Abstract
Several clinical trials of bone marrow cell infusion in patients with liver cirrhosis (LC) have shown clinical improvement, despite conflicting results from animal models. We investigated serial pathological features and the clinical impact after autologous bone marrow infusion (ABMI) in patients with advanced LC. Ten patients with advanced LC due to chronic hepatitis B virus infection underwent ABMI. Serological tests, MRI, and liver biopsies were performed, and quality of life was assessed by a questionnaire. Median serum albumin and hemoglobin levels increased significantly after ABMI. All patients showed an improvement in quality of life, with no serious adverse events. Liver volume, measured by MRI, increased in 80% of the patients, and ascites decreased after ABMI. Child-Pugh scores were also significantly improved at 6 months after ABMI. In the serially biopsied livers, a gradually increasing activation of the hepatic progenitor cell (HPC) compartment, including HPC activation (ductular reaction) and HPC differentiation (intermediate hepatocyte), reached a peak after 3 months, with continued proliferation of hepatocytes, and returned to baseline levels after 6 months. There was no significant change in grade or stage of liver fibrosis or stellate cell activation after ABMI. ABMI is suggested to improve liver function and to activate the progenitor cell compartment. Although clinical improvement was sustained for more than 6 months, histological changes in the liver returned to baseline 6 months after ABMI. Further comparative studies are warranted.
Keywords
Introduction
Liver cirrhosis (LC) is the end stage of various chronic liver diseases and is extremely difficult to treat. In addition to substitution of parenchyma by fibrous tissue, the gradual loss of cellular function results in the impairment of homeostasis. Until now, liver transplantation has been the only effective method of cure, but due to the several limitations of transplantation, such as a lack of donors, surgical complications, rejection, and high cost, regenerative therapy has been suggested to provide alternative choices using less invasive procedures.
In general, organ regeneration requires the presence of a progenitor cell population in the absence of a resident cell proliferation (15). However, in the liver physiological turnover and regeneration occur predominantly through replication of native hepatocytes. Progenitor-dependent regeneration takes place only if parenchymal hepatocytes are severely damaged and unable to regenerate efficiently, as in cirrhosis (17). Thus, as an external source of hepatocytes, mature hepatocytes, fetal hepatocytes, oval cells (hepatoblasts in humans), hematopoietic stem cells, mesenchymal stem cells, multipotent adult progenitor cells, or embryonic stem cells have been used for regeneration in liver disease models (4).
After Peterson and colleagues (19) first reported bone marrow cells (BMCs) as a potential source of oval cells, supportive data about bone marrow-derived hepatocytes in animals (12,28) and humans (1) have been published. Although recent advances have documented that bone marrow cells can also differentiate into muscle (5), heart (16), pancreas (9), and lung (8) cells, controversy still exists regarding the role of BMCs in organ regeneration, including in the liver. Researchers who have a negative opinion on the plasticity of BMCs insist that little evidence exists for the use of bone marrow-derived hepatocytes in the replacement of injured liver (10), and that no data have demonstrated transdifferentiation to hepatocytes or a major role in regeneration (14,30,31). On the other hand, BMCs have been reported to be a possible source of functional hepatic stellate cells and myofibroblasts contributing to fibrosis (23).
However, in a murine model, transplanted BMCs populated and differentiated into albumin-producing hepatocytes, via hepatoblast intermediates (27). Furthermore, in humans, BMC infusion elevates serum albumin levels (26), and, in mice or rats, it reduces liver fibrosis (7), corrects liver dysfunction (2), and improves survival rate (25). Based on these results, a clinical trial of autologous BMC infusion (ABMI) has been performed (18,24), and it was shown that ABMI administered through a peripheral vein or the hepatic artery improves liver function in patients with LC (13,26).
Although recent clinical trials have shown transient improvements in liver function, there has been no report about the duration of effect and what happens in the liver after cell transplantation. The aims of this study were to investigate the clinical impact and serial histological changes that contribute to liver regeneration and to determine how long such changes were sustained after ABMI in patients with advanced LC.
Materials and Methods
Patients
Patients were enrolled between November 2006 and February 2008. All patients had biopsy-proven LC. Child-Pugh class B or C patients aged between 18 and 75 years with total bilirubin ≤3.0 mg/dl, platelet count ≥0,000/μl, and no viable hepatocellular carcinoma (HCC) on magnetic resonance imaging (MRI) were eligible. Patients were excluded from the study if they had problems with organs other than the liver, required general anesthesia, or had any uncontrolled malignancies. We advised all patients to maintain their current medications, except diuretics, during the study period. After ABMI, doses of diuretics were adjusted according to the patient's condition. Antiviral therapy was individualized, according to the guidelines. No patient was given albumin during the study.
BMC Harvest and Infusion
BMCs (500–750 ml) were harvested from both ilia according to standard procedures under general anesthesia and were collected in a plastic bag containing heparin. After fat and bony particles were removed by filtration, collected cells were moved to a cell-processing device. The automatic cell washer 2991 (Gambro, Lake-wood, CO, USA) was used for collecting mononucleated cells (MNCs), as well as depleting red blood cells and plasma in a closed system. The final concentrated cell product was made up to a final volume of about 100 ml. Five milliliters of the final cell product was subjected to trypan blue dye exclusion testing, bacterial culture, cell counting, and fluorescence-activated cell sorting analysis by flow cytometry (Cytomics FC 500; Beckman Coulter, Inc., Fullerton, CA, USA). Because no specific cell marker has been proven useful in estimating contributing cells for hepatic regeneration, hematopoietic stem cell markers (CD34, CD45, CD133; Miltenyi Biotech, Inc., Auburn, CA, USA) and an epithelial cell marker (CD 117; Immunotech, Beckman Coulter, Marseille, France) were used to assess cell composition. At 4–8 h after harvesting the BMCs, the final MNC preparation was administered into a peripheral vein over a 1-h infusion.
Clinical Follow-up Protocol
Before ABMI, laboratory tests with MRI and liver biopsy were conducted. After ABMI, patients were discharged within 5 days and followed up every week for 2 weeks, and monthly for 6 months. Laboratory tests, body weight check, and a questionnaire for performance status, subjective well-being, and quality of life were checked at every visit. Performance status was scored using a 0–100% grading scale. Subjective well-being and quality of life were scored using a 1–7 grading scale. Indocyanine green clearance was also measured before and 1, 3, and 6 months after the procedure.
MRI Evaluation
All MRI to measure liver volume and ascites, and to screen for HCC, was performed using 1.5-T MR scanners (Gyroscan Intera; Philips Medical Systems Best, The Netherlands). Routine T1-weighted gradient echo, T2-weighted turbo spin echo, T2*-weighted gradient echo, and three-phase, dynamic three-dimensional (3D) T1-weighted images after injection of gadobenate dimeglumine (dose: 0.1 mmol/kg of body weight; Multi-Hance; Bracco SpA, Milan, Italy) were obtained.
The total liver volume was measured with commercially available image postprocessing software (Voxel plus 2; Mevisys, Daeduk, Korea) using the summation-of-areas technique with a 10-mm reconstruction thickness by one radiologist. Right perihepatic ascites was estimated by a 3D measurement [the largest length (x) of ascites x the greatest perpendicular length (y) x sum of the vertical length (z)] on a picture archiving and communication system (PACS) monitor by another radiologist. The radiologist was unaware of the patient history, laboratory results, or biopsy findings.
Liver Biopsy, Tissue Preparation, and Immunohistochemistry
Liver biopsies were performed before and 1,3, and 6 months after ABMI using an ultrasound-guided 16-gauge gun, if the patient consented. Transjugular liver biopsy was attempted as an alternative biopsy in cases with massive ascites or a high risk of bleeding.
The biopsied liver tissues were routinely processed and hematoxylin and eosin (H&E) stained. Tissue sections (4 μm) of formalin-fixed, paraffin-embedded liver specimens were deparaffinized with xylene and rehydrated through graded alcohols. After washing in distilled water, the sections were immersed in 3% hydrogen peroxide to block endogenous peroxidase activity. Antigen retrieval was performed by boiling the sections in 100 mM sodium citrate (pH 6.0) for 15 min in a microwave oven. Primary monoclonal antibodies [cytokeratin 7, clone OV-TL 12/30, Dako, Glostrup, Denmark, 1:100 dilution; proliferating cell nuclear antigen (PCNA), clone PC10, Dako, 1:60 dilution; p21WAF1/Cip1, Clone SX118, Dako, 1:50 dilution] and α-smooth muscle actin (Clone 1A4, Dako, 1:100 dilution) were applied for 30 min at room temperature, followed by washing in PBS. Incubation with the secondary antibody was carried out using the Dako EnVision Rabbit/Mouse kit for 30 min at room temperature. Sections were then developed with diaminobenzidine (Dako) and counterstained with hematoxylin.
The hepatic progenitor cell (HPC) compartments, including HPC activation (ductular reaction) and HPC differentiation (intermediate hepatocyte), were assessed on cytokeratin 7-stained sections (22). Positively stained ductular cells and intermediate hepatoyctes were counted in at least five non-overlapping high-power fields (HPFs; x400), and the average of these scores/HPF was taken. The proliferating hepatocytes were evaluated in PCNA-stained sections; the hepatocytes with nuclear PCNA expression were counted in at least five non-overlapping HPFs, and the average of these scores/HPF was taken. The hepatocytes in replicative arrest were evaluated on p21-stained sections and the p21 labeling index was determined as the percentage of positive nuclei per total number of nuclei counted in at least five non-overlapping HPFs. Stellate cell activation was evaluated in smooth muscle actin-stained sections. Activated hepatic stellate cells were evaluated semiquantitatively, as follows: grade 0, no positive cells; 1+, rare positive cells that required a careful search at high power; 2+, scattered positive cells readily identified at medium power; 3+, scattered or clustered positive cells apparent at low power; 4+, widespread positive cells apparent at low power.
Statistical Analyses
Changes in clinical indices from baseline to months after ABMI were analyzed. Wilcoxon's signed-ranks test with Bonferroni correction was used to compare the values of paired samples. Descriptive statistics were also used. Changes in proportion from baseline to last follow-up were plotted and compared. The analyses were performed using the SPSS 13.0 statistical package (SPSS Inc., Chicago, IL, USA). All statistical analyses were based on two-tailed hypothesis tests with a significance level of p < 0.05.
Ethical Considerations
The protocol for the clinical trial conformed to the ethical guidelines of the Declaration of Helsinki and was approved by the Institutional Review Board (No. 4-2006-0087; approved June 20, 2006) of Severance Hospital in the Yonsei University Health System. Written informed consent was obtained from all patients. This investigational study was performed with the cooperation of Yamaguchi University Graduate School of Medicine in Japan, under a Memorandum of Understanding. The costs related to this study were supported by a grant for the Liver Cirrhosis Clinical Research Center, Seoul, Korea.
Results
Patient Characteristics
Between November 2006 and February 2008, 12 patients were screened. Ten patients were finally enrolled; one patient was excluded due to continuous hyperbilirubinemia, and another patient decided to withdraw due to family opposition. The baseline demographic features and clinical characteristics are listed in Table 1. The median age was 56 (range 43–64) years old. Three patients were male and seven were female. All patients had a history of ascites, and four patients had ascites at enrollment. All patients had Child-Pugh class B cirrhosis. Eight of the 10 patients had been on antiviral treatments, with oral nucleoside/nucleotide analogs. All patients were positive for HBsAg and negative for antibodies against hepatitis C.
Patient Characteristics at Screening
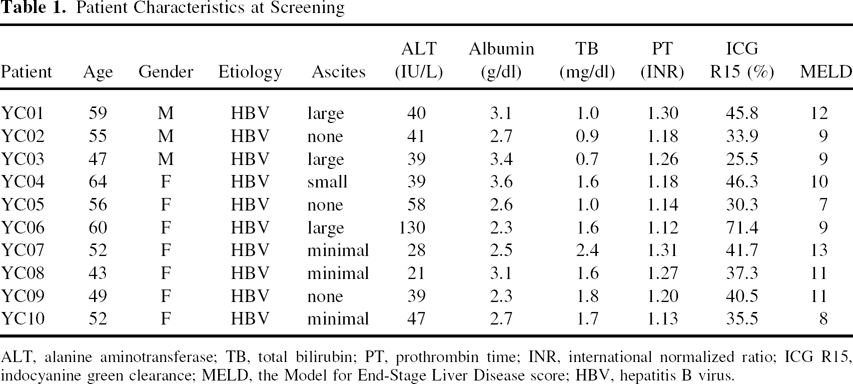
ALT, alanine aminotransferase; TB, total bilirubin; PT, prothrombin time; INR, international normalized ratio; ICG R15, indocyanine green clearance; MELD, the Model for End-Stage Liver Disease score; HBV, hepatitis B virus.
Characteristics of Processed Total Mononucleated Cells
Infused MNC characteristics and composition, according to cellular markers, are summarized in Table 2. The median infused MNC count per body weight was 0.995 × 108 cells/kg (range, 0.48–1.48 × 108). The median infused volume of concentrated MNCs was 112.5 ml (range, 88–150). CD45-positive hematopoietic stem cells were the major cell component (median, 79.85%). CD133-positive hematopoietic stem cells composed 1.05% of total infused cells. CD34-positive stem cells and CD117-positive epithelial cells represented 0.53% and 1.0% of infused cells, respectively.
Infused Mononuclear Cell Characteristics

Changes in Clinical Data
Serial clinical data from each patient were recorded. The initial median albumin level at screening was 2.7 g/ dl. The median albumin level during the 6 months before ABMI was 2.9 g/dl, and this slightly decreased with conservative management, including use of antiviral agents. Beginning at the first month after ABMI, the serum albumin levels gradually increased and reached at peak level to 3.5 g/dl at 9 months, which was maintained at 12 months after ABMI. Significant increase was noted at 6 months after ABMI (Fig. 1A). The median hemoglobin level showed a gradual but significant increase at each time point, from 11.7 g/dl at baseline to 13.8 g/dl at 12 months after ABMI (Fig. 1B). The median cholesterol level improved significantly at 6 and 12 months after ABMI (Fig. 1C). The median prothrombin time tended to improve; a significant difference was seen only at 6 months after ABMI (Fig. 1D). Serum alanine aminotransferase, total bilirubin, and α-fetoprotein levels showed no statistically significant change. The Child-Pugh score improved significantly after 6 months (Fig. 1E); however, there was no change in MELD scores. Performance status reflecting objective daily activity, subjective scoring about feeling of well-being (Fig. 1F), and quality of life increased significantly at 6 months, at 6 and 12 months, and at 6 months after ABMI, respectively.

Serial changes in clinical parameters. Serum albumin (A), hemoglobin (B), serum cholesterol (C), prothrombin time (D), Child-Pugh score (E), and feeling of well-being (F) were serially checked according to the follow-up schedule. Gradual but significant improvements in serum albumin (A), hemoglobin (B), and serum cholesterol (C) were observed. The Child-Pugh score improved significantly at 6 months after ABMI (E). Feeling of well-being improved (F) significantly up to 12 months after ABMI. *p < 0.05, Wilcoxon signed ranks test with Bonferroni correction.
Changes in Liver Volume and the Amount of Ascites, by MRI
Hepatic volumes before and at 6 months after ABMI were compared. The median liver volume increased by 5% after 6 months of ABMI (Fig. 2). Patient 1's liver, in particular, grew by 42% in the 6 months after ABMI.

Changes in liver volume before and at 6 months after ABMI. MRIs taken in the sixth month were analyzed to compare liver volume. Significant liver volume enlargement was observed after 6 month of ABMI. *p < 0.05, Wilcoxon signed ranks test.
The relative amount of ascites between baseline and the last visit was also compared. Among four patients with ascites at baseline, three showed a decreased amount of ascites in spite of reduction or discontinuation of diuretics. The amount of diuretics required was reduced in all patients without weight gain or peripheral edema. None of the patients developed new ascites.
Liver Pathological Features in Serially Biopsied Liver
The HPC compartment, including HPC activation (ductular reaction) and HPC differentiation (intermediate hepatocytes), which showed a positive reaction for cytokeratin-7, was evaluated at baseline and in serial liver biopsies after ABMI (Fig. 3). The baseline liver biopsy in all patients showed a low level of HPC activation and differentiation. The HPC compartment showed a gradual increase in all patients after ABMI. It peaked at 3 months after ABMI and there were 1.4–6.8-fold increases, when compared with the baseline liver biopsy. Liver biopsy at 6 months after ABMI showed a decrease in the HPC compartment to baseline levels. This pattern of HPC compartment activation was consistent in all patients (Fig. 4).

Progenitor cell compartment in the baseline and serially biopsied liver tissue. Ductular reactions and intermediate hepatocytes, which showed positive expression of cytokeratin 7, were counted as the progenitor cell compartment. Before ABMI, the progenitor cell compartment showed a low level of activation (A). The progenitor cell compartment increased gradually at 1 month after ABMI (B) and was markedly activated at 3 months after ABMI (C). At 6 months after ABMI (D), activation of the progenitor cell compartment had decreased to baseline levels. Positive staining is shown as the brown color (original magnification 200x).
The proliferating hepatocytes showed nuclear expression of PCNA. The mean number of proliferating hepatocytes per HPF gradually increased after ABMI and peaked at 3 months after ABMI (Fig. 4B). The number of hepatocytes in replicative arrest was evaluated using the p21 labeling index (Fig. 4C). The p21 labeling indexes were very low, and the maximal value was less than 3.5% in all serially biopsied liver tissues; there was no significant change before and after ABMI.

Serial changes in (A) progenitor cell compartment activation, (B) hepatocyte proliferation, and (C) portion of senescent hepatocytes. (A) Progenitor cell compartment increased significantly to a peak at 3 months after ABMI and decreased at 6 months, with a consistent pattern in all patients. *p < 0.05, Wilcoxon signed ranks test with Bonferroni correcction. (B) Mean proliferating hepatocytes, using PCNA labeling index, tended to increase, but there was no statistical significance. (C) p21 labeling indexes were counted to evaluate hepatocytes in replicative arrest. Indexes were maintained at a very low level without significant change. CK, cytokeratin; HPF, high-power field; PCNA, proliferating cell nuclear antigen; LI, labeling index.
All liver biopsies at baseline and after ABMI showed cirrhosis with mild necroinflammatory activity. There was no significant change in grade or stage of cirrhosis in the serially biopsied livers after ABMI. Additionally, stellate cell activation was grade 2+ in most biopsies, and there was no significant alteration in the activation of stellate cells among baseline and serially biopsied livers after ABMI in any patient.
Toxicity and Complications
No serious adverse events occurred during or after ABMI. Two patients complained of pain at the puncture site on the first day, which subsided without medication.
Discussion
We found that ABMI could induce clinical improvement and increase HPC and hepatocyte proliferation in patients with advanced cirrhosis. To our knowledge, this is the first report to show serial clinicopathological changes, such as activation of the liver progenitor cell compartment, and to reveal how long these alterations were sustained after ABMI. We also demonstrated clinical improvements, consistent with previous data (3,13,26).
Clinical trials of ABMI in chronic liver disease have begun in a few institutes, although the basic mechanism of hepatic regeneration is unclear in the human liver. Peripheral infusion of concentrated autologous BMCs has been reported to improve liver function in patients with cirrhosis (13,26). In this study, we also found that it could improve liver function and quality of life, with objective liver volume increases in patients with advanced LC. The increase in liver volume was especially marked in patient 1, who showed a final liver volume 1.42-fold larger than the initial volume. This patient had undergone a right lobectomy due to HCC 16 months before the ABMI, which was a distinct clinical history among these patients. His liver volume showed no significant changes during the 12 months prior to the ABMI. When hepatocyte loss, such as that caused by partial hepatectomy in experimental models and clinical situations, evokes a rapid regenerative response to restore liver volume (6), we suspect that a suboptimal regeneration after a previous partial resection of the liver may alter the effect of ABMI on the patient's liver growth. Although in our study total MNCs were infused via a peripheral administration route at a different time point long after the operation, the portal administration of autologous CD133-positive BMCs before an operation also accelerates liver regeneration and is a novel therapy supporting hepatic resection (3).
Serum albumin as the most important marker of hepatic function and hemoglobin showed a gradual increase and reached a significant level from 1 month after ABMI. These findings were sustained up to 12 months, although liver biopsies showed that significant activation of progenitor cell compartment had lasted only for 3 months. Serum cholesterol and prothrombin time as other markers representing hepatic function also showed improvement. In four patients who had ascites initially, the amount decreased, even after reducing the dose of diuretics. This is also an indirect marker of hepatic functional improvement. Quality of life and daily activities improved in all patients, and all experienced a better sense of well-being and complained less about fatigue, although we could not completely rule out a placebo effect. In this study, among various clinical parameters, because serum albumin and ascites were improved remarkably, Child-Pugh score had improved significantly, but not MELD score. As factors constituting Child-Pugh score include albumin and ascites, not MELD score but Child-Pugh score had improved. Although there was no remarkable change in liver pathology except progenitor cell compartment, functional improvement was noted. Activation of progenitor cell compartment after ABMI seen in liver pathology might result in increase of functional hepatocytes. And we can guess these hepatocytes could contribute clinical improvements, such as increase in albumin.
All of the patients had hepatitis B virus-related cirrhosis. Eight patients had been receiving antiviral treatment before the procedure, and two patients started antiviral therapy after the procedure. Under antiviral treatment, the viral level was well controlled in most patients. Because there was some doubt that antiviral therapy contributed to the clinical improvement, we compared serum albumin levels during the 6 months prior to ABMI and 6 months after ABMI. There was no improvement in serum albumin levels during the 6 months prior to ABMI, whereas levels rather prominently increased in the same period after ABMI. Furthermore, two patients who started antiviral therapy after ABMI did not contribute more significantly to the changes relative to the remainders who had previously received antiviral therapy. Thus, ABMI seemed to have a major effect on these changes. However, we cannot exclude additive desirable effects of antiviral therapy on these clinical and histological changes, because antiviral treatment has been shown to improve clinical state and liver fibrosis. If there is synergistic or additive effect of ABMI and antiviral therapy, ABMI would be more effective especially in HBV-related cirrhosis.
We found that the HPC compartment, including HPC activation (ductular reaction) and HPC differentiation (intermediate hepatocytes), showed a gradual increase after ABMI in serially biopsied livers. The ductular reaction represents activated progenitor cells, which are thought to correspond to oval cells or oval-like cells in rodents. These cells can differentiate toward functioning hepatocytes through intermediate cells, which are phenotypically between hepatocytes and cholangiocytes (20,21,29). Activation of the HPC compartment has been well documented in acute, subacute, and chronic severe liver injury, when the regeneration of mature hepatocytes is impaired and lost (11). All cirrhotic patients in this study showed a low level of activation of the HPC compartment in the baseline liver biopsy that clearly increased after ABMI. Activation of the HPC compartment peaked at 3 months after ABMI, with a 1.4–6.8-fold increase compared to the baseline liver biopsy. After 6 months, they returned to baseline levels.
Another important cell in liver regeneration is the proliferating hepatocyte. The PCNA labeling index of mature hepatocytes showed a gradual increase, with a peak at 3 months after ABMI. The maximal value was five times higher than the baseline biopsy. These proliferating mature hepatocytes are considered to contribute to clinical improvement because mature hepatocytes are functional and synthesize albumin. These proliferating hepatocytes could be from the activation of quiescent mature hepatocytes or could have originated from the HPC. There were increases in both HPC activation (ductular reaction) and HPC differentiation (intermediate hepatocytes) in all cases of serially biopsied liver tissues after ABMI; thus, at least some portion of the proliferating hepatocytes apparently originated from the HPC. Future studies using cellular tracers would be helpful in identifying the origin of the activated progenitor cells, to establish whether they come from bone marrow. In contrast, the p21 labeling index was very low in all of the biopsied livers, and the maximum p21 labeling index was less than 2.5% after ABMI. Thus, there seemed to be little effect of replicative arrest in liver regeneration in these patients.
The reversibility of liver fibrosis is another interesting issue in ABMI; improvements in liver fibrosis have been reported after bone marrow transplantation in animal experiments (25). Unfortunately, the pathological feature(s) supporting fibrosis reversal was unclear in the biopsied livers. There is a possibility of liver fibrosis reversal after ABMI, because the small biopsied sample may not sufficiently represent the subtle changes in fibrosis, considering sampling error and the uneven distribution of liver fibrosis and large cirrhotic nodules. In addition, 6 months were not sufficient to show the change of fibrosis. However, there was no significant change in stellate cell activation in the serially biopsied liver tissues.
In this study, the effect of ABMI was demonstrated, which can be used in future general clinical application, and there were no serious adverse events. The administration route and cellular processing method were chosen to minimize the risk of complications. Direct injection into the liver or infusion through the hepatic artery, portal vein, or splenic vein may be other options for cellular administration. Other modifications such as clonal selection, cellular tracing, expansion by culture, and repeated injection are important issues for cell therapy. The results of this study can be a basis for further advances and modification.
Our results suggest that autologous BMCs infused via a peripheral vein contribute to regeneration of the liver and improve hepatic function via progenitor cell compartment activation and hepatocyte proliferation, which lasts for 6 months or less. Because of the small number of patients, we cannot generalize these findings. However, if further studies reveal benefits, ABMI may be helpful in patients with advanced LC as a bridge therapy before liver transplantation. Further comparative studies are warranted.
Footnotes
Acknowledgments
The authors thank S. M. Yoon for technical assistance, S. O. Ki for coordination of the trial and K. J. Song for statistical advice. This study was supported by a grant of the Good Health R&D project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (No. A050021).
