Abstract
Maintenance of freshly isolated porcine liver cells in vitro is limited for a short period of time. Therefore, establishment of easy handling cell lines is extremely important for in vitro study for liver cells and their possible utilization for cell differentiation and growth of stem cells. Porcine liver cells were transduced with a retroviral vector SSR#69 expressing SV40T, one of SSR#69-immortalized porcine liver cell lines, JSNK-1, was established and characterized. Morphology of JSNK-1 cells was spindle shaped. When the cells became confluent, JSNK-1 cells revealed hills-and-valleys pattern. In the presence of vitamin A, JSNK-1 cells showed big droplets inside the cytoplasm, which were positive with PAS staining. JSNK-1 cells showed the gene expression of collagen type 1α1, collagen type 1α2, FLT-1, β-actin, and SV40T. Immunostaining study revealed that JSNK-1 cells produced collagen, vimentin, and α-smooth muscle actin. JSNK-1 cells possessed the characteristics of the liver stellate cells. JSNK-1 cells produced hepatocyte growth factor (HGF) in a time-dependent manner. When cocultured with iPS cells towards the hepatic differentiation, JSNK-1 cells facilitated their hepatic differentiation in terms of albumin production. In conclusion, JSNK-1 cells would be valuable in the study of liver stellate cell pathophysiology and contribute to the optimization of hepatic differentiation of iPS cells.
Introduction
Hepatic stellate cells (HSCs) constitute a mesenchymal cell type and display unique features as morphology and function. HSCs store more than 80% of total body vitamin A and maintain a blood homeostasis of vitamin A. HSCs are located between parenchymal cell plates and sinusoidal endothelial cells and extend well-developed long processes surrounding sinusoids in vivo as pericytes and are responsible for microcirculation in the liver and scavenger functions (18). They are also the predominant cell types producing extracellular matrices (ECM) components as well as ECM-degrading metalloproteases, indicating their prime role in ECM remodeling in both normal and pathologic liver conditions (7).
The isolation of porcine liver cells allows us in vitro studies, but the procedure needs time and is laborious, and sometimes results in only small cell yields. Moreover, terminally differentiated liver cells have a limited proliferating potential and eventually enter senescence, thereby limiting their use for the basic study. One solution is the construction of cell lines, produced by an immortilization technique. One approach to get a clonal liver cell line is the transduction of primary liver cells with genes from DNA tumor virus, such as simian virus 40 large T antigen (SV40T) (6). Therefore, we used a retroviral vector expressing SV40T to immortalize the freshly isolated porcine liver cells. In the present study, we successfully established a porcine hepatic stellate cell line JSNK-1.
In both the developing and mature adult liver, cell–cell interactions are imperative for coordination of sophisticated liver functions (1). Coculture of hepatocytes with HSCs has been shown to help maintain differentiated phenotype of hepatocytes in vitro, such as albumin and cytochrome P-450-assosited proteins (1, 17).
Thus, we evaluated the effect of our newly developed cell line JSNK-1 on the enhancement of hepatic differentiation of mouse induced pluripotent stem (iPS) cells in vitro. Coculture of JSNK-1 cells increased functionality of iPS cells in terms of albumin production.
Materials and Methods
Structure of a Recombinant Retroviral Vector SSR#69
The retroviral vector SSR#69 has been previously described (24) in detail. SSR#69-producing Ψ Crip cells were cultured with Dulbecco's modified Eagle medium (DMEM) (Gibco, Invitrogen, CA) containing 10% new-born calf serum, 100 U/ml penicillin-G, and 100 μg/ml streptomycin sulfate.
Isolation and Transduction of Porcine Liver Cells with SSR#69
All of the animal procedures were approved by Okayama University Institutional Animal Care and Use Committee. Porcine liver cells were isolated using a modified four-step retrograde perfusion method with dispase and collagenase digestion, as previously reported (11). Briefly liver samples were first cannulated into the main blood vessels on the cut surface and flushed with EDTA in buffer solution at 37°C. The tissue was then continuously perfused with a prewarmed digestion buffer solution (collagenase IV and dispase II). Upon sufficient digestion, the liver capsule was mechanically disrupted and emerging cell suspension was filtered through a 250-μ mesh and then centrifuged at low speed (50 × g, 2 min, 4°C). We had regularly employed a cell incubation step of 10 min using the DNAse 1 containing buffer solution, during which cell clumps break up and damaged cells were digested. Then the resulted cell suspension was filtered through 75-μm nylon mesh and cells were harvested by low-speed centrifugation (50 × g, 75 s, 4°C). This was followed by washes using cold buffer solutions and filtered through 75-μm mesh and another centrifugation (50 × g, 75 s, 4°C). Finally, resulting cell clumps were suspended in Williams' medium E. Viability was assessed by a trypan blue exclusion test. The freshly isolated porcine liver cells were cultured in Williams' medium E (Gibco) containing 10% fetal bovine serum (FBS) (Hyclone, New Zealand), 1% penicillin-streptomycin solution (Sigma-Aldrich, Irvine, UK), and 1% l-glutamine (MP Biomedicals, OH) and subjected to the transduction with SSR#69 three times. One week after the final transduction, the cells were cultured with the medium containing 100 μg/ml hygromycin. One of the emerging clones, referred as to JSNK-1, was investigated in the present study.
Growth of JSNK-1 Cells
JSNK-1 cells were cultured with Williams' medium E medium supplemented with 10% FBS, 100 U/ml penicillin-G, and 100 μg/ml streptomycin sulfate. The cells grew steadily in vitro for more than 60 passages without showing any morphological changes. JSNK-1 cells (passage 30) were examined using an inverted phase contrast microscope Olympus IX51 (Olympus, Tokyo, Japan). To examine cellular proliferation of JSNK-1 cells (passage 30), a growth curve was obtained by an MTS assay, as previously reported (14).
Immunofluorescent Study for SV40T, α-Smooth Muscle Actin (α-SMA), Collagen Type 1 and Type IV, and Vimentin in JSNK-1 Cells
JSNK-1 cells were subjected to the immunostaining study for SV40T, α-smooth muscle actin (α-SMA), collagen type 1, collagen type IV, and vimentin. After JSNK-1 cells attached to slides well, the cells were fixed in 4% paraformaldehyde solution for 10 min at 37°C. After fixation, the cells were washed twice with PBS containing 0.1% Triton X-100 (EMD Chemicals, NJ), and then blocked with blocking solution (PBS containing 0.02% rabbit serum and 0.1% Triton X-100) for 10 min at 37°C. Cells were then labeled with a mouse monoclonal IgG α-SMA antibody (Upstate Biotechnology, NY), or mouse monoclonal IgG collagen type I antibody (Genetex Inc., CA), or rabbit collagen type IV antibody (LSL, Tokyo, Japan), or mouse monoclonal IgG vimentin antibody (Genetex Inc., CA), as previously reported (13, 15). Tetramethylrhodamine isothiocyanate-conjugated goat-poly anti-mouse IgG (Sigma-Aldrich, Irvine, UK), HRP-conjugated goat-poly anti-mouse IgG antibody (Bethyl Laboratories Inc., MI), or HRP conjugated goat-poly anti-rabbit IgG antibody (Stressgen Bio-reagents, MI), or FITC conjugated goat-poly anti-mouse IgG antibody (Bethyl Laboratories Inc.), were used as the secondary antibodies, respectively. The fluorescence microscope (Model Axiophot FL, Carl Zeiss, Inc., Oberkochen, Germany) was used to visualize the localization of these examined molecules. The Hoechst nuclear staining was performed to identify the nuclear fluorescence of JSNK-1 cells.
Electron Microscopic Examination
JSNK-1 cells cultured with Willams' medium E in the presence of retinol of 0–10 μM (Sigma-Aldrich) were subjected to transmission electron microscopy (TEM). Ultrathin sections of the samples were stained with uranyl and observed under Hitachi H-7100, as previously reported (23).
Gene Expression of SV40T and HSC Markers in JSNK-1 Cells
Reverse-transcription polymerase chain reaction (RT-PCR) was carried out for the gene expression analysis for SV40T and HSC markers (collagen type 1ot1, collagen type 1ot2, and FlT-1) in JSNK-1 cells at passages 30 and 50. A porcine β-actin gene served as internal control. Total RNA was extracted from JSNK-1 cells using TRIzol (Invitrogen, CA) reagent according to manufacturer's instructions and served as templates, as previously reported (19). Reverse transcription (RT) was performed at 22°C for 10 min, followed by 42°C for 20 min, using the specific primers. RT-PCR was performed with AmpliTaq gold DNA polymerase, Gene Amp PCR gold buffer, and MgCl2 solution (Applied Biosystems, CA). PCR products were resolved on 2.5% agarose gels and visualized by ethidium bromide staining. Normal porcine liver cells were used as a positive control and SSR#69-immortalized human hepatocyte NKNT-3 cells (9) were used as a negative control, respectively. Primers used were as follows: SV40T [422 base pair (bp)]: sense 5′-CAGGCATAGAGTGTCTGCA-3′ and antisense 5′-CAACAGCCTGTTGGCATATG-3′; FLT-1 (331 bp): sense 5′-ACTACCAGATGGACAACGGC-3′ and antisense 5′-ATACACACGTGGTGCTGGAA-3′; collagen type 1ot1 (243 bp): sense 5′-CCTGGACGCCATCAAA GTCTTC-3′ and antisense 5′-TGGGAAGCCTCAGTG GACATC-3′; collagen type 1ot2 (297 bp): sense 5′-GC CTAGCAACATGCCAATCT-3′ and antisense 5′-CAG GTCCTTGGAAACCTTGA-3′ β-actin (218 bp): sense 5′-TGGACTTCGAGCAGGAGATG-3′ and antisense 5′-GTGTTGGCGTAGAGGTCCTT-3′.
Measurement of Hepatocyte Growth Factor (HGF) Production of JSNK-1 Cells
To measure the production of hepatocyte growth factor (HGF), 1 × 106 of JSNK-1 cells were plated in T75 flasks. Two days after plating, the cells were cultured with Willams' medium E without FBS for 48 h. The production of HGF in the culture medium of JSNK-1 cells was measured by porcine hepatocyte growth factor ELISA kit (Cusabio Biotech, Wuhan, China) according to the manufacturer's instructions.
Coculture of Mouse iPS Cells and JSNK-1
Mouse iPS cells were kindly provided by Riken Cell Bank (cell No. APS0001, cell name iPS-MEF-Ng-20D-17, Lot No. 006) (16). The culture of iPS cells was performed as previously reported (5). Knockout DMEM-based embryoid body (EB) formation medium, consisting of Knockout DMEM (Gibco), 15% knock out serum replacement (KSR) (Gibco), 1% nonessential amino acids, 1% 2-mercaptoethanol (Gibco), 1% pencillin/streptpmycin, and 1% l-glutamic acid was used for EB formation of iPS cells. The cells were cultured free floating with ultra-low attachment six-well plates (Corning, NY) at a density of 1 × 105 cells/ml. EBs at day 5 were then resuspended in Knockout DMEM medium consisting of 1% penicillin/streptomycin, 1% l-glutamic acid, 100 ng/ml activin A (R&D systems, MN), 100 ng/ml basic fibroblast growth factor (bFGF) (Peprotech, NJ) to induce definitive endoderm (DE) for an additional 3 days. KSR was supplemented at days 6–7 and the concentrations were varied: 0.2% for day 6 and 2.0% for day 7. Half of the medium was replaced daily. At day 8 the medium was replaced by Knockout DMEM supplemented with 10% KSR, 1% nonessential amino acids, 1% l-glutamic acid, penicillin/streptomycin, 1% dimethyl sulfoxide (DMSO) (Sigma-Aldrich), and coculture of JSNK-1 cells started using cell inserts. The coculture was conducted for 8 days (days 8–15) and iPS cells were cultured with dexamethasone (40 ng/ml) for an additional 3 days (days 16–18). At day 19, the culture medium was collected and analyzed for albumin production by an Assay Max Mouse Albumin ELISA kit (Assay Pro, MO) according to the manufacturer's protocol.
Assay for Oncogenecity of JSNK-1 Cells
To evaluate the tumorigenicity of JSNK-1 cells, 1 × 107 cells were inoculated into the thighs of three different severe combined immunodeficiency (SCID) mice. As a control, 1 × 106 human liver cancer PLC/PRF/5 cells were transplanted at the opposite side. The mice were observed for at least 3 months after transplantation.
Statistical Analyses
Mean values are presented with SDs. Differences between groups were estimated by Student's t-test analysis. All differences were deemed significant at the level of p < 0.05.
Results
Morphology of JSNK-1 Cells
One of SSR#69-transduced immortalized porcine liver cell lines, JSNK-1, was longitudinally spindle shaped (Fig. 1A). When the cells became confluent, JSNK-1 cells displayed hills-and-valleys pattern (Fig. 1B), which is one of the in vitro characteristics of HSCs.

Morphology and growth of JSNK-1 cells. (A) JSNK-1 cells were longitudinally spindle shaped. Scale bar: 200 μm. (B) At confluence, the cells display hills-and-valleys formations, which is typical of HSCs under phase-contrast microscopy. Scale bar: 200 μm. (C) JSNK-1 cells grew steadily in vitro and proliferated at more than passage 60. (D) TEM revealed abundant cytoplasmic inclusions, resembling fat droplets, in JSNK-1 cells. Scale bar: 250 μm. (E) Bigger fat droplets appeared in the cytoplasm when the concentration of retinol increased at 10 μM. Scale bar: 250 μm. (F) These cytoplasmic inclusions were positive for Oil Red O staining, and confirmed to be fat droplets (original magnification 400x).
The cells grew steadily in vitro (Fig. 1C). JSNK-1 cells cultured in the medium containing retinol (5 μM) showed many fat droplets in their cytoplasm (Fig. 1D) and when the concentration of retinol increased at 10 μM big fat droplets appeared in the cytoplasm of JSNK-1 cells (Fig. 1E). These fat droplets were positively stained with an Oil Red O reagent (Fig. 1F). These findings indicate that JSNK-1 cells possessed HSC characteristics.
JSNK-1 cells showed the protein expression of SV40T, α-SMA, Collagen Type 1 and Type IV, and Vimentin
Immunofluorescence study for SV40T revealed that JSNK-1 cells uniformly expressed SV40T in the nuclei. Normal human fibroblasts, used as a control, were uniformly negative for SV40T expression. These data indicated that the emerging JSNK-1 cells were not derived from spontaneous long-surviving subpopulations in the parental cultures, but an SSR#69-immortalized cell clone. The presence of α-SMA, collagen type 1 and type IV, and vimentin was confirmed in JSNK-1 cells (Fig. 2).

Immunofluorescent study for α-SMA, collagen type 1 and type IV, and vimentin in JSNK-1 cells. JSNK-1 cells produced α-SMA (A), collagen type 1 (B), collagen type IV (C), and vimentin (D) in their cytoplasm. Scale bar: 50 μm.
Gene Expression of SV40T and HSC Markers in JSNK-1 Cells
HSC markers were examined in JSNK-1 cells at two different time points (passage 30 and passage 50). JSNK-1 cells showed the stable gene expression profile of collagen type 1α1 and type 1α2 and FLT-1. The gene expression of SV40T was also confirmed in JSNK-1 cells (Fig. 3).

The gene expression of HSC markers and SV40T in JSNK-1 cells. RT-PCR was performed to determine the gene expression of HSC markers, collagen type 1α1 and collagen type 1α2, FLT-1, and SV40T at passages 30 and 50. Equal loading of PCR product was verified by detecting the β-actin gene mRNA. JSNK-1 cells stably expressed these HSC markers and SV40T. The data are representative of passage 50.
JSNK-1 Cells Produced HGF
Production of HGF was analyzed in JSNK-1 cells. The cells produced HGF into the culture medium and the amount increased in the time-dependent manner (Fig. 4A, B). The amount of HGF produced was 71.95 ± 23.86 pg/ml for 24-h culture and 156.33 ± 28.53 pg/ml for 48-h culture, respectively.
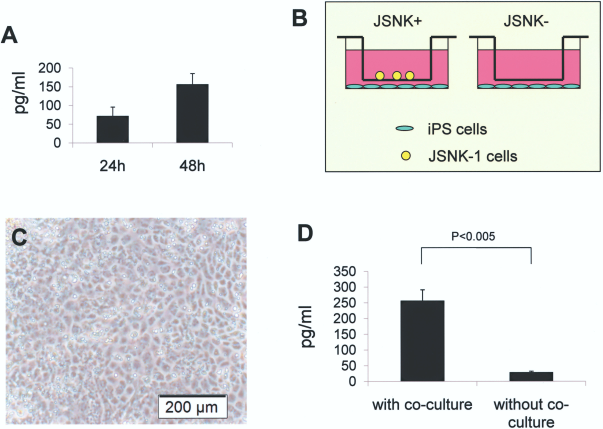
HGF production of JSNK- 1 cells and the effect of coculture of iPS cells with JSNK-1 cells. (A) HGF production of JSNK-1 cells. JSNK-1 cells produced HGF in time-dependent manner. (B) Schematic drawings of coculture of iPS and JSNK-1 cells. The gelatin-coated 12-well plates and cell culture inserts were used for coculture of iPS and JSNK-1 cells. (C) When cocultured with JSNK-1 cells, iPS cells showed maintenance of polygonal shape and abundant cytoplasmic granules under a phase contrast microscope. Scale bar: 200 μm. (D) Albumin production of iPS cells significantly increased when cocultured with JSNK-1 cells in comparison of a single culture of iPS cells.
Morphology and Albumin Production Capacity of iPS-Derived Hepaocyte-Like Cells in the Coculture of JSNK-1 Cells
iPS-derived hepaocyte-like cells, when cocultured with JSNK-1 cells, showed hepatocytic morphology, such as polygonal shape, well-defined margins, showing abundant cytoplasmic granules, under phase contrast microscope (Fig. 4C). iPS-derived hepatocyte-like cells significantly increased albumin production when cocultured with JSNK-1 cells. The production of albumin of iPS cells was 29.0 ± 3.2 pg/ml for a single culture of iPS cells and 256.9 ± 34.2 pg/ml for coculture with JSNK-1 cells, respectively (Fig. 4D).
Oncogenetic Assay of JSNK-1 Cells
For oncogenetic study, JSNK-1 cells (1 × 107) were transplanted into three SCID mice. No tumor developed at the injection site of JSNK-1 for at least 3 months after transplantation, whereas PLC/PRF/5 cells (1 × 106) formed tumors around 3 weeks after inoculation. These results demonstrated that JSNK-1 was not a tumorigenic clone.
Discussion
The liver is one of the largest organ in the body that performs various functions in the body, including synthesis of serum proteins, regulation of nutrients, production of bile, and the metabolism and conjugation of toxic compounds for excretion in the bile or urine. Although hepatocytes are the predominant cells in the liver, other cell types, such as sinusoidal endothelial cells, cholangiocytes, Kupffer cells, and HSCs, are equally important in coordinating liver functions. HSCs, being the nearest neighbor of hepatocytes, play vital role in microcirculation in the liver, proliferation and maintenance of differentiation of hepatocytes, vitamin A homeostasis, and scavenger functions. So the role of HSCs cannot be elucidated when considering the differentiation of stem cells to hepatocytes and prospective clinical cell therapies for liver disorders. In recent decades, due to advancement in regenerative medicine, cell therapies are becoming the attractive choice for various liver disorders. For such therapies, we will need the continuous supply of cells on an extremely large scale. Unfortunately, there is severe shortage of human donor livers for liver cell isolation; thus, porcine liver cells may be an alternative for cell therapies, such as hepatocyte transplantation and extracorporeal bioartificial liver support (12). As primary porcine liver cells are very difficult to maintain for the long term, especially to induce proliferation in vitro culture, there is compelling need of easy handling cell lines (8). Therefore, we tried to retrovirally transduce primary porcine liver cells with SV40T and successfully established a clonal cell line JSNK-1. JSNK-1 cells showed a hills-and-valleys pattern at confluence in culture (Fig. 1B) and TEM revealed big fat droplets (Fig. 1D, E) in their cytoplasm, which was confirmed by PAS staining (Fig. 1F). JSNK-1 cells were positive for the protein expression of SMA, collagen, and vimentin (Fig. 2). JSNK-1 cells posses HSC characteristics. We believe that JSNK-1 may be a very useful tool for the study of HSCs.
Recently, iPS cells had been successfully established from mouse and human fibroblasts (10, 20). iPS cells are pluripotent and are able to proliferate unlimitedly in vitro. This technical breakthrough in creating iPS cells from somatic cells has noteworthy implications for overcoming the immunological rejection and the ethical issues associated with the derivation of embryonic stem cells from embryos. In the near future scientists will be interested in the use of hepatocytes derived from iPS cells to treat liver diseases. Towards the goal, it is extremely important to establish a reliable and reproducible hepatic differentiation-inducing method for iPS cells. Therefore, we investigated the effect of JSNK-1 cells on the hepatic differentiation of iPS cells in vitro. As shown in Figure 4C and D, coculture of JSNK-1 cells maintained the favorable morphology of iPS-derived hepatocyte-like cells and increased the production of albumin 10-fold. The exact mechanism of coculture of JSNK-1 cells is now under investigation. One explanation is that enhanced hepatic differentiation of iPS cells was due to the presence of HGF produced by JSNK-1 cells. HGF is a mesenchymally derived potent mitogen for hepatocytes and acts as a growth factor for a broad spectrum of tissues and cell types (2, 4, 19). We also speculate that lipid droplets in JSNK-1 cells may contribute to hepatic differentiation for iPS cells. Lipid droplets in HSC contain not only vitamin A, but they also contain triglycerides, cholesterol, and phosopholipids (25). The differentiation-inducing action of vitamin A has been well investigated (3); more recently unsaturated fatty acids have been recognized as important signals in diverse processes such as differentiation, development, and proliferation (21). Moreover, HSC express another protein molecule named as epimorphine, which induces in vitro differentiation of hepatocytes (22). Hence, JSNK-1 cells can be useful for iPS cells towards their hepatic differentiation.
Further evaluation including identification of cytokines and growth factors that are produced by JSNK-1 cells is really essential before applying human iPS cells for the clinical use. JSNK-1 cells were not tumorigenic when injected into SCID mice for at least 3 months. However, unexpected malignant transformation of JSNK-1 cells for long-term culture in vitro cannot be completely denied. Accumulation of the redundant data on the safety issues will be needed for JSNK-1 cells, even though the cells are used for coculture.
In conclusion, we have established a porcine hepatic stellate cell line and observed the enhanced hepatic differentiation of iPS cells when JSNK-1 cells were cocultured.
Footnotes
Acknowledgment
This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science, and Culture (to N.K), Japan.
