Abstract
Human amnion epithelial cells (hAECs) exert potent antifibrotic and anti-inflammatory effects when transplanted into preclinical models of tissue fibrosis. These effects are mediated in part via the secretion of soluble factors by hAECs which modulate signaling pathways and affect cell types involved in inflammation and fibrosis. Based on these reports, we hypothesized that these soluble factors may also support liver regeneration during chronic liver injury. To test this, we characterized the effect of both hAECs and hAEC-conditioned medium (CM) on liver repair in a mouse model of carbon tetrachloride (CCl4)-induced fibrosis. Liver repair was assessed by liver fibrosis, hepatocyte proliferation, and the liver progenitor cell (LPC) response. We found that the administration of hAECs or hAEC-CM reduced liver injury and fibrosis, sustained hepatocyte proliferation, and reduced LPC numbers during chronic liver injury. Additionally, we undertook in vitro studies to document both the cell–cell and paracrine-mediated effects of hAECs on LPCs by investigating the effects of co-culturing the LPCs and hAECs and hAEC-CM on LPCs. We found little change in LPCs co-cultured with hAECs. In contrast, hAEC-CM enhances LPC proliferation and differentiation. These findings suggest that paracrine factors secreted by hAECs enhance liver repair by reducing fibrosis while promoting regeneration during chronic liver injury.
Introduction
Chronic liver diseases such as viral hepatitis, alcoholic liver disease, and nonalcoholic steatohepatitis are associated with persistent hepatic inflammation and fibrosis, which over time, may develop into liver cirrhosis. For patients with cirrhosis, treatment options are severely limited, and a liver transplant remains the only cure for end-stage disease. Liver transplants are complex, costly, and limited by the availability of donor organs. Therefore, there is an urgent need for antifibrotic therapies for patients with chronic liver disease 1 . One such alternative that has demonstrated promise in preclinical models is placental-derived human amnion epithelial cell (hAEC) therapy 2 .
The hAECs are a readily available, nontumorigenic, and immunogenically privileged cell type that possess potent anti-inflammatory and antifibrotic properties 3 –6 . They can attenuate inflammation and reduce fibrosis when transplanted in models of liver, lung, cardiac, epidermal, or neurological injury 3,6 –9 . These therapeutic effects are mediated in part by the secretion of soluble factors that modulate pathways and affect cells involved in inflammation and fibrosis 10 –12 . Importantly, the beneficial effects of hAECs have been demonstrated in models of chronic injury where hAECs or their secretome were administered after inflammation and fibrosis were established 5,6,12 . Furthermore, hAEC therapy has been reported to improve tissue function independent of cell engraftment suggesting that they promote tissue repair and regeneration 5,6,8 . However, the mechanisms underlying the therapeutic efficacy of hAECs, particularly how they affect liver regeneration, are not fully understood.
Liver regeneration occurs through two different pathways. Typically, hepatocytes are restored through self-replication; however, when hepatic injury progresses to chronicity, the regenerative capacity of hepatocytes is impaired 13 . In this setting, the liver progenitor cell (LPC) compartment is activated to augment liver repopulation. LPCs are a population of bipotential epithelial cells that emerge from the bile canaliculi and proliferate and differentiate into hepatocytes and/or cholangiocytes, the two important functional cell types of the liver parenchyma 14 . This response is activated during persistent liver injury especially where there are chronic inflammation and fibrosis 15 . Accordingly, both of these regenerative pathways need to be examined to fully understand the effect of hAEC therapies on liver regeneration during chronic liver injury.
hAECs may modulate liver regeneration through a number of possible mechanisms including cell–cell mediated responses and the secretion of paracrine factors by hAECs. Additionally, both mechanisms may involve direct interaction among hAECs, LPCs, and hepatocytes, as well as changes to inflammatory cells that have direct effects on LPCs 16 and hepatic stellate cells that produce collagen which underpins the fibrotic response 17 . We report data from experiments designed to better understand the interplay between the liver cell types, identify potential mediators, and show the signaling pathways involved.
We propose that hAECs act by direct cell–cell interaction as well as through secreted cytokines and chemokines; not only on hepatocytes but also LPCs. To test our hypothesis, we characterized the effect of both hAEC and hAEC-conditioned medium (CM) on liver repair in a mouse model of carbon tetrachloride (CCl4)-induced fibrosis. Importantly, previous studies show hAECs and hAEC-CM exert anti-inflammatory and antifibrotic effects in this model 6,12 . Given that in vivo studies cannot distinguish between an effect of hAECs on LPCs that is mediated through modifying the inflammatory response and direct interaction between the two cell types, we thus undertook in vitro studies to document the effect of hAECs on an established LPC line 18 .
Materials and Methods
hAEC Isolation
Amnion membranes were collected from healthy women with a normal singleton pregnancy undergoing cesarean section at term. The Monash Health Human Research Ethics Committee approved the collection and use of human amnion (Monash Health HREC approval numbers: 01067B, 12223B). Informed written consent was obtained from each patient prior to surgery. Isolation of hAECs was performed as previously described 19,20 . Viable hAECs were sorted by flow cytometry with cytokeratin 7 and 8/18 (Dako, Glostrup, Denmark) used to assess cell purity. Only batches with cells >98% positive for cytokeratins with typical cobblestone morphology in culture were used. Cells were frozen in fetal bovine serum (FBS; Gibco, Thermo Fisher Scientific, Australia) with 5% dimethyl sulfoxide (DMSO; Sigma Aldrich, Saint Louis, MO, USA) and later assessed for viability, mycoplasma, bacterial, and fungal contamination.
hAEC-CM Production
hAEC-CM was produced as previously described 10 . Briefly, cryopreserved primary hAECs from four to five randomly selected donors were thawed, pooled to equal ratios, and seeded at a density of 1 × 107 cells in a 175 cm2 flask coated with rat tail collagen IV (1 mg/ml; Roche, Mannheim, Germany). hAEC cultures were grown in Dulbecco’s modified Eagle medium (DMEM)/F12 supplemented with 10% FBS and 10 ng/ml epidermal growth factor (EGF; Gibco). Once 90% confluence was achieved, cells were serum starved for 72 h in DMEM/F12, the medium collected, spun down, passed through a 0.2 µm filter (Sartorius, Gottingen, Germany), and then stored at −80°C until needed.
Liver Fibrosis Model
Approval to undertake animal experiments was obtained from the Monash University Animal Ethics Committee (approval number: MMCB 2013-32). Mice were purchased from Monash Animal Services, Melbourne, Australia. Six-week-old male C57/BL6 mice were given twice weekly intraperitoneal injections with 1 µl/g body weight CCl4 (Merck, Darmstadt, Germany) mixed with olive oil 1:10 as previously described for a total for 12 weeks 21 . Following 8 weeks of CCl4 treatment, animals received either a single dose of 2 × 106 hAEC in 200 µl of normal saline or hAEC-CM in 350 µl aliquots, via tail vein, three times weekly until the end of the study (Supplemental Fig. 1). Animals that were administered hAECs received an even mixture consisting of five different hAEC donors. Prior to their administration, hAECs were thawed to room temperature, pooled at equal ratios, and washed in sterile phosphate-buffered saline. The suspension was then centrifuged and resuspended to 2 × 106 hAECs per 200 µl of normal saline. hAEC-CM was also thawed and preloaded into 1 ml syringes.
hAEC-CM was administered thrice weekly to model the effect of soluble factors being continually produced by the injected cells. Control groups consisted of CCl4 only, DMEM/F12 (three times weekly for 4 weeks beginning at week 8), and no treatment. DMEM/F12 (with no FBS or EGF) was used as a control for hAEC-CM-treated animals as this was the base medium for hAEC-CM production. Animals were culled 4 weeks post-treatment, with blood and liver tissue harvested. Serum biochemistry was performed by Monash Health Pathology.
Immunohistochemistry
Liver tissue was fixed in 10% neutral buffered formalin (NBF; Australian Biostain, Victoria, Australia) and embedded in paraffin. Sections were cut at 4 µM, dewaxed, and then antigen retrieval performed. Primary antibodies consisted of mouse antimouse alpha-smooth muscle actin (α-SMA; Sigma Aldrich), rabbit antimouse Ki67 (Abcam), Wide Spectrum Screening Cytokeratin (Pan-CK; Agilent, Victoria, Australia) and A6 (a gift from Dr Valentina Factor). Antigen retrieval for α-SMA and Ki67 consisted of heat-induced epitope retrieval. Briefly, sections were submerged in 0.1 M tri-sodium citrate heated to 100°C for 20 min and then cooled for 20 min. For Pan-CK and A6, sections were treated for 10 min with Proteinase K (Agilent). Endogenous peroxidases were then neutralized with 1%–3% hydrogen peroxide for 10 min, followed by the application of a blocking solution for 30 min. CAS protein block was used for α-SMA staining (Life Technologies, Camarillo, CA, USA), while the Dako Serum-free protein block (Agilent, Santa Clara, CA, USA) was used for Ki-67, Pan-CK, and A6 staining. Next, primary antibodies diluted in Dako Antibody Diluent (Agilent) were applied overnight at 4°C. To visualize α-SMA staining, the VectaStain Elite ABC Kit (Vector Laboratories, Burlingame, CA, USA) was used, while for Ki67, A6, and Pan-CK, staining was visualized by the Universal LSAB + Kit/HRP (Agilent). Finally, slides were counterstained with hematoxylin. Staining was quantitated as follows: α-SMA by manual scoring, at 200× magnification for the number of positive cells over five nonoverlapping fields of view. For Ki67, Pan-CK, and A6, slides were scanned at 40× using the Aperio ScanScope digital slide scanner (Aperio Technologies, Vista, CA, USA), and 15 random nonoverlapping fields of view captured per section. Positive cells were quantified using Inform software (Perkin Elmer) as described previously 22 . Importantly, this algorithm excluded cholangiocytes that lined the bile ducts.
Immunofluorescence
Tissue sections (4 µM thick) were dewaxed, and then heat-induced epitope retrieval performed. Sections were blocked with the Dako Serum-free protein block (Agilent) for 30 min. Next, F4/80 primary antibody diluted in Dako Antibody Diluent (Agilent) was applied overnight at 4°C. The following day, sections were incubated in Alexa-Fluor-594 goat-antirat IgG (Invitrogen, A11007) diluted 1:400 in Tris-buffered saline for 1 h at room temperature. Sections were stained with Hoechst (Sigma, Cat 14533) for 10 min at room temperature and mounted with Gelvatol medium. Fluorescence was imaged, and the number of positive cells quantified using the Cellinsight CX 7 High-Content Screening Platform (Thermo Fisher Scientific).
Morphometric Analysis of Fibrosis
Liver fibrosis was quantified by determining the area of collagen stained by Sirius Red relative to the total area as previously described 21 . Briefly, sections were incubated for 90 min in Picro Sirius Red (Direct Red 80, 0.1% wt/vol in saturated picric acid; Sigma Aldrich) and washed in acetic acid:water (1:200). Fifteen consecutive nonoverlapping fields were acquired from each mouse liver using a Leica DMLB microscope (Leica Microsystems, Ltd, Heerbrugg, Switzerland) at 200× magnification. Images were digitized, and the fibrosis area was measured by computer-assisted morphometry using Scion Image for Windows (Version Alpha 4.0.3.2, Scion Corporation, Frederick, MD, USA).
Derivation of Murine Liver Progenitor Cell Line
The LPC line used for the in vitro experiments was derived from the liver of day 14 TAT-GRE lacZ transgenic mouse embryos by the “plate and wait” method described by Strick-Marchand 23 . This method selects LPCs that undergo spontaneous immortalization and form large colonies following extended culture. Colonies were then selected, passaged, and expanded. These LPC lines have been characterized previously by bipotentiality, morphology, proliferative capacity, and expression of LPC markers 23 –25 .
LPC Co-Culture with hAECs and hAEC-CM Treatment
LPCs and hAECs were co-cultured in six-well plates at a 1:1 ratio with a total of 100,000 cells seeded per well. A culture consisting of only LPCs served as a control. These cultures were maintained in DMEM/F12 supplemented with 30 ng/ml insulin growth factor-II (IGF-II), 50 ng/ml EGF, 10 μg/ml insulin, 100 U/ml penicillin and streptomycin, and 10% FBS. For hAEC-CM treatment, LPCs were seeded in tissue culture plates (six, 24, and 96 well) at a density of 8.3 × 103 cells/cm2 in Roswell Park Memorial Institute Medium (RPMI) 1640 medium GlutaMAX (Gibco) with 10% FBS, 30 ng/ml IGF-II (Sino Biological Inc., Beijing, China), 50 ng/ml EGF, and 10 μg/ml insulin (Sigma Aldrich). Medium was changed 3 days later to control medium, hAEC-CM, or control medium consisted of RPMI GlutaMax, 50% Dulbecco’s phosphate-buffered saline (Gibco) containing 5% FBS, 30 ng/ml IGF-II, 50 ng/ml EGF, 10 μg/ml insulin, and 100 U/ml penicillin and streptomycin (Gibco); hAEC-CM was RPMI GlutaMax, 50% hAEC-CM, 5% FBS, 30 ng/ml IGF-II, 50 ng/ml EGF, 10 μg/ml insulin, and 100U/ml penicillin and streptomycin. Cultures were maintained at 37°C in 95% humidity and 5% carbon dioxide with the medium changed every 3 days.
Proliferation
LPC proliferation was assessed using the colorimetric bromodeoxyuridine (BrdU) ELISA (Roche) according to the manufacturer’s instructions. LPC cultures treated with hAEC-CM or control medium for 3 and 10 days were incubated with 10 μM BrdU labeling medium for 5.5 h. The absorbance at 450 nm measured using a microplate reader (Tecan, Männedorf, Switzerland).
Real-Time PCR
To quantify gene expression levels in cultured LPCs, RNA was isolated using the Qiagen RNeasy mini kit (Qiagen Pty Ltd, Hilden, Germany) according to the manufacturer’s instructions. Following elution, the RNA concentration was determined using the Nanodrop ND-100 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA), and 1 µg RNA was used to generate cDNA, as per manufacturer’s instructions, using the High-Capacity cDNA Reverse Transcription Kit (Life Technologies, Foster City, CA, USA). Gene expression was assessed at the level of mRNA using Power Sybr Green (Life Technologies) on the Rotor Gene 3000 light cycler (Qiagen Pty Ltd, Sydney, Australia) using the following cycle conditions: 95°C for 10 min, followed by 40 cycles of 95°C for 15 s and 60°C for 60 s. Following 8 days of treatment, expression of alpha-fetoprotein (AFP) and albumin was measured. For co-culture experiments, changes in gene expression were normalized using a mouse-specific glyceraldehyde 3-phosphate dehydrogenase primer set, while for hAEC-CM experiments 18S ribosomal RNA was used. Primers were purchased from MicroMon (Victoria, Australia); for details see Table S1.
Generation and Analysis of Microarray Data
Gene expression profiles of LPCs treated for 16 days with hAEC-CM and control medium were generated using the Agilent SurePrint G3 Mouse Gene Expression 8 × 60 K slides and the GeneSpring 10 program (Agilent, Santa Clara, CA, USA). Array files were normalized against the signal of the array from control (untreated) LPCs, using quantile normalization such that each chip has the same intensity distribution. To detect differentially expressed genes, a significance analysis was applied where a
The dataset of differentially expressed genes was uploaded to the Database for Annotation, Visualization and Integrated Discovery (DAVID) for the Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) pathway analysis. DAVID used an EASE score (a modified Fisher’s exact test) to calculate
Assessment of Hepatocyte Function
Urea synthesis
Cell culture supernatants from day 17 cultures were analyzed by Southern Cross Pathology (Melbourne, Australia) for urea content. These were normalized to total protein, and the urea content in medium subtracted from the sample values.
X-Gal staining
X-Gal staining was performed on day 8 cultures using a previously published protocol 27 . Staining was visualized and recorded at 20× using a Nikon Eclipse TS100 inverted microscope (Nikon Corporation, Tokyo, Japan). Images captured under bright field and phase contrast were overlayed using ImageJ.
β-galactosidase
β-galactosidase (β-gal) activity was measured on day 17 and day 24 cultures using the Mammalian β-gal Assay Kit (Pierce Biotechnology, Rockford, IL, USA) both as an on-plate assay according to the manufacturer’s instructions and also following cell harvest with both normalized against cell number. Absorbance was measured at 409 nm on the Tecan microplate reader (Tecan).
Periodic acid Schiff
Day 17 cultures were stained for glycogen using periodic acid–Schiff. Cells in 24-well plates were fixed with NBF for 10 min, covered with 0.5% periodic acid solution for 5 min, incubated with Schiff’s Reagent DeTomasi (HD Scientific Supplies, NSW, Australia) for 15 min, and counterstained with Hoechst dye 33342 (Molecular Probes, Eugene, Oregon, USA). Images of both stained and unstained cells were captured using the Olympus IX71 inverted microscope.
Statistical Analysis
Experiments were repeated a minimum of three times. Murine studies were conducted with 4–7 animals in each group. Statistical analysis was performed using GraphPad Prism 6.0e (GraphPad Software, La Jolla, California, USA). Statistical significance was determined using the unpaired
Results
hAECs and hAEC-CM Sustain Hepatocyte Regeneration and Reduce LPC Numbers During Chronic Liver Injury
Mice exposed to CCl4 for 12 weeks showed significantly increased numbers of Ki67+ hepatocytes (Fig. 1A). The administration of hAECs, control medium, or hAEC-CM did not significantly alter the number of Ki67+ hepatocytes when compared with the CCl4 only group. Furthermore, there was no significant difference between the control medium and hAEC-CM groups. Next, we determined the number of LPCs using the markers Pan-CK and A6. CCl4 treatment significantly increased the number of Pan-CK+ and A6+ LPCs (Fig. 1B, C). Compared with the mice given CCl4 only, the administration of hAECs reduced A6+ and Pan-CK+ LPC by twofold and threefold, respectively (

Effects of hAEC and hAEC-CM on hepatocyte proliferation and LPC numbers in vivo. (A) The average number of Ki67+ hepatocytes per field of view. Ki67+ hepatocytes were distinguished by their large size and high levels of DAB-positive nuclei. CCl4 treatment resulted in a significant increase in the number of Ki67+ hepatocytes which was not significantly changed by the administration of hAEC, hAEC-CM, or control medium. (B and C) The average number of A6+ and Pan-CK+ LPCs per field. LPCs were distinguished from ductal cells by morphological differences in hematoxylin and DAB staining. All three treatment cohorts had a significantly reduced LPC response compared with the CCl4 only group. **
hAEC-CM Reduces Hepatocellular Injury
Treating mice with CCl4 for 12 weeks resulted in a 26.8-fold (

hAEC-CM reduces serum ALT and AST during chronic liver injury. (A) Quantification of serum ALT showed a significant increase following 12 weeks of CCl4 treatment. The administration of hAEC-CM significantly decreased ALT levels when compared with control and DMEM/F12 cohorts. (B) Serum AST level significantly increased following 12 weeks of CCl4 administration. Treatment with hAEC-CM significantly reduced serum AST relative to the control and DMEM/F12 cohorts. *
hAEC-CM Reduce Liver Fibrosis and Macrophage Numbers
Liver fibrosis area (LFA) was assessed by the extent of Picrosirius red staining as a percentage of liver tissue and the number of αSMA+ cells per field. LFA was increased by 4.5-fold (

Antifibrotic and anti-inflammatory effects of hAEC and hAEC-CM in vivo. (A) Quantification of Picrosirius red staining of collagen. Mice with established fibrosis induced by CCl4 were given hAEC or hAEC-CM. hAEC and hAEC-CM treated mice had a significantly reduced fibrosis area. (B) The average number of α-SMA positive HSCs per field of view. Treatment with hAEC-CM resulted in fewer activated HSC. (C) The average number of F4/80 positive macrophages per field of view. hAEC-CM significantly reduced the number of hepatic macrophages. *
The number of hepatic macrophages was assessed by the number of F4/80+ cells per field (Fig. 3C). The average number of F4/80+ cells increased 26-fold (
hAEC-CM Promotes LPC Proliferation
The effects of hAEC-CM on LPC proliferation were determined by BrdU labeling. LPC cultures were treated with hAEC-CM, and BrdU incorporation was determined following 3 and 10 days of hAEC-CM treatment. At day 3, LPC cultures increased BrdU incorporation by 14% compared with the untreated LPC cultures (

hAEC-CM induces LPC proliferation. LPCs were cultured from 3 to 10 days in control medium, hAEC-CM, or differentiation. BrdU incorporation was used to determine LPC proliferation. (A) After 3 days, BrdU incorporation was slightly increased in hAEC-CM treated LPCs. (B) By 10 days, proliferation increased by twofold in hAEC-CM-treated LPC. ****
hAEC-CM Promotes LPC Differentiation into Hepatocytes
We investigated the effect of hAEC-CM and hAEC co-culture on LPC differentiation toward a hepatocyte lineage following 8 days of culture. A fivefold increase in the expression of AFP (

hAEC-CM stimulates LPC differentiation toward hepatocyte (A–D). LPC gene expression of AFP and albumin was assayed following 8 days culture in control medium, hAEC-CM, and co-culture with hAECs. Compared with the control medium, hAEC-CM significantly increased the expression of AFP and albumin by 5.4 and 8.13-fold, respectively. In contrast, the gene expression of AFP and albumin was not changed by co-culture with hAECs. (E) X-gal staining of 8-day cultures detected β-gal positive LPCs when cells were maintained in hAEC-CM. Images were taken at 20×. Scale bar = 100 μm. *
hAEC-CM Enhances Hepatocellular Functions by LPCs
We evaluated the effect of hAEC-CM on LPCs in terms of functional abilities that are performed by mature hepatocytes. Urea production, a surrogate marker of nitrogen metabolism, was measured following 16 days of exposure to hAEC-CM. The concentration of urea in the supernatant of hAEC-CM treated cultures was 22-fold greater than the controls and fivefold greater than the differentiation medium (
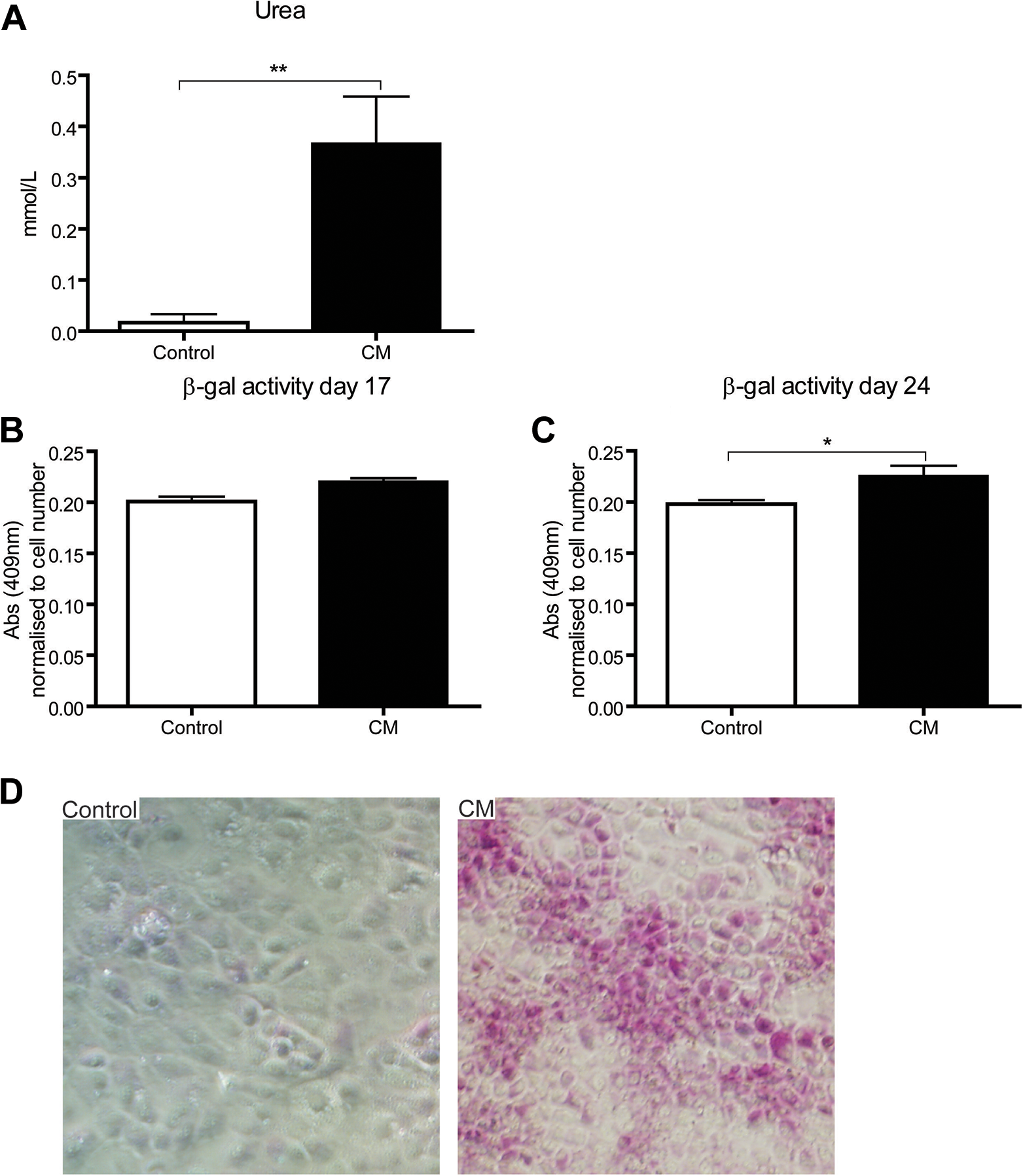
hAEC-CM enhances hepatocyte functions in LPCs. LPCs cultured in control medium and hAEC-CM were assayed for hepatocellular functions. (A) Compared with control, hAEC-CM increases LPC urea excretion by eightfold. (B and C) β-gal activity, a marker of LPC differentiation to mature hepatocytes, was assessed following 17 and 24 days. No significant difference in β-gal activity was observed between the day 17 cultured LPCs. LPC maintained for 24 days in hAEC-CM displayed a 13.8% increase in enzyme activity compared with control. (D) PAS staining performed on day 17 culture show that LPCs were treated with hAEC-CM. Images were taken at 10×. *
hAEC-CM Increases LPC Expression of Genes Involved in Metabolism Pathways.
KEGG pathway analysis of the differentially expressed genes shows that genes upregulated in LPCs by hAEC-CM are associated with multiple hepatic pathways, including drug, glutathione, and arachidonic acid metabolism, as well as steroid biosynthesis and bile acid secretion. Genes downregulated by hAEC-CM were associated with pathways involved in cell proliferation and phosphoinositide 3-kinase signaling (Fig. 7). The results of the GO pathway analysis revealed that the genes upregulated in LPCs by hAEC-CM were significantly enriched in biological processes, including multiple metabolic processes, negative regulation of cell growth, and apoptosis. The downregulated differentially expressed genes were enriched in pathways associated with cell proliferation (Fig. 8).
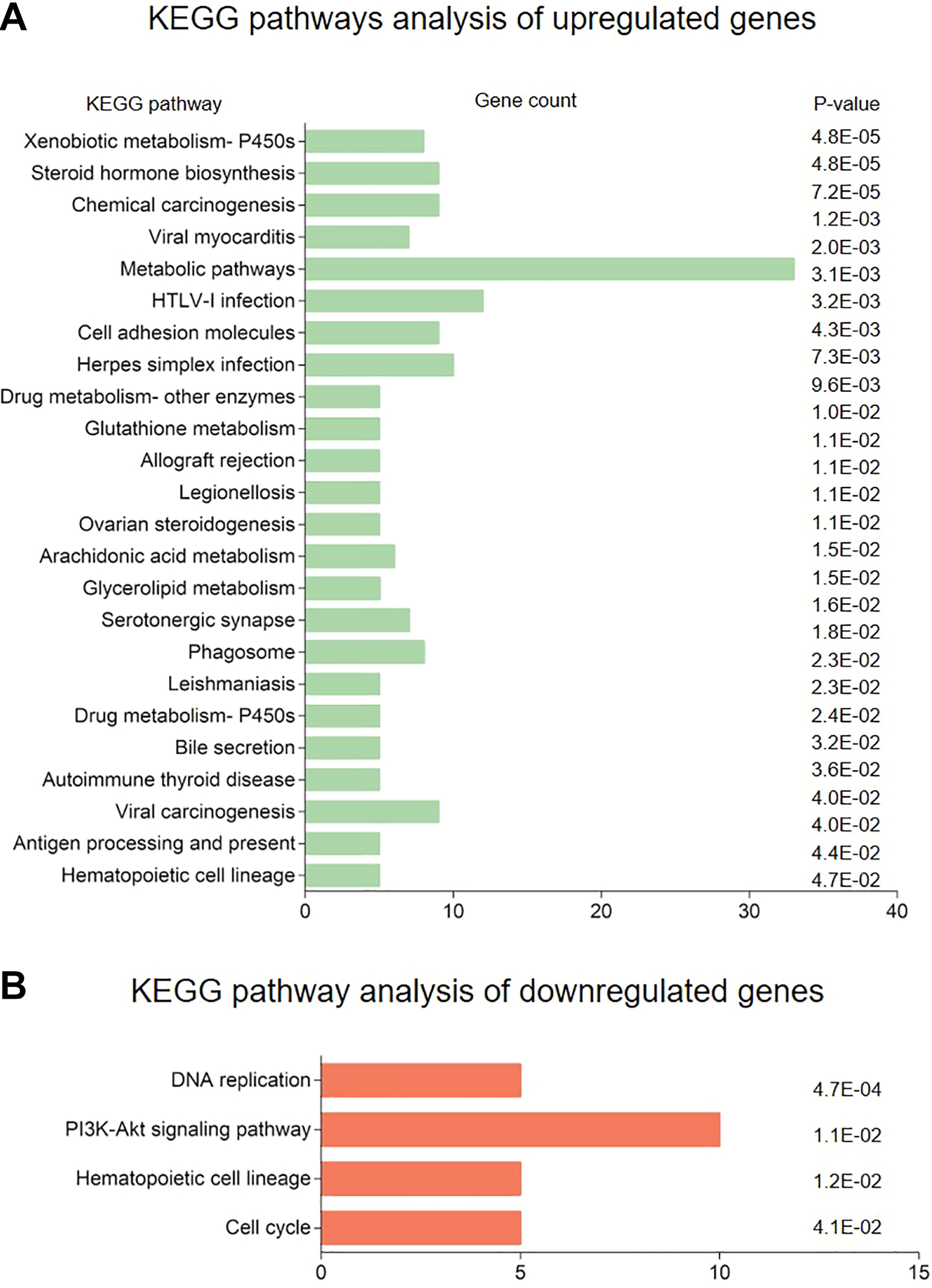
KEGG pathway analysis of differentially expressed genes. KEGG pathway analysis of genes (A) upregulated and (B) downregulated in LPC following 16 days culture in hAEC-CM. The Database for Annotation, Visualization and Integrated Discovery online tool was used to identify overrepresented pathways (

Enriched GO biological function of differentially expressed genes. GO pathway analysis of genes (A) upregulated and (B) downregulated in LPCs following 16 days culture in hAEC-CM. The Database for Annotation, Visualization and Integrated Discovery online tool was used to identify overrepresented biological processes (
Discussion
Previous studies documented antifibrotic effects with the administration of hAECs or hAEC-CM to a chronic liver injury model 6,12 . However, these studies focused on their effects from inflammatory and fibrotic perspectives of liver repair. Liver repair during chronic liver injury involves a complex interplay between parenchymal, fibrotic, and immune cell types 28 . Elements of this response, in particular inflammation, are key drivers of both hepatocyte and LPC-mediated regeneration 16,29 –34 . Given that hAECs suppress the inflammation during chronic liver injury, it is important to ascertain the effect of hAEC-based therapies on both hepatocytes and LPCs 6,8,12 . Thus, we undertook this study to ascertain the effect of hAECs and factors they produce (hAEC-CM) on both of these cell populations in a mouse model of chronic liver injury.
We established that in addition to reducing liver inflammation and fibrosis, the administration of hAECs during chronic liver injury sustains hepatocyte proliferation while reducing the LPC response. This reduction in LPC response can be explained by the decrease in hepatic macrophages. Macrophages are a crucial source of LPC mitogens such as TWEAK and IL-6; in fact, suppression of macrophage recruitment during liver injury attenuates the LPC response 16,30,35 . Similarly, the LPC response correlates with the severity of liver fibrosis. Therefore, the antifibrotic effect of hAECs would indirectly suppress LPC activity 17 . Overall, these results show that hAEC therapy reduces the extent to which LPCs participate in liver regeneration by modulating inflammation and fibrosis.
In general, the effect of hAEC-CM on hepatocytes, LPCs, and liver fibrosis were similar to the whole hAECs. However, hAEC-CM significantly reduced hepatocellular injury and the number of activated HSCs and macrophages, suggesting it may be more effective than hAECs at reducing liver injury and fibrosis. We previously showed that hAECs and hAEC-CM exert antifibrotic effects in a CCl4 model of liver fibrosis similar to those here 6,11,12 . However, an important difference between this and previous studies is that we have directly compared the two approaches. This provides a new perspective that it is primarily a paracrine mechanism that is responsible for the therapeutic effects of hAECs. Additionally, we show that hAEC-CM therapy may have additional therapeutic advantages compared with whole-cell therapy.
We observed a reduced number of LPCs in the absence of a reduction in liver injury, fibrosis, and macrophage numbers in the mice given control medium. Control medium contains folic acid and pantothenic acid which have been reported to reduce oxidative stress in hepatocytes and hepatic expression of inflammatory cytokines, including tumor necrosis factor-alpha (TNFα) and interleukin-6 (IL-6) during liver injury 35 –37 . Hepatic TNFα and IL-6 levels were significantly reduced by the administration of the control medium (Supplemental Fig. 4). Hence, this mechanism may be responsible for the reduced LPC response in mice given control medium. However, unlike hAEC-CM, the effects of control medium on liver repair overall cannot be regarded as beneficial since injury and fibrosis persisted unabated. It follows that if hAEC-CM suppresses the LPC response in vivo by the same mechanisms as control medium, a further reduction in their numbers resulting from the reduction in liver injury, fibrosis, and macrophage infiltration be expected. Therefore, the fact that LPC numbers were similar in the hAEC-CM and control medium cohorts suggests that hAEC-CM maintains the LPC response while simultaneously reducing injury, fibrosis, and inflammation. Furthermore, a recent study by Katsuda and colleagues (2017) described the in vivo differentiation of LPCs to hepatocytes and 75%–90% repopulation of chronically injured livers in transgenic urokinase-type plasminogen activator-SCID (cDNA-uPA/SCID) mice. The outcomes from this study indicate that exogenously transplanted bipotential LPCs can repopulate chronically injured livers. Taken together with the outcomes from our current study, the data suggest that hAECs may influence the expansion of the LPC compartment, differentiation of LPCs to mature hepatocytes, and eventual repopulation of the chronically injured liver.
To clarify the effects of hAEC-CM on LPC activity, we performed a series of in vitro studies on an established LPC line 23 . We showed that LPCs cultured in hAEC-CM displayed greater BrdU incorporation indicating increased proliferation. Additionally, we demonstrated that hAEC-CM promotes LPC differentiation toward a hepatocyte lineage. Exposure to hAEC-CM increased LPC expression of AFP and albumin. Both of these genes are upregulated early during the differentiation of LPCs to hepatocyte, suggesting more LPCs were differentiating down this lineage 38 . Analysis of hepatocellular functions by LPCs revealed that long-term exposure to hAEC-CM induces urea and glycogen synthesis and the expression of a β-gal reporter gene 27 . Additionally, KEGG pathway enrichment and GO biological function annotation analysis of differential expression data showed that genes upregulated in LPC by hAEC-CM were mainly enriched in pathways associated with hepatic metabolism and synthesis 39,40 . Collectively, these findings indicate that soluble factors secreted by hAECs directly induce LPC to proliferate and differentiate into functional hepatocytes in culture.
In contrast to LPCs cultured in hAEC-CM, LPCs co-culture with hAECs did not significantly alter AFP or albumin expression nor were β-gal positive LPCs observed by X-gal staining. However, caution must be taken when directly comparing the hAEC-CM and co-culture experiments since their conditions are inherently different. For instance, the consumption of nutrients by hAECs may have a negative effect on LPC differentiation during co-culture. Nevertheless, these results do suggest that interactions between hAECs and LPCs do not require direct cell–cell contact.
The apparent contradictory effects of hAEC-CM on LPCs in vivo compared with the in vitro are informative. It suggests that the immunosuppressive effect of hAEC-CM is more important than its direct effect on LPCs. The overall effect of hAEC-CM on the LPC response in vivo will be the sum of direct and indirect pathways, which either promote or attenuate their expansion. Therefore, these in vitro findings support the notion that the decrease in LPC numbers in vivo caused by hAEC-CM is the result of immunosuppression and increased differentiation.
In this study, we provide evidence that soluble factors secreted by hAECs support liver repair during chronic injury by a combination of reducing fibrosis, yet maintaining parenchymal regeneration. These therapeutic properties combined with their availability, abundance, and safety supports their case as excellent candidates for therapeutic use in liver pathologies. Future studies should investigate the efficacy of hAECs in animal models that represent alternative etiologies of chronic liver disease in contrast to CCl4 exposure. For instance, models that recapitulate nonalcoholic steatohepatitis in humans could provide insight into the clinical utility of hAECs in this context. Nonetheless, our results highlight the therapeutic-c potential of hAECs and advocate for their development as a novel antifibrotic therapy for treating chronic liver diseases.
Supplemental Material
Supplemental Material, Appendix_A - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Appendix_A for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, F480_staining - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, F480_staining for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, Ki67_A6_and_PANCK_IHC - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Ki67_A6_and_PANCK_IHC for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, Supp_fig_4_cytokines - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Supp_fig_4_cytokines for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, Supp_Fig_5_FN14_and_GP130 - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Supp_Fig_5_FN14_and_GP130 for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, Supp_fig_6_Heatmap_of_markers - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Supp_fig_6_Heatmap_of_markers for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Supplemental Material
Supplemental Material, Treatment_outline - Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury
Supplemental Material, Treatment_outline for Human Amnion Epithelial Cells Produce Soluble Factors that Enhance Liver Repair by Reducing Fibrosis While Maintaining Regeneration in a Model of Chronic Liver Injury by Alexander Hodge, Neil Andrewartha, Dinushka Lourensz, Robyn Strauss, Jeanne Correia, Mihiri Goonetilleke, George Yeoh, Rebecca Lim and William Sievert in Cell Transplantation
Footnotes
Acknowledgments
The authors thank Professor Scott Friedman (Mount Sinai Hospital, NY, USA) for his kind gift of LX-2 cells and Dr Valentina Factor (National Institute of Health, MD, USA) for the A6 antibody. We also acknowledge the facilities, scientific and technical assistance of the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy, Characterization & Analysis, UWA.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded by an Australian National Health and Medical Research Council grant (GNT1064247).
Footnotes
Prior conference presentation of some of this submitted material:
Hodge, A., Lourensz, D., Tchongue, J., Lim, R., Yeoh, G., Sievert, W.
Awarded Presidential poster of distinction
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
