Abstract
Human amnion-derived cells are considered to be a promising alternative cell source for their potential clinical use in tissue engineering and regenerative medicine because of their proliferation and differentiation ability. The cells can easily be obtained from human amnion, offering a potential source without medical intervention. It has been proven that human amnion-derived cells express immunosuppressive factors CD59 and HLA-G, implying that they may have an immunosuppressive function. To assess the immunosuppressive activity, we investigated the effect of human amnion-derived cells on NK cell and monocyte function. Amnion-derived cells inhibited the cytotoxicity of NK cells to K562 cells. The inhibition depended on the NK/amnion-derived cell ratio. The inhibition of NK cytotoxicity was recovered by continuous culturing without amnion-derived cells. The inhibition of NK cytotoxicity was related to the downregulation of the expression of the activated NK receptors and the production of IFN-γ, as well as the upregulation of the expression of IL-10 and PGE2 in human amnion-derived cells. The addition of antibody to IL-10 or PGE2 inhibitor tended to increase NK cytotoxicity. IL-10 and PGE2 might be involved in the immunosuppressive activity of amniotic cells toward NK cells. Amniotic cells also suppressed the activity of cytokine production in monocytes analyzed with TNF-α and IL-6. These data suggested that amniotic cells have immunosuppressive activity.
Introduction
The amniotic membrane is a tissue of fetal origin and is composed of three major layers: a single epithelial layer, a thick basement membrane, and an avascular mesenchyme. It is adjacent to the trophoblast cells and lines the amniotic cavity in which the fetus can grow without distortion by pressure from surrounding structures. Although the fetus has semiallogeneic antigens recognized by the mother, pregnancy is established normally in most cases. It is thought that a special immunologic mechanism may protect the fetus from maternal aggression. Furthermore, it is reported that a kidney allograft avoided allograft rejection in one case when immunosuppression was discontinued during and after pregnancy (8), which implied that an active immunoregulatory mechanism may be generated. Based on this, there has been increasing interest in the investigation of the immunosuppressive activity of amnion-derived cells in vivo and in vitro (23–25,33,50,53). The human amniotic membrane contains two principal types of cells: amniotic epithelial cells isolated from epithelial layer and amniotic mesenchymal cells isolated from the avascular mesenchymal layer. Emerging evidence suggests that both of them retain proliferation and multipotent/pluripotent characteristics (18,47,49,50,57), making the amniotic membrane a promising and very attractive source of cells for regenerative medicine. The amnion membrane can provide a sufficient cell supply without any substantial ethical issues, increasing the attractiveness of amnion-derived cells for cell transplantation.
CD59, a negative regulator of complement activation, was reported to be expressed in amnion-derived cells (12), indicating that they may possess a mechanism to escape from complement attack by inhibiting formation of membrane attack complex. It has also been shown that amnion-derived cells express the nonpolymorphic, nonclassical human leukocyte antigen G (HLA-G), but lack the polymorphic antigens HLA-A, HLA-B, and HLA-C (class IA), and HLA-DR (class II) (48,49). HLA-G was thought to protect the fetus from maternal uterine NK cells and is considered to be an important immunosuppressive factor during pregnancy (39). These findings suggest that amnion-derived cells may be immunologically inert and would have a reduced risk of rejection upon transplantation. There are many potential cell sources for regenerative medicine, including bone marrow-derived mesenchymal stem cells, tissue-specific progenitor cells, embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs). Although their biological potentials have been demonstrated, all of them still have some infeasibility for clinical applications. Bone marrow-derived mesenchymal stem cells have been shown to ameliorate tissue damage and to improve function after lung injury (28), myocardial infarction (16,17), renal injury (30), and bone injury (15). However, the decrease in cell number depending on the patient's age is one of the limitations (27,37). Tissue-specific progenitor cells are very hard to isolate and grow in vitro. Also, transplantation needs a large cell supply (42). ESCs and iPSCs are considered to be the most promising stem cells because of their tremendous differentiation ability. However, with the pluripotency comes genetic instability, which leads to concerns of tumorigenicity (14,20) and mutation (11). Furthermore, their expansion and maintenance to obtain a therapeutically sufficient number of cells requires time, effort, and cost. As a promising alternative source, use of the amnion membrane can resolve all of these problems. Therefore, human amnion-derived cells have started to be appreciated as an attractive and promising alternative source in the field of regenerative medicine. If amnion-derived cells have immunosuppressive activity, the problem of immune rejection could be overcome by the use of amnion-derived cells.
Materials and Methods
Cell Culture
A human amniotic membrane was peeled mechani cally from the chorion of a placenta obtained from a patient undergoing cesarean section with informed consent. The study and the use of the amnion were approved by the Research Ethics Committee of University of Toyama. Fresh amniotic epithelial cells (fHAEs) and fresh amniotic mesenchymal cells (fHAMs) were isolated from human fresh amniotic membrane by sequential trypsin (Sigma-Aldrich, St. Louis, MO, USA) and collagenase (Wako Pure Chemical Industries, Ltd., Osaka, Japan) digestion as previously described (52). Immortalized human amniotic mesenchymal cells (iHAMs) and immortalized human amniotic epithelial cells (iHAEs) were established previously (47,57). HAMα is a proliferation subpopulation purified from fHAM (female) (32,51). Human umbilical vein endothelial cells (HUVECs, female) were obtained from Lonza (Walkersville, MD, USA). K562 cells (female) were purchased from ATCC (Manassas, VA, USA). fHAM, iHAM, HUVECs, and K562 cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich) supplemented with 10% fetal bovine serum (FBS; PAA Laboratories GmbH, Pasching, Austria), 1% penicillin–streptomycin mixed solution (Nacalai Tesque, Kyoto, Japan), and 1% 200 mmol/L
Cytotoxicity Assay
The cytotoxicity of NK cells was evaluated by the 5,6-carboxyfluorescein diacetate succinimidyl ester (CFSE) dilution method. Briefly, NK cells were plated (1 × 105 cells) in flat-bottomed 12-well tissue culture plates (Greiner Bio-One GmbH) with or without amnion-derived cells or HUVECs with numbers 2.5 × 104, 5 × 104, 1 × 105, 2 × 105, 5 × 105, 1 × 106 cells for 1 day, 3 days, or 5 days. NK cells were collected as effectors in the cytotoxic assays. K562 cells were labeled with 4.5 μM CFSE (Sigma-Aldrich) in 0.01% FBS/Dulbecco's PBS– (Wako Pure Chemical Industries, Ltd.) at room temperature for 8 min and continuously incubated at 37°C for 10 min. Labeled cells were washed three times with 2% FBS/PBS and then were coincubated with NK cells (with NK/K562 ratios 1:2) at 37°C for 4 h. The cytotoxicity of NK cells against K562 cells was assessed by fluorescence-activated cells sorter analysis using FACS Calibur (BD Bioscience, Franklin Lakes, NJ, USA) by quantifying apoptosis of K562 with 3,8-diamino-5-[3-(diethylmethyl-ammonio)propyl]-6-phenyl-phenanthridinium diiodide solution (propidium iodide; Dojindo, Kumamoto, Japan). IL-10 neutralizing antibody (Abcam, Cambridge, UK), indomethacin (Sigma-Aldrich), or neutralizing antibody to thioredoxin (ADF11, a kind gift from Dr. Yoshiyuki Matsuo and Dr. Junji Yodoi) was added to the coculture. Cytotoxicity of NK cells was investigated at 3 days.
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Amnion-derived cells were cultured with NK cells at a ratio of 10:1 for 1 day. Amnion-derived cells and NK cells were separated for RNA isolation with ISOGEN II (Nippon Gene Co., Ltd., Tokyo, Japan). cDNA was synthesized from 1 μg of total RNA using ReverTra Ace® qPCR RT Master Mix (Toyobo, Osaka, Japan). cDNA was subjected to PCR using a Taq PCR Core Kit (QIAGEN, Tokyo, Japan). The primer sequences, annealing temperature, and amplification cycle were as follows: NKp30 (forward: CTCATCTTGATCATGGTCCATCC, reverse: TGAGGATGTTCTTTCTCCACCAC; 57°C; 27 cycles); NKp44 (forward: CTTCAGCACTTGTGTGCATCAG, reverse: TCACGGTTTTCCACCATATGTC; 55°C; 35 cycles); NKp46 (forward: ATCTGAGCGATGTCTTCC ACACTC, reverse: AGACCAGGCATGGTTGTTATA GGAG; 59°C; 40 cycles); NKG2D (forward: TGAG AGTAAAAACTGGTATGAGAGCCA, reverse: GCAT GCAGATGTATGTATTTGGAG; 57°C; 38 cycles); CD69 (forward: CATAGCTCTCATTGCCTTATCAGT, reverse: CCTCTCTACCTGCGTATCGTTT; 60°C; 30 cycles); IFN-γ (forward: TGCAGGTCATTCAGATGTAG, reverse: AGCCATCACTTGGATGAGTT; 59°C; 30 cycles); IL-4 (forward: GCGATATCACCTTACAGGAG, reverse: TTGG CTTCCTTCACAGGACA; 50°C; 40 cycles); IL-6 (forward: AAATTCGGTACATCCTCGAC, reverse: CAGG AACTGGATCAGGACTT; 50°C; 34 cycles); IL-8 (forward: CTTGGCAGCCTTCCTGATTT, reverse: CTCAG CCCTCTTCAAAAACT; 51°C; 34 cycles); IL-10 (forward: AGATCTCCGAGATGCCTTCA, reverse: TTTCGTATC TTCATTGTCATGTA; 50°C; 40 cycles); HLA-G (forward: TATGCCTACGATGGCAAGG, reverse: CTACA GCTGCAAGGACAACC; 50°C; 22 cycles); COX-2 (forward: AACAGGAGCATCCTGAATGG, reverse: GGTC AATGGAAGCCTGTGAT; 50°C; 32 cycles); TGF-β (forward: CAAGTGGACATCAACGGGTT, reverse: GCTC CAAATGTAGGGGCAGG; 62°C; 26 cycles); GAPDH (forward: GTCAACGGATTTGGTCGTATTG, reverse: CATGGGTGG AATCATATTGGAA; 57°C; 30 cycles).
Enzyme-Linked Immunosorbent Assay (ELISA)
NK cells were cultured with amnion-derived cells at a 1:10 ratio for 3 days. The supernatant was collected to detect the secretion of IL-10 or prostaglandin E2 (PGE2) by enzyme-linked immunosorbent kit (eBioscience, San Diego, CA, USA and Cayman Chemical Company, Ann Arbor, MI, USA) according to the manufacturers’ instructions.
Monocyte Assay
PBMCs were plated (1 × 105 cells) in flat-bottomed six-well tissue culture plates (Greiner Bio-One GmbH) in the presence of amnion-derived cells (1 × 106 cells) with lipopolysaccharides (LPS, 0.01 μg/ml; Sigma-Aldrich) as a stimulator for 4 h (9). Brefeldin A (5 μg/ml; Sigma-Aldrich) was added to stop the reaction among PBMCs, LPS, and amnion-derived cells, and to inhibit the protein secretion. After 4-h inhibition, PBMCs were collected to be analyzed for the expression of TNF-α (APC anti-human TNF-α; BioLegend, San Diego, CA, USA) and IL-6 (APC anti-human IL-6; BioLegend) on monocytes labeled by CD14 (CD14IOM2; Beckman Coulter, Brea, CA, USA) by fluorescence-activated cells sorter analysis with BD FACS CantoTMII (BD Bioscience, Franklin Lakes, NJ, USA).
Statistical Analysis
The Student's
Results
Amnion-Derived Cell-Mediated Inhibition of NK Cytotoxic Activity
To assess the immunosuppressive activity of amnion-derived cells, NK cytotoxic activity was analyzed by targeting K562 cells after coculturing for 1, 3, or 5 days with or without HUVECs (Fig. 1A) (35), iHAM cells (Fig. 1B), iHAE cells (Fig. 1C), or HAMα cells (Fig. 1D). iHAM, iHAE, and HAMα cells inhibited NK cytotoxic activity with an amnion-derived cell/NK cell ratio of 10:1 at day 3, declining with lower amnion-derived cell content, whereas in Figure 1A, HUVECs did not inhibit NK cytotoxic activity. However, the NK cytotoxic activity was little decreased at day 1. The NK cytotoxic activity decreased artificially at day 5. This may have been because of damaged NK activity through coculturing without changing the media for a long time. fHAM cells suppressed NK cytotoxicity significantly, and fHAE cells also had the tendency to suppress NK cytotoxicity (data not shown). Furthermore, the suppression of iHAM, fHAM, and HAMα to NK cytotoxicity was much more than iHAE and fHAE. In summary, amnion-derived cells suppressed NK cytotoxic activity to K562 cells in a dose-dependent manner.

Amnion-derived cell-mediated inhibition of NK cytotoxic activity. NK cytotoxic activity was analyzed after coculturing for 1, 3, or 5 days with or without HUVECs (A) or iHAM (B), iHAE (C), HAMα (D) at an effector/target ratio of 1:10. The cytotoxic activity of NK cells cultured with HUVECs or amnion-derived cells was expressed as a percentage of K562 dead cells with respect to NK cells alone (100%). The results are representative of three independent experiments and are shown as the means ± SE.
Inhibition by Amnion-Derived Cells to the Expression of Activated NK Receptors and NK Cell Function
NK cell activation depends on the balance between activating and inhibitory signals mediated by cell surface receptors (34). To further analyze the inhibition of NK cytotoxic activity by amnion-derived cells, the expression of NK-activated receptors NKp46, NKp30, NKp44, NKG2D, and CD69, which are involved in the NK cell activation and target cell killing (7,22,56), was analyzed by RT-PCR. Significant downmodulation of NK receptors NKp30, NKp44, NKp46, and CD69 after coculturing with amnion-derived cells was observed (Fig. 2). The expression of NKG2D was partly decreased after coculturing with amnion-derived cells. The expression of NK-activated receptors also was decreased in NK cells after coculturing with fHAM, fHAE, and HAMα (data not shown). These data suggested that the amnion-derived cell-mediated inhibition of NK cytotoxic activity correlated with downmodulation of NK-activated receptors. Together with cytotoxic activity, cytokine production is another main NK cell function. To better characterize the inhibitory effect exerted by amnion-derived cells, IFN-γ production by NK cells after coculturing was analyzed by RT-PCR (10). The expression of IFN-γ in NK cells significantly decreased after stimulation with iHAM and iHAE cells. Also, the expression of IFN-γ in NK cells decreased after stimulation with fHAM, fHAE, and HAMα (data not shown). These data indicated that amnion-derived cells can inhibit not only NK cytotoxic activity but also cytokine production. IFN-γ, as a proinflammatory, was involved in innate immunity and can trigger immunosuppressive activity (2,21,44). The decrease in production of IFN-γ in NK cells further confirmed that amnion-derived cells have immunosuppressive activity.
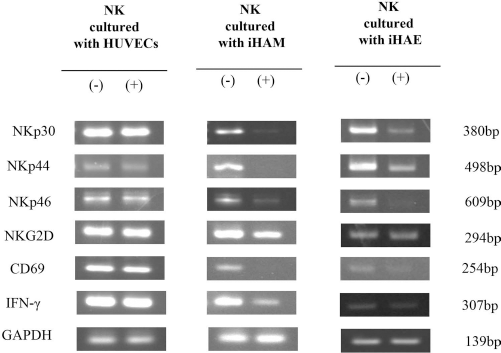
Expression of activated NK cell receptors and cytokine. The expression of activated receptors (NKp30, NKp44, NKp46, NKG2D, and CD69) and cytokine (IFN-γ) in NK cells cultured with or without HUVECs (as control) or amnion-derived cells for 1 day was investigated by RT-PCR. GAPDH was used as a loading control.
Increase in the Expression of Anti-inflammatory Factors in Amnion-Derived Cells by Interaction with NK Cells
To further investigate the factors involved in this immunosuppressive activity, the expression of immunosuppressive factors in amnion-derived cells was analyzed. Amnion-derived cells were harvested, and RNA was isolated for RT-PCR after coculturing with NK cells for 1 day. The expression level of IL-4, IL-6, IL-8, HLA-G, and TGF-β was not changed, while the expression of IL-10 and COX-2 was remarkably increased after coculturing with NK cells (Fig. 3). The expression of IL-10 and PGE2 also was increased in fHAM and fHAE cells after coculturing with NK cells (data not shown).

Expression of immunosuppressive factors. The expression of immunosuppressive factors IL-4, IL-6, IL-8, IL-10, HLA-G, COX-2, and TGF-β in amnion-derived cells cultured with or without NK cells for 1 day was investigated. GAPDH was used as a loading control.
The Detection of IL-10 and PGE2 by ELISA
The secretion of IL-10 and PGE2 protein from amnion-derived cells was analyzed by ELISA. The secretion of IL-10 in the supernatant from amnion-derived cells cultured without NK cells was very low but significantly increased after coculturing with NK cells (Fig. 4A). The secretion of PGE2 in fHAE cells and HAMα cells cocultured with NK cells was significantly increased compared to that without NK cells (Fig. 4B). The production of PGE2 from iHAM, iHAE, and fHAM cells after coculturing with NK cells had the tendency to increase. Moreover, the amount of PGE2 in iHAM, fHAM, and HAMα cells is much higher than that in iHAE or fHAE cells. This result is consistent with the result of the NK cytotoxicity assay. IL-10 and PGE2 may be involved in the immunosuppressive activity of amnion-derived cells.

Detection of IL-10 and PGE2 by ELISA. The production of IL-10 (A) or PGE2 (B) by amnion-derived cells cultured with or without NK cells for 3 days was investigated by ELISA. Statistically significant difference between amnion-derived cells cultured alone and cocultured with NK cells: *
Blocking of IL-10, PGE2, and Thioredoxin with Specific Inhibitors
The addition of anti-IL-10 antibody to the amnion-derived cells and NK cell coculture had the tendency to increase the NK cytotoxicity that had been decreased by coculturing with amnion-derived cells (Fig. 5A). The addition of indomethacin, a single inhibitor specific to PGE2, blocked the suppression to NK cells from interaction with amnion-derived cells (Fig. 5B). The neutralizing antibody to thioredoxin also had a tendency to restore the decreased NK cytotoxicity by amnion-derived cells (data not shown).
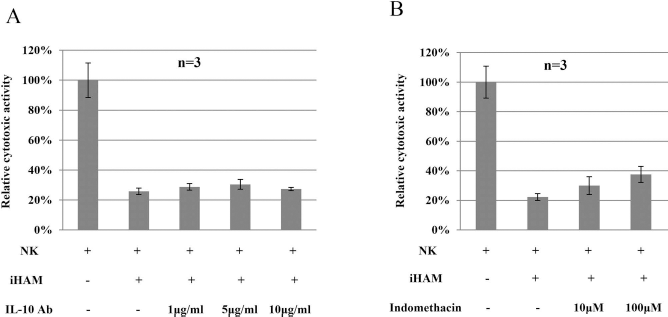
The block of IL-10 and PGE2 with specific inhibitors. IL-10 neutralizing antibody (A) was added at concentrations of 1 μg/ml, 5 μg/ml, or 10 μg/ml, and indomethacin (B) was added at concentrations of 10 μM or 100 μM to the NK and iHAM coculture. Cytotoxicity of NK cells was investigated at day 3.
Recovery of NK Cytotoxic Activity
To confirm the immunosuppressive activity of amnion-derived cells, a recovery assay was performed. After interaction with amnion-derived cells for 3 days, NK cells were cultured continuously in the absence of amnion-derived cells for 2 days prior to exposure to K562 cells (35). Compared to inhibition of 3-day coculture of amnion-derived cells and NK cells, extending the incubation without amnion-derived cells significantly restored NK cytotoxic activity (Fig. 6). Therefore, the decreased NK cytotoxic activity after interaction with amnion-derived cells was not because NK cells were damaged by coculturing. Amnion-derived cells suppressed NK cytotoxic activity.

Recovery of NK cytotoxic activity. FACS analysis of NK cytotoxic activity was performed after culturing with iHAM (A), iHAE (B), or HAMα (C) for 3 days and without amnion-derived cells for 2 days. The cytotoxic activity of NK cells was expressed with respect to NK cells alone (100%). Statistically significant difference between 3-day inhibition assay and 3+2-day recovery assay: *
Amnion-Derived Cell-Mediated Inhibition of Cytokine Production in Monocytes
As immune system cells, monocytes play an important role in immunomodulation. To further confirm the immunosuppressive activity of amnion-derived cells, the inhibition of monocyte cytokine production mediated by amnion-derived cells was also investigated. The expression level of TNF-α in monocytes after coculturing with iHAM, iHAE, and HAMα cells was significantly decreased (Fig. 7A). The expression level of IL-6 after coculturing with iHAM and HAMα cells was significantly decreased (Fig. 7B). The expression of IL-6 after coculturing with iHAE cells also had tendency to decrease. Interaction with amnion-derived cells decreased the production of cytokine TNF-α and IL-6 in LPS-stimulated monocytes. The data showed that amnion-derived cells suppressed the cytokine production of monocytes by coculturing. This result suggested that amnion-derived cells have immunosuppressive activity.

Amnion-derived cell-mediated inhibition of cytokine production in monocytes. PBMCs from different donors were plated in the presence of amnion-derived cells with LPS. Four hours later, brefeldin A was added to stop the reaction. The secretion of TNF-α (A) and IL-6 (B) on monocytes cultured with or without iHAM, iHAE, or HAMα cells was analyzed by FACS. The secretion of TNF-α and IL-6 was expressed with respect to monocytes alone (100%). Statistically significant difference of TNF-α and IL-6 on monocytes with or without amnion-derived cells: *
Discussion
Amnion-derived cells have been demonstrated as a promising alternative source, for their immunosuppressive activity (23–25,33,50,53). Although the mechanism underlying the immune modulation of amnion-derived cells remains to be elucidated, it is now clear that these cells suppress allogeneic immune responses. Several studies have recently shown that amniotic membrane-derived cells display immunomodulatory properties similar to those described for mesenchymal cells obtained from other sources such as bone marrow and adipose tissue (5,23,50,53). Similar to other MSCs, the mechanism of immune modulation by amnion-derived cells was yet to be clarified. Here we reported a more accurate analysis of amnion-derived cell-induced inhibition of NK cell function and defined the molecular basis of their inhibitory effect (4). First, we showed that amnion-derived cells sharply inhibited the NK-mediated cytotoxic activity. Inhibition of expression of NKp30, NKp44, NKp46, NKG2D, and CD69, which are involved in the NK cytotoxicity to K562 cells, occurred in NK cells cultured with amnion-derived cells. The expression of IL-10 and COX-2 in human amnion-derived stem cells was increased after culturing with NK cells. In addition, the secretion of IL-10 and PGE2 in the supernatant from NK and amnion-derived cell coculture was increased. This observation is particularly evident in the inhibition of NK cytotoxic activity by using neutralizing IL-10 antibody or single inhibitor specific to PGE2 or neutralizing thioredoxin antibody. The decreased NK cytotoxicity by interaction with amnion-derived cells was recovered by continuous culturing in the absence of human amnion-derived cells; this means that the decreased NK cytotoxicity is recoverable, implying that the decreased NK cytotoxicity is not because the NK cells were damaged by coculturing. Moreover, amnion-derived cells also suppressed the activity of cytokine production in monocytes analyzed by TNF-α and IL-6.
In our study, amnion-derived cells exerted an inhibitory effect on NK cell and monocyte activation and function. These data showed that amnion-derived cells have immunosuppressive activity and that IL-10 and PGE2 may be involved in the immunosuppressive activity of amnion-derived cells.
IL-10 is a well-known cytokine involved in cell regulation and promotion of proliferation and activation of regulatory cells or anti-inflammatory cells. Previous studies have demonstrated that IL-10 production was increased in a mixed lymphocyte population cocultured with human bone marrow-derived MSCs (hBM-MSCs) (1,3,19,21,38) and in plasmacytoid dendritic cells (DCs) cocultured with BM-MSCs and human adipose tissue-derived MSCs (hAD-MSCs) (55). However, other groups found no change in IL-10 production when hBM-MSCs and PBMCs were cocultured, or IL-10 was not detected in the anti-inflammatory of AD-MSCs (26,36). Some previous studies also investigated induction of IL-10 production by umbilical cord lining-derived cells (6). Our data showed increased levels of IL-10 in the amnion-derived cells and supernatant from coculture of amnion-derived cells and NK cells. Our results showed that the addition of IL-10 antibody in the NK cells and amnion-derived cell coculture has the tendency to recover the decreased NK cytotoxicity. So, it is considered that IL-10 was involved in the function of amnion-derived cells to NK cells.
PGE2, one of the immune modulation candidates synthesized from arachidonic acid by COX-1 and COX-2 enzymes, regulates the maturation and antigen presentation of DC and inhibits T-cell proliferation and cytokine production (54). According to Aggarwar and Pittenger (1), the PGE2 production pattern is bell shaped and occurs in a time-dependent manner, indicating that this factor might be related to early immune suppression by MSCs. Our results also showed that COX-2 mRNA and PGE2 production increased in amnion-derived cells when they were cocultured with NK cells. Some previous studies showed that PGE2 is involved in the immunosuppression of MSC to NK cell cytotoxicity (10,45). Also, it is reported that culture with mixed lymphocyte reaction (MLR) increases the release of PGE2 in umbilical cord tissue-derived cells (13). Moreover, blocking PGE2 production in the NK cell/MSC coculture counteracted the decreased NK cytotoxicity (10), and blocking PGE2 production fully blocked it in the umbilical cord tissue-derived mesenchymal stem cell/MLR coculture (13). Our results showed that blocking PGE2 partly recovered the decreased NK cytotoxicity. It suggested that PGE2 may play an important role in the immunosuppressive activity of amnion-derived cells to NK cells. Therefore, the immunomodulation of amnion-derived cells is a complicated procedure, which involves many factors (31), among which IL-10 and PGE2 play important roles (40). Further studies will be necessary to determine the detailed mechanism of the immunomodulation.
iHAM, fHAM, and HAMα cells exhibited more inhibition to NK cytotoxicity than iHAE and fHAE, consistent with more decreased expression in activated NK receptors and IFN-γ and more significant increase in production of IL-10 and COX-2, suggesting that amnion-derived mesenchymal cells exerted more effective immunosuppressive properties than amnion-derived epithelial cells. Monocyte assay indicated that amnion-derived mesenchymal cells exerted more impairment to monocyte response than amnion-derived epithelial cells. The results suggested that the capacity of immunosuppressive activity may depend on the origin of the cells. HAMα indicated the most immunosuppression on NK and on monocyte and the most secretion of IL-10 and PGE2 after coculturing with NK cells in amnion-derived mesenchymal cells. HAMα is a proliferative subpopulation from fHAM and has shown stem cell characteristics and differentiation ability. HAMα can be considered as the most promising potential cell source for cell-based therapy. Although there is a small difference of immunosuppressive activity among mesenchymal, epithelial, and subpopulations of mesenchymal cells, the amnion-derived cells overall have immunosuppressive activity and would be a good source for cell therapy.
In conclusion, immunosuppressive activity of amnion-derived cells on NK cells and monocytes was demonstrated. Soluble factors IL-10 and PGE2 produced by amnion- derived cells may suppress allogeneic immune responses. These findings support the hypothesis that these cells have potential therapeutic use. Further study is needed to identify the detailed mechanisms responsible for the immunomodulatory effects of amnion-derived cells. Amnion-derived cells will be transplanted into a mouse model for further analysis of immunosuppressive activity or anti-inflammatory effects.
Footnotes
Acknowledgments
