Abstract
Umbilical cord mesenchymal stromal cells (UC-MSCs) are emerging as leading stem cells in regenerative medicine due to their high proliferative capacity, potent immunomodulatory effects, and non-invasive collection. However, the absence of standardized guidance on optimal passage number and tissue-specific characterization criteria limits clinical translation, raising concerns about variability in potency, genomic stability, and safety. This review synthesizes evidence on how in vitro passaging alters UC-MSC properties, including purity, proliferation, senescence, genomic integrity, differentiation, and immunomodulation. Preclinical data suggest that UC-MSCs tolerate extended passaging better than MSCs derived from other sources, with several functional attributes preserved up to later passages. Clinical evidence indicates that early to middle passages (P3–P5) achieve the best balance between scalability and therapeutic efficacy. We further propose an updated immunophenotypic framework incorporating tissue-specific positive and negative markers to enhance clinical-grade characterization. Establishing harmonized passage guidelines and potency assays is essential to maximize reproducibility, safety, and the translational potential of UC-MSC therapies.

Introduction
Umbilical cord-derived mesenchymal stromal cells (UC-MSCs) have gained recognition as promising stem cells in regenerative medicine, attributed to their high proliferative and differentiation potential coupled with low immunogenicity and immunomodulatory properties 1 . UC-MSCs modulate major immune effectors and exert anti-inflammatory effects through both cell-to-cell contact and the secretion of bioactive factors 2 . Leveraging these properties, the clinical applications of UC-MSCs encompass a diverse range of diseases, including autoimmune disorders, neurological injuries, and age-related degeneration3–5. With the increasing use of UC-MSCs in clinical trials, it is imperative to ensure consistent cell quality and safety for clinical-grade preparations. This goal can be partly achieved through the standardization of cell characterization protocols that support regulatory compliance, product reproducibility, as well as therapeutic efficacy and safety 6 .
A critical factor influencing the characteristics of UC-MSCs is the number of cell passages during in vitro expansion. Studies have demonstrated that cell passage numbers influence biological properties, including immunophenotype, proliferation rate, differentiation potential, genetic stability, cytokine secretion, and immunomodulatory effects as a result of in vitro aging7,8. Despite this, international guidelines on optimal passage selection for therapeutic use remain absent, and most studies evaluate MSCs properties at a single passage rather than across successive passages 7 . This lack of harmonization introduces risks of inconsistent product quality, variable therapeutic potency, and challenges in regulatory approval, thereby limiting clinical translation.
Moreover, the immunophenotypic profiling that is defined by the expression of surface markers remains the most widely accepted criterion for MSCs identification and validation 9 . The immunophenotypic characterization of human MSCs is delineated according to the guidelines established by the International Society for Cellular Therapy (ISCT), which stipulates that MSCs should express positive markers and lack negative markers to maintain their purity and lack of non-MSCs population 10 . Yet, the current MSCs positive markers exhibit overlaps with fibroblast profiles and do not necessarily constitute indicators of their clinical functionality11,12. Moreover, negative marker identification has largely been limited to the exclusion of hematopoietic and immune cell populations 10 . While current MSCs characterization guidelines provide a useful framework for standardization that facilitates comparisons across studies, the concerns about the sufficiency for clinical decision-making raise as these guidelines may not fully capture the complexity of MSCs identity and function, which require further refinement and standardization 13 .
In this review, we critically evaluate the effect of passage numbers on the biological characteristics and functional suitability of UC-MSCs in clinical implications, while proposing additional immunophenotypic markers that can be integrated into updated characterization guidelines for UC-MSCs. By addressing these gaps, we aim to clarify the relationship between passage-dependent alteration in UC-MSCs and their therapeutic potency to facilitate the development of standardized and clinically relevant criteria for clinical-grade UC-MSCs characterization and application.
Methodology
This narrative review examines the impact of passage number on the biological and functional properties of umbilical cord mesenchymal stem cells (UC-MSCs). A targeted search of the PubMed database was conducted for peer-reviewed articles published within the last five years using keywords including “umbilical cord,” “Wharton’s jelly,” “mesenchymal stem cells,” “mesenchymal stromal cells,” “passage number,” and “immunomodulation.” To minimize selection and reporting bias, all eligible articles meeting the search criteria were considered regardless of country of origin, study design, sample size, or direction of findings. Studies were included based on their relevance to UC-MSC expansion, passage-dependent characteristics, and immunomodulatory outcomes. Data from both preclinical and clinical studies were qualitatively synthesized to identify recurring themes, consensus, and existing knowledge gaps regarding passage-associated changes.
Umbilical cord mesenchymal stromal cells (UC-MSCs)
Stem cells are defined by their capacity for self-renewal and differentiation. Their developmental potential, or “potency,” ranges from totipotent zygotes, which can form all embryonic and extraembryonic tissues, to pluripotent embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), which can differentiate into cells of all three germ layers but not placental tissue. In contrast, mesenchymal stromal cells (MSCs) and hematopoietic stem cells (HSCs) are multipotent, restricted to specific lineage differentiation14,15. Although pluripotent stem cells hold great therapeutic promise, their clinical use is limited by risks of tumorigenicity, immunogenicity, and ethical concerns related to embryo-derived ESCs16,17. MSCs, by comparison, present a more practical alternative. They can be readily isolated from adult and perinatal tissues, demonstrate reduced tumorigenic risk, and possess low immunogenicity due to minimal expression of MHC-II and co-stimulatory molecules, features that enable their safe allogeneic application18,19.
Among MSC sources, the umbilical cord (UC) has gained particular attention as it is obtained from postnatal tissue usually discarded as medical waste, making it a non-invasive and ethically acceptable source 20 . UC-MSCs can be isolated from four main compartments: Wharton’s Jelly (WJ), perivascular region (PV), sub-amnion, and amnion membrane 21 (Fig. 1). Of these, WJ is the most widely studied; fibroblast-like cells with MSC characteristics were first reported from this tissue 22 . WJ-MSCs are advantageous over other types of stem cells due to their high proliferation rate, non-invasive sources, strong stemness, broad multilineage differentiation potential, and strong immunomodulatory properties, which lowers the risk of rejection, reinforcing their suitability for clinical translation23,24.

Graphical illustration of the umbilical cord showing the arteries and vein, as well as the four main compartments: the amnion membrane, Wharton’s jelly, sub-amnion, and perivascular region. This illustration was created by the authors using Canva Pro.
Clinical-grade isolation and expansion protocols
The translation of UC-MSCs from research to therapeutic use requires isolation and expansion strategies that are safe, reproducible, and GMP-compliant. Two main approaches are commonly employed: explant culture and enzymatic digestion, each shaping heterogeneity and influencing passage-dependent outcomes.
Explant culture relies on MSC migration from tissue fragments, with outgrowth typically observed within 8-10 days. It is simple, cost-effective, and avoids enzyme-induced stress, often yielding cultures of higher purity and reduced heterogeneity at early passages25,26. However, tissue detachment and floating fragments can reduce yields, though refinements such as pre-incubation and anchoring devices have improved adherence and proliferation27,28.
Enzymatic digestion accelerates isolation by degrading the extracellular matrix, enabling earlier proliferation (6–8 days) and higher initial cell numbers. This efficiency is primarily attributed to the use of enzymes including collagenase, hyaluronidase and trypsin, which effectively break down the UC tissue components25,29. Yet, enzyme exposure can compromise viability, and early passages frequently show heterogeneous populations with increased hematopoietic contamination 30 . Optimizing digestion time, enzyme concentration, and seeding density is therefore essential to preserve quality 31 .
Hybrid protocols, combining short enzymatic digestion with explant culture, aim to balance yield and purity while minimizing enzymatic stress. Although promising, their applicability to GMP-grade production remains under investigation 32 . Standardizing isolation protocols will be critical to ensure that passage-dependent findings can be consistently reproduced across laboratories and translated into GMP-compliant therapies.
Immunophenotypic characterization of UC-MSCs
The International Society for Cell and Gene Therapy (ISCT) set forth minimal criteria in 2006 to define MSCs, which include (a) adherence to the plastic surface under culture conditions; (b) absence of hematopoietic and vascular epithelial cell surface markers (CD14, CD19, CD34, CD45, and HLA-DR) and positive expression of mesenchymal markers (CD73, CD90, CD105); (c) capacity for multilineage differentiation in vitro; (d) self-renewal ability and minimal immunogenicity 10 . While these criteria provide a consensus on the general MSC definition, these criteria were initially established for BM-MSCs, a relatively uniform source primarily composed of MSCs and hematopoietic stem cells 33 . MSCs from different tissue sources carry dissimilar immunophenotypes that are excluded from current guidelines 34 . Although ISCT had presented amendments and suggestions on immunosuppressive properties, tissue origin and nomenclature 13 , a tissue-specific guideline should be proposed to fully capture cellular heterogeneity, functional diversity, and safety requirements, including additional markers and functional assays to better define clinically relevant MSCs populations 35 .
In MSC therapy, UC-MSCs have emerged as the second most frequently utilized MSCs in clinical trials 36 . In line with this effort, the ISCT has recently published ISO standardization documents focused on the biobanking of MSCs from Wharton’s Jelly and bone marrow, underscoring the importance of tissue-specific guidance in ensuring quality, consistency, and clinical translatability 37 . Compared to bone marrow, UC tissue offers a more diverse cellular environment which presents additional challenges for isolation and characterization 38 . Leukocytes, endothelial cells, and epithelial markers such as cytokeratin and cell-cell junctions have been detected in UC-MSC isolates, indicating cellular heterogeneity during isolation27,39. Enhanced identity profiling and negative selection of non-MSC markers should be implemented to minimize non-MSC contamination from the cord lining.
Therefore, a specific marker panel is proposed based on the latest consensus MSC definition and guidelines for MSC therapy, allowing for a comprehensive and detailed identification of UC-MSCs (Table 1). Among the markers listed, CD90, CD105, CD73 and CD45 are the canonical markers for MSC identification from any source 40 . Aligned with the encouragement of tissue-specific immunophenotyping, endothelial and epithelial markers as well as pericyte markers are included in this proposed panel to allow accurate UC-MSCs identification as a perinatal MSC source with minimum biological contamination39,41,42.
Canonical and proposed cell surface markers for UC-MSCs characterization.
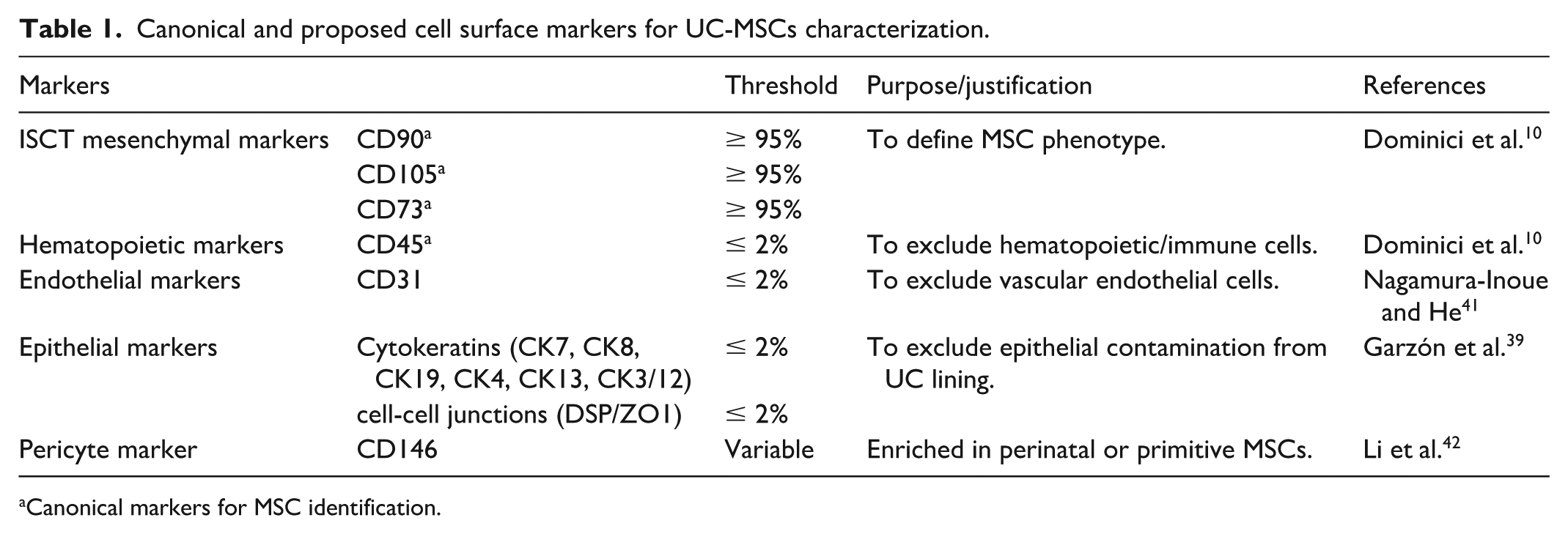
Canonical markers for MSC identification.
Biological advantages of UC-MSCs
A key advantage of UC-MSCs lies in their youthful origin. As postnatal cells are minimally exposed to environmental stress, UC-MSCs circumvent donor age-related decline seen in autologous bone marrow-derived (BM-MSCs) and adipose-derived MSCs (AD-MSCs), in which cellular senescence, reduced growth kinetics, and altered secretome profiles compromise therapeutic efficacy43–45. Studies consistently report that UC-MSCs exhibit a shorter population doubling time and higher expansion potential than adult MSC sources 46 . UC-MSCs also demonstrate potent immunomodulatory activity, mediated through direct cell-cell interactions and secretion of anti-inflammatory cytokines, making them attractive for the treatment of immune-mediated disorders 47 . Compared with BM-MSCs, UC-MSCs display enhanced ability to suppress T-cell proliferation, modulate macrophage polarization, and reduce pro-inflammatory cytokine release 48 .
The richness of the secretome released by UC-MSCs further enhances the regenerative capacity. These secretomes of UC-MSCs is rich in hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), fibroblast growth factor-2 (FGF2), brain-derived neurotrophic factor (BDNF), and tissue inhibitor of metalloproteinase (TIMP), among others, compared with adult MSCs46,49. These factors regulate the immune system, promote tissue regeneration, angiogenesis, and anti-inflammatory effects, underscoring their broad therapeutic potential. However, studies have indicated that the composition and potency of secretome may decline with extensive passaging, as reflected in cellular senescence and metabolic changes8,50,51. Therefore, maintaining low passage of UC-MSCs or optimizing culture conditions is essential to preserve the paracrine activity. Taken together, the proliferative resilience, immunomodulatory strength, and robust paracrine signaling of UC-MSCs make them a particularly attractive cell source for regenerative medicine, including applications in neurodegenerative, autoimmune, cardiovascular, and musculoskeletal diseases5,52. Maintaining these properties during large-scale expansion is critical for clinical translation. Thus, the influence of passage number on these advantageous characteristics warrants further examination.
Passage number: Definition and its biological effects
Subculturing, or passaging, refers to the transfer of cells into new culture vessels to sustain proliferation and prevent nutrient depletion and contact inhibition 53 (Fig. 2). Regular passaging is essential in large-scale MSC manufacturing, ensuring sufficient yields for clinical dosing, maintaining culture purity, and preserving functional potency 54 . Without timely passaging, cultures undergo growth arrest and senescence, compromising therapeutic applicability 55 .

Schematic overview of the cell culture expansion workflow illustrating cell growth and propagation across multiple culture plates. This illustration was created by the authors using Canva Pro.
For clinical translation, enzymatic detachment methods must balance efficiency with cell integrity. Recombinant enzymes such as TrypLE are widely adopted due to their xeno-free formulation, low cytotoxicity, and ease of inactivation25,56. Accutase, although capable of gentle detachment, has been linked to reduced viability and higher apoptosis rates in UC-MSCs57,58. Optimization of enzyme concentration, incubation time, and cell density is critical, as excessive enzymatic exposure alters surface marker expression, impairs adhesion, and reduces immunomodulatory function59,60. Thus, both the timing of passage and the parameters of enzymatic digestion directly affect the viability and functionality of UC-MSCs.
Passage phases are generally classified into three ranges: early passage (P1–P3), representing primary expansion and initial characterization; middle passage (P4–P6), typically considered optimal for clinical and preclinical use; and late passage (≥P7), often studied for safety and senescence61–64. Most GMP-compliant protocols restrict expansion to P6, as this range achieves clinically relevant yields while minimizing functional decline.
Evidence from BM-MSCs shows that functional alterations accumulate with prolonged culture. For example, extracellular vesicles derived from mid-passage BM-MSCs (P5) demonstrated superior tendon-healing effects compared to late-passage cells (P12) 65 . Similarly, immunomodulatory profiles vary with passage; Connard et al. 66 reported comparable T-cell suppression across passages, though cytokine secretion shifted at P6 and P9. These findings highlight that both therapeutic potency and secretome composition are passage-dependent. Importantly, donor age appears to affect MSC tolerance to passaging. Autologous BM-MSCs from elderly donors often display senescence as early as P367,68, limiting their scalability. In contrast, UC-MSCs are more resilient, with stable morphology, proliferation, and immunophenotype reported until ≥P10 69 . This resilience underscores their potential as a scalable allogeneic product, provided that passage-dependent changes in purity, proliferation, senescence, genomic stability, differentiation capacity, and immunomodulation are carefully characterized.
Impact of increasing passage number
Understanding how these characteristics evolve across early, middle, and late passages is essential to determine the optimal passage range for clinical applications. The following subsections provide current evidence on how passaging shapes each of these properties and highlight the implications for manufacturing and therapeutic use of UC-MSCs.
Effect of passage number on purity
The purity of MSC preparations is a critical determinant of safety and therapeutic efficacy, as contaminating cell types such as endothelial or epithelial cells may introduce immunogenicity or pro-inflammatory responses 10 . In UC cultures, early passages often contain heterogeneous populations. For instance, De Bruyn et al. 70 reported that up to 31% of UC-MSC primary isolates (P1) were contaminated by endothelial cells, but these contaminants declined substantially after subsequent passages. Similarly, supplementation with defined culture media and repeated passaging selectively expands MSCs, reducing non-MSC populations71,72.
Phenotypically, UC-MSCs across passages consistently maintain the canonical ISCT marker profile (CD73, CD90, CD105 positive; CD34, CD45, HLA-DR negative), with no significant deviations reported even at higher passages63,73. However, studies suggest that middle passages (P3–P5) display enhanced purity, evidenced by increased proportion of cells expressing MSC-associated markers such as CD90 and CD105, coupled with a reduction in cells expressing the endothelial marker CD31 74 .
Collectively, available data indicate that passaging not only sustains but can improve UC-MSC purity by progressively eliminating non-MSC contaminants. Nonetheless, systematic cross-passage analyses of both MSC and non-MSC markers, for instance, endothelial, epithelial, and hematopoietic remain limited. Establishing standardized marker panels for purity assessment across passages will be essential to ensure consistency in clinical-grade UC-MSC preparations.
Proliferative capacity across passages
Proliferation refers to the capacity of UC-MSCs to divide and expand in culture, a property that is essential for producing the high cell numbers required for therapy. Clinically, systemic administration often demands doses of 105-107 MSCs/kg, which translates into billions of cells per patient 75 . Achieving these numbers requires extended in vitro expansion, making proliferative capacity a critical determinant of scalability and function. Proliferative capacity is typically measured by population doubling time (PDT), cumulative population doublings (CPD), and expression of stemness-associated genes such as SOX2, c-MYC, and NANOG76,77. Across studies, UC-MSCs consistently show strong proliferation from early through middle passages. The shortest PDT and greatest fold expansion occur at P4–P5, indicating peak growth efficiency at this stage73,78. This trend likely reflects cellular adaptation to in vitro conditions. Proliferative stability appears robust across UC compartments; Wharton’s Jelly, perivascular, and membrane-derived MSCs display comparable growth rates from P1 to P8 79 .
Long-term analyses confirm sustained expansion, with UC-MSCs continuing to accumulate CPDs up to P20, though functional monitoring is required to detect the onset of senescence 80 . Importantly, middle-passage cells are not only faster growing but also exhibit elevated stemness marker expression, reinforcing their proliferative advantage 79 . Clinically, UC-MSCs expanded to P5 under human platelet lysate supplementation can yield 1011-10 12 cells sufficient for multiple therapeutic doses while preserving immunological properties81,82. Thus, proliferation is the foundation of UC-MSC clinical scalability. Evidence indicates that middle passages (P3–P5) strike the optimal balance, combining rapid expansion, high stemness, and stable phenotype, thereby enabling the generation of clinically relevant cell doses.
Onset of senescence with serial passaging
Despite their high proliferative capacity, UC-MSCs undergo replicative senescence after a finite number of divisions. Senescence negatively impacts stemness, proliferation, differentiation, migration, and cytokine secretion, thereby limiting therapeutic potency8,83. Senescence is regulated mainly through the p53/p21 and p16/Rb signaling pathways, and can be detected by morphological changes, molecular markers, and functional decline 84 .
Morphologically, UC-MSCs maintain spindle-shaped fibroblast-like features through early (P3) and middle passages (P5), but progressively adopt an enlarged, flattened phenotype in late passages (≥P7)73,85. Some studies, however, report delayed senescence, with typical morphology retained until ≥P18, reflecting donor and culture-dependent variability79,86.
Molecular signatures confirm these trends, where late-passage UC-MSCs exhibit reduced expression of OCT4/ POU5F1, NANOG, SOX2, and telomerase activity, alongside increased levels of p16, p21, and p5387,88. Functional assays reveal declining proliferation and cumulative cell yield beyond P10–P12, consistent with activation of senescence pathways. Senescence-associated β-galactosidase (SA-β-Gal) activity also rises significantly in passages ≥P14, correlating with p21 upregulation62,86,89,90. Epigenetic modulation has shown promise in delaying senescence, with low-dose histone deacetylase inhibitors (e.g., trichostatin A, largazole) maintaining self-renewal and proliferative activity89,91. Taken together, UC-MSCs retain their proliferative and functional phenotype during early and middle passages but undergo progressive senescence in late passages, manifested by morphological changes, stemness suppression, and senescence marker activation. Their relative resilience compared to BM-MSCs where senescence may appear as early as P3 in aged donors 67 , supports their feasibility for scaled allogeneic use but underscores the need for strict passage monitoring to ensure clinical viability.
Genomic stability across passages
Genomic stability refers to the preservation of chromosomal structure and integrity during in vitro culture, a prerequisite for ensuring the safety of UC-MSCs in clinical applications. Prolonged ex vivo expansion increases the risk of DNA replication errors, chromosomal abnormalities, and mutational events, raising concerns of tumorigenic transformation92,93.
Evidence suggests that UC-MSCs display strong resilience to genomic instability across multiple passages. Blázquez-Prunera et al. 94 , demonstrated that UC-MSCs retained a normal diploid karyotype up to P8 under xeno-free conditions. Similarly, other studies reported normal chromosomal numbers and absence of malignant transformation even at very late passages (≥P15)62,95. Comparative analyses also indicate that UC-MSCs are more genomically stable than BM-MSCs, in which donor age and extended culture more readily induce aneuploidy 96 . Collectively, these findings highlight that UC-MSCs maintain genomic integrity across early and middle passages, and in many cases beyond, reinforcing their suitability for clinical expansion. Nonetheless, systematic monitoring of karyotype and genome-wide alterations remains essential, particularly when scaling production for therapeutic use.
Passage-dependent differentiation capacity
The differentiation capacity of UC-MSCs underpins their therapeutic efficacy, as this capacity represents the MSCs stemness that correlates to the paracrine signaling mechanisms in tissue regeneration 97 . Passage number directly influences this capacity by modulating stemness, mitochondrial function, and membrane properties.
In general, early to middle passages preserve robust trilineage potential, whereas late passages exhibit functional decline. For osteogenesis, middle-passage UC-MSCs (P3–P8) show increased expression of RUNX2 and BGLAP, enhancing bone-forming ability 79 , while osteogenic differentiation decreases sharply at P15, likely due to senescence-associated mitochondrial dysfunction98–100. Interestingly, increased membrane capacitance in mid-passage UC-MSCs correlated with upregulation of COL1A1, further promoting osteogenesis 101 . Adipogenic and chondrogenic differentiation generally follow similar trends, declining in late passages, although occasional studies report increased adipogenesis at P15 98 . Beyond trilineage capacity, disease-relevant differentiation has also been explored. Cardiogenic markers such as GATA4 peak at P7, while NKX2-5 expression rises at P14, indicating passage-dependent variability in cardiac differentiation 102 . For neural induction, UC-MSCs are commonly used at P3–P5, though enhanced neurogenic differentiation has been reported at P10–P11 63 .
Paracrine signaling is also passage-dependent. VEGF, FGF, KGF, and HGF secretion increases from P2 to P6, supporting angiogenesis and tissue repair 103 . Moreover, IL-17 expression peaks at P4–P5, potentially enhancing homing and migration to sites of injury104,105. Together, these findings indicate that middle passages (P3–P6) are optimal for preserving trilineage and paracrine function. Late passages may retain certain specialized differentiation capacities, for instance, cardiogenic and neurogenic, but supporting evidence remains limited and requires further study.
Changes in immunomodulatory function with passaging
The immunomodulatory function of UC-MSCs is a cornerstone of their therapeutic utility, enabling them to suppress inflammatory responses and promote immune tolerance. Mechanisms include inhibition of T-cell proliferation, modulation of Th1/Th17 activity, and enhancement of Treg populations, primarily through cytokine secretion106,107. Passage number significantly influences these effects. Early (P2) and middle-passage (P5) UC-MSCs show comparable immunosuppressive activity, effectively inhibiting Th1/Th17 cells and enhancing Tregs. However, these functions decline in late passages (≥P8), with diminished T-cell suppression and reduced Treg induction88,108.
Cytokine profiling provides mechanistic insight. Early to middle passages sustain high levels of TGF-β and IL-10, key mediators of immunosuppression109,110. IFN-γ expression peaks around P4, coinciding with optimal immunomodulatory activity, before declining at later passages105,111. IFN-γ also regulates lipid mediators such as ceramide-1-phosphate and phosphatidylcholine, which support immune cell modulation 112 . These strongly highlight that UC-MSCs have the strongest immunomodulatory capacity during early and middle passages, with progressive decline at later stages. This pattern mirrors clinical observations of reduced efficacy in prolonged culture, underscoring the importance of passage monitoring when manufacturing UC-MSC therapies.
In summary, cumulative evidence indicates that serial passaging exerts measurable and passage-dependent effects on the biological attributes of UC-MSCs, including culture purity, proliferative kinetics, senescence onset, genomic integrity, differentiation potential, and immunomodulatory properties. These characteristics collectively determine the safety, scalability, and therapeutic potency of UC-MSCs, underscoring the importance of identifying an optimal passage range for clinical translation. However, the translational significance of these in vitro observations can only be fully appreciated when considered in the context of clinical studies. Passage number is a critical variable in manufacturing workflows, with direct implications for regulatory compliance, therapeutic efficacy, and dosing feasibility in systemic applications. To bridge experimental insights with clinical practice, the subsequent table summarizes evidence from clinical trials that have employed UC-MSCs at defined passage stages across diverse organ systems and disease indications, thereby contextualizing laboratory findings within real-world therapeutic outcomes (Table 2).
Summary of clinical trials utilizing UC-MSCs across different passage numbers and their therapeutic indications across organs and systems.

ADL: Activity of Daily Living, ALP: Alkaline Phosphatase, ANA: Antinuclear Antibody, ASIA: American Spinal Injury Association, BILAG: British Isles Lupus Assessment Group, EDSS: Expanded Disability Status Scale, EQ: EuroQol, GGT: Gamma-glutamyltransferase, HbA1c: Hemoglobin A1c, IBDQ: Inflammatory Bowel Disease Questionnaire, LVEDV: Left Ventricular End-diastolic Volume, LVEF: Left Ventricular Ejection Fraction, LVESV: Left Ventricular End-systolic Volume, MELD: Model for End-Stage Liver Disease, PASI: Psoriasis Area & Severity Index, PCS: Physical Component Summary, PET: Positron Emission Tomography, PPG: Postprandial Plasma Glucose, SF-36: 36-Item Short Form Survey, SLEDAI: Systemic Lupus Erythematosus Disease Activity Index, SPET: Single-photon Emission Computed Tomography, SSDAI: Sjögren’s Syndrome Disease Activity Index, TEWL: Transepidermal Water Loss, VAS: Visual Analogue Scale, WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
Prospects and perspectives
UC-MSCs represent a promising cell source in regenerative medicine, owing to their high proliferative capacity, potent immunomodulatory effects, and encouraging outcomes across diverse clinical indications. Evidence consistently indicates that passage number critically impacts their biological attributes, including proliferation, senescence onset, genomic stability, differentiation potential, and immunomodulatory activity. Early to middle passages (P3–P5) generally provide the optimal balance between safety, potency, and scalability. However, the primitive state of UC-MSCs suggests that slightly later passages may also be feasible under tightly controlled conditions.
Despite this potential, several limitations remain. Available data in the literature are confounded by heterogeneity in donor characteristics, isolation protocols, and culture conditions, while many preclinical and clinical studies report outcomes at single passage points rather than across sequential passages. Moreover, incomplete reporting of passage numbers in clinical trials, together with the absence of globally accepted guidelines, restricts the ability to define universal benchmarks for UC-MSC manufacturing and application.
Future research should prioritize systematic evaluation of UC-MSCs across successive passages using validated potency assays such as flow cytometry-based immunophenotyping, cytokine profiling, or functional immunosuppression assays to establish quantitative correlations between in vitro characteristics with therapeutic efficacy. In addition, integrating advanced multi-omics platforms could provide a deeper insight into passage-dependent molecular shifts, enabling the identification of predictive potency markers relevant to clinical outcomes. In parallel, harmonized international guidelines for UC-MSC culture, passage selection, and reporting are urgently needed to ensure reproducibility, regulatory compliance, and patient safety. Eventually, bridging regulatory frameworks with real-world clinical observations will ultimately refine the balance between cell yield and functional quality, advancing UC-MSC-based regenerative therapies from laboratory optimization to reliable bedside implementation. As clinical translation advances, it becomes evident that both therapeutic success remains the ultimate benchmark, highlighting the need to define efficacy through validated functional outcomes instead of passage count alone. By addressing these challenges, the field can move toward standardized, evidence-based practices that unlock the full therapeutic potential of UC-MSCs in regenerative medicine.
Conclusion
Among various types of MSCs, UC-MSCs possess several unique properties that render them favorable for regenerative therapy. UC-MSCs have high proliferation capacity, trilineage differentiation capacity and immunomodulatory activity. Middle passages generally provide an optimal balance between potency, stemness and safety, supporting more reliable therapeutic outcomes in various clinical indications. However, several pre-clinical studies reported that UC-MSCs can maintain these properties and support specialized differentiation, which requires a standardized validation to determine the clinically acceptable expansion limits. Establishing systematic and evidence-based guidance on passage number will be important for regulatory decision-making and for ensuring consistent safety and efficacy in future UC-MSCs therapies.
Footnotes
Acknowledgements
The authors sincerely thank their respective institutions for providing academic support and access to scholarly resources throughout the preparation of this review. We gratefully acknowledge Yakin Splendor Global Holding Berhad for its support and encouragement, which facilitated the collaborative efforts among authors working across different entities. We also extend our appreciation to colleagues and experts who contributed valuable insights and constructive feedback that helped improve the quality of this manuscript. The author(s) acknowledge the use of Grammarly (Grammarly Inc.), an AI-assisted editing tool for improving spelling, grammar, clarity and readability, during the preparation of the manuscript. After the use of the tool, author(s) have reviewed and edited the text and takes full responsibility for the final content. No scientific content, data, analyses, or interpretations were generated, modified, or influenced using artificial intelligence. The authors take full responsibility for the accuracy, originality, and integrity of the work.
Ethical considerations
Not applicable
Author contributions
L.F and G.R contributed to the original draft preparation, writing, review, and editing of the manuscript. M.E.A.M., N.M.F., M.S.S, and A.S reviewed and edited the manuscript. P.H. conceptualized and supervised the study, provided critical revisions and editions, and approved the final version of the manuscript for submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Yakin Splendour Sdn Bhd.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of Yakin Splendour Sdn Bhd., Yakin Health Management Sdn Bhd., and Yakin Medic Sdn Bhd., which operate in the field of stem cell technologies. This study was funded by Yakin Splendour Sdn Bhd. The funder had no role in the conceptualization, design, data interpretation, writing, or conclusions of this review. The authors declare no additional financial or non-financial competing interests related to this work, which is intended solely for academic knowledge dissemination.
Data availability statement
The original contributions presented in the study are included in the article; further inquiries can be directed to the corresponding author.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
