-type mice to evaluate the potential role of bone marrow stem cells to provide new islet cells to the graft. No β-cells with green fluorescence were seen in the graft when wild-type islets were transplanted into MIP-GFP mice. When wild-type islets were transplanted into β-actin GFP mice, no β-cells with GFP staining could be identified in the grafts. Similarly, no endocrine cells with GFP staining could be identified in the grafts after injection of β-actin GFP bone marrow cells into wild-type islet-transplanted wild-type mice. This study provides further support for the concept that recipient precursor cells do not produce new β-cells in grafts of transplanted islets.
Introduction
There has been great interest in whether there are circulating precursor or adult stem cells that replenish the endocrine pancreas. Bone marrow cells have been thought by some to be capable of differentiating into not only hematopoietic cells but also multiple cell types such as cardiomyocytes, hepatocytes, neural cells, and endothelial cells (1,3,13,17,18,22,29,30,34). However, others find little evidence for such plasticity (35). One study provides strong arguments that bone marrow-derived cells only adopt a hepatocyte phenotype as a consequence of rare cellular fusion events (36). The ability of bone marrow cells to differentiate into pancreatic endocrine cells is also controversial. Jiang et al. reported that bone marrow multipotent adult progenitor cells (MAPCs) have potential to differentiate into β-cells (14), and others find that mesenchymal stem cells can express β-cell markers (21,26). Ianus et al. provided some evidence that transplanted bone marrow stem cells transdifferentiate into β-cells without cell fusion (11), but these findings have not been confirmed (10,19,33). Hess et al., however, did find that bone marrow cell injections could improve hyperglycemia induced by low-dose streptozocin (STZ), but not by serving as β-cell precursors (10). Others find similar beneficial effects (2,9,12,20,32). There are also several papers reporting that bone marrow-derived cells transdifferentiate to become endothelial cells but not β-cells in STZ-induced diabetic mice (4,23). In these in vivo studies, bone marrow-derived cells were found in sites of injured tissue, suggesting that they may interact with some injury factor to enhance β-cell mass and/or function.
Previously, we reported the vulnerability of transplanted islets during the peritransplant period in a syngeneic model, presumably due to the combined effects of hypoxia and hyperglycemia (6). During this period, both the apoptotic and protective genes are unregulated in the transplanted islets (31). After an initial marked disruption of islet architecture, an impressive remodeling takes place by 14 days after transplantation. We know that β-cell replication takes place in islet grafts to provide new β-cells (24,25), but it is uncertain if replenishment can be provided by recipient-derived cells.
In this study, we investigated whether recipient circulating cells from bone marrow or elsewhere, or cells from tissues surrounding the grafts could contribute new β-cells to the islet grafts, a model of islet injury. Two different kinds of transgenic mice that express green fluorescent protein (GFP) were employed, MIP-GFP and β-actin GFP mice. MIP-GFP mice express enhanced GFP (EGFP) only in β-cells under the control of mouse insulin 1 gene promoter (8). β-Actin GFP mice express EGFP under the control of chicken β-actin promoter and cytomegalovirus enhancer in all of the tissues with the exception of hair, nails, and erythrocytes (28). Using these two transgenic strains, we show that recipient cells are highly unlikely to serve as precursors for β-cell replenishment in transplanted islets.
Materials and Methods
Animals
C57BL/6 mice and chicken β-actin promoter GFP mice [C57BL/6-TgN(ACTbEGFP)10sb] were purchased from The Jackson Laboratories (Bar Harbor, ME) at 6–8 weeks of age. MIP-GFP mice were generated and kindly provided by Manami Hara (University of Chicago) (8). All mice were bred under conventional conditions in an air-conditioned room with free access to tap water and standard pelleted chow. β-Actin GFP and MIP-GFP mice were generated by mating male β-actin GFP and MIP-GFP mice with female C57BL/6 mice. MIP-GFP mice had been backcrossed with C57BL/6 mice for more than eight generations, and no immune rejection was observed after transplantation. Therefore, all of the mice used in the experiment had the same genetic background (C57BL/6) and are syngeneic, sufficiently identical, and immunologically compatible as to allow for transplantation to each other. Diabetes was induced by intraperitoneal injection of 180–250 mg/kg streptozocin (STZ, Sigma, St. Louis, MO) freshly dissolved in an equivalent volume of 0.05 M citrate buffer (pH 4.5) into the 6–8-week-old mice. Diabetes was confirmed by the presence of hyperglycemia with fed blood glucose levels higher than 350 mg/dl. Glucose levels were determined with a portable glucose meter (Precision, Abbott Laboratories, Bedford, MA). All animal experiments were approved by the Joslin Institutional Animal Care and Use Committee (IACUC).
Islet Isolation, Bone Marrow Cell Isolation, Islet Transplantation, and Bone Marrow Cell Injection
The islet isolation method has been described previously (7). Briefly, pancreases were distended with 3 ml of cold M199 medium (Gibco BRL, Grand Island, NY) containing 1.5 mg/dl of Liberase™ (Roche Diagnostics, Indianapolis, IN), excised, and incubated in a stationary bath at 37°C. Islets were separated by density gradients (Histopaque-1077; Sigma) and handpicked under a stereomicroscope. Bone marrow cells were obtained from the femurs and tibias of donors at 4–6 weeks of age. The ends of the removed bones were cut and bone marrow cells were collected by forcing Hank's solution with 1% fetal calf serum into a needle inserted in the bone shaft. Red blood cells were lysed by cell lysis buffer (Sigma) and filtrated through 70-μm pore cell strainer (BD Biosciences, MA) to purify bone marrow cells. Bone marrow cells were centrifuged, resuspended in 1000 μl Hank's solution, and then placed on ice until transplanted. Cell numbers were counted under a microscopy using Burker-Turk plate (VWR, Bridgeport, NJ).
Islet transplantation and bone marrow injections were carried out 10 days after STZ injection. Islets isolated from syngeneic mice were transplanted under the left kidney capsule of diabetic mice as previously described (6). Briefly, 300 islets were packed in a capillary tube. Recipients were anesthetized by methoxyflurane, and the left kidney was exposed through a lumbar incision. A capsulotomy was performed in a lower pole of the kidney, and the capillary tube was advanced under the capsule to the upper pole, where the islets were injected. In simultaneous bone marrow injection experiments, 0.6–1.0 × 107 bone marrow cells isolated from β-actin GFP mice were injected into retro-orbital cavity through a 26-gauge fine needle. After transplantation, blood glucose levels were measured twice a week between 1000 and 1200 h. Blood was obtained by snipping tails with a sharp razor blade.
Analysis of mRNA Expression
Islet grafts were removed with left kidney from the recipient mice using pentobarbital anesthesia. The grafts were then dissected from the kidney, and after homogenization, total RNA was extracted using RNeasy kit (Quiagen, Valencia, CA). cDNA was synthesized by reverse transcription (RT), using 1 μg total RNA and Superscript II reverse transcriptase (Invitrogen, Carlsbad, CA). Insulin cDNA and EGFP cDNA were amplified by PCR using Goldstar Taq DNA polymerase (Eurogentec, San Diego, CA) and specifically designed forward and reverse primers 5′-TCTTCTACACACCCATGTCCC-3′ and 5′-GGTGCAGCACTGATCTAC-3′, respectively, for insulin, and 5′-TGGAAACTGCAGCTTCAG-3′ and 5′-GTCCAGCTCGACCAGGATGG-3′ for EGFP, respectively. Genomic DNA contamination was checked by PCR for insulin using extracted total RNA without RT and cDNA after RT as samples for 40 cycles, and examining the product size by electrophoresis. If genomic DNA contamination was found, the total RNA sample before RT was treated by DNase for 30 min, and then RT was carried out.
Histology
The islet graft surrounded by kidney tissue fixed was fixed in 4% paraformaldehyde for 2 h, then embedded in paraffin and sliced into 3-μm sections. After deparaffinization, diluted primary antibodies were added to sections following blocking and the sections were incubated overnight at 4°C. The next day, primary antibodies were washed and the matching mix of fluorophore-conjugated secondary antibodies was added for 1–3 h, and then excess secondary antibodies were washed off with PBS. Sections were evaluated and photomicrographs were taken using a confocal microscope (LSM-410, Carl Zeiss, Germany).
Antibody identity, source, and dilution are as follows: guinea pig anti-insulin (1:200, Linco Research Immunoassay, St. Charles, MO), guinea pig anti-glucagon (1: 3000, Linco Research Immunoassay), and rabbit anti-GFP (1:1000, Clontech, Mountain view, CA). The conjugated secondary antibodies are: Texas red-conjugated donkey anti-guinea pig IgG, and FITC-conjugated donkey anti-rabbit IgG (1:200 dilution for all, Jackson ImmunoResearch Laboratories, West Grove, PA).
Statistical Analysis
Results are presented as mean ± SEM. Statistical significance was evaluated with the unpaired Student's t-test. A value of p < 0.05 was considered significant.
Results
Experimental Design and Animal Characteristics
To evaluate the possibility that the host-derived precursor cells replenish endocrine cells in islet graft, we used the experimental plan shown in Figure 1. Diabetic mice were produced with STZ, and 10 days after injection when diabetes was confirmed, 300 islets were transplanted under the kidney capsule. Five types of islet transplants using three mouse strains (wild-type, MIP-GFP, and β-actin GFP mice) were performed (Fig. 1B). Briefly, MIP-GFP islets were transplanted into wild-type mice and wild islets were transplanted into MIP-GFP mice. The blood glucose levels were normalized after the transplantation throughout the observation period in the latter group (Fig. 1C). Similarly, using β-actin GFP mouse, two converse combinations of groups were designed. When β-actin GFP islets were transplanted into wild-type diabetic mice, blood glucose levels decreased slowly until 4 weeks after transplantation, suggesting that β-actin GFP islets are less efficacious than wild-type islets; the latter immediately normalized blood glucose levels after transplantation when transplanted into β-actin diabetic mice (Fig. 1D). Questions could be raised about xenogeneic GFP protein eliciting an immune response, but only rare macrophages were found. It seems unlikely that an immune response would have accounted for the lack of rapid achievement of normoglycemia. However, we cannot rule out the possibility that GFP protein somehow reduced insulin production from the transplanted islet cells.
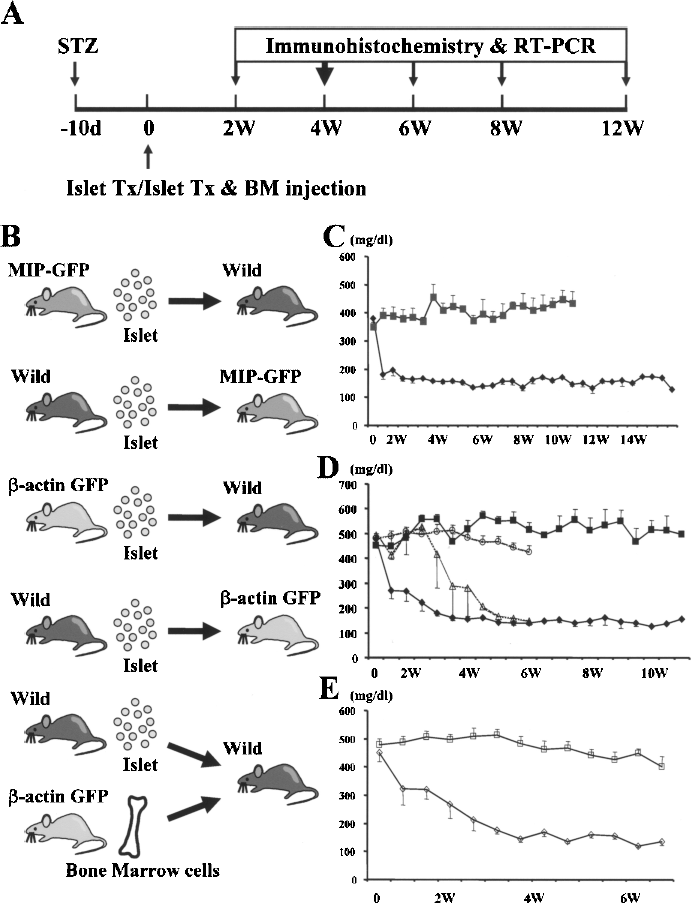
Time course of experiments of mice receiving islets and/or bone marrow cells. (A) Experimental design for the induction of diabetes, islet/bone marrow transplantation, and evaluation. (B) Combinations of the donors and the recipients. (C) Comparison of blood glucose levels of STZ-treated MIP-GFP mice (closed squares) and MIP-GFP mice transplanted with islets from wild-type B6 mice (closed diamonds), n = 8 and 9, respectively. As discussed in Results, the grafts from these mice were examined to assess the GFP-FITC staining. On the basis of the heterogeneous staining that we found, we decided to rely more heavily on β-actin GFP. (D) Comparison of blood glucose levels of STZ-treated wild-type mice (closed squares), STZ-treated β-actin GFP mice (open circles), β-actin GFP diabetic mice transplanted with wild-type islets (closed diamonds), and wild-type diabetic mice transplanted with β-actin GFP islets (open triangles), n = 10, 6, 10, and 3, respectively. (E) Comparison of blood glucose levels in STZ-treated wild mice (open squares), and STZ-treated wild mice that received simultaneous β-actin GFP bone marrow injection and wild-type islet transplantation (open triangles), n = 6 and 7, respectively. The y-axes for (C), (D), and (E) are glucose mg/dl.
We made another group in which wild-type islets were transplanted under the kidney capsule of wild-type mice, and simultaneously β-actin GFP bone marrow was transplanted into the retro-orbital space. Blood glucose levels gradually decreased after transplantation and remained normal throughout the rest of the observation period (Fig. 1E). Four to 10 weeks after transplantation, the kidney containing the islet graft was removed; then each graft was evaluated histologically or evaluated for mRNA expression by RT-PCR.
Transplantation of Islets From MIP-GFP Mice and β-Actin GFP Mice Into B6 Wild-Type Mice
At first, as a pilot study, we transplanted MIP-GFP islets into wild-type mice. In this model, β-cells in islet grafts without green fluorescence could be considered to be recipient derived. However, as shown in Figure 2A–C, even when stained with anti-GFP labeled by FITC there were many β-cells that had minimal if any GFP-FITC staining, but stained strongly for insulin. Therefore, we looked at the islets in native pancreas of MIP-GFP mice. Similarly, even in pancreatic islets, some β-cells have weak or no green fluorescence (Fig. 2D–F). The explanation for this heterogeneous GFP staining in β-cells of MIP-GFP mice is unclear. Of note, the GFP-FITC staining of β-cells in the graft and the native pancreas was typically stronger in nuclei than cytoplasm.

GFP expression in the islets of MIP-GFP mice and in the transplanted islet grafts. (A–F) GFP staining labeled by anti-GFP antibody with FITC, insulin staining labeled by anti-insulin antibody with Texas-Red and the merged image in the graft of MIP-GFP islets transplanted into wild-type mouse (A, B, C). GFP, insulin staining labeled by FITC and Texas-Red, and the merged image of the MIP-GFP pancreatic islet (D, E, F). The area surrounded by dotted line shows the cells stained with insulin but do not express GFP. (G, H, I) GFP staining labeled by FITC and Texas-Red, and the merged image of β-actin GFP islets transplanted into wild-type mouse. Scale bar: 10 μm.
To follow up on results obtained from transplantation with MIP-GFP islets, we examined the expression level of GFP in β-actin GFP islets by transplanting β-actin GFP islets into wild-type mice and staining with GFP labeled by FITC. Comparison of direct GFP fluorescence and FITC-stained GFP fluorescence showed no evident differences in staining intensity between cells (Fig. 2G–I). Thus, β-actin mouse tissues have enough GFP expression to evaluate the origin of the cells when transplanted into wild-type mice and have some advantage over the MIP-GFP mice.
Wild-Type Islet Transplantation Into MIP-GFP Mice
As a different approach, we transplanted wild-type islets into MIP-GFP mice. In this model, only the host-derived β-cells have green fluorescence; therefore, if β-cells in the graft have green fluorescence, they are from host-derived precursor cells. The islet grafts were evaluated histologically at 3 and 6 weeks after transplantation. In total, 48 sections, 8 to 15 sections from each of four mice, were examined and more than 25,000 insulin-positive cells were examined (Fig. 3A); no GFP-positive cells could be found in the graft (Fig. 3B). Moreover, no overlap of green and red (insulin) was found in the merged images of GFP and insulin (Fig. 3D). A few green cells could be found, but these cells were not stained with insulin and appeared to be in blood vessels, suggesting they are blood cells with autofluorescence or were stained for GFP nonspecifically.

Wild-type islets were transplanted into MIP-GFP mice. (A) GFP labeled by FITC. (B) Insulin staining labeled by Texas-Red and (C) the merged image of the graft. Scale bar: 10 μm. (D) The number of the GFP-positive cells and the total examined cells. Total cell numbers were calculated by counting nuclei of the insulin-positive cells in the graft in two sections and multiplying the average with the total numbers of the examined sections in each animal. (E) Insulin and GFP mRNA expression levels in the graft at 6 weeks after transplantation. PCR cycle numbers are 30 for insulin and 35 for GFP.
Insulin and GFP expressions in the graft were also examined at the mRNA level with RT-PCR (Fig. 3E), and no GFP mRNA was detected 6 weeks after islet transplantation, indicating that no new β-cells were provided to the graft by the recipient.
Islet Transplantation Using β-Actin GFP Mice
Next, β-actin GFP islets were transplanted into wild-type mice and, as a converse combination, wild-type islets were transplanted into β-actin GFP mice. Both β-cells and α-cells were evaluated with immunostaining. In this situation, all insulin-positive cells had GFP signal when β-actin GFP islets were transplanted into wild-type mice (Fig. 4). GFP signals seemed to be missing in some α-cells, raising the possibility that some of the α-cells were replenished by recipient-derived cells or, more likely, that GFP expression levels were lower in those α-cells (Fig. 4B). Therefore, in order to confirm the result obtained from insulin staining and to examine the possibility of α-cells being replenished by the recipient, the same evaluation was performed using wild-type islets transplanted into β-actin GFP mice. A total of 32 sections (5 to 8 sections from each of 5 mice), which was approximately 24,000 cells of all types in the graft were examined; in this model, several GFP-positive cells seen in the graft, but none were stained for insulin (Fig. 4C, D). The majority of these GFP-positive cells had a long narrow shape, suggesting they were endothelial or fibroblastoid cells. Only two GFP-positive round-shaped cells in the 24,000 cells were found adjacent to a cluster of β-cells seen in Figure 4C; however, these cells were not stained with insulin, suggesting that they were macrophages derived from the recipient. There was one cell that seemed to co-stain for GFP (green) and glucagon (red), as seen in Figure 4E. However, we suspect this was a mast cell that had autofluorescence, which allowed it to appear both red or green, depending upon which filter was used. We found such occasional cells in another study and confirmed the mast cell identity with histamine staining (data unpublished).

Graft of β-actin GFP islets transplanted into wild-type mice and wild-type islets transplanted into β-actin GFP mice. (A, C) GFP and insulin staining and merged image of the graft of (A) β-actin GFP islets transplanted into wild-type mice, and (C) wild-type islets transplanted into β-actin GFP mice. GFP was labeled with FITC and insulin was labeled with Texas-Red. (B, D, E) α-Cells were examined by glucagon staining labeled with Texas-Red in the graft of (B) β-actin GFP islets transplanted into wild-type mice and (D, E) wild-type islets transplanted into β-actin GFP mice. (E) From a different field of (D). Scale bar: 10 μm.
Bone Marrow Cell Injection From β-Actin GFP Mouse Into Wild-Type Mice
To examine if bone marrow stem cells replenish islet grafts, β-actin GFP bone marrow cells were injected into the retro-orbital space of wild-type mice. Injected bone marrow cells had survived in the blood until at least 3 weeks after injection when confirmed by PCR; however, 6 weeks after injection, GFP gene expression was no longer detected (Fig. 5A). Therefore, histological experiments were carried out within 4 weeks after transplantation. In this model, GFP cells were rarely seen in the graft, and the majority of the GFP-positive cells had narrow shape. Twenty-two sections from three mice with an approximately 16,500 cells in the graft were examined, but no GFP-positive cells were double stained with insulin or glucagon, indicating that neither β-cells nor α-cells were replenished by bone marrow-derived cells (Fig. 5B).

β-Actin GFP bone marrow cells were injected into wild-type mouse with simultaneous wild-type islet transplantation. (A) RT-PCR was performed for GFP mRNA using peripheral blood as a sample. Samples were taken 3 and 6 weeks after bone marrow injection. PCR cycle number was 35. (B) GFP and insulin staining labeled with FITC and Texas-Red and merged image. α-Cells were examined by glucagon staining labeled with Texas-Red. Scale bar: 10 μm.
Discussion
Our results provide evidence that recipient-derived precursor cells do not replenish endocrine cells in grafts containing transplanted islets. Bone marrow transplantation has been reported to have an antidiabetic effect in STZ diabetic mice (2,9,10,12,20,32), and several groups provided evidence that bone marrow cells contain not only hematopoietic stem cells but mesenchymal stem cells that can differentiate into various tissue-specific cells and even into β-cells or β-like cells (11,14). Not only bone marrow cells, but also tissue-specific cells such as splenocytes and hepatocytes, were reported to have potential to replenish the decreased mass of β-cells in diabetic state (16,37). However, efforts to confirm the splenocyte work have been unsuccessful (27). These findings and questions led us to explore the possibility that circulating bone marrow stem cells and/or the tissues surrounding the graft could become a source of new β-cells in islet grafts. To detect the origin of newly formed cells, we used two types of transgenic mice. Wild-type islets were transplanted into MIP-GFP mice to search for green recipient-derived β-cells in islet grafts. Knowing that a small population of β-cells of MIP-GFP mice express little if any GFP, we examined a very large number of cells (25,000 β-cells) in transplanted grafts and also determined mRNA GFP expression levels. We found not a single cell that expressed both GFP and insulin; moreover, no GFP mRNA was found. We cannot rule out the possibility that a rare recipient cell might have expressed insulin but not GFP. Recently, other studies have failed to find that bone marrow can serve as a precursor for β-cells in the pancreas (4,11,19). Our results are unique because they focus upon transplanted islets, but they add to the growing body of evidence that circulating precursor cells do not become β-cells in either the native pancreas or islet grafts. Our negative results with MIP-GFP mice were supported by the experiments using β-actin GFP mice.
For the stimulation of differentiation of stem cells or precursor cells, it is possible that metabolic demand or events that occur during injury could be influential. During the period immediately after islet transplantation, a very dynamic process occurs with cell death, tissue remodeling, and revascularization (6). This is accompanied by falling glucose levels as the diabetic state is reversed. Interestingly, the normal architecture of the pancreatic islet is not reestablished in islet grafts (15). Taking all of these acute changes into account, it would not be surprising to find factors that could promote regeneration. The majority of our evaluation was done at 4 weeks after transplantation when the reconstruction of islet graft was thought to have been established. However, we also examined the grafts 2, 6, 8, and 12 weeks after transplantation, and no recipient-derived endocrine cells were found (data not shown). We did not perform transplants in euglycemic recipients; this condition could have conceivably created a different remodeling and regeneration dynamic.
To focus on the contribution of bone marrow cells as a stem cell source, we studied bone marrow cell injections into STZ diabetic mice. For permanent bone marrow transplantation, endogenous bone marrow cells should be removed before transplantation by irradiation or chemicals to make space for the transplanted cells. However, because the mice were undergoing consecutive surgeries, we were concerned that irradiation might lead to fatal consequences. Therefore, we chose to inject littermate bone marrow cells without irradiation to increase circulating bone marrow cells. Persistence of the injected bone marrow cells was confirmed by RT-PCR with a detection of the expression of GFP mRNA in peripheral blood 3 weeks after injection, and in islet grafts, bone marrow-derived green fibroblast-like cells and/or endothelial-like cells were seen.
Our data are compatible with the reports of others, that bone marrow cells do not improve hyperglycemia directly by differentiating into β-cells by themselves, but initiate pancreatic tissue regeneration through some other mechanism that could involve endothelial cells (4,5,10). However, as in endogenous pancreas, it is possible that bone marrow cells have a beneficial effect on transplanted islets. We must be cautious about assuming that our findings in grafts located in the kidney can be equated with grafts in the liver. While the similarities in most studies are impressive, it is possible that regenerative capacity differs between the two sites. In conclusion, our data suggest that β-cells in islet grafts may be replenished by their own replication or by precursor cells from the donor, not by bone marrow cells or tissue precursor cells from the recipient.
Footnotes
Acknowledgments
We thank Jennifer Lock for her technical assistance. This study was supported by grants from the National Institute of Health (U19DK61251), the Juvenile Diabetes Research Foundation, and the Diabetes Research and Wellness Foundation. Help was also provided by the Advanced Microscopy Core of Joslin Diabetes and Endocrinology Research Center (DERC) supported by the National Institutes of Health (DK36836). Yoshiyuki Hamamoto is the recipient of the grant from Sumitomo Life Social Welfare Services Foundation (Osaka, Japan).
