Abstract
Intraorbital transection of the optic nerve (ON) always induces ultimate apoptosis of retinal ganglion cells (RGCs) and consequently irreversible defects of vision function. It was demonstrated that transplanted olfactory ensheathing cells (OECs) in partially injured spinal cord have a distant in vivo neuroprotective effect on descending cortical and brain stem neurons. However, this study gave no answers to the question whether OECs can protect the central sensitive neurons with a closer axonal injury because different neurons respond variously to similar axonal injury and the distance between the neuronal soma and axonal injury site has a definite effect on the severity of neuronal response and apoptosis. In the present study, we investigated the effect of transplanted OECs on RGCs after intraorbital ON transection in adult rats. Green fluorescent protein (GFP)-OECs were injected into the ocular stumps of transected ON and a significantly higher number of surviving RGCs was found together with a consistent marked increase in the mRNA and protein levels of BDNF in the ON stump and retina in the OEC-treated group at 7 days, but not 2 and 14 days, time point when compared to the control group. Our findings suggest that OEC transplantation induces the expression of BDNF in the ocular ON stump and retina and delays the death of axotomized RGCs at a certain survival period.
Keywords
Introduction
Intraorbital transection of the optic nerve (ON) always induces ultimate death of retinal ganglion cells (RGCs) (3,29) and consequently irreversible defects of vision function. Deprivation of target-derived neurotrophic factors has been thought to be one of the major explanations for such RGC death (2). Many strategies, including application of neurotrophic factors, have been demonstrated to be able to protect axotomized RGCs (5,33).
Olfactory ensheathing cells (OECs), a sort of macroglial cell found in the nasal olfactory mucosa and olfactory bulb, can secrete neurotrophins such as nerve growth factor (24), brain-derived neurotrophic factor (BDNF) (17,31), and neurotrophin-4/5 (NT-4/5) (4). Previous investigations also showed that conditioned medium of adult rat OECs protected PC12 cells from Zn+ insult in vitro (30) and transplanted OECs in the partially injured spinal cord have a distant in vivo neuroprotective effect on descending cortical and brain stem neurons (20,25). However, no study has investigated the effects of transplanted OECs on RGC survival, although OECs have been shown to enhance axonal regeneration of axotomized RGCs (16). Because the distance between the soma and axonal injury site of a neuron has a definite effect on the severity of neuronal response and apoptosis (29), more neurons die with a closer axonal injury (2). In the present study, we transplanted OECs into the ocular stump of intraorbitally transected ON in adult rats to see whether OECs protect injured RGCs with a very close axonal injury.
In the present study, the number of surviving RGCs increased at day 7 after OEC transplantation and a significantly promoted mRNA and protein levels of BDNF were observed simultaneously within the ocular ON stump and retina. The mechanisms of these effects of OEC transplants on RGC survival will be discussed.
Materials and Methods
Preparation of Donor OECs for Transplantation
Transgenic Sprague-Dawley rats, 13 days old (purchased from Laboratory Animal Services at Rutgers University and obtained as the gift from Dr. Wutian Wu at the University of Hong Kong), expressing green fluorescent protein [GFP, CZ-004, SD-Tg(CAG-EGFP) Cz-004Osb; SLC, Shizuoka, Japan] were anesthetized with an IP injection of sodium pentobarbital (60 mg/kg body weight; Ovation Pharmaceuticals, USA). The olfactory bulbs were removed. The outer nerve fiber and granular layers of the olfactory bulb were separated and the tissues were dissociated with collagenase A and D and papain (Sigma, USA). The separated cells were then cultured with Dulbecco's modified Eagle medium/nutrient mixture F-12 (DMEM/F12; Gibco, USA) supplemented with 10% fetal bovine serum (Gibco-Invitrogen, USA) for 10 days. Mixed fibroblasts were removed with a differential adhesion method and the culture contained mostly (90%) p75-positive cells, indicative of OECs. To prepare the cells for transplantation the cultured cells were thoroughly washed by DMEM/F12, dissociated with 0.125% trypsin (Gibco-Invitrogen, USA), washed again in DMEM/F12 to remove trypsin, and suspended at a density of 105/μl in DMEM/F12.
ON Transection and OEC Transplantation
Animal Care and Use Committees at the Fourth Military Medical University and Rutgers University approved all animal protocols. The left ONs of 126 female wild-type Sprague-Dawley rats, 72 days old (Laboratory Animal Center at the Fourth Military Medical University and Laboratory Animal Services at Rutgers University), were exposed via an intraorbital approach under deep sodium pentobarbital anesthesia. After the dural sheath was opened, the ON was completely transected 1.5 mm from the optic disc using small iris scissors (Adler, Germany). Care was taken to maintain blood supply to the retina intact throughout the operation. Immediately after ON transection, 1 μl GFP-OEC suspension or the same volume of DMEM/F12 medium was injected into the ocular ON stump using a glass micropipette in 63 experimental and 63 control animals.
Retrograde Labeling of Surviving RGCs
A small piece of gelform soaked in 5% FluoroGold (FG, Fluorochrome, USA) was applied onto the ocular ON stump in 36 animals (n = 18 for each of the experimental and control groups) immediately after OEC transplantation to retrogradely label the surviving RGCs (6,10). These 36 animals were killed with an overdose of pentobarbital anesthesia 2, 7, or 14 days after ON transection (n = 6 for each time point in the experimental and control groups) and the left eyes with the ocular stumps were enucleated. The retinas were placed in 4% paraformaldehyde in 0.1 M phosphate buffer (PB; pH 7.4) with four radial cuts dividing the retinas into four quadrants, postfixed in the fixative for 1 h, rinsed in 0.1 M PB, and then flat-mounted in 50% glycerin (Sigma, USA) with the RGC side up on slides. FG-labeled RGCs were counted along the median line of the four quadrants starting from the optic disc to the peripheral border of the retina with an eyepiece grid of 500 × 500 μm2 at 500-μm intervals under a BX51 fluorescence microscope (Olympus, Japan) using a 355–425 nm ultraviolet filter. The mean density of labeled RGCs in each retina was then multiplied by the area of the retina to obtain the mean number of labeled RGCs in the retina.
Hoechst 33342 Staining of the Stumps
Nine experimental and nine control animals were killed with a lethal overdose of sodium pentobarbital 2, 7, or 14 days after ON transection (n = 3 for each time point) and perfused through the heart with cold saline followed by 4% paraformaldehyde. The ON stumps were dissected from the eyes and serially sectioned (16 μm) along the longitudinal axis of the ON. After rinsing in phosphate-buffered saline (PBS), the sections in all groups were incubated with Hoechst 33342 (5 μg/ml; Molecular Probes) for 15 min, coverslipped after rinsing in PBS, and observed under the fluorescence microscope for the morphology and distribution of transplanted OECs.
Real-Time Quantitative Polymerase Chain Reaction (RT-PCR)
Another 18 experimental and 18 control animals were euthanized 2, 7, or 14 days after ON transection (n = 6 for each time point). The ocular ON stump and the whole retina were removed and homogenized in Trizol (Sigma, USA). The mRNA was extracted and reverse transcribed with SuperScript II and Oligo(dT) in a PE2400 PCR instrument (PerkinElmer, USA). RT-PCR was performed on all the mentioned trophic factors, using synthesized cDNA, specific primers, and SYBR Green fluorescent marker. The reaction was carried out using LightCycler 2.0 (Roche Applied Science, USA) with a 20-μl final volume containing 50 nmol/L of primers, 10 ng of cDNA, nucleotides, Taq DNA polymerase, and the SRBR Green I PCR product. The conditions of PCR reaction were: initial denaturation phase of 94°C for 5 min followed by denaturation at 94°C for 45 s, annealing at 58°C for 60 s, and extension at 72°C for 60 s, for 40 cycles. The changes in fluorescence of SYBR Green I dye in each cycle were monitored by the system software. Gene expression was determined by the LightCycler Relative Quantification software. Primer sequences were: BDNF: forward 5′-GCG GCA GAT AAA AAG ACT GC-3′, reverse 5′-GCC AGC CAA TTC TCT TTT TG-3′; ciliary neurotrophic factor (CNTF): forward 5′-GCC TTG ACT CAG TGG ATG GT-3′, reverse 5′-AGG CAG AAA CTT GGA GCG TA-3′; glial cell-derived neurotrophic factor (GDNF): forward 5′-GAC TTG GGT TTG GGC TAT GA-3′, reverse 5′-AAC ATG CCT GGC CTA CTT TG-3′; leukemia inhibitory factor (LIF): forward 5′-AGA AGG TCC TGA ACC CCA CT-3′, reverse 5′-GCC TGG ACC ACC ACA CTT AT-3′; NGF: forward 5′-CAT GGG GGA GTT CTC AGT GT-3′, reverse 5′-GCA CCC ACT CTC AAC AGG AT-3′; neurotrophin-3 (NT-3): forward 5′-GAT CCA GGC GGA TAT CTT GA-3′, reverse 5′-AGC GTC TCT GTT GCC GTA GT-3′; β-actin: forward 5′-AGC CAT GTA CGT AGC CAT CC-3′, reverse 5′-CTC TCA GCT GTG GTG GTG AA-3′.
Western Blot Assay
Based on the outcomes of RT-PCR assay, the protein level of BDNF, showing significant changes of BDNF associated with transplanted OECs, was chosen to be assessed in all groups. Thus, 18 experimental and 18 control animals were euthanized at 2, 7, or 14 days after ON transection (n = 6 for each time point). Unfixed ocular ON stumps with the whole retinal tissue were freshly dissected in lysis buffer (1% Triton X-100, 10 mM EDTA, and 0.1% SDS in PBS) with 1/100 protease inhibitor, homogenized, and centrifuged at 14,000 rpm for 20 min. The supernatant was subjected to the Western blot analysis. Proteins were separated by 15% sodium dodecy1 sulfate-poly acrylamide gel electrophoresis and transferred onto nitrocellulose membranes (0.22-μm pores). The blots were blocked in 5% dried milk in TBST and processed for immunostaining with a primary antibody against BDNF (1:300; Sigma) overnight at 4°C. Bound antibodies were visualized with an anti-mouse IgG secondary antibody (1:10,000). Immunoreactive bands were visualized by enhanced chemiluminescence. The relative densities of the immunoreactive bands were determined and normalized with respect to actin, using a densitometric analysis.
Statistical Analyses
Data were analyzed with the Bonferroni tests after one-way analysis of variance (ANOVA), using Microcal Origin software (Version 7.0, Originlab Corp, USA). Results were expressed as mean ± SEM.
Results
The Number of Surviving RGCs
No GFP-OECs were detected in the retinas in the experimental and control groups at all time points. The mean number of surviving RGCs at 2 days after ON transection in the control animals was 118,237 ± 3,224, which sharply dropped to 51,252 ± 2,280 (p < 0.01) at 7 days and 12,219 ± 575 at 14 days. Taking the number at 2 days as the baseline, 43% and 10% of the 2-day RGC population survived at 7 and 14 days. In OECs-treated animals that survived for 2, 7, and 14 days, the numbers of surviving RGCs were 120,663 ± 3,710, 80,590 ± 4,637, and 12,643 ± 1,262, suggesting an increase in percentages of surviving RGCs to 67% at 7 days but dropped to 10% at 14 days. An OEC treatment therefore increased number of RGCs (p < 0.01) transiently at 7 days when compared to the control group (Fig. 1).
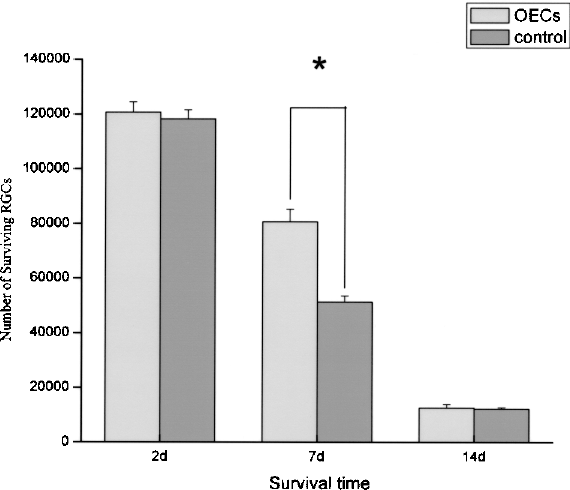
Histogram illustrating the mean numbers of surviving RGCs in the experimental and control groups at different survival time points. The asterisk indicates a significant difference (p < 0.01) in the RGC number between the OECs-treated and control groups at day 7. Error bars = SEM.
Morphology of Transplanted GFP-OECs in the ON Stumps
Numerous GFP-positive OECs were present within the ON stump at 2 days after transplantation. Most of these cells were fusiform and some were elliptical or cycloid. Nuclei stained with Hoechst 33342 could be observed in OECs (Fig. 2A, B). At 7 days, the number of OECs had declined remarkably and most OECs became irregular in shape (Fig. 2C, D). No GFP-OECs could be found in the stump at 14 days (Fig. 2E, F).
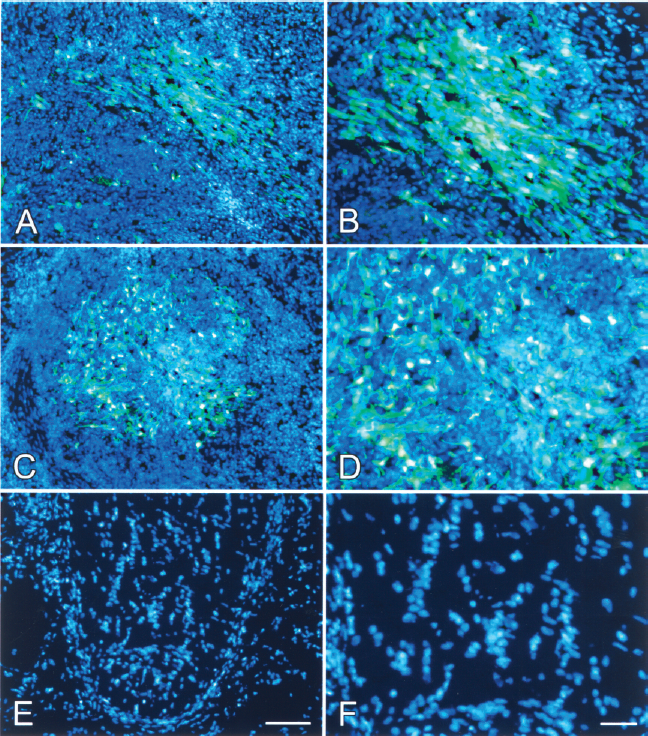
Photomicrographs of fluorescent labeled sections of the remaining optic nerve. Many GFP-OECs (green) with Hoechst-stained nuclei (blue) distributed along the longitudinal axis of the ON stump 2 days after OEC transplantation (A). The number of transplanted GFP-OECs decreased sharply and the cell morphology became deteriorative when the survival time increased to 7 days (C). No GFP-OECs were revealed in the ON stump at day 14 (E). (B, D, F) Amplified magnified images of (A, C, E). Scale bars: 50 μm.
RT-PCR Assay
When compared to the control groups, mRNA levels of BDNF (p < 0.01) within the ON stump and retina in the OECs-treated groups were significantly higher at 7 days, but not 2 and 14 days (p > 0.05) (Fig. 3). As for the other five factors assayed, mRNA levels (p > 0.05) did not differ significantly between the experimental and control groups at all time points (Fig. 4).

Histogram showing the mRNA level of BDNF in the OECs-treated and control groups at different survival time points. The asterisk indicates a significantly higher level of BDNF (p < 0.01) in the OECs-treated group compared with that in the control group at the day 7 time point. Error bars = SEM.

Histogram showing the mRNA levels of CNTF, GDNF, LIF, NGF, and NT-3 in the experimental and control groups at all different survival time points. No significant differences (p > 0.05) in the mRNA levels were shown between OECs-treated and control groups at all time points. Error bars = SEM.
Western Blot Assay
Western blot assay also showed a significantly higher protein level (p < 0.05) of BDNF in the ON stump and retina in the OEC-treated group at 7 days, when compared to the control group (Fig. 5).

Western blot assay and densitometric analysis showing changes of the protein level of BDNF at different time points. (A) Quantitative analysis of Western blot results showed that the protein level of BDNF in OECs-treated group (A, C, E) was significantly higher than that in the control group (B, D, F) at day 7. (B) OEC-treated and control groups at 2, 7, and 14 day time points, respectively. *p < 0.05. Error bars = SEM.
Discussion
The number and corresponding percentage of surviving RGCs showed no significant changes within 3 days after intraorbital ON transection, but declined to approximately 50% and 10% of the 2-day RGC population at 7 and 14 days (3). In the present study, the number of surviving RGCs in the control group at day 2 was similar to the normal RGC population in adult rats (9,19), and is often used as a value of surrogate number representing the normal RGC population (6,10). Also, the reduced percentages of surviving RGCs (43% and 10% at day 7 and 14) in the control groups are similar to those reported in a previous study (3).
Several investigations have reported that transplanted OECs survive, migrate, and integrate within the host central nervous system (1,11,21–23), including the ON where transplanted OECs can survive for over 6 months after transplantation (16). In the present study, injected OECs survived in the ocular ON stump for at least 7 days but the number of transplanted cells declined so that no surviving OECs could be detected in the ON stump at 14 days. The simplest explanation for such a short OEC survival is immune rejection of the cells by recipient animals and deleterious microenvironment of the degenerating ON stump. The complete loss of OECs at 14 days and deteriorative morphology of OECs at 7 days is consistent with delayed immune rejection that occurs with heterologous OEC transplants into central nervous system tissues. We used a higher percentage of OEC transplant (~90% OEC suspension), compared to ~50% OEC-embedded matrix in a previous study (16).
Deprivation of target-derived neurotrophic factors is a frequently cited reason for RGC apoptosis after ON transection (2). Application of NGF, BDNF (8,12,28), NT-4/5 (18,27), CNTF (7,13), fibroblast growth factor (FGF), or GDNF (14,15,26,32) into the eye or ON stump at an early stage can protect axotomized RGCs. Transplanted OECs express many neurotrophic factors, such as NGF, BDNF, NT-4/5, and GDNF, which may promote neuronal survival in injured brain and spinal cord. In the present study, OEC-transplanted retinas had 67% surviving RGC at 7 days, a significantly higher percentage than 43% in the control group, suggesting an effective neuroprotection of 24% more RGCs after OEC transplantation. The outcomes from RT-PCR and Western blot assays demonstrated a significantly higher mRNA and protein levels of BDNF, the only one among five other neurotrophic factors examined (NGF, NT-3, CNTF, LIF, and GDNF), which is elevated in OECs-treated animals at 7 days. The neurotrophin BDNF correlated with the survival of OECs. A similar correlation had been observed previously between neuroprotection of axotomized descending projection neurons and a higher BDNF expression at 7 days after OEC transplant in partially transected spinal cord (25). The higher BDNF levels may have been secreted by the transplanted OECs or from endogenous cells induced by OEC transplantation.
At 14 days, the percentage of surviving RGCs in the OECs-treated animals dropped to 10%, a percentage similar to that in the control animals, hence indicating that OECs did not rescue axotomized RGCs, but rather delay RGC death. The transplanted OECs did not survive for more than 7 days in the ON stump to secrete BDNF or stimulate the production of endogenous BDNF by 14 days. At 14 days, not only were the cell survival numbers low but BDNF levels were correspondingly low. In 2006, Sasaki et al. reported no difference in BDNF levels in the spinal cord transplanted with OECs or nontransplanted control spinal cords at 4 weeks after transplantation (25).
Our study indicates that the transplanted OECs have a transient neuroprotective effect. These new findings are consistent with another study in which BDNF enhanced the neuronal survival after OEC transplantation into the injured spinal cord (25). Further studies should be considered to investigate the unknown source of highly expressed BDNF 7 days after OEC transplantation and strategies including immunosuppressive agents to promote a longer survival of transplanted OECs in the injured ON. Thus, it would be possible to rescue more injured RGCs at a prolonged survival time and reach the goal of clinical application.
Footnotes
Acknowledgments
Supported by Natural Science Foundations of China (Grant numbers: 30872829, 30571998, and 30901049) and Army Medical Research Foundation of China (Grant number: 06H039).
