Abstract
Cell escape occurs after intramyocardial injection for treatment of myocardial infarction (MI) and then the migrated cells might be entrapped by extracardiac organs. We investigated the fate of migrated bone marrow-derived mesenchymal stromal cells (MSCs) and their impact on lung, liver, and spleen. MI model was created by coronary artery ligation in female Lewis rats. Three weeks after the ligation, bromodeoxyuridine (BrdU)-labeled male MSCs were directly injected into the infarcted area in the cell transplantation group (n = 22). The same volume of phosphate-buffered solution (PBS) was injected in the control group (n = 21). In the sham group (n = 10) intramyocardial injection of the same volume of PBS was performed in healthy rats. Four weeks later, echocardiography was performed and the cell retention was evaluated by quantitative real-time polymerase chain reaction (qRT-PCR). Immunohistochemistry study was performed to identify the migrated cells. Heart function was improved after the cell injection. qRT-PCR results showed the percentage of retained cells in heart, spleen, liver, and lung ranked 3.63 ± 0.48%, 0.77 ± 0.13%, 0.68 ± 0.10%, 0.62 ± 0.11%, respectively, after cell transplantation. The implanted MSCs that escaped to liver, spleen, and lung did not differentiate into fibroblast, myofibroblast, or alveolar epithelial cells. However, the migrated MSCs in liver expressed functional hepatocyte marker. In conclusion, cell migration after intramyocardial injection did not result in deterioration of lung, liver, and spleen function. Our study might pave the way for new safety investigation of emerging cell resources and their impact on target and untargeted organs.
Introduction
Bone marrow-derived mesenchymal stromal cells (MSCs), which contain multipotent mesenchymal stem cells, have been a popular cell resource for cardiac regeneration in recent years (18). The therapeutic potency of MSC transplantation for the treatment of myocardial infarction (MI) has been well demonstrated in experimental and clinical studies. Intramyocardial injection is considered as the most effective route for the precise cell delivery to the target areas. However, many cells still migrated from the injection sites to the extracardiac organs, and these cells tended to be entrapped by the filter organs in pulmonary or systemic circulation (7,9,27). Until now, little is known about the impact of escaped progenitor cells on extracardiac organs. Therefore, in the present study we address 1) the cell retention after intramyocardial injection in a chronic MI model and 2) the fate and impact of escaped MSCs on liver, spleen, and lung.
Materials and Methods
Animals
Seventy syngenic Lewis rats (Vital River Laboratory Animal, Inc., Beijing, China) weighing 200–250 g were enrolled in this study. All animals received humane care and the animal protocols complied with the institution's guidelines. All animal experimental procedures were approved by the Ethics Committee for Animal Study in Fu Wai Hospital.
Cell Culture
MSCs were isolated from male Lewis rats (60 g body weight) and cultured as previously reported (12). In brief, bone marrow cells were obtained by flushing the bone marrow cavity of the femora and tibiae and then cultured with Dulbecco's modified Eagle's medium (DMEM, Gibco, Grand Island, NY) supplemented with 10% fetal bovine serum. To track the transplanted cells, cultured expanded MSCs were labeled with bromodeoxyuridine (BrdU, Sigma, St. Louis, MO) before transplantation (26). In brief, 50 μl of 0.4% BrdU was added to each culture dish and incubated with the cells for 48 h.
Myocardial Infarction and Cell Transplantation
MI was induced in 60 female rats by left anterior descending artery (LAD) ligation as previously described (11). Three weeks later, the qualified rats, with left ventricular ejection fraction (LVEF) lower than 60% and left ventricular fractional shortening (LVFS) lower than 30%, were randomly divided into two groups. MSCs (3 × 106) were intramyocardially injected into the border zone of the infarcted area in the cell transplantation group (n = 22). The same volume of phosphate-buffered solution (PBS) was injected in the control group (n = 21). Four weeks postinjection, there were 20 and 15 rats left in the cell transplantation and control groups, respectively. All 10 healthy rats in the sham group survived the open-chest operation.
Echocardiography Examination
Two-dimensional echocardiography was performed at 3 weeks after MI and 4 weeks after cell injection (28). Left ventricular end-diastolic diameters (LVEDd) and left ventricular end-systolic diameters (LVESd) were measured at the middle papillary muscle level and LVFS and LVEF were calculated as follows: LVFS(%) = (LVEDd - LVESd)/LVEDd x 100 and LVEF(%) = [(LVEDd3 - LV ESd3)/LVEDd3] x 100. All measurements were averaged on three consecutive cardiac cycles and were analyzed by two independent observers.
Measurement of Lung Function and Inflammatory Factors in Lung Tissue
Four weeks after cell injection, the rats (n = 15 in the cell transplantation group, n = 15 in the control group, n = 10 in the sham group) were placed in a sealed whole body plethysmograph and mechanically ventilated with AniRes2003 lung function system (AniRes2003, Beijing SYNOL High-Tech Co. Ltd, Beijing, China). Lung function was measured as forced vital capacity (FVC), forced expiratory volume in 0.2 s at the beginning expiration of total volume (Fev0.2), and Fev 0.2/FVC%.
After completing the lung function examination, the rats were sacrificed and the lung tissue homogenized. Tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) levels were measured with antibody enzyme-linked immunosorbent assays (ELISA) with commercial ELISA kits (Rapid Bio Lab, Calabasas, CA). All reagents, samples, and working standards were prepared according to the manufacturer's instruction. The optical density was measured at 450 nm wavelengths by a Model 680 Microplate Reader (Bio-Rad Laboratories, Hercules, CA, USA).
The myeloperoxidase (MPO) activity (an indicator of neutrophil infiltration) was measured according to the manufacturer's instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Blood and Tissues Harvest
After completing the heart and lung function examination, the rats were weighted and the blood samples was collected from abdominal aorta through an abdomen opening under anesthesia, and then the rats were sacrificed under deep anesthesia. The heart, liver, spleen, lung, and kidney were harvested, weighed, and fixed in 10% formaldehyde neutral buffer solution.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
Four weeks after cell injection, the numbers of engrafted male cells in the myocardium, liver, spleen, lung, and kidney of 5 rats were evaluated by RT-PCR amplification of the male cell-specific sex-determining region on Y-chromosome (SRY) gene. Quantitative real-time PCR with the ABI Prism 7300 Sequence Detection System (Applied Biosystem, Foster City, CA) using the Master Mix SYBR Green Kit (Applied Biosystems) was used to quantify donor male rat cells. The genomic DNA taken from implanted donor male MSCs was used to obtain a standard curve. Genomic DNA was extracted by using a Wizard® genomic DNA purification kit (Pro-mega, Madison, WI). Primers used for detection of rat male-specific sry gene were as follows: (sense 5-CATC GAAGGGTTAAAGTGCCA-3; antisense 5-ATAGTG TGTAGGTTGTTGTCC-3). The qRT-PCR protocol consisted of an initial denaturation step at 95°C for 10 min, followed by 40 cycles of 95°C for 15 s, and 60°C for 1 min (21).
To obtain a standard curve, genomic DNA of 3 × 106 MSCs was extracted, and DNA was dissolved in 90 μl of water. A 3-μl sample corresponding to 105 copies of Y-chromosomal DNA was diluted to produce samples, which had a range from 101 to 105 copies. A serial 10-fold dilution of the DNA from male rat MSCs was tested five times in separate experiments.
Assessment of Liver and Spleen Function
Liver and spleen weight/body weight ratios were evaluated. Concentration of liver injury markers in blood, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin (TB), direct bilirubin (DBIL), and albumin (ALB), was analyzed using UniCel® DxC 800, a Synchron® Clinical System (Beckman Coulter, Los Angeles, CA). Routine blood tests, including red blood cell (RBC), white blood cell (WBC), platelet (PLT), and hemoglobin (Hgb), were examined using Sysmex XT-1800i (Sysmex, Kobe, Japan). The level of immunoglobulin M (IgM) in serum was analyzed using IMMAGE® Immunochemistry Systems (Beckman Coulter).
Tissue Immunohistochemistry Examination
Paraffin sections were immunostained for ALB and α-fetoprotein (AFP) (Zymed, San Francisco, CA) to identify functional hepatocytes. The sections were also immunostained for α-smooth muscle actin (α-SMA) and vimentin (Zymed) to identify myofibroblasts and fibroblasts. To identify alveolar epithelial cells, sections were immunostained with pan-cytokeratin (Santa Cruz Biotechnology, Santa Cruz, CA). A double staining kit (Zymed) was used to distinguish simultaneous expression of BrdU and the targeted proteins listed above. The paraffin sections were dehydrated in a graded ethanol series after deparaffinization with xylene. Antigen retrieval was performed by placing the slides in a boiling sodium citrate buffer (10 mM sodium citrate, pH 6) for 20 min and allowing the slides to cool for another 20 min in the same buffer. Endogenous peroxidase was then quenched with 3% hydrogen peroxide for 10 min. After rinsing with PBS, sections were incubated with the primary mouse monoclonal antibodies against BrdU at a dilution of 1:200 (Zymed) overnight at 4°C. The slides were then gently washed in PBS, and incubated with peroxidase-conjugated secondary antibody used diaminobenzidine (DAB) for color development. Subsequently sections were placed in boiling sodium citrate buffer again for antigen retrieval, then incubated overnight at 4°C with the antibodies directed against ALB (1:300), AFP (1:500), α-SMA (1:200), vimentin (1:500), or pan-cytokeratin (1:500), respectively, and then rinsed three times with PBS. For the negative control, PBS was substituted for primary antibody. The sections were then incubated with peroxidase-conjugated secondary antibody used the AEC (3-amino-9-ethylcarbazole) staining solution for color development and applied cover slip with glycerol gelatin. The immunostaining were assessed under a microscope (Olympus BX61, Tokyo, Japan).
Statistical Analysis
Numerical values are expressed as mean ± SD. Multiple comparisons between three groups was performed using a one-way analysis of variance (ANOVA) followed by Bonferroni test. Comparisons of parameters between two groups were made by Student's t-test. Values were analyzed using the statistical package SPSS 13.0 (SPSS, Inc., Chicago, IL). A value of p < 0.05 was considered to be statistically significant.
Results
Surgical Mortality
Twelve rats died within the first 24 h after MI surgery. Five rats died either post-cell (n = 2) or PBS (n = 3) injection in the MI rats. There was no death in the sham group. In addition, 5 rats were excluded owing to the unqualified ejection fraction after echocardiography exam, which was performed 3 weeks post-MI surgery. Therefore, there were 43 qualified rats subjected to cell transplantation among which 5 died during the cell or PBS injection.
Distribution of Implanted Cells
A standard curve was generated by serially diluting rat male genomic DNA prepared from male MSCs and quantified SRY gene by using real-time PCR. Threshold cycle of SRY gene in male MSCs has a good relation with gradually diluted cells (R2 = 0.9978) (Fig. 1). Four weeks after MSCs transplantation, the percentage relative to the initial number of injected cells in heart, liver, spleen, and lung was 3.63 ± 0.48%, 0.68 ± 0.10%, 0.77 ± 0.13%, and 0.62 ± 0.11%, respectively. We did not detect any implanted male cells in the kidney.

Quantification of transplanted male MSCs. (a) Real-time amplification plot showing change in normalized reporter dye fluorescence (Rn) versus number of amplification cycles in sample containing serially diluted male MSCs genomic DNA. (b) Standard curve generated from data in (a) and showing relationship between threshold cycle (Ct) and number of male MSCs.
Evaluation of Heart Function
As shown in Table 1, LVEF and LVFS in the cell transplantation group were higher than that in the control group (p < 0.01), indicating a significantly improved heart function 4 weeks following MSCs injection.
Left Ventricular Function at Baseline and 4 Weeks After Cell Transplantation

LVEDd: left ventricular end-diastolic diameter; LVESd: left ventricular end-systolic diameter; LVEF: left ventricular ejection fraction; LVFS: left ventricular fractional shortening.
p < 0.01 versus control group at 4 weeks after injection.
Escaped Cells in Lung and the Impact on Lung Function
Vimetin and α-SMA antibody were applied to identify fibroblast and myofibroblast, respectively. Pancytokeratin was applied to identify alveolar epithelial cells. There was no colocalization of cells with brown nucleus (indicating BrdU-positive donor cells) and red cytoplasmic coloration (indicating vimentin-, α-SMA-, or pan-cytokeratin-positive expression) (Fig. 2a–c).
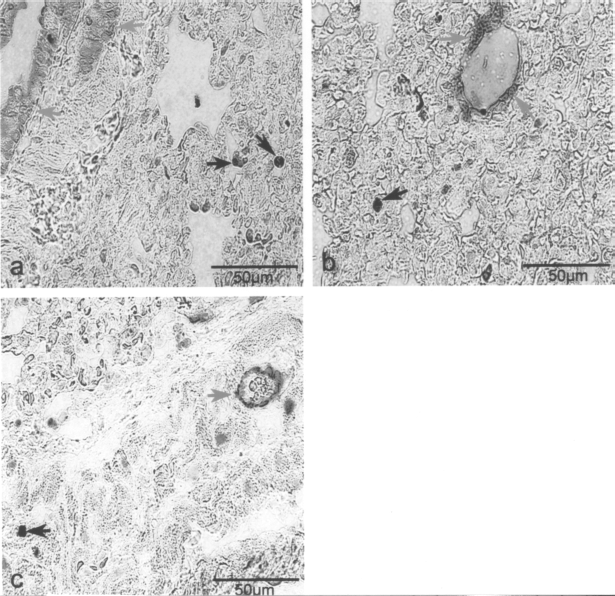
MSCs in lung at 4 weeks after cell transplantation. (a) Double staining with bromodeoxyuridine (BrdU) (indicated by black arrow) and pan-cytokeratin (indicated by red arrow). (b) Double staining with both BrdU (indicated by black arrow) and vimentin (indicated by red arrow). (c) Double staining with BrdU (indicated by black arrow) and α-smooth muscle actin (α-SMA) (indicated by red arrow). Scale bars: 50 μm.
There was no significant difference, in terms of lung weight/body weight ratio (p > 0.05), among three groups. There was no statistical difference with FVC, Fev0.2, and Fev 0.2/FVC% (p > 0.05), level of TNF-α and IL-1β, and MPO activity in lung tissue between the cell transplantation and control groups (p > 0.05) (Table 2).
Lung Function Assessment of the Different Groups 4 Weeks After MSCs Transplantation

TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; MDA, malondialdehyde; MPO, myeloperoxidase; FVC, forced vital capacity; Fev0.2, forced expiratory volume in 0.2 s at the beginning expiration of total volume. There were no differences between various groups.
Escaped Cells in Liver and the Impact on Liver Function
The escaped cells were disorderly distributed throughout the liver tissue. As shown in Figure 3a and b, labeled MSC expressed ALB and AFP; furthermore, the BrdU-positive cells did not coexpress the fibroblast or myofibroblast markers (vimentin and α-SMA) (Fig. 3c, d).

MSCs in liver at 4 weeks after cell transplantation. (a) Double staining with bromodeoxyuridine (BrdU) (indicated by black arrow) and albumin (ALB) (indicated by red arrow). (b) Double staining with both BrdU (indicated by black arrow) and α-fetoprotein (AFP) (indicated by red arrow). Some BrdU-positive cells were also stained positively for ALB or AFP (indicated by blue arrow). (c) Double staining with BrdU (indicated by black arrow) and vimentin (indicated by red arrow). (d) Double staining with BrdU (indicated by black arrow) and α-smooth muscle actin (α-SMA) (negative expression). The labeled escaped MSCs did not express vimentin or α-SMA. Scale bars: 50 μm.
Serum ALB level was significantly increased in the cell transplantation group compared to that in the controls (p = 0.04). In contrast, ALT level was significantly decreased in the cell transplantation group compared to the control group (p = 0.04). There was no significant difference among the three groups, in terms of the liver weight/body weight ratio, and the level of AST, ALP, TBIL, and DBIL (Table 3).
Liver Function Assessment of the Different Groups 4 Weeks After MSCs Transplantation
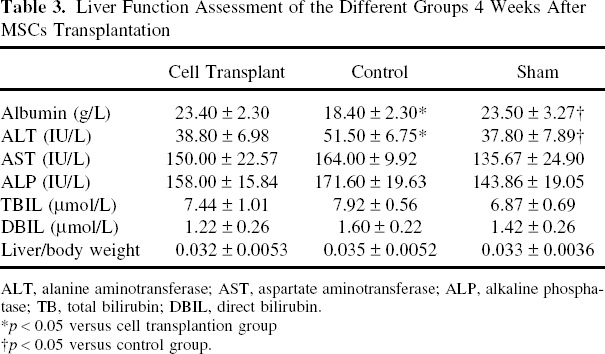
ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; TB, total bilirubin; DBIL, direct bilirubin.
p < 0.05 versus cell transplantion group
p < 0.05 versus control group.
Escaped Cells in Spleen and the Impact on Spleen Function
There was no colocalization of cells with brown nucleus (indicating BrdU-positive donor cells) and myofibroblast or fibroblast-specific markers of α-SMA (Fig. 4A) and vimetin (Fig. 4b).

MSCs in spleen at 4 weeks after cell transplantation. (a) Double staining with bromodeoxyuridine (BrdU) (indicated by black arrow) and α-smooth muscle actin (α-SMA) (indicated by red arrow). (b) Double staining with BrdU (indicated by black arrow) and vimentin (indicated by red arrow). Scale bars: 50 μm.
The spleen weight/body weight ratio was significantly decreased in the cell transplantation group compared to the control group (p = 0.04). The IgM level in serum was elevated in the control group compared with the sham group (p = 0.03) but not different with the cell transplantation group (p = 0.09). There were no marked differences, in terms of WBC, RBC, PLT, and Hgb, among these three groups (p > 0.05) (Table 4).
Spleen Function Assessment of the Different Groups 4 Weeks After MSCs Transplantation

RBC, red blood cell; WBC, white blood cell; PLT, platelet; Hgb, hemoglobin; IgM, immunoglobulin M.
p < 0.05 versus cell transplant group.
p < 0.05 versus control group.
Discussion
Cell transplantation has emerged as an attractive approach for cardiac regenerative therapy. The progenitor cells can be delivered into the injured myocardium by intracoronary infusion or intramyocardial injection. Although direct intramyocardial injection is more specific with less systemic redistribution, locally injected cells still migrate to the extracardiac organs, especially to the spleen, liver, and lung (8,10,27). Previous studies have shown that the transplanted bone marrow cells could be a two-edged sword with unexpected negative impact on the liver (5,20). Therefore, the biosafety of escaped progenitor cells should be considered after cell injection.
The major findings of present study were that 1) the majority of injected MSCs resided in the myocardium and ultimately improved compromised heart function, 2) injected MSCs migrated to the liver, expressed the markers of functional hepatocyte, and partly improved liver function, and 3) MSCs were also retained in the spleen and lung and did not deteriorate spleen and lung function.
We found that heart function was improved after MSCs transplantation, and our result is consistent with other studies (16,17). However, 4 weeks after cell transplantation, the total number of cell retention in liver, spleen, and lung was over half of the amount of that in the heart. Furthermore, no escaped cells were founded in the kidney. We have proved that the escaped cells in extracardiac organs were originally injected cells instead of migrated macrophages (21,27). After intramyocardial injection some implanted cells would be drained through collateral channels and ventricular lumen, pumped into systemic and pulmonary circulation, and eventually entrapped by the filter organs (8,10,27). Because the liver, spleen, and lung are the most important filter organs in systemic and pulmonary circulation, we focused our study on the impact of escaped cell on these three organs.
In previous air or fat embolism study, the lung weight/body weight ratio, inflammatory factors, and MPO activity would be dramatically increased (14,15). In a recent intravenous MSCs infusion study, the series histological examination showed that the infused cells could form emboli in lung and resulted in augmentation of many inflammatory transcripts (13). Although the entrapped cells were found in the lung, there was no evidence of embolism and augmented inflammatory reaction after cell intramyocardial injection. Furthermore, there was no evidence of ventilation dysfunction according to the lung function examination. In the bleomycininduced lung fibrosis model, the infused MSCs could differentiate into alveolar epithelial-like cells and repair lung fibrosis (12,19). However, in this study, the entrapped MSCs did not differentiate into any major cell types which composed lung parenchyma. We presumed that in our MI model, there was no organic injury to the lungs; therefore, there was no appropriate microenvironment for the activation of milieu-dependent differentiation of MSCs.
It has been reported that administration of MSCs could rescue liver injury in chronic models of fibrogenesis (7,9). However, Di Bonzo and associates found that injected MSCs resulted in relatively rare hepatocelluar differentiation and hold more pro-fibrogenic potential (5). Russo and his colleagues provided similar evidence that the predominant source of myofibroblasts in the chronic injured liver was differentiated from transplanted MSCs (20). MI-induced chronic heart failure is a systemic clinical syndrome with liver congestion and impaired liver function (1,3). We did not find any evidence of liver fibrogenesis, but expression of the markers of functional hepatocyte was observed in a few escaped MSCs. In addition, the improved heart function could also contribute to the subsequently enhanced liver function (increased ALB and decreased ALT level).
Cell fusion has been suggested as an explanation for stem cell plasticity. It has been reported that neo-hepatocytes, which derived from transplanted bone marrow cells, could arise from cell fusion instead of differentiation (2,23). One limitation of the present study was that we did not distinguish the cell differentiation and fusogenicity. However, cell fusion usually occurred under some special conditions such as injury (2) and the frequency of spontaneous fusion resulting in bone marrow-derived hepatocytes was as low as 10−4 to 10−6 (24). Recently, Colletti and coworkers reported that the change of phenotype of transplanted MSCs was mainly attributed to true differentiation rather than cell fusion or donor-to-recipient mitochondrial/membrane transfer (4). Therefore, the details of the mechanism by which bone marrow-derived hepatocytes are derived should be further investigated.
The spleen is the main blood filter organ in the systemic circulation and traps the majority of escaped cells. Because there were no specific markers of functional spleen cells, we applied vimentin and α-SMA to identify fibroblasts and myofibroblasts for the potential cell differentiation and did not find fibrosis differentiation of BrdU-positive cells. The spleen weight/body weight ratio may increase to a certain degree owing to the spleen congestion after MI. After cell transplantation, it was decreased along with the restored heart function. Meanwhile, patients with chronic heart failure were characterized by systemic inflammation (25). In the present study, we found the IgM level was enhanced owing to the MI-induced heart failure in the control group; however, there was no significant difference between the sham and cell transplantation groups at 4 weeks after cell injection. The CD20+ CD21+ B lymphocyte, which worked as an important producer of IgM, is mainly localized in the spleen (22). It has been reported that the level of IgM would be decreased in patients with splenectomy after trauma (6). Furthermore, there were no significant difference in WBC, RBC, PLT, and Hgb level among the three groups. Taken together, there was no evidence of hypo- or hypersplenism observed, although many MSCs were found reside in the spleen.
In present study, MSCs weere chosen as a cell resource for injection. We did not find escaped stromal cells' fibroblast differentiation and the evidence of deterioration of lung, liver, and spleen function. Clinical safety and efficacy studies with MSCs are already being done worldwide (17). These studies mostly focused on the improved heart function and major cardiac events. Although our short-term animal study confirmed the safety of escaped MSCs in various extracardiac organs, long-term observation of these escaped cells would still be warranted. Our study might pave the way for new safety investigation of the emerging cell resource and their impact on untargeted organs.
Footnotes
Acknowledgments
This study was supported by National 863 Project (2006AA02A106), the Fok Ying Tung Education Foundation (121043), and the Natural Science Foundation of China (81070099). The authors thank Dr. Zhi-qiang Jia for his excellent language editing.
