Abstract
Cell therapy is a proven and efficient method for treating multiple diseases. For both basic research and clinical practice, the development of noninvasive in vivo imaging methods is essential for monitoring the trafficking or homing of transplanted cells. One attractive approach for the effective imaging of transplanted cells is the efficient labeling of cells with a contrast agent. In this study, we developed a novel contrast agent of magnetic resonance imaging (MRI), TMADM-02. TMADM-02 was efficiently transduced into cells without toxicity. However, the aggregation of TMADM-02 was observed because of its low stability in culture medium. Therefore, TMADM-02 may have led to a false-positive test result. In future studies, we should verify not only the efficiency of labeling cells but also the stability of the contrast agent of MRI for clinical applications.
Introduction
Cell therapy is a proven and efficient method for treating multiple diseases including diabetes, hematological and central nervous system (CNS) disorders. Remarkable advances have been made in islet, bone marrow, and stem cell transplantation (15, 17, 19–22). However, their interrelationships and interactions in vivo remain mostly unknown. For both basic research and clinical practice, the development of noninvasive in vivo imaging methods is essential to monitor the trafficking or homing of transplanted cells. Currently, a range of technologies including positron emission tomography (PET) and fluorescence and bioluminescence optical imaging is used for the cellular and molecular imaging applications (1, 10, 23). Magnetic resonance imaging (MRI) has been used as a valuable noninvasive detection method in diagnostics and functional imaging. MRI of contrast agent-labeled cells has become a useful tool for studying cell trafficking in vivo (5–8, 16, 18, 25).
One of the key steps for the effective imaging of transplanted cells is the efficient labeling of cells with a contrast agent. Supermagnetic iron oxide nanoparticles (SPIO) were developed as detection markers in MRI and were used to track lymphocytes or to image liver metastases by showing the displacement of normal tissue phagocytes (9, 27). The most convenient method for cell labeling is a simple incubation of cells with the chosen contrast agent. This technique requires the addition of various transfection reagents, such as polycationic transfection agents, lipofectamine (4), poly-l-lysine (26), and protamine sulfate (3). All the reagents are positively charged, and coat the negatively charged MR contrast agents to create positively charged compounds. This facilitates the attraction and binding of positively charged contrast agents to the negative cell membranes. Although the mechanisms by which positive agents enter the cell have not been completely elucidated, they probably include a disruption, bending, and invagination of the cell wall. The increased localization of contrast agents to the cell membrane increases the encapsulation of the contrast agents into the cytoplasm (2). The primary advantage of the incubation labeling method is its simplicity. The primary disadvantage is the prolonged incubation time that is required. The process can take hours or days, depending on the cell type, transfection reagents, and MR contrast agents used. The prolonged incubation time can also alter the cellular properties and increase the risk of bacterial contamination.
In this study, we developed a positively charged nanoparticle for efficient labeling without the need for any additional reagents and investigated its efficiency in labeling cells.
Materials and Methods
Magnetic Nanoparticles
Alkali-treated dextran-coated, magnetic iron oxide nanoparticles (ATDM) and trimethylamino dextran-coated, magnetic iron oxide nanoparticles (TMADM-02) were kindly provided by the MEITO Sangyo Co., Ltd. (Kiyosu, Japan).
Cell Line
MIN6 cells were routinely grown in sterile plastic flask in Dulbecco's modified Eagle's medium (DMEM) containing 25 mM glucose supplemented with 15% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin and 5 μ/L β-mercaptoethanol at 37°C in a humidified atmosphere of 5% CO2.
Cell Labeling in Adhesion State
MIN6 cells (5.0 × 105 cells) were seeded in each well of a six-well plate (BD Biosciences) with 2 ml of culture medium for 12 h, and were confirmed to adhere to the bottom of the plate. MIN6 cells were incubated for 1 h at 37°C with TMADM-02 reconstituted in DMEM. At the end of the uptake experiments, MIN6 cells were washed three times in phosphate-buffered saline (PBS). The measurement of cellular toxicity was performed by the manual counting method based on the trypan blue exclusion procedure.
Cell Labeling in Floating States
MIN6 cells were detached from the plates with Trypsin-EDTA and incubated for 2 h at 37°C with TMADM-02 reconstituted in DMEM. At the end of the uptake experiments, MIN6 cells were washed three times in PBS. The measurement of cellular toxicity was performed by the manual counting method based on the trypan blue exclusion procedure.
Estimation of Iron Content in MIN6 Cells
The iron content of MIN6 cells labeled with each nanoparticles was measured by photon correlation spectroscopy (PCS), using Nuclear Magnetic Resonance Sequence (NMR) (Bruker mq20 Series NMR Analyzer: Bruker, Milton, Ontario, Canada). At the end of the uptake experiment, the labeled cells were collected in 500 μl deionized water and then were homogenized. The volume was made up to 3 ml with deionized water and then was analyzed by pulse NMR.
Results
Cell Labeling with the Novel Contrast Agent of MRI
In order to determine the labeling efficiency of the novel contrast agent of MRI, 5 × 105 MIN6 cells were incubated for 2 h at 37°C with each nanoparticle (50 μg Fe/ml) reconstituted in culture medium. TMADM-02 was efficiently transduced into the cells because of their positive charge (Table 1). On the other hand, the negatively charged nanoparticles, ATDM, were transduced to a lesser extent into the cells because of this condition of the labeling cells. These data suggest that TMADM-02 may be useful for labeling cells.
Characteristic Features of the Contrast Agents

Concentration Studies of Iron Uptake Levels in MIN6 Cells Labeled with TMADM-02
In order to determine the appropriate concentration of the nanoparticles for labeling the cells with TMADM-02, MIN6 cells were incubated for 2 h at 37°C with several concentrations of the contrast agent. The uptake of the nanoparticle reached its peak with 50 μg Fe/ml (Fig. 1).

Concentration studies of the iron uptake levels in MIN6 cells labeled with TMADM-02 in the adhesion state. MIN6 cells (5.0 × 105 cells) were incubated for 2 h at 37°C with TMADM-02 reconstituted in culture medium with 0.5, 5, 25, 50, and 100 μg Fe/ml. After incubation, the iron uptake was measured using NMR.
Time Course Studies of Iron Uptake Levels in MIN6 Cells Labeled with TMADM-02
In order to determine the treatment time of the nanoparticles for labeling the cells with TMADM-02, MIN6 cells were incubated at 37°C with 50 μg Fe/ml of TMADM-02 for several hours. The iron uptake reached its peak over a 2-h time course (Fig. 2).

Time course studies of iron uptake levels in MIN6 cells labeled with TMADM-02 in the adhesion state. MIN6 cells (5.0 × 105 cells) were incubated at 37°C with TMADM-02 reconstituted in culture medium with 50 μg Fe/ml for 0.5, 1, 2, and 3 h. After incubation, the iron uptake was measured using NMR.
Cell Number-Dependent Iron Uptake Levels in MIN6 Cells Labeled with TMADM-02
In order to evaluate the relationship of uptake with the cell number, MIN6 cells were incubated at 37°C with 50 μg Fe/ml of TMADM-02 at varying numbers of cells. The iron uptake increased in proportion to the number of cells (Fig. 3).

Cell number-dependent iron uptake levels in MIN6 cells labeled with TMADM-02 in the adhesion state. MIN6 cells (0.1, 0.5, 1.0, 5.0, and 10.0 × 105 cells) were incubated for 2 h at 37°C with TMADM-02 reconstituted in culture medium with 50 μg Fe/ml. After incubation, the iron uptake was measured using NMR.
Comparison of Transduction Efficiency of TMADM-02 in Adhesion and Floating State
MIN6 cells (5.0 × 105 cells) were incubated for 2 h at 37°C with TMADM-02 reconstituted in culture medium with 50 μg Fe/ml in adhesion and floating states. The uptake of the nanoparticles was higher in floating state in comparison to the adhesion state (Fig. 4).

Comparison of the transduction efficiency of TMADM-02 in the adhesion and floating states. MIN6 cells (5.0 × 105 cells) were incubated for 2 h at 37°C with TMADM-02 reconstituted in culture medium with 50 μg Fe/ml in the adhesion and floating states. After incubation, the iron uptake was measured using NMR.
Stability of the Nanoparticles in Culture Medium
The stability of the nanoparticle in the culture medium was evaluated. TMADM-02 aggregated in the medium (Fig. 5).
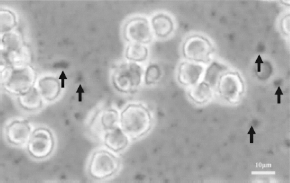
Aggregation of TMADM-02 in culture medium. MIN6 cells were incubated for 2 h at 37°C with 50 μg Fe/ml of TMADM-02 in culture medium. The arrows show the aggregated TMADM-02.
Discussion
The evaluation of homing, migration, and functional integration of locally transplanted cells by noninvasive, readily available, and reproducible techniques is warranted for the further development of restorative treatment strategies (9). MRI fulfills many of these requirements, can provide detailed anatomic information in a noninvasive manner, and is currently being used to characterize the histopathological and morphologic phenotypes (11–14, 25). One attractive approach is cell labeling with a contrast agent of MRI. Several groups have reported the use of supermagnetic iron oxide nanoparticles (SPIO), one of the T2 magnetodendrimer agents for MRI (3, 24). However, this technique requires the addition of positively charged transfection reagents, which cause cytotoxicity. In addition, the prolonged incubation times decrease the cellular viability. Therefore, this technique is not a practical cell-labeling method for clinical applications.
In this study, we developed a novel positively charged contrast agent of MRI, TMADM-02, for the magnetic labeling of transplanted cells. We used modified raw materials of the iron nanoparticle contrast agent Resovist® (ATDM) for cell labeling, which has already been clinically approved for use in humans. TMADM-02 was efficiently transduced into cells without the induction of cytotoxity. On the other hand, negatively charged ATDM was not transduced into the cells. These data suggest that the positively charged nanoparticles may be useful for cell labeling without the need for any additional agents.
In conclusion, TMADM-02 was efficiently transduced into cells. However, the aggregation of TMADM-02 may have led to a false-positive test result because of its low stability. In future studies, we should confirm not only the efficiency of labeling the cells but also the stability of the contrast agent of MRI for clinical applications.
Footnotes
Acknowledgments
The authors wish to thank Ms. Rina Yokota (Nagoya University) for assistance. This work was supported in part by the Juvenile Diabetes Research Foundation International (JDRFI); the Japanese Ministry of Education, Science and Culture; the Japanese Ministry of Health, Labour and Welfare; and Baylor All Saints Health Foundation.
