Abstract
There is currently no effective treatment method available for liver fibrosis. We therefore evaluated the use of Wharton's jelly stem cells (WJSCs; the major umbilical cord stem cell population) to treat chemically induced liver fibrosis via intraperitoneal injection of thioacetamide. WJSCs were transplanted into liver-damaged rats via the portal vein and the treatment was evaluated by assessing serum biochemistry and histopathology. Transplanted WJSCs were distributed in the fibrotic area and around blood vessels, and hepatic recovery was accelerated. Serum prothrombin time significantly recovered, and serum albumin also improved at 21 days posttransplantation; collagen accumulation also decreased at 14 days. Thus, human WJSCs promoted recovery after chronic liver damage. Using immunohistochemical analyses, we determined that transplanted WJSCs produce albumin, hepatocyte growth factor (HGF), and metalloproteinase (MMP) after transplantation to chemically injured liver, indicating that WJSC may help to decrease liver collagen and thus may be useful for treating liver fibrosis.
Introduction
According to the World Health Organization (WHO) census, chronic liver diseases are a serious worldwide health problem (18). Hepatitis B, hepatitis C, alcohol, and certain chemicals can result in liver fibrosis and cirrhosis, and ultimately in liver failure. Currently, liver transplantation remains the only curative option for patients with liver failure (11).
The potential role for stem cell therapy in the treatment of liver diseases has lately become topical in medical research because of the self-renewal characteristics and differentiation potential of stem cells. Stem cells derived from bone marrow or umbilical cord blood have the potential to become various kinds of cells (10,31—33) and hepatocyte-like cells (3,21,25,27,30,37,39). In addition, in vivo studies indicate that transplanted stem cells can differentiate into hepatocyte-like cells and express specific markers such as albumin, tryptophan 2, 3-dioxygenase, and cytokeratin 18 (CK18) (5,27,35). Transplantation of rat stem cells accelerates an animal's recovery following liver damage (2,14,15,29,38).
Excess deposition of collagen is the main characteristic of liver fibrosis induced by chronic viral hepatitis, alcohol abuse, and drug toxicity. Metalloproteinase plays an important role in digesting collagen in fibrotic liver, and studies have revealed that increased expression of metalloproteinase can help to degrade collagen in the fibrotic liver (26,28). Further, other stem cells (e.g., mouse embryonic stem cells) can secrete metalloproteinase to degrade collagen, but this has not been clearly established for Wharton's jelly stem cells.
Wharton's jelly from human umbilical cord is an important source of stem cells (22). Wharton's jelly stem cells (WJSCs) are easily isolated and expanded ex vivo, and under certain conditions they have the potential to differentiate into neuron-like and cartilage-like cells (22), and can be adipogenic, cardiogenic, chondrogenic, and osteogenic (36). To date, however, neither the hepatic differentiation potential of WJSC nor their potential in the treatment of liver fibrosis has been thoroughly described. Therefore, the purpose of this study was to evaluate the feasibility of WJSC therapy for managing thioacetamide-induced liver damage. A rat liver fibrosis model was established, and both liver biochemical function and the resulting histopathology were assessed to monitor the recovery from liver damage.
Materials and Methods
Cells and Cell Culture
The WJSCs used in this study were originally isolated and expanded from a donated human umbilical cord with the donor's consent, according to a previously reported protocol (36). The WJSCs isolation was approved by the Institutional Review Board of Buddhist Tzu Chi General Hospital. The cells obtained were cultured in Iscove's modified Dulbecco's medium (IMDM; Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (HyClone, Logan, UT, USA), 10 ng/ml basic fibroblast growth factor (R&D, Minneapolis, MN, USA), 2 mM L-glutamine, and 100 U/L penicillin-streptomycin (Invitrogen, Carlsbad, CA, USA) in a 37°C incubator with 5% CO2. Cells with passage number 10 to 20 were used in this study.
Surface Phenotype Characterization
WJSC surface markers were characterized by flow cytometry (FC500; Beckman Coulter, Brea, CA, USA) after being labeled with various antibodies, including those against human CD105 (Santa Cruz Biotechnology, Santa Cruz, CA, USA); CD13, CD14, CD29, CD34, CD44, CD45, HLA-DR, and IgG2a (Dako, Carpinteria, CA, USA), and CD49b, CD49d, CD73, and CD90 (Becton Dickinson, Franklin Lakes, NJ, USA). Secondary antibodies conjugated with fluorescein isothiocyanate were purchased from Chemicon (Temecula, CA, USA).
In Vitro Hepatic Differentiation
In vitro hepatic differentiation was tested by a modified coculture protocol (13). With a trans-well membrane (pore size 0.4 μm; Becton Dickinson) separating the two cell populations, 3 × 104 WJSCs were cocultured with 50 mg liver tissue in each well of a six-well plate. The liver tissue was obtained from male C57BL/6 mice that had been exposed to a hepatotoxic reagent [thioacetamide (TAA) Sigma-Aldrich, St. Louis, MO, USA] at 350 mg/kg, intraperitoneally, 24 h prior to the coculture experiment. On day 2 and day 4 during the coculture, total RNA from WJSC was extracted to examine the expression of liver-specific genes. Polymerase chain reaction (PCR) was performed using specific primers as follows: albumin—(F) 5′-TGCTTGAATGTGCTGAT GACAGGG-3′ and (R) 5′-AAGGCAAGTCAGCAGG CATCTCATC-3′; tryptophan 2,3-dioxygenase—-(F) 5′-TGGTACTCTCCTCAATCTGCTG-3′ and (R) 5′-CTC TGGATTGACTGTGGAAGT-3′; α-fetoprotein—-(F) 5′-ATACAGAGACTTCAGGAGC-3′ and (R) 5′-GTGAA GAGGGAAGACATAACTG-3′; hepatocyte growth factor (HGF)—-(F) 5′-CAGATCATCCATTGCATTCG-3′ and (R) 5′-ACTCCAGAGGCATTTCCATG-3′; metalloproteinase (MMP)—-(F) 5′-TgAATgCCCTTgATgTC ATCCT-3′ and (R) 5′-ACACCTACACCAAgAACTT C-3′; CYP7A1—-(F) 5′-GAGAAGGCAAACGGGTGA AC-3′ and (R) 5′-ATCGGGTCAATGCTTCTGTG-3′; nanog—-(F) 5′-TGCCTCACACGGAGACTGTC-3′ and (R) 5′-TGCTATTCTTCGGCCAGTTG-3′; oct4—-(F) 5′-CTTGCTGCAGAAGTGGGTGGAGGAA-3′ and (R) 5′-CTGCAGTGTGGGTTTCGGGCA-3′; ckit—-(F) 5′-AT GAGAGGCGCTCGCGGCGC-3′ and (R) 5′-AGCTTG GCAGGATCTCTAAC-3′; glyceraldehyde 3 phosphate dehydrogenase—(F) 5′-GGGCTGCTTTTAACTCTGG T-3′ and (R) 5′-GCAGGTTTTTCTAGACGG-3′. PCR was performed for 35 cycles, with each cycle consisting of denaturation for 30 s at 95°C, annealing for 30 s at 56—60°C (based on the primer set used), and elongation for 60 s at 72°C, with a final 10-min incubation at 72°C. The amplified cDNAs were analyzed by gel electrophoresis.
Establishment of the Rat Liver Fibrosis Model
In this study, Wistar Kyoto (WKY) rats were obtained from LASCO CO., LTD (Taipei, Taiwain). All procedures followed the ethical guidelines and were approved by the Institutional Animal Care and Use Committee of Dong-Hwa University.
To establish the chronic liver fibrosis model in rat, 20 adult male WKY rats (320 ± 20 g) were used. Twelve out of 20 rats were intraperitoneally injected with 200 mg/kg TAA (Sigma-Aldrich) once every 3 days for 60 days (i.e., 20 injections) as the fibrosis model group. In the control group (8 out of 20 rats), normal saline was used in place of the TAA. At day 64 (i.e., 4 days after the last injection), these rats were sacrificed and cardiac blood samples were collected. These samples were analyzed with a biochemical analyzer (Integra 800; Roche, Holliston, MA, USA) to measure the liver function index, which included glutamate oxaloacetate transaminase (GOT), glutamate pyruvate transaminase (GPT), serum albumin, and prothrombin time.
Afterwards, the histopathology of liver tissue samples obtained from the sacrificed rats was also analyzed. Liver tissue samples were fixed in 3.7% formaldehyde and then embedded in paraffin. Serial 3-μm sections of the embedded tissues were stained with hematoxylin and eosin or Masson's trichrome. Liver fibrosis is the excessive accumulation of collagen in liver, and therefore the accumulation of collagen is an important index for evaluating liver fibrosis (6). Masson's trichrome stain is frequently used to identify increases in collagenous tissue in fibrotic liver. For Masson's trichrome stain, sectioned samples were placed in Bouin's solution (Sigma-Aldrich) at 56°C for 1 h and then were stained sequentially with the following solutions: Mayer's hematoxylin solution (Sigma-Aldrich) for 5 min, Biebrich scarlet-acid fuchsin solution (Sigma-Aldrich) for 15 min, phosphomolybdic acid-phosphotungstic acid (Sigma-Aldrich) for 15 min, and aniline blue (Sigma-Aldrich) for 5 min.
Cell Transplantation Experiment
WJSCs (2 × 107) that had been cultured in 75-cm2 T-flasks as described above were detached by trypsinization and collected by centrifugation. These cells were then resuspended in normal saline to a concentration of 2thinsp;x 106 cells/ml.
The cell transplantation protocol was modified from that reported previously (1,38). Thirty rats with TAA-induced fibrotic livers were used for cell transplantations at day 64 (i.e., 4 days after the last TAA injection) as described in the establishment of the rat liver fibrosis model section. These rats were randomly divided into two groups for the transplantation experiment. Fifteen rats in the WJSC group were anesthetized with ether and injected with 1 × 106 WJSCs (in 0.5 ml of normal saline) via the portal vein. The other 15 rats (sham group) were treated with 0.5 ml normal saline only.
After transplantation, five rats each from the WJSC and sham groups were sacrificed weekly for 3 weeks. The biochemical liver function index and liver histopathology were also analyzed at each time point.
Monitoring Transplanted WJSCs
Monoclonal antibodies specific for human albumin, human mitochondria, metalloproteinase, and human hepatocyte growth factor were used to monitor the transplanted WJSCs. Sectioned paraffin-embedded liver samples (as described above) were subjected to immunohistochemistry with anti-human albumin (1:1000; Sigma-Aldrich), anti-metalloproteinase (1:200; Abcam, Cambridge, MA, USA), and anti-human hepatocyte growth factor (1:200, Abnovus, Walnut, USA). Subsequently, an immunohistochemistry kit (InnoGenex, San Ramon, CA, USA) was used to visualize the immune complexes. For immuno-fluorescence staining, sectioned paraffin-embedded liver samples were incubated overnight with anti-human mitochondria (1:50; Chemicon, Billerica, MA, USA). After removing the primary antibody, rhodamine-conjugated anti-mouse secondary antibody (Chemicon, Billerica, MA, USA) was added for 2 h, and the samples were then evaluated by fluorescence microscopy.
Quantification of Liver Fibrosis
For quantification of liver fibrosis in each experimental group, Liver fibrosis was quantified using the method reported by Bruck et al. (7). According to their method, the liver sections were stained with Masson's trichrome. The Masson's trichrome-stained liver tissue slices were photographed and scored semiquantatively by three independent pathologists. The degrees of liver fibrosis of different experimental group were shown as the average of 10 different fields within each slide, which had been classified on a score of 0—3. A higher fibrosis score signifies a more serious degree of liver fibrosis. By using this method, the liver fibrosis degree in each experimental group was determined.
Statistical Analysis
All data were shown as mean with SD. For the comparison of different treatments in two groups, the data were analyzed by using the Student's t-test. Values of p < 0.05 were considered significant. The Bonferroni correction was used for the multiple comparisons.
Results
Characterization of WJSCs
WJSC morphology and surface phenotype were evaluated by microscopy and flow cytometry, respectively.
The surface phenotypes of WJSCs isolated for this study were similar to that described previously for WJSCs (35). The WJSCs obtained in this study did not express hematopoietic markers CD14, CD34, or CD45 but did express the mesenchymal stem cell markers CD29, CD44, CD49b, CD49d, CD73, CD90, and CD105, the myeloid marker CD13, and the major histocompatibility complex marker HLA-DR (Fig. 1A—M). In addition, these cells showed a fibroblast-like morphology (Fig. 1N).

Surface phenotype and typical morphology of Wharton jelly stem cells (WJSCs) observed in this study. Immunohistochemical staining with antibodies against (A) CD13, (B) CD14, (C) CD29, (D) CD 34, (E) CD44, (F) CD45, (G) CD49b, (H) CD49d, (I) CD73, (J) CD90, (K) CD105, (L) HLA-DR, and (M) IgG2a-FITC as a positive control. (N) Morphology of WJSCs. Left panel: low-density culture; right panel: high-density culture. Scale bar: 50 μm.
The expression of hepatic-related WJSC genes, and thus the ability of these cells to undergo hepatic differentiation, was examined by coculturing WJSCs with TAA-damaged mouse liver tissue. RT-PCR showed that WJSCs cultured under the experimental conditions expressed liver-specific genes including CK18, albumin, tryptophan 2,3-dioxygenase (TO), α-fetoprotein (AFP), and CYP7A1 (Fig. 2). Further, the expression of stem cell genes, including nanog, oct4, and ckit, were decreased while WJSCs were cocultured with TAA-damaged liver. These results suggest that the WJSCs had the potential to differentiate into hepatocyte-like cells in vitro. In addition, the gene expression of HGF and MMP were increased while WJSCs differentiate into hepatocyte-like cells.
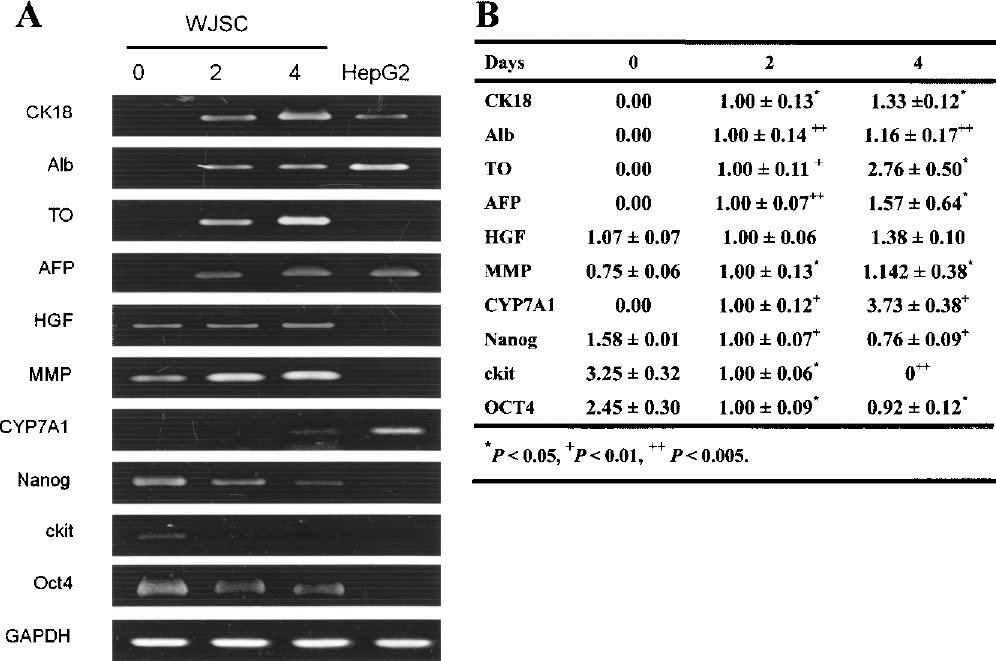
The expression of liver-specific genes in WJSCs as a measure of their hepatic differentiation. The WJSCs were cocultured with thioacetamide (TAA)-treated liver tissue and the total RNA was extracted from WJSCs at day 0, 2, and 4, respectively. (A) The expression of hepatic-specific genes was analyzed by RT-PCR and gel electrophoresis. (B) The semiquantitative result from RT-PCR. The gene expression at day 2 or day 4 was compared with gene expression at day 0, respectively. *p < 0.05, +p < 0.01, ++p < 0.005. The Bonferroni correction was used for the multiple comparisons.
Establishment of a Liver Fibrosis Model and Evaluation of WJSC Cell Therapy
To confirm the liver fibrosis model, 12 of 20 rats were treated with TAA every 3 days for 60 days as the liver fibrosis rat group. The other rats (8 of 20) were treated with normal saline as a normal rat group. Rats in both groups were sacrificed at day 64 to examine the liver function index and to score histopathology. In evaluation of WJSC cell therapy, 30 rats were subjected to TAA treatment as described for the liver fibrosis model. At day 64, rats were divided into two groups and either WJSCs (WJSC group) or saline (sham group) was introduced into the liver-damaged rats (n = 15 for each group) through the portal vein. Five rats from each group were evaluated at days 7, 14, and 21 after transplantation, as described in Materials and Methods.
In the liver fibrosis rat group, the GOT and GPT levels, compared to the normal rat group, were increased (GOT from 110 ± 16 to 1034 ± 361 U/L, and GPT from 77 ± 10 to 185 ± 50 U/L), indicating liver damage (Table. 1). Decreased albumin (from 4.11 ± 0.43 to 3.43 ± 0.08 g/dl) and significantly increased prothrombin time (from 9.4 ± 0.11 to 11.5 ± 1.0 s) were also found in the blood. The biochemical results in the TAA-induced liver fibrosis experiments were consistent with previous reports (1,16,24,38), suggesting significantly reduced liver function in these animals. In the WJSC group and the sham group, biochemical liver function indices were also examined (Table. 2). GOT and GPT levels declined to near-normal values in both the WJSC and sham groups at day 7 after transplantation. GOT and GPT levels did not differ significantly between groups. The observed low GOT and GPT levels may be attributable to the short half-lives of GOT and GPT (i.e., 17 and 47 h, respectively) (17).
Plasma Biochemical Value of Normal Rats and Liver Fibrosis Rats

Data represent mean ± SD. The normal rats group did not receive any treatment and the liver fibrosis rats group was injected with 200 mg/ kg once every 3 days for 60 days. After 60 days, the normal rats or liver fibrosis rats were sacrificed and the cardiac blood samples were collected from normal rats or liver fibrosis rats, respectively. The biochemical values, GOT (glutamate oxaloacetate transaminase), GPT (glutamate pyruvate transaminase), albumin, and prothrombin time, of cardiac blood samples were analyzed with a biochemical analyzer.
p < 0.01, the liver fibrosis rats compared to the normal rats.
p < 0.05, the liver fibrosis rats compared to the normal rats.
Plasma Biochemical Value of Wharton Jelly Stem Cell Group (WJSC-Transplanted Liver Fibrosis Rats) or Sham Group (Saline-Treated Liver Fibrosis Rats)

Data represent mean ± SD. Fifteen thioacetamide (TAA)-induced liver fibrosis rats were transplanted with 1 × 106 WJSCs (in 0.5 ml normal saline) via the portal vein as WJSC group. Another 15 TAA-induced liver fibrosis rats were injected with 0.5 ml normal saline via portal vein only as sham group. After WJSC transplantation or normal saline treatment, five rats each from the WJSC or sham groups were sacrificed on day 7, 14, and 21 and the cardiac blood samples were collected. The biochemical values, GOT (glutamate oxaloacetate transaminase), GPT (glutamate pyruvate transaminase), albumin, and prothrombin time, of cardiac blood samples were analyzed with a biochemical analyzer. WJSC, TAA-induced liver fibrosis rats transplanted with WJSC; sham group, TAA-induced liver fibrosis rats treated with 0.5 ml normal saline.
p < 0.01, WJSC at 21 days compared to the sham at day 21.
Although the GOT and GPT levels did not differ between groups, prothrombin time indicated significant differences between the two groups at 21 days and the albumin was approaching normal levels in the WJSC group at 21 days. Prothrombin time was significantly different between the WJSC group and the sham group 21 days after transplantation (9.5 ± 0.1 vs. 9.9 ± 0.2, p < 0.01). At 21 days posttransplantation, serum albumin in the WJSC group had recovered to a greater extent than in the sham group (3.84 ± 0.22 vs. 3.52 ± 0.14). These changes suggest that liver function had recovered after 21 days of WJSC therapy (Table. 2).
Hematoxylin and eosin staining of normal rat liver tissue sections indicated that there was no damage, as expected (Fig. 3A). In comparison, tissue vacuolation, necrosis, and the degeneration of cell nuclei were observed in liver sections from TAA-induced liver fibrosis rats (Fig. 3B). Histopathologic data also suggested that WJSC transplantation ameliorated liver damage; recovery of tissue degeneration and vacuolation was evident in the WJSC group at day 21, whereas the sham group still showed serious inflammation and necrosis (Fig. 3C, D).
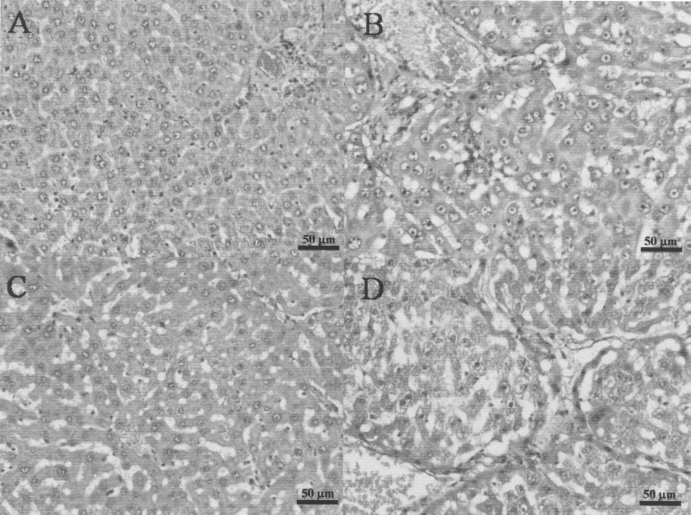
Hematoxylin and eosin staining of rat liver sections. (A) Liver section of a normal rat. (B) Liver section of a liver fibrosis rat (Rats were injected with TAA 200 mg/kg once every 3 days for 60 days). (C) Liver section of WJSC group at day 21 after WJSC transplantation. (D) Liver section of sham group at day 21 after normal saline injection. Scale bars: 50 μm.
Masson's trichrome stain revealed severe accumulation of collagen in livers from the liver fibrosis rat group (Fig. 4). The quantification of the degree of liver fibrosis in the liver fibrosis rat group was significantly higher than in the normal rat group (Table. 3). This suggested that TAA injection indeed induced liver fibrosis in rats. “Bridging,” which results from accumulated collagen that connects two blood vessels, was found in most areas in the liver fibrosis group. This phenomenon was very similar to previous liver fibrosis reports (4,6,16,20) and was not found in the control group (Fig. 4A, B).

Masson's trichrome staining of liver sections. (A) Liver sections of a normal rat. (B) Liver section of liver fibrosis rat. (C) Liver section of WJSC group at day 7. (E) Liver section of WJSC group at day 14. (G) Liver section of WJSC group at day 21. (D) Liver section of sham group at day 7. (F) Liver section of sham group at day 14. (H) Liver section of sham group at day 21. Scale bars: 200 μm.
The Quantity Score of Liver Fibrosis in Each Experimental Group

Values shown as mean ± SD. Normal rats: Rats did not receive any treatment; liver fibrosis rats: Rats were injected with TAA 200 mg/kg once every 3 days for 60 days; sham group: TAA-induced liver fibrosis rats were injected with 0.5 ml saline via portal vein; WJSC group: TAA-induced liver fibrosis rats were transplanted with WJSCs via portal vein.
p<0.01 the WJSC at day 14 compared to the sham at day 14 and the WJSC at day 21 compared to the sham at day 21.
Using Masson's trichrome staining to assess collagen expression, there was no difference in collagen content at day 7 in the 10 rats analyzed from each group (Fig. 4C, D, Table 1). Collagen degradation was found at day 14 in the WJSC group (Fig. 4E, F, Table 1). The WJSC group rarely showed collagen accumulation at day 21 (Fig. 4G, Table 1), whereas the sham group still showed substantial collagen accumulation (Fig. 4H, Table 1) at 21 days after transplantation.
Monitoring Transplanted Cells
To follow the transplanted human cells in this study, antibodies specific for human albumin, human mitochondria, metalloproteinase, and human HGF were used to stain sectioned liver samples. The human albumin-positive cells were detected in the WJSC group up to 21 days after transplantation (Fig. 5A, B) but in the sham group the albumin-positive cells were not detected (Fig. 5C, D). This result suggested that the transplanted WJSCs differentiated into albumin-secreting hepatocyte-like cells in the damaged livers of the rats from the WJSC group. Immunohistochemical staining for metalloproteinase revealed many metalloproteinase-positive cells in the peri-venule and bile duct areas in the WJSC group (Fig. 6A). However, metalloproteinase-positive and human mitochondria-positive cells were not observed in the sham group (Fig. 6B). The metalloproteinase-positive cells in the WJSC group were also costained with human mitochondria (Fig. 6C) but were not observed in the sham group. This indicated that these metalloproteinase-positive cells were transplanted WJSCs. Further, the human HGF-positive cells were observed and along the collagen fiber in the damaged liver of the WJSC group (Fig. 7A) but were not observed in the sham group. This indicated that the transplanted WJSCs could express human HGF in damaged liver.
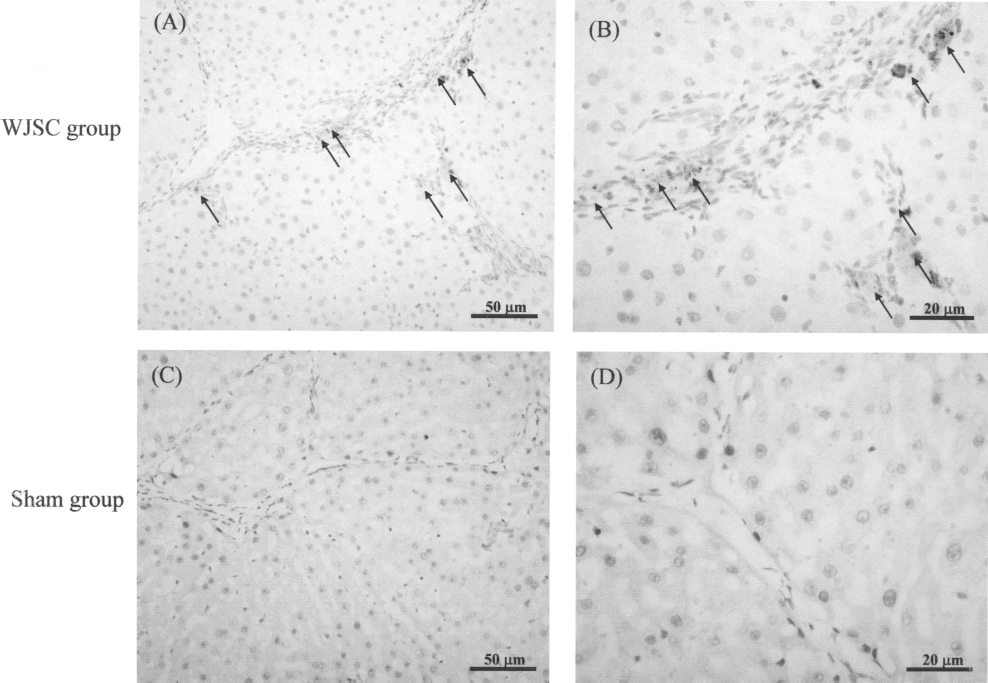
Human albumin immunostaining of WJSC group and sham group liver sections at day 21 after transplantation. (A, B) The liver section of WJSC group; (C, D) the liver section of sham group. Antibodies specific for human albumin was used. Brown staining indicates the human albumin-positive cells. Scale bars: (A, C) 50 μm; (B, D) 20 μm.

Immunostaining for metalloproteinase and mitochondria in liver sections of WJSC group and sham group at day 21 after transplantation. (A) The immunostaining for metalloproteinase in liver sections from a WJSC rat. (B) The immunostaining for metalloproteinase in liver sections from a sham rat. (C) The immunostaining for human mitochondria in liver sections from a WJSC rat. (D) The immunostaining for human mitochondria in liver sections from a sham rat. Black and white arrows: metalloprotease and human mitochondria-positive cells. Arrowheads: metalloprotease-positive bile duct cells. Scale bars: 10 μm.
Discussion
We have established a model system for studying liver damage following periodic injections of TAA for 60 days. In this study, WJSCs had the potential to differentiate into hepatocyte-like cells when cocultured with chemically damaged mouse liver in vitro. The biochemical liver function index and histopathological analysis from in vivo experiments revealed that the human WJSCs promoted the recovery of TAA-induced hepatic damage and thus may have potential cell therapy applications.
There are several explanations for the amelioration of TAA-induced liver damage facilitated by WJSCs. First, immunostaining of WJSCs incorporated into the TAA-induced fibrotic liver indicated that WJSCs could differentiate into albumin-producing hepatocyte-like cells (Fig. 5A, B). The in vitro study showed that WJSCs expressed liver-specific genes when cocultured with chemically damaged liver tissue (Fig. 2). These hepatocyte-like cells that differentiated from WJSCs may play an important role in liver function recovery. Moreover, in the animal study, the WJSC-derived hepatocyte-like cells were likely responsible for facilitating the recovery of TAA-induced cell damage. Second, HGF might play a role in the acceleration of liver function recovery. HGF can facilitate damaged liver regeneration because of its antiapoptotic effects of signaling through c-met (8,9,19,23,40). The WJSCs used in this study could express HGF mRNA (Fig. 2) and secrete HGF in fibrotic liver (Fig. 7). This led us to speculate that the WJSCs could secrete HGF and accelerate liver recovery. Third, the collagen content in the WJSC group decreased every week; at day 21 posttransplantation, there was little collagen in liver sections. By comparison, the sham-treated group showed no collagen decrease (Fig. 4, Table 1). Rat mesenchymal stem cells have a potential therapeutic effect on the fibrotic process through their ability to minimize collagen deposition in addition to their capacity to differentiate into hepatocytes (1,24). Our study is the first to describe a similar reduction in collagen deposition in response to exposure to WJSCs.
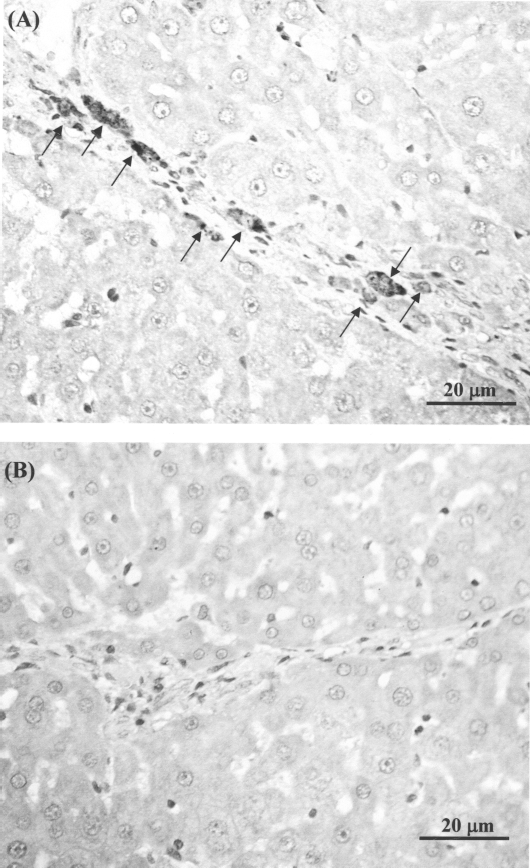
Human hepatocyte growth factor immunostaining of WJSC group and sham group liver sections at day 21 after transplantation. (A) The liver section of WJSC group. (B) The liver section of sham group, which was only treated by normal saline. Antibodies specific for human hepatocyte growth factor (HGF) was used. Brown staining and arrows indicate the human HGF-positive cells. Scale bar: 20 μm.
The matrix metalloproteinase family plays an important role in collagen degradation (11,12). Since Whar-ton's jelly secretes certain matrix metalloproteinases (34), it is reasonable to presume that transplanted WJSCs can secrete metalloproteinase, which would help to degrade collagen in the fibrotic liver. Indeed, immunohistochemical staining of the TAA-damaged liver 21 days after WJSC transplantation revealed that metalloproteinase expression was significantly increased in WJSC (Fig. 6). Increased metalloproteinase expression was observed in the peri-venule area in our study, where WJSCs were often observed. Interestingly, metalloproteinases were also present in the proliferating bile ducts where no WJSC were observed. One possible explanation is that WJSCs generated a microenvironment that induced metalloproteinase expression in bile ducts. Both sources of induced metalloproteinase expression may contribute to the degradation of collagen fibers in TAA-induced liver fibrosis. Further investigation is required to elucidate the precise mechanism of collagen degradation that occurs after WJSC transplantation.
In conclusion, our results demonstrate that WJSC could different into hepatocyte-like cells in vitro. The WJSC also could express MMP and HGF in vitro. In addition, in an in vivo study WJSC differentiated into hepatocyte-like cells. Besides, WJSC could express HGF and metalloproteinase, an attribute that also was observed in the surrounding bile duct, in vivo. The expression of HGF and metalloproteinase in undifferentiated WJSC as well as the WJSC-mediated differentiation of cells into hepatocyte-like cells facilitated regeneration of damaged liver in TAA-induced liver fibrosis. WJSC, the major stem cell population of the umbilical cord, are thus a promising source of cells for the treatment of liver fibrosis.
Footnotes
Acknowledgments
This work was supported by a Grant from Buddhist Tzu Chi General Hospital (TCRD94-16) and Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH99-TD-B-11-004). This work was also supported by grants (No. 09/426K9) from the Gwo Xi Stem Cell Applied Technology, Hsinchu, Taiwan, ROC.
