Abstract
We have recently reported that small-sized bone marrow cells (BMCs) isolated by counterflow centrifugal elutriation and depleted of lineage markers (Fr25lin-) have the capacity to differentiate and contribute to regeneration of injured islets. In this study, we assess some of the characteristics of these cells compared to elutriated hematopoietic progenitors (R/O) and whole BMCs in a murine model of streptozotocin-induced chemical diabetes. The GFPbrightCD45+ progeny of whole BMCs and R/O progenitors progressively infiltrate the pancreas with evolution of donor chimerism; are found at islet perimeter, vascular, and ductal walls; and have a modest impact on islet recovery from injury. In contrast, Fr25lin- cells incorporate in the islets, convert to GFPdimCD45-PDX-1+ phenotypes, produce proinsulin, and secrete insulin with significant contribution to stabilization of glucose homeostasis. The elutriated Fr25lin- cells express low levels of CD45 and are negative for SCA-1 and c-kit, as removal of cells expressing these markers did not impair conversion to produce insulin. BMCs mediate two synergistic mechanisms that contribute to islet recovery from injury: support of islet remodeling by hematopoietic cells and neogenesis of insulin-producing cells from stem cells.
Keywords
Introduction
Hematopoietic stem and progenitor cells (HSPCs) derived from various sources have been considered as candidates for regeneration of the endocrine pancreas in type 1 diabetes. Intense work dedicated to islet regeneration suggests two major approaches to substitution of the injured tissue and restoration of insulin production: neogenesis from stem cells and support of endogenous recovery. A proof of concept for neogenesis is the successful induction of insulin production in a variety of cells in vitro, including bone marrow-derived mesenchymal stromal cells (MSCs) (35,48), adherent and nonadherent bone marrow and blood fractions (49,65), and umbilical cord blood (UCB) cells (9,13,62). However, in vivo conversion of cells derived from hematopoietic compartments remains controversial (13). On the one hand, murine cells derived from the bone marrow (23) and spleen (36) and human UCB cells (21) incorporate in the islets of irradiated recipients, as also observed after UCB cell grafting in neonate immunodeficient mice (69). Insulin production has been detected in particular subsets of stem cells after islet injury (36,46): very small embryonic-like (VSEL) cells corresponding to a cluster of differentiation 45-negative stem cell antigen 1 (lymphocyte antigen 6 complex, locus A; Ly6a)-positive chemokine C-X-C motif receptor 4-positive lineage-negative (CD45-SCA-1+CXCR4+lin-) phenotype (22,55) and small-sized elutriated cells (elutriated flow rate 25 ml/min; Fr25) devoid of lineage markers (Fr25lin-) (24). Altogether these data suggest that neogenesis of insulin-producing cells is a rather low-efficiency mechanism of islet repair, provided that the autoimmune reaction is controlled or abrogated (26).
On the other hand, most studies have shown beneficial effects of cellular and cytokine interventions on islet remodeling and recovery from injury without significant conversion to produce insulin (8,12,19,33,39,40,42,45,66,68). Hematopoietic cells play a general supportive role for endogenous islet recovery through activation of pancreatic progenitors, expansion of β-cells, and conversion of α-cells (11,44,66,70). These supportive activities have been attributed to MSCs, immune cells, hematopoietic, and endothelial progenitors derived from the bone marrow and UCB (12,13), which generally came short of restoring glycemic control (1,6,7,18,27,39,41,47,56). The first mechanism involves direct and indirect support of revascularization and neovascularization, which has been demonstrated for whole bone marrow cells (BMCs) (8,19,39,64), adherent BMC subsets (39,40,45), lineage-negative (lin-) (19,47), and c-kit+lin- progenitors (19). The second mechanism involves local immunomodulation attained by MSCs (7,33,40,42), and one or multiple BMC infusions (6,19). Combined vascular and immunogenic activities have been also achieved by bone marrow activation with 5-fluorouracil (8), mobilization (64), and cotransplantation (43). The third mechanism is support of islet stroma, which is an additional mechanism of MSC activity (7,40), and local provision of growth factors such as hepatocyte growth factor and insulin-like growth factor-1 (1,12,27,42).
We have used a subset of small-sized BMCs isolated by counterflow centrifugal elutriation (Fr25lin-) to demonstrate significant incorporation in chemically injured islets (24). These stem cells convert to express markers specific of hepatocytes, epithelia, and glia in vitro and in vivo (15,16,28,37) in addition to durable multilineage hematopoietic reconstitution (30,31). Another fraction isolated by elutriation contains primarily large-sized progenitors in various stages of differentiation, endowed with radioprotective activity due to short-term reconstituting capacity (30,31). This study aims to resolve some of the controversies regarding the role of bone marrow-derived cells in islet recovery. We focused on the involvement of various subsets in two major mechanisms of islet recovery from injury: neogenesis of insulin-producing cells and support of islet remodeling. We found that these mechanisms are mediated by distinct bone marrow subsets including small-sized stem cells and large-sized progenitors, without apparent possibility of prospective identification using putative HSPC markers.
Materials and Methods
Animal Preparation, Diabetes, and Transplantation
Mice used in this study were C57Bl/6J (B6, H2Kb, protein tyrosine phosphatase, receptor type Cb; CD45.2), B6.SJL-Ptprca Pepcb/BoyJ (H2Kb, protein tyrosine phosphatase, receptor type Ca peptidase Cb; CD45.1), and C57BL/6-TgN(ACTbEGFP)1Osb [enhanced green fluorescent protein (GFP) under the control of a chicken β-actin promoter and cytomegalovirus enhancer, H2kb, CD45.2]; they were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and housed in a barrier facility. The Institutional Animal Care Committee of the Schneider Medical Center approved all procedures (#022B6229 dated 4/1/ 2009). Diabetes was induced in female mice (aged 6–8 weeks) by five daily consecutive intraperitoneal injections of 60 μg/g streptozotocin (STZ; Calbiochem, Darmstadt, Germany). Blood glucose levels were monitored with a standard glucometer (Accu-Chek Sensor; Roche Diagnostics, Basel, Switzerland) in mice fed ad libitum at constant daytime hours (9–11 AM). Diabetes was considered at glucose levels exceeding 250 mg/dl in two consecutive measurements. Before cell transplantation (day 0), recipients were sublethally irradiated at 675 rad (total body irradiation) using an X-ray irradiator (RadSource 2000, Suwanee, GA, USA) at a rate of 106 rad/min (day -1). For transplantation, cells suspended in 0.2 ml phosphate-buffered saline (Sigma-Aldrich, St. Louis, MO, USA) were infused into the lateral tail vein.
Cell Isolation
Whole BMCs (wBMCs) from wild-type or GFP-positive male donors (aged 6–8 weeks) were harvested by flushing of medullar cavities of femur, tibia, and iliac bones. Single cell suspensions (5 × 108 cells) were loaded into the chamber of a counterflow centrifuge (Beckman Instruments, Palo Alto, CA, USA) operating at a constant speed of 3,000 rpm (1,268 × g). Fractions were collected in 200 ml at an elutriation flow rate of 25 ml/min to isolate the smallest subset of nucleated cells (Fr25), and the largest cells were collected in the rotor off position (R/O). Fr25 cells were lineage-depleted by incubation with saturating amounts of rat anti-mouse monoclonal antibodies (mAb) against CD5 (clone 53–7.3, T-cells), B220 (clone RA3-3A1/6.1, B-lymphocytes), GR-1 (clone RB6-8C5, granulocytes), Mac-1 (clone M1/70, macrophages), extracted from hybridoma cell lines (ATCC, Manassas, VA, USA) and purified TER119 (erythroid; eBioscience, San Diego, CA, USA). Antibody-bound cells were incubated with goat anti-rat secondary antibodies conjugated to magnetic beads at a ratio of four to five beads per cell (Dynal Biotech, Oslo, Norway) and were collected by exposure to a magnetic field.
Flow Cytometry
Measurements were performed with a Vantage SE flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) on cells that underwent red cell lysis (34). Hematopoietic chimerism was determined by GFP fluorescence or mAb against minor antigens CD45.1 (clone A20; eBioscience) and CD45.2 (clone 104; eBioscience) and the antibodies mentioned above. Data were acquired using several combinations of fluorochromes, including fluorescein-isothyocyanate (FITC), phycoerythrin (PE), allophycocyanin (APC), and peridinin chlorophyll protein complex (PerCP) (all from BD Pharmingen). In all measurements, nonspecific binding was prevented by addition of 1 μl mouse serum.
Determination of Blood Insulin
Serum from mice was collected by centrifugation and assessed in 96-well microtiter assay plates (Millipore, Billerica, MA, USA) using the rat/mouse insulin enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Abingdon, UK). Absorbance at 450 nm and 590 nm was determined using an ELISA PowerWave-10 in a plate reader (BioTeK, Winooski, VT, USA). Insulin standards were used to determine a calibration curve.
Tissue Preparation
Mice were sacrificed by CO2 asphyxiation, and infusion of ice-cold fixative containing 1.5% fresh paraformaldehyde and 0.1% glutaraldehyde (Sigma-Aldrich) was performed with a miniperistaltic pump (P720; Instech, Plymouth Meeting, PA, USA) through a blunt 20-gauge needle (Victorg, Kanpur, India) placed in the left ventricle. Excised pancreata were placed in this medium for 2 h at 4°C for additional fixation, immersed in 30% sucrose overnight (Sigma-Aldrich), embedded in optimal cutting temperature media (Sakura Finetek, Torrance, CA, USA), and frozen in isopentane (Sigma-Aldrich) suspended in liquid nitrogen. Tissue was sectioned (3–6 μm) with a cryotome (Thermo Shandon, Runcorn, Cheshire, UK).
Immunofluorescence and FISH
Immunofluorescence was designed according to several considerations to allow detection of the donor cells with variable GFP fluorescence intensities (25). Nuclei were labeled with Hoechst-33342 (1:1,000; Molecular Probes, Eugene, OR, USA), and sections were mounted in antifade medium (Dako, Glostrup, Denmark). Serial cryosections were immunostained with primary antibodies for 1 h and counterstained with respective secondary antibodies for 30 min at room temperature: polyclonal mouse-anti-proinsulin (1:20; R&D Systems), goat-anti-pancreatic and duodenal homeobox 1 (PDX-1; 1:5,000, kindly provided by Shimon Efrat), biotinylated anti-mouse CD45 (1:100; Biolegend, San Diego, CA), and biotinylated rabbit-anti-GFP (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA). These primary antibodies were counterstained with FITC-labeled donkey anti-rabbit (1:200; Jackson Immunoresearch, West Grove, PA, USA) and goat anti-rat mAb (1:200; Santa Cruz), Alexa Flour 568-conjugated donkey anti-goat (1:500), and cyanine 3 (Cy3)-conjugated rat anti-goat (1:200; Molecular Probes). Biotinylated primary antibodies were counterstained with Cy3-conjugated streptavidin (1:400) and Cy5-conjugated streptavidin (1:500) purchased from Jackson Immunoresearch.
The Y chromosome in male donor cells was visualized in fresh sections or immunostained slides after treatment with 0.025% pepsin (Invitrogen, Grand Island, NY, USA) for 30 min at 37°C (25). Nuclear probes (Applied Spectral Imaging, Migdal Haemek, Israel) were denatured, and probe hybridization was performed overnight, followed by sequential wash in 0.4× saline sodium citrate buffer (SSC; Sigma-Aldrich) and 2× SSC stringency solution supplemented with 0.1% NP4O detergent (Sigma-Aldrich). Slides were washed and mounted with antifade containing 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich).
Images were acquired with an Axioplan 2 (C. Zeiss, Göttingen, Germany) fluorescence microscope equipped with an Apotome using AxioVision 4.5 and LSM 510 software, respectively. Images were pseudocolored and RGB reconstructed using Adobe Photoshop software (San Francisco, CA, USA).
Real-Time PCR and Western Blots
Western blot for PDX-1 used rabbit anti-PDX-1 polyclonal antibodies (1:500; Chemicon, Billerica, MA, USA) and peroxidase-conjugate goat anti-rabbit IgG (1:10,000; Jackson Immunoresearch). RT-qPCR was used to study the expressions of CD45, c-kit, and Sca-1. Elutriated fractions were frozen in liquid nitrogen, total RNA was extracted with TRIzol (Invitrogen), followed by reverse transcription into cDNA using random hexamers (Amersham Biosciences, Cardiff, UK), and Moloney murine leukemia virus reverse transcriptase (Promega, Madison, WI, USA). cDNA was analyzed with a Sequence Detection System (ABI Prism 7900; Applied Biosystems, Foster City, CA, USA). Gene expression was measured by normalization of cDNA input levels against mouse β-actin. Reactions were performed for forward and reverse primers in Master Mix buffer (SYBR Green I; Applied Biosystems) according to the PCR cycling conditions previously described (4,16). Standard curves were obtained using untreated mouse cDNA for each gene PCR assay and quantified by the comparative Ct method (4,15). Primers used for analysis are delineated in Table 1.
Primers

Pdx-1, pancreatic and duodenal homeobox 1; CD45, cluster of differentiation 45; Sca-1, stem cell antigen-1.
Statistical Analysis
Data are presented as means ± standard deviations for each experimental protocol. Results in each experimental group were evaluated for reproducibility by linear regression of duplicate measurements. Differences between the experimental protocols were estimated with a post hoc Scheffe t-test, and significance was considered at p < 0.05.
Results
Cellular Composition and Hematopoietic Function of Elutriated Subsets
Counterflow centrifugal elutriation selects subsets of BMCs according to density: small-sized cells (Fr25) and large-sized progenitors collected in the R/O position, each one of the subsets accounting for ~20% of the sample (30,31,53). These cell populations differ substantially in contents of the various lineages (Fig. 1), with myelomonocytic progenitors predominantly found in the R/O subset (Fig. 1B) and erythroid and lymphoid cells in the Fr25 subset (Fig. 1A). Depletion of the various lineages from the Fr25 subset results in an enriched population of small-sized stem cells (Fr25lin-) displaying variable CD45 expression (Fig. 1C). These subsets have distinct hematopoietic activities: R/O cells mediate short-term reconstitution, while Fr25lin- cells are devoid of radioprotective activity, and their slow activation mediates long-term durable multilineage reconstitution (30,31). Therefore, we validated our elutriation technique in a competitive engraftment assay; both subsets were grafted into lethally irradiated syngeneic hosts with constitutive expression of GFP (Fig. 1D). At 3 weeks posttransplantation (short term), the donor hematopoietic progeny was predominantly of R/O cell origin (CD45.2+GFP-), whereas at 16 weeks posttransplant (long term), donor chimerism originated primarily from the Fr25lin- subset (CD45.1+GFP-). The differential radio-protective activities of these subsets isolated by elutriation were also evident from the significant mortality of mice grafted with Fr25lin- cells alone and survival of all recipients of R/O progenitors (Table 2A).

Composition and characteristics of elutriated BMC subsets. (A) Fractional composition of hematopoietic lineages within the elutriated rotor off (R/O; n = 5) and flow rate of 25 ml/min (Fr25; n = 13) subsets: T-cells (CD5), B lymphocytes (B220), granulocytes (GR-1), macrophages (Mac-1), erythroid (Ter-119), and lineage-negative cells (lin-). (B) Demonstrative plots of GR-1 expression in the R/O and Fr25lin- subsets. (C) Analysis of cell size, hematopoietic lineage markers, and CD45 in wBMCs (n = 28), R/O progenitors (n = 9), and the Fr25 subset before (n = 38) and after depletion of lineage-positive cells (Fr25lin-, n = 21). (D) Irradiated (850 rad) GFP+ recipients were grafted with 5 × 105 minor antigen disparate Fr25lin- cells (GFP- CD45.1) and 5 × 105 R/O cells (GFP- CD45.2). Chimerism was determined by gating on GFP- cells after 3 and 16 weeks for identification of the progeny of the two subsets. PE, phycoerythrin; APC, allophycocyanin.
Consequences of Cellular Transplants
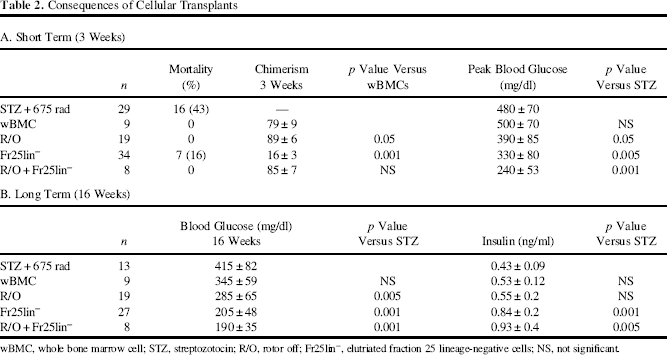
wBMC, whole bone marrow cell; STZ, streptozotocin; R/O, rotor off; Fr25lin-, elutriated fraction 25 lineage-negative cells; NS, not significant.
Differential Effects of Cell Subsets on Recovery From Chemical Diabetes
To evaluate the impact of transplantation of various cell subsets on the course of chemical diabetes, we used sublethal irradiation, attempting to shed light on the mechanisms of BMC transplantation on islet recovery from injury. This established model (8,14,19,22,24,25,40,64,68) includes selective injury by five consecutive doses of 60 μg/g STZ (days -6 to -2) followed by sublethal irradiation (675 rad) and cell grafting after 1 day (Fig. 2A). Under these conditions, the mice develop severe hyperglycemia (Fig. 2B) with significant mortality (Table 2A), both parameters being effectively ameliorated by cotransplantation of Fr25lin- cells and R/O progenitors. Histological analysis of the islets revealed incorporation of GFPdim cells in the inner sections of the islets and the presence of GFPbright cells at the islet perimeter (Fig. 2C).
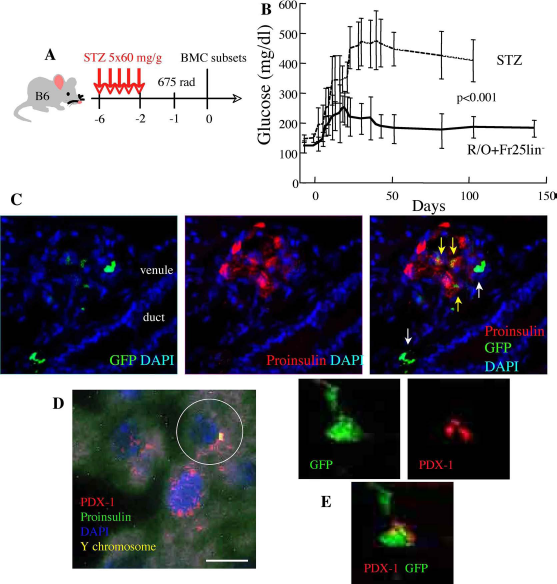
Incorporation and functional conversion of Fr25lin- cells. (A) Experimental design: chemical diabetes was induced by five consecutive doses of 60 μg/g STZ, sublethal irradiation at 675 rad, and syngeneic cell transplantation in C57/Bl6 mice. (B) Blood glucose levels of sublethally irradiated diabetic mice (STZ, n = 13) and after cotransplantation of 106 R/O progenitors and 106 Fr25lin- cells (n = 8). (C) Histological analysis of islets (identified by proinsulin staining) of recipients of Fr25lin- and R/O cells discloses GFPdim cells incorporated in the inner islet areas (yellow arrows) and GFPbright cells surrounding the islets (white arrows) juxtaposed to ducts and venules (scale bar: 20 μm). (D) Positive identification of male cells (Y chromosome, encircled) incorporated in the inner islet regions of a female recipient, coexpressing pancreatic and duodenal homeobox 1 (PDX-1) and proinsulin (scale bar: 10 μm). (E) Demonstrative confocal images of coexpression of nuclear PDX-1 and cytosolic GFP after transplantation of Fr25lin- cells in diabetic mice (scale bar: 5 μm). DAPI, 4′,6-diamidino-2-phenylindole (nuclear stain).
To dissociate between the contributions of the two subsets, they were individually grafted into diabetic mice. Male Fr25lin- cells, identified by a genomic marker, produced proinsulin, incorporated in the inner regions of the islets (Fig. 2D), expressed PDX-1 (Fig. 2E), and mediated a significant reduction in blood glucose levels (Table 2A). Notably, reduced blood glucose levels in recipients of Fr25lin- cells alone and in conjunction with R/O progenitors are attributed to the corresponding increase in insulin levels at 16 weeks posttransplant (Table 2B). In variance, transplantation of R/O progenitors and whole bone marrow cells had little impact on glucose homeostasis (Fig. 3A) and insulin levels (Table 2B) despite early evolution of donor chimerism (Table 2A). Donor R/O progenitors localized to the islet perimeter, particularly adjacent to the vascular pedicle, and retained bright GFP intensity and were negative for PDX-1 staining (Fig. 3B). Similar qualitative analysis after transplantation of wBMCs showed significant numbers of GFPbright donor cells surrounding the islets, ducts, and pancreatic vasculature (Fig. 3C). These cells were largely negative for proinsulin (Fig. 3C), and the vast majority expressed the pan-hematopoietic marker CD45 (Fig. 3D), corroborating the absence of significant conversion to adopt endocrine phenotypes and produce insulin. These findings recapitulate prior reports of incorporation in injured pancreata of grafted wBMCs and hematopoietic progenitors (6–8,11,14,19,39,40,46,47,64,66,70).
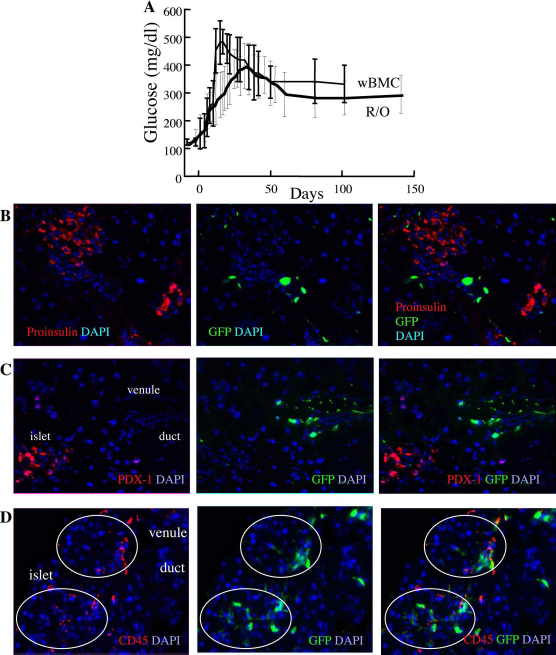
Consequences of transplantation of whole BMCs and progenitors. (A) Blood glucose levels in mice with chemical diabetes after transplantation of 5 × 106 syngeneic whole BMCs (n = 9) and 106 R/O progenitors (n = 19). (B) Grafted R/O progenitors surrounding the injured islets (white arrows) express bright GFP and do not stain for proinsulin (scale bar: 40 μm). (C, D) GFP-bright cells within the vasculature and surrounding the ducts detected after transplantation of whole BMCs. These cells do not express the transcription factor PDX-1 (C, scale bar: 20 μm) and express the panhematopoietic marker CD45 (D, scale bar: 40 μm).
Fr25lin- cells are in fact a minor subset of BMCs, questioning whether the numbers of stem cells within the wBMC grafts were too low for detection of conversion. To evaluate this possibility, sublethally irradiated mice with chemical diabetes were grafted with 3 × 107 BMCs, which contain an equivalent of 106 Fr25lin- cells (~20% Fr25 cells of which ~15% are collected after lineage depletion). The outcome of these transplants was not different from the long-term outcome of mice grafted with 5 × 106 BMCs (Table 2B), indicating that the low Fr25lin- numbers were not responsible for the poor outcome of wBMC transplants.
Hematopoietic Progenitor Markers Do Not Delineate Islet-Engrafting Cells
Controversial evidence on neogenesis of insulin-producing cells has been presented by studies using hematopoietic progenitor markers such as SCA-1 and c-kit, ranging from positive identification of differentiation (22,46) to negative results (19,68). The positive identification of Fr25lin- cell conversion to produce insulin (24) was used to address the possible use of these markers for prospective identification of the candidate stem cells that participate in islet regeneration. As shown in prior studies (59), Sca-1 and c-kit are variably expressed in the Fr25 subset, and lineage depletion results in further decrease in cell surface expression of these markers and CD45 (Fig. 4A). Considering that progenitor commitment and priming might be detectable only at the transcriptional level (50), mRNA encoding these markers was assessed in the small-sized cells. Elutriation of Fr25 cells and lineage depletion corresponded to decreased levels of mRNA encoding c-kit and Sca-1 (Fig. 4B), showing that progenitors expressing these markers are not enriched by the elutriation procedure. In the next stage, we sought to assess the possible participation of progenitors demarcated by these markers in islet regeneration by their depletion from the Fr25lin-subset. Transplantation of SCA-depleted and kit-depleted cells had no significant impact on recovery from chemical diabetes mediated by the Fr25lin- cells (Fig. 4C), indicating that these hematopoietic markers are neither obligatory nor characteristic of bone marrow-derived cells that convert to adopt endocrine phenotypes.

Characterization of hematopoietic progenitor markers in Fr25lin- cells and their contribution to islet recovery from injury. (A) Expression of CD45, c-kit, and stem cell antigen 1 (SCA-1) on the surface of freshly elutriated Fr25 and lineage-depleted (Fr25lin-) cells. Data are normalized against expression in wBMCs (n = 5). (B) Measurements of c-kit and SCA-1 expression in the corresponding subsets as determined by flow cytometry. (C) Transcriptional profiles of CD45, c-kit, and SCA-1 in Fr25 and Fr25lin- cells as determined by real-time PCR. Data are normalized against mRNA contents in wBMCs (n = 3). (D) Blood glucose levels after day 100 in sublethally irradiated diabetic mice (STZ + rad, n = 13) and after transplantation of 106 Fr25lin- cells (n = 27), and equal number of the Fr25lin- subset depleted of c-kit (n = 10) and SCA-1 (n = 10). PerCp, peridinin chlorophyll protein complex.
Discussion
Transplantation of murine BMCs augments islet recovery from chemical injury through two main mechanisms. The major mechanism is neogenesis of insulin-producing cells from small-sized stem cells within the bone marrow. The minor mechanism is indirect support of islet remodeling by hematopoietic progenitors that generate early hematopoietic chimerism, along with activities of mesenchymal cells and endothelial progenitors (7,12,13,40,47). Because autologous hematopoietic transplants do not abrogate auto-immunity indefinitely (3,34) due to excessive conditioning (2), MSCs might offer an alternative to immunogenic control of the autoimmune disorder (12,33) in particular derived from UCB (13).
Our data emphasize several distinct activities of small-sized stem cells and large-sized progenitors in a model of toxic injury to the pancreatic islets. First, competitive engraftment assays showed early reconstitution from R/O progenitors endowed with radioprotective activity and delayed functional reconstitution from small-sized Fr25lin-cells, which become the predominant source of durable mulilineage hematopoiesis (30). Therefore, consistent with sequential engraftment of various bone marrow-derived progenitors (31,38,53), elutriation dissociates functionally distinct subsets of stem cells and progenitors. Second, the patterns of cell incorporation in the injured islets disclose differential affinities to central and peripheral regions of the injured islets, which also correlate with the intensity of GFP expression. Fr25lin- cells engraft primarily in the inner regions of the islets and downregulate GFP expression, as a feature of active differentiation (24,25). In variance, hematopoietic progenitors and their progeny are found in the vasculature, ductal walls, and at islet perimeter, sustaining bright GFP signals. The activities of these cells are largely supportive of islet remodeling, including neovascularization (8,19,39,44,47,64), immunomodulation (6,7,12,39,42), stabilization of the stroma (7,40), and provision of cytokines (1,27). Third, the therapeutic efficacy of the two subsets attributes clear advantage of the Fr25lin-cells in stabilization of glucose levels through sustained increase in insulin levels. In contrast, R/O progenitors and whole BMC grafts share limited capacity to elevate insulin levels, corroborating the partial and transient reduction in blood glucose levels mediated by hematopoietic progenitors (6,7,14,18,27,39,41,47,56).
Small-sized nucleated BMCs differentiate into various nonhematopoietic lineages in vivo, expressing markers specific of hepatocytes, astrocytes, endothelium, retinal neurons, and endocrine pancreas in response to selective injury (15,16,24,28). The most convincing evidence of such differentiation is the evolution of multiple epithelial cell types following transplantation of single Fr25lin-cells, which also demonstrates the self-renewal capacity of these primitive precursors in vivo (37). The characteristics of differentiating Fr25lin- cells in the injured islets include upregulation of PDX-1, downregulation of tacking markers such as GFP, production of proinsulin, and secretion of insulin. Since Fr25lin- cells represent a small fraction of nucleated BMCs, the numbers of candidate stem cells might be insufficient in wBMC grafts used for hematopoietic reconstitution (6–8,14,19,22,39, 40,47). However, transplantation of large BMC numbers, containing equivalent fractions of the elutriated Fr25lin-subset, resulted in undetectable morphological and functional incorporation in the islets. It is therefore the way these cells are prepared for transplantation rather than their numbers that determine their capacity to convert and produce insulin. Possibly, extraction of Fr25lin- cells by elutriation exposes them to stress that terminates their dormant state and primes them to differentiation. The small-sized elutriated cells are mitotically quiescent in the host bone marrow (38), have no radioprotective activity under limiting dilution conditions (60), and initiate indefinite multilineage hematopoiesis after several weeks (28,29,53). In fact, epithelial and hepatic differentiation within days (28,37) and endocrine, glial, and neural differentiation within weeks (15,16,24) precede in time the engagement of these cells in long-term hematopoietic reconstitution. The plausible explanation is that these residents of a dedicated hematopoietic compartment such as the bone marrow require stronger inhibitions of hematopoietic differentiation in order to preserve a pool of long-term reconstituting HSPCs. This may be the cause of their slow initiation and delayed hematopoietic activity.
The two distinct mechanisms of BMC support of islet regeneration and remodeling question the differentiation capacity of hematopoietic progenitors positively identified by cell surface markers. R/O progenitors, wBMCs, and their progeny sustained bright GFP signals and were located at the islet perimeter, the vascular pedicles, and surrounding the ducts, similar to the location reported for progenitors expressing c-kit and SCA-1 (19,46,68). Exclusion of Fr25lin- cells expressing either one of these progenitor markers had no significant impact on endocrine differentiation, thus they cannot be used for prospective identification of candidate stem cells (8,19,39,64). In addition, the early activity of c-kit and SCA-1 signaling is dispensable in neogenesis of insulin-producing cells, without excluding the possibility of their upregulation and activation in later stages of differentiation. Our findings are consistent with the contention that the most primitive precursors residing in the bone marrow (29) express neither SCA-1 nor c-kit (10,52). These markers are expressed by committed hematopoietic progenitors responsible for both short-and long-term reconstitution (51,61,67), but are not essential or characteristic of long-term repopulating cells (59). In fact, c-kit and SCA-1 are neither unique nor specific to hematopoietic differentiation and are often expressed in pancreatic (17,57), endodermal (20), hepatic (20,54,58) and neural progenitors (67), and MSCs (5,63).
Analysis of CD45 expression reinforced the contention that endocrine differentiation is restricted to the most primitive progenitors (stem cells) within the bone marrow. This pan-hematopoietic marker was detectable in GFPbright cells surrounding the islets (19,39,40,47), but not in insulin-producing cells derived from the Fr25lin- cells (24), CD45-SCA-1+CXCR4+lin- (VSEL) (22,55), and splenocytes (36). Although the Fr25lin- subset shares with VSEL cells the small size, absence of lineage markers and CD45-/low expression, Fr25lin- cells converting to produce insulin are largely characterized by a SCA-1 negative phenotype.
The competitive nature of engraftment may be one of the restrictions of the limited capacity of distinct subsets of hematopoietic precursors to convert into insulin-producing cells. Expression of progenitor markers defines a certain degree of commitment, and their primary activity in irradiated hosts is hematopoietic reconstitution. We observed that total body irradiation is rather detrimental to quantitative incorporation of Fr25lin- cells in the ischemic retina (4), due to the high affinity of these cells to the bone marrow (32,38). It is undetermined yet whether the grafted Fr25lin- cells migrate directly to sites of injury or are primed in the bone marrow and are trapped in the aplastic marrow space of irradiated hosts.
The current data emphasize two distinct mechanisms of adult bone marrow-derived cell participation in islet recovery from chemical injury. Hematopoietic cells, MSCs, and endothelial progenitors mediate immune, vascular, stromal, and cytokine-mediated mechanisms of support of islet remodeling after injury with modest impact on glucose homeostasis (1,6–8,14,18,19,22,27,39,40,46,47,56,64). A major contribution to glycemic control is conferred by neogenesis of insulin-producing cells from primitive precursors residing in the adult bone marrow, which can be isolated by counterflow elutriation. These regenerative cells do not express lineage and hematopoietic progenitor markers, display variable CD45, silence GFP, express PDX-1, produce proinsulin, convert, and secrete insulin. The beneficial effects of multiple cell types with distinct mechanisms on islet remodeling and regeneration are promising toward development of cell-based therapeutic approaches to type 1 diabetes.
Footnotes
Acknowledgments
We thank Dr. Saul Sharkis and Dr. Michael Collector for the outstanding support, discussion, and conceptual contribution to this study. Funding was provided by a generous grant from the Leah and Edward M. Frankel Trust for bone marrow transplantation and the Zanvyl and Isabelle Krieger Fund, Israel Academy of Sciences and Humanities (1371/08), the Eldor-Metzner Clinician Scientist Award, Chief Scientist, Israel Ministry of Health (3-3741), and a Pilot Grant from North American Neuro-Ophthalmology Society. Author contributions: Performance of experiments: S.I., N.G., T.S., N.A. Collection of data: S.I., N.G., T.S., I.Y., J.S. Analysis and interpretation: N.G., I.Y. Manuscript writing: J.S., N.A. Conception and design: N.A. The authors declare no conflicts of interest.
