Abstract
While the problem of organ shortage has not yet been solved, the number of patients who need to be treated with dialysis due to end-stage renal disease (ESRD) is increasing each year. With the aim of eliminating dialytic therapy as much as possible, the opportunities for organ donation from expansive criteria donor (ECD) or marginal donors due to cardiac death have been increasing. With the purpose of extracting organs in a state in which the function is preserved as much as possible, we reexamined the conditions of tissue disorders resulting from temporary ischemia of the organs as well as changes in tissue function and the effects on the preservation of renal function over time by using rat models in order to clinically utilize erythropoietin, which has inhibitory effects on ischemia-reperfusion disorder, as has been conventionally reported. With 8- to 9-week-old Wister male rats, after the right kidney had been resected under general anesthesia, the left renal artery was clamped to inhibit the blood flow for 45 min. At 30 min before inhibiting the blood flow and after releasing the inhibited blood flow, 100 U/kg of recombinant human erythropoietin (rhEPO) was administered via the inferior vena cava and the abdominal cavity, and then the tissues and blood samples were extracted at 6 and 24 h after the release. The renal tissue specimens were evaluated using H&E staining and TUNEL staining in order to observe differences in the expression of apoptosis as well as the renal function and changes in the emergence of active oxygen were investigated by using samples that had been obtained from drawn blood. Moreover, we examined the degree of renal dysfunction by means of neutrophil gelatinase-associated lipocalin (NGAL) in the spot urine samples. The changes in renal function, which were observed according to the serum creatinine level, showed that the renal function was preserved with a significant difference in the rhEPO administration group. The liver deviation enzymes, which had also shown increases in the serum as well as the occurrence of renal dysfunction, showed clear decreases in the serum, even though changes with a significant difference were not observed in the rhEPO administration group. The active oxygen did not show changes before and after ischemia-reperfusion nor changes due to the rhEPO administration. When examining the status of apoptosis in the tissues, apoptosis was shown to be inhibited due to the rhEPO administration. It is believed that the main preservation effects of rhEPO are the elimination of cytopathy/cell death, as derived from the resulting ischemic condition that extends to the target organ before ischemia occurs. In this examination, no direct effects of rhEPO administration on the emergence of active oxygen were observed. It is therefore suggested that there is a possibility of preserving the renal function in marginal donors with a longer agonal stage by effectively using rhEPO.
Introduction
Bonsdoff et al. (1) referred to biological fluid component that has increased erythropoietic effects as “erythropoietin” in 1948, and Jacobson clarified that erythropoietin is produced in the kidneys (7). It is known that erythropoietin mostly affects colony forming unit-erythroid and promotes maturation in addition to inhibiting apoptosis and adjusting the production of blood cells (18). Furthermore, another effect of erythropoietin that is believed to be effective in a renal anemia condition is the inhibition of neocytosis in which the produced premature blood cells are destroyed (24). However, according to Garcia et al. in 1988, 5 out of 6 nephrectomy models showed that the administration of erythropoietin ameliorated anemia but induced hypertension and exacerbated renal function (3). Subsequent studies (10, 25) and a prospective randomized control study (RTC) (11) showed that the sufficient control of hypertension ameliorated anemia and, in turn, protected renal function and inhibited the progress of renal dysfunction (4), so thereafter erythropoietin obtained the status of a medical agent, which substantially contributed to the renal anemia of patients with renal dysfunction. For a long time, erythropoietin has garnered attention only with regard to its erythropoietic function and not regarding its other effects. However, Sakanaka et al. proved that erythropoietin that is produced in the brain has a function of protecting nerve cells (19), and Parsa et al. reported the effects of erythropoietin on the size of myocardial infarction (17). Furthermore, it is suggested that it has local physiological effects on the genital organs or the digestive tract (20). These effects proved that a plurality of cellular protection signals are activated by linking erythropoietin with its receptors, and among these, phosphatidylinositol 3-kinase (PI3K) is important. The activated PI3K further activates Akt kinase (protein kinase B), which plays a major role in inhibiting cell death in the intracellular signaling of cytokine, and inhibits the apoptosis or oncosis of tissues resulting from ischemia or reperfusion disorder.
Moreover, in the area of organ transplantation, serious organ disorder caused by ischemia-reperfusion disorder is an important factor in obtaining the function of the transplanted organ after transplantation, so it is expected that the administration of erythropoietin would eliminate such disorders.
It is necessary for donor kidneys for transplantation to be perfused with a perfusion solution and for the donated organ to be cooled immediately after cardiac arrest as well as to handle it so as to not clot the blood. Therefore, the donated organ is inevitably susceptible to tissue disorders due to an ischemic condition or reperfusion. In the current situation of transplantation in which the problem of organ shortage has not yet been resolved, the potential likelihood of receiving an organ donation from an expansive criteria donor (ECD) with a longer agonal stage and deviations from the criteria of general organ donation has been increasing. On the other hand, there is no means of appropriately observing the biological activation level of a donated kidney in the agonal stage, so it is difficult to examine in real time because the generally performed tests such as for changes in the serum creatinine level causes time differences between the test results and the activity level. Consequently, in addition to the conventionally used serum creatinine level, we examined neutrophil gelatinase-associated lipocalin (NGAL), which has recently garnered attention as a biomarker in urine. It is said that NGAL in urine is effective as a biomarker for acute renal disorder and its sensitivity to renal disorders is significantly high (16).
In this study, with the purpose of achieving renal function after transplantation, we examined the possibility of utilizing erythropoietin in order to maintain graft viability at the agonal stage before the organ is donated by a donor, by using rat ischemia-reperfusion models, and discussed the significance of NGAL in spot urine as a biomarker for donation-related renal tissue disorders.
Materials and Methods
Animal Model
We purchased 32 Wister/ST male rats with a weight range of 250–300 g from Japan SLC, Inc. (Shizuoka, Japan) and raised them for a week for use in a subsequent experiment. In performing the experiment, we observed the Guidelines for Animal Experiments in Kanazawa Medical University. The rats were randomly divided into four groups (the right kidney of each of the rats was resected): 1) a group without ischemia-reperfusion by administrating saline (single kidney control group, n = 8); 2) a group with only ischemia-reperfusion treatment (IRI group, n = 8); 3) a group in which rhEPO was administered 30 min before ischemia-reperfusion (EPOpre30 group, n = 8); and 4) a group in which rhEPO was administered at the time of ischemia-reperfusion (EPOjust0 group, n = 8). The control group, the EPOpre30 group, and the EPOjust0 group were initially intravenously administered 500 μl of saline and a 100 IU/kg of recombinant human erythropoietin (Chugaipharm., Tokyo, Japan) via the inferior vena cava by 30 min before the ischemia and concurrently with the ischemia. The EPOpre30 group was injected 30 min before the right nephrectomy and the avascularization of the left renal arteriovenous, and the EPOjust0 group was injected concurrently with the avascularization. After administering inhalation anesthesia to all of the rats using diethyl ether (Wako Pure Chemical Industries, Ltd., Osaka, Japan), 30 mg/kg of sodium pentbarbital (Kyoritsu Pharm., Ltd., Tokyo, Japan) was intraperitoneally administered. After avascularization of the left renal arteries and veins was ensured by using an artery clip via a transabdominal approach, the color of the kidney was macroscopically observed to have become dark red. The avascularization time was set to 45 min and, thereafter, the blood flow was resumed/reperfused by removing the artery clip wherein the kidney was macroscopically observed to have returned to a red color. With the aim of extracting the tissue, rats were sacrificed at 6 and 24 h after the reperfusion. At the time of extracting the tissue, we washed the blood from the tissue with about 15 ml of saline using a 24-gauge indwelling needle in the blood vessel from the abdominal aorta.
The Blood Biochemical Test and the Measurement of Serum Active Oxygen
The blood biochemical test targeted creatinine (Cr), serum urea nitrogen (BUN), asparate aminotransferase (AST), and alanine aminotransferase (ALT), and we measured these at three time points: before the ischemic treatment and at 6 and 24 h after the ischemic reperfusion (using only the left kidney).
For the active oxygen level, 140 μl of a 0.1 M NaOAc buffer was added to each well of a 96-multiwell plate, after which 5 μl of serum that was to be used for the blood biochemical test as a sample as well as 5 μl of standard hydrogen peroxide solution for the creation of an analytical curve were added, and subsequently incubation was performed at 37°C for 5 min. After 100 μl of a mixture of R1 solution and R2 solution (1:25, prepared immediately before use) was added to each well and incubated on a plate reader set to 37°C for 1 min, the absorbance at 505 nm was measured at intervals of 15 s, up to 180 s. In accordance with the upward slope of each measured value from 60 to 180 s after the measurement was started, the total reactive oxygen species (ROS) in the sample was calculated and represented in logarithm. R1 was prepared by dissolving N,N-diethyl-para-phenylene-diamine, DEPPD) in a 0.1 M NaOAc buffer with a pH of 4.8 to total 100 mg/ml. R2 was prepared by dissolving ferric chloride (Sigma Aldrich Co., St. Louis, MO, USA) in a 0.1 M NaOAc buffer to total 4.37 mM.
The Measurement of NGAL in Urine (Sandwich ELISA Analysis of NGAL Expression in Urine)
The urine samples were extracted at three time points—before the ischemic treatment and 6 and 24 h after the ischemic reperfusion (using only the left kidney)—in accordance with the extraction of blood samples. The samples were centrifuged immediately after being extracted, and the supernatant was preserved at −70°C until use. The NGAL was measured in accordance with the accompanying procedural instructions by using a Rat NGAL ELISA Kit (BioPorto Diagnosis A/S, Denmark) in Sandwich ELISA analysis employing streptoavidin. The samples that had been frozen and preserved were thawed at room temperature and diluted to 500 times for the test. For all of the samples, the average was derived in triplicate. The standard curve and the absorbance of the samples were measured with a microplate reader (model 680 microplate reader, Bio-Rad Laboratories, Inc., CA, USA) at a wavelength of 450 nm and were analyzed using Microplate Manager Software (ver. 5.2PC, Japan Bio-Rad Laboratories, Inc., Tokyo, Japan).
Terminal Deoxynucleotidyl Transferase-Mediated dUTP Nick End-Labeling (TUNEL) Staining
The resected kidney was longitudinally divided into two, immersed into a 10% formalin solution, embedded in paraffin, sliced into 6-μm slices with a microtome, and mounted on a glass slide. After being deparaffinized with xylene, it was subjected to hydrophilic treatment with ethanol at multiple levels of concentration to complete the preparation before staining. The TUNEL staining was performed in accordance with the accompanying procedural instructions using the Apoptosis in situ Detection Kit Wako (Wako Pure Chemical Industries, Ltd., Osaka, Japan). After again being dehydrated with ethanol at multiple levels of concentration, it was subjected to penetration treatment with xylene for encapsulation with a hydrosoluble encapsulant (Biomeda Corp., CA, USA). We employed an Olympus BX50 (Olympus Corporation, Tokyo, Japan) microscope for microscopic observation and captured images with a DP71SET-A (Olympus Corporation, Tokyo, Japan) digital camera for microscopes. For the analysis of the images, the DP2-BSW (Olympus Corporation, Tokyo, Japan) analytical software program was used.
Statistics
The significance of differences between control and test values was determined by the two-tailed multiple t-test with Bonferroni correction, wherein 0.05 or less was designated as significant.
Results
Blood Biochemical Findings
As a result of the blood biochemical test (Table 1), the group in which the rhEPO was administered 30 min before the ischemic procedure had a significantly inhibited increase in the serum creatinine level in comparison to the group without the rhEPO administration and the group in which the rhEPO had been administered immediately after the ischemic treatment and at 6 and 24 h after the ischemia-reperfusion. Moreover, the BUN was also significantly inhibited from increasing in the group in which the rhEPO had been administered immediately after the ischemic treatment (Fig. 1).
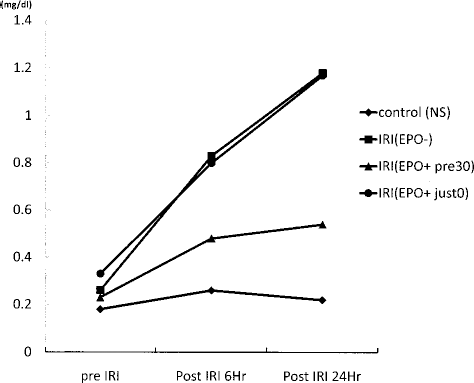
Changes in the serum creatinine levels at 6 and 24 h after ischemia-reperfusion in each group.
The Serum Creatinine Level, Urea Nitrogen, and Liver Deviation Enzyme Level Before and After Ischemia-Reperfusion in Each Group

In addition to the serum creatinine level and BUN, a significant difference was not obtained, but the liver deviation enzyme (AST/ALT) was observed to have a lower increase from the level before the ischemic procedure in the group that was administered 30 min before, in comparison to the groups that were administered immediately after the ischemic procedure and had no administration.
Serum Active Oxygen Levels
At the time when the biochemical investigation was performed, the serum active oxygen level was measured. With this measurement method, generally from 100 to 200 units of active oxygen were observed in normal adult serum. However, in our examination, the active oxygen level in the rat blood was also within a similar range, wherein a specific difference did not occur among the changes in either group. The serum active oxygen level 6 h later was in a slightly downward tendency in both groups in comparison to that before the ischemic procedure (Fig. 2).

The serum active oxygen levels at 6 h before and after ischemia-reperfusion. Serum active oxygen was measured with the total ROS assay system by means of the phenytoin reaction method using a 96-multiwell plate.
Results of the NGAL in the Urine
For the changes in the NGAL that were measured with spot urine, increases in the urine NGAL, which are believed to have invaded due to the sham OP, were observed even in the control group in which the ischemic procedure was not performed, while in the ischemia-reperfusion group increases in the urine NGAL were significantly observed in the non-rhEPO administration group. On the other hand, in the group in which rhEPO was administered 30 min before the ischemic procedure, increases in the urine NGAL were observed more than that of the control group, but they had decreased greater than that in the ischemia-reperfusion group without rhEPO administration (Fig. 3).

The concentration of NGAL in spot urine using ELISA. Urine NGAL before and 6 and 24 h after ischemia-reperfusion between the group without EPO administration and the group that received administration 30 min before the ischemia-reperfusion compared to the control group. ∗p < 0.05 versus the group without EPO administration.
TUNEL Staining and Evaluation
In order to histogenetically check the rhEPO of renal protection from ischemia-reperfusion disorder, we performed TUNEL staining and compared the number of positive cells in the TUNEL staining at 24 h after the ischemic reperfusion, between the rhEPO administration group and the group without administration. In fact, the renal tissues in the ischemia-reperfusion disorder model expressed a number of cells in which apoptosis occurred in the tubular epithelium (Fig. 4). In the rhEPO administration group, the apoptosis of cells was observed to have significantly decreased (Fig. 5).
Discussion
In this study we observed the effects of rhEPO preserving the renal function in renal ischemia-reperfusion models using rats and examined its usability as a biomarker for acute renal disorder resulting from ischemia-reperfusion by measuring the NGAL in spot urine. In the examination using rat models, the rhEPO appeared to have significantly inhibited the ischemia-reperfusion renal disorder but also showed the effects of rhEPO that could not be expected depending on the administration timing. Specifically, this examination showed that the renal dysfunction was significantly inhibited in the group in which the rhEPO had been administered 30 min before the ischemic procedure, while the renal function was exacerbated in the group in which the rhEPO had been administered at the same time as the ischemia as well as in the group without the rhEPO administration. The protection mechanism of the rhEPO due to tissue disorders, as a direct effect, acts to protect tissue by causing glutathione peroxydase activities to increase in the renal tissues, promoting the production of HO-1 (heme oxygenase-1), and inhibiting the intracellular oxidation stress (9), and as for the indirect effects, rhEPO is believed to facilitate endothelial progenitor cell (EPC) inducibility into the marrow, cause an increase in the expression of VEGF (vascular endothelial growth factor) as an physiologically active substance, and act to protect the renal function (8). Therefore, it is necessary for the rhEPO to have reached the target organ before ischemia-reperfusion disorder occurs, so it is believed that, also in this examination, in the group in which rhEPO was administered at the same time as the ischemia, the rhEPO had not reached the organ before the disorder occurred, so tissue disorder progressed at exactly the same degree as in the group without such administration. In the examination of ischemic renal dysfunction models, it was reported that the renal function was not improved by the administration of rhEPO after the ischemia-reperfusion (15, 21), so it is believed that the timing of administration in which the effects of the rhEPO are expected should be before the ischemia. In this study, we examined the serum concentration of active oxygen, but changes due to the rhEPO administration were not observed. As mentioned above, the effect of the rhEPO is not to decrease the active oxygen but rather to inhibit a disorder from the produced active oxygen, and this examination result also demonstrated such point.
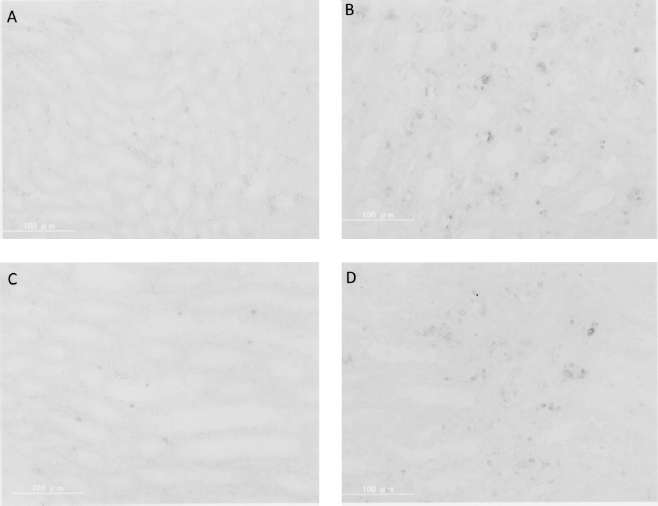
TUNEL staining in the renal tissue at 24 h after ischemia-reperfusion. (A) The control group; (B)the group with only ischemia-reperfusion (EPO-); (C) the group in which EPO was administered 30 min before ischemia-reperfusion (EPO+ pre30); and (D) the group in which EPO was administered concurrently with ischemia-reperfusion (EPO+ just0).

The number of positive cells in TUNEL staining at 6 and 24 h after ischemia-reperfusion. The number of positive nuclear staining cells in TUNEL staining was represented in the average value from three viewpoints in the control group, the group without ischemia-reperfusion and EPO administration (EPO-), the group in which EPO was administered 30 min before ischemia-reperfusion (EPO+ pre30), and the group in which EPO was administered concurrently with ischemia-reperfusion (EPO+ just0).
Another noteworthy point regarding rhEPO is the effect of inhibiting apoptosis. As for molecules that inhibit changes into apoptosis due to ischemia, Bcl-2 is well known, and the inhibition of apoptosis in the renal tubular epithelial cells was also observed in Bcl-2 transgenic mice (23). Further applications are expected, but the clinical application thereof still appears to be distant. As shown in our results, decreases in apoptosis-positive cells were significantly observed in the rhEPO administration group. In this study as well, in the cases of the renal function, apoptotic-positive cells were observed in the group that was administered immediately after the ischemia-reperfusion with almost no difference compared to the group without the administration. Apoptosis, among the forms of cell death, is a so-called programmed death that a biological body selectively undergoes, and the control becomes activated when the tissue or organ is exposed to serious disorder. Therefore, it is a very useful function and is effective for organs that would be controlled after a serious disorder is avoided. Essentially, the erythropoietin that is produced in vivo, as one type of molecule that is activated by HIF1-α (hypoxia inducible factor 1-α) resulting from a low oxygen condition at the tissue cellular level, other than a hematogenic effect, it is in charge of the function of reversing changes from autopahgy to apoptosis that progress due to disorders resulting from low oxygen, thus allowing for a powerfully inhibiting transition to apoptosis through the administration of rhEPO (14).
Although the standard test method of renal function for evaluating the degree of renal disorder is to test the serum creatinine level, creatinine essentially does not increase to a clinically critical concentration within 1 or 2 days, and the effects at the time of a disorder can be retrospectively expected as residual renal function, but it is actually difficult to show the degree of a disorder that occurs at that point. It has been elucidated in urine NGAL monitors with acute renal dysfunction that occurs after open heart surgery in children that the concentration of NGAL in the urine increases within a few hours after acute renal dysfunction occurs, and therefore it is optimal as a biomarker for acute renal dysfunction (13). We conducted this study with a view of its possibility as a real-time biomarker for acute renal dysfunction at the agonal stage in kidney marginal donors, which has been anticipated in the medical field of renal transplantation. As a result of the study, we recognized that it appears to be a very promising biomarker for renal dysfunction supported by changes in creatinine.
While its utilization for a hematogenic effect by the EPO that has been established with a long history and the effects of tissue protection capability that were recently observed and reported have garnered attention, there are also some negative aspects that cause concern regarding the administration thereof. Some cancer cells, specifically renal cancer cells, express EPO receptors, so there is concern that, when the EPO is activated, cellular proliferative ability would increase and cancer proliferation would progress due to the apoptosis inhibition ability or VEGF (5). Indirectly, there is a possibility of shrinking the renal vessels and reducing the physiological function via endothelin (22). The level of administration that we set was the same as the general clinical level, but with high-dosage rhEPO administration as has previously been studied (15), there is concern that erythrocytosis may be caused, the blood may become more viscous, and the rate of the occurrence of thrombosis may increase. Recently, when targeting patients without anemia in order not to target the hematogenic effect of such an EPO, the cellular protection effect in nerve cells due to the EPO desialylated so as to not demonstrate a strong hematogenic effect has reported (2). Moreover, it has been reported that the carbamoylated EPO of other derivatives also does not have hematogenic activity but does have a cellular protection effect in vitro (12) and an effect of protecting against ischemia-reperfusion disorder in the kidneys (6). This is still at a stage that requires further examination, but it is expected to be one alternative for ensuring a tissue protection effect in the future.
In conclusion, rhEPO appears to inhibit ischemia-reperfusion disorder while also preventing apoptosis from being induced in general clinical usage. The measurement of NGAL in spot urine suggests that there is a possibility of using this marker as a real-time biomarker in the kidneys at the agonal stage. We will continue our examinations with the aim of clinically utilizing marginal graft protection via rhEPO in marginal donors with a longer agonal stage.
