Abstract
Since the successful demonstration of the Edmonton protocol, islet transplantation has advanced significantly on several fronts, including improved pancreas preservation systems. In this study, we evaluated two different types of organ preservation solutions for human islet isolation. Modified Celsior (Celsior solution with hydroxyethyl starch and nafamostat mesilate; HNC) solution and modified Kyoto (MK) solution were compared for pancreas preservation prior to islet isolation. Islet yield after purification was significantly higher in the MK group than in the HNC group (MK = 6186 ± 985 IE/g; HNC = 3091 ± 344 IE/g). The HNC group had a longer phase I period (digestion time), a higher volume of undigested tissue, and a higher percentage of embedded islets, suggesting that the solution may inhibit collagenase. However, there was no significant difference in ATP content in the pancreata or in the attainability of posttransplant normoglycemia in diabetic nude mice between the two groups, suggesting that the quality of islets was similar among the two groups. In conclusion, MK solution is better for pancreas preservation before islet isolation than HNC solution due to the higher percentage of islets that can be isolated from the donor pancreas. MK solution should be the solution of choice among the commercially available solutions for pancreatic islet isolation leading to transplantation.
Introduction
Pancreatic islet transplantation is a promising option for the treatment of the type 1 diabetic patients (33, 34). Since the Edmonton protocol was reported, more than 600 type 1 diabetics in more than 50 institutions have undergone islet transplantation to treat their disease. However, barely half of the processed pancreata effectively meet the criteria for clinical transplantation in most centers. Furthermore, current isolation techniques usually recover fewer than half of the islets from a given pancreas, necessitating islet transplantation from two or more donors to achieve euglycemia (17, 27, 34). The outcome of islet transplantation can be affected by the conditions under which pancreata are recovered and stored prior to isolation.
Donor pancreata are usually preserved with University of Wisconsin (UW) solution, an intracellular solution containing hydroxyethyl starch (HES). Some centers have used the two-layer method (TLM), which employs oxygenated perfluorochemical (PFC) and UW solution (6, 14, 15, 19). Although UW solution is currently considered by most centers to be the gold standard of preservation solution, it has several disadvantages: it is chemically unstable, it must be cold stored until use, and its short shelf life makes it expensive. In addition, its highly viscous nature complicates the initial organ flush (39). Especially in the field of islet isolation, UW solution inhibits the activity of collagenase, an enzyme blend for pancreatic digestion (4, 32), and it strongly affects islet yield. Recently, we reported that pancreas preservation with modified Kyoto (MK) solution is better for islet yield in porcine islet isolation than UW solution (25). MK solution contains trehalose, gluconate, and ulinastatin. Trehalose has a cytoprotective effect against stress, and gluconate acts as an extracellular oncotic agent, which prevents cells from swelling (1). Ulinastatin is a trypsin inhibitor and eliminates trypsin activity during pancreas preservation (25). MK solution also has a high sodium/low potassium composition with comparatively low viscosity, while UW solution has high potassium, which causes vasospasms and high viscosity and both of these may inhibit perfusion before preservation (40). The advantages of MK solution are trypsin inhibition and less collagenase inhibition (23, 25). However, MK solution has not yet been evaluated using human pancreata.
Recently, Celsior solution has been used as an alternative solution for organ preservation. Celsior is an extracellular solution deprived of colloid and was initially developed for heart preservation (16). Preliminary clinical studies showed no differences between UW and Celsior for lung (31), liver (2), and kidney (5) preservation. Hubert et al. recently reported on the application of Celsior solution for in situ perfusion of the donor before human and pig islet isolation (7). Their data showed that in situ perfusion of UW solution is superior to Celsior solution. In contrast to UW, Celsior induced cell swelling and pancreas edema after only 4 h of cold storage. The components of Celsior solution are in part similar to MK solution (a high-sodium/low-potassium composition with comparatively low viscosity) and in part similar to UW solution (including lactobionate acid and glutathione). Because Celsior solution lacks HES, which is an oncotic agent and protects cell swelling, we added HES to Celsior solution in this study. We also added nafamostat mesilate, one of the trypsin inhibitors, in Celsior because one of the advantages of MK solution is trypsin inhibition by ulinastatin. Nafamostat mesilate has a higher level of trypsin inhibition than ulinastatin (Noguchi et al.; submitted data).
In this study, we compared the effects of MK solution and modified Celsior (Celsior solution with HES and nafamostat mesilate; HNC) solution on human pancreatic islet isolation.
Materials and Methods
Preservation Solution
We used MK solution (Kyoto solution, Otsuka Pharmaceutical Factory, Inc., Naruto, Japan; ulinastatin, Mochida Pharmaceutical, Tokyo, Japan), HNC solution (Celsior, Sangstat, Lyon, France; nafamostat mesilate, Torii Pharmaceutical, Tokyo, Japan), and PFC (Apollo Scientific Ltd., UK). The components and other features of the solutions are shown in Table 1.
Composition and Characteristics of Each Preservation Solution
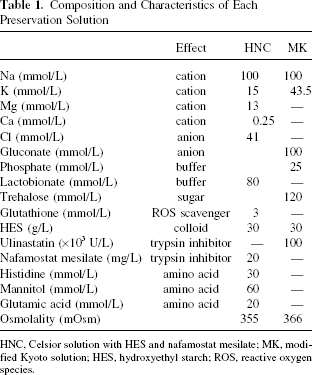
HNC, Celsior solution with HES and nafamostat mesilate; MK, modified Kyoto solution; HES, hydroxyethyl starch; ROS, reactive oxygen species.
Measurement of Trypsin Inhibition Ability of Solutions
In order to assess the trypsin inhibition of HNC, MK, or phosphate-buffered saline (PBS, control), 3 ml of 0.3 mM N-benzoyl-l-arginine ethylester reagent (BAEE reagent; Sigma Chemical Co., St. Louis, MO) was incubated for 5 min at 25°C and then 5 μl of 1 mg/ml trypsin and 45 μl of HNC, MK solution, or PBS were added. Trypsin activity was measured by absorption spectrophotometry (λ253 nm) using BAEE reagent for the trypsin substrate, as described previously (15). Absorbance was measured at 1, 2, 3, 4, 5, and 6 min. One BAEE unit was defined as a change in optical density of 0.001/min.
Human Islet Isolation
The islet isolation protocol was approved by the Institutional Review Board of Baylor Health Care System. All pancreata were procured using a standardized technique to minimize warm ischemia. UW solution (ViaSpan, DuPont Pharmaceuticals, Wilmington, DE) was used for in situ perfusion of the donor. The pancreas was excised immediately after the liver and before the kidneys and placed on ice. After the removal of the spleen and duodenum, we immediately inserted a cannula into the main pancreatic duct. The pancreas was weighed and 1 ml/g pancreas weight of preservation solution was infused through the intraductal cannula (12, 24). Pancreata were placed into an HNC/PFC or MK/PFC two-layer preservation container at 4°C for less than 8 h, until the islet isolation procedure.
Human islet isolation was conducted as previously described (12, 18, 22) according to the standard Ricordi technique (29) with modifications later introduced in the Edmonton protocol (13, 14, 26, 28, 34). In brief, after decontamination of the pancreas, the ducts were perfused in a controlled fashion with a cold enzyme blend of Liberase HI (1.4 mg/ml; Roche Molecular Biochemicals, Indianapolis, IN) or Serva collagenase NB1 with neutral protease (SERVA Electrophoresis GmbH, Heidelberg, Germany). The distended pancreas was then cut into 7–9 pieces, placed in a Ricordi chamber, and shaken gently. While the pancreas was being digested by recirculating the enzyme solution through the Ricordi chamber at 37°C, we monitored the extent of digestion with dithizone staining by taking small samples from the system. Once digestion was confirmed to be complete, dilution solution (Mediatech, Inc., Manassas, VA) was introduced into the system, and then the system was cooled to stop further digestive activity. The digested tissue was collected in flasks containing 25% HSA and washed with fresh medium to remove the enzyme. The phase I period was defined as the time between placement of the pancreas in the Ricordi chamber and the start of collecting the digested pancreas. The phase II period was defined as the time between the start and end of collection. Islets were purified with a continuous density gradient of iodixanol (Optiprep®, Sigma-Aldrich, St. Louis, MO)-based solution as previously reported (12, 22) in an apheresis system (COBE 2991 cell processor, Gambro Laboratories, Denver, CO).
Islet Evaluation
The crude number of islets in each diameter class was determined by counting islets after dithizone staining (3 mg/ml final concentration; Sigma Chemical Co.) using an optical graticule. The crude number of islets was then converted to the standard number of islet equivalents (IE; diameter standardizing to 150 μ m) (30). The islet recovery was defined as the percentage of IE recovered after purification compared to the IE before purification. Islet viability after purification was assessed using a double fluorescein diacetate/propidium iodide (FDA/PI) staining to visualize living and dead islet cells simultaneously (14, 30, 34). Fifty islets were inspected and their individual viability was determined visually, followed by calculation of their average viability (14).
Islet function was assessed by monitoring the insulin secretory response of the purified islets during glucose stimulation according to a procedure described by Shapiro and colleagues (34). Briefly, 1200 IE were incubated with either 2.8 mM or 25 mM glucose in RPMI-1640 for 2 h at 37°C and 5% CO2. The supernatants were collected and insulin levels were determined using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (ALPCO Insulin ELISA kit; ALPO Diagnostics, Windham, NH). The stimulation index was calculated by determining the ratio of insulin released from islets in high glucose concentration to the insulin released in a low concentration. The data were normalized to total DNA. The data were expressed as mean ± SE.
Determination of ATP Production
To measure adenosine triphosphate (ATP) production, isolated islets in each group were cultured overnight with CMRL plus 5% FBS and washed twice with ice-cold PBS and solubilized. The amount of ATP was then measured using an ATP assay system (ATP-lite, Perkin Elmer, Groningen, Netherlands), according to the manufacturer's instructions. The data were normalized to total DNA. The data were expressed as mean ± SE.
In Vivo Assessment
Nude mice were rendered diabetic by a single IP injection of streptozotocin (STZ) at a dose of 220 mg/kg. Hyperglycemia was defined as a glucose level of >350 mg/dl detected twice consecutively after STZ injection. The 2,000 IE human islets obtained from each group were transplanted into the renal subcapsular space of the left kidney of a diabetic nude mouse, as previously described (20, 21). During the 30-day posttransplantation period, the nonfasting blood glucose levels were monitored three times per week. Normoglycemia was defined when two consecutive blood glucose level measurements showed less than 200 mg/dl. No statistical differences in either pretransplantation blood glucose levels or pretransplantation body weight were observed between the two groups of mice. Mouse studies were approved by the Institutional Animal Care and Use Committee (IACUC) of Baylor Health Care System.
Statistics
The differences between each group were considered significant if the value was p < 0.05 using an unpaired Student's t-test with Bonferroni correction or the Kaplan-Meier log-rank test.
Results
Activity of Trypsin Inhibition in Preservation Solutions
The trypsin inhibitor in MK solution is ulinastatin and in HNC is nafamostat mesilate. We examined the activity of trypsin inhibition in each preservation solution. Compared with PBS (control), MK solution and HNC solution significantly inhibited trypsin activity PBS: 32.8 ± 0.9 BAEE units; HNC: 1.7 ± 0.4 BAEE units; MK: 10.7 ± 0.3 BAEE units) (Fig. 1). These data suggest that ulinastatin and nafamostat mesilate work well in each preservation solution and that these solutions may reduce trypsin activity during pancreas preservation.

Measurement of trypsin inhibition. BAEE reagent (3 ml of 0.3 mM) was incubated for 5 min at 25°C and then 5 μl of 1 mg/ml trypsin was added with either 45 μl of HNC (n = 4), MK solution (n = 3), or PBS (control; n = 3). Trypsin activity was measured by absorption spectrophotometry (λ253 nm) using BAEE reagent for the trypsin substrate. Absorbance was measured at 1, 2, 3, 4, 5, and 6 min. One BAEE unit was defined as a change in optical density of 0.001/min. Data are expressed as the mean ± SE.
Islet Isolation Characteristics
The characteristics of the human pancreata are shown in Table 2. There were no significant differences in age, body weight, body mass index (BMI), pancreas size, or cold ischemic time (CIT) between the two groups.
Pancreas Characteristics

Data are expressed as mean ± SE. There were no significant differences between the two groups.
There was no significant difference in islet yield before purification between the HNC group and the MK group (HNC: 7134 ± 2608 IE/g, MK: 7047 ± 1092 IE/g) (Fig. 2). Islet yield after purification was significantly higher in the MK group compared with the HNC group (HNC: 3091 ± 344 IE/g, MK: 6186 ± 985 IE/g) (Fig. 2B). Other islet characteristics are shown in Table 3. The phase I period was significant longer in the HNC group compared with the MK group (HNC: 22.7 ± 2.4 min, MK: 15.3 ± 0.8 min) (Table 3). The data suggest that HNC solution can inhibit collagenase activity. Higher undigested tissue volume and percentage of embedded islets in the HNC group also support its collagenase inhibition.

Islet yield before purification (A) and after purification (B). The MK group had significantly improved islet yield after purification compared to the HNC group. Data are expressed as the mean ± SE.
Islet Characteristics

Data are expressed as mean ± SE.
Postpurification recovery (%) = IE after purification/IE before purification x 100.
There were significant differences between the two groups (p < 0.05).
The rate of postpurification recovery was significantly higher in the MK group than in the HNC group (HNC: 51.3 ± 10.9%, MK: 87.8 ± 5.4%). The lower recovery rate after purification in the HNC than the MK group is due to a higher percentage of embedded islets in the HNC group. Even if iodixanol-based purification was used, it was extremely difficult to collect a large number of embedded islets. Because the islet yields before purification were similar between the two groups, and the purification methods were also similar, the large numbers of embedded islets from the HNC solution (because of collagenase inhibition) must have affected the islet yield after purification, as opposed to what was seen in the MK group. The lower recovery rates in the HNC group than the MK group also supports this.
Quality of the Isolated Islets
To assess the islet quality in each group in vitro, the ATP content of isolated islets was measured. There was no significant difference in ATP content between the two groups (HNC: 101.9 ± 7.9 pmol/μg DNA; MK: 112.7 ± 7.0 pmol/μg DNA) (Fig. 3A). To assess the islet graft function of each group in vivo, 2,000 IE from each group were transplanted below the kidney capsule of STZ-induced diabetic nude mice. The blood glucose levels of four of the five mice (80.0%) receiving islets from the HNC group and five of the six mice (83.3%) from the MK group decreased gradually and reached normoglycemia. The blood glucose levels remained stable thereafter and returned to pretransplantation levels after kidney-bearing islets were removed (30 days after transplantation) (Fig. 3B). The attainability of posttransplantation normoglycemia was similar between the two groups. These data suggest that the quality of islets was similar between the two groups.

Cellular ATP content and in vivo bioassay for islet function. (A) The ATP concentration of the cell lysate after islet isolation was measured using an ATP assay system. The ATP was normalized to DNA concentration (HNC group n = 5; MK group n = 5) Data are expressed as the mean ± SE. (B) Normoglycemic percentage of STZ-induced diabetic nude mice after islet transplantation. Immediately after isolation, 2,000 IE islets were transplanted below the kidney capsule of diabetic nude mice. Normoglycemia was defined as two consecutive posttransplant blood glucose levels of less than 200 mg/dl (HNC group n = 5; MK group n = 6).
Discussion
Our study shows that MK solution is a more effective cold-storage solution for the pancreas preservation step of islet isolation than HNC solution. It has been previously reported that UW, one of the standard preservation solutions used for abdominal organ procurement, inhibits collagenase activity during the digestion phase of islet isolation. This results in poor islet yield and islets of poor viability (4, 32). The components in UW solution that were found to be most inhibitory were magnesium, low sodium, high potassium, HES, and adenosine. Allopurinol in combination with either lactobionate or glutathione was also markedly inhibitory. The most inhibitory solution tested was a combination of three components: raffinose, glutathione, and lactobionate (4). HNC solution includes magnesium, HES, glutathione, and lactobionate. On the other hand, MK solution has only HES (from the UW solution). It has also been shown that the Na+/K+ ratio, adenosine, allopurinol, and glutathione are not important for the cold storage of pancreatic digests prior to islet purification (3). Moreover, we previously reported that trehalose and ulinastatin inhibit collagenase digestion less than UW solution (25).
Trypsin from pancreatic acinar cells destroys islets in the process of islet isolation. Previous studies have shown that trypsin inhibition by Pefabloc during human pancreas digestion improves islet yield and reduces the fraction of embedded islets (8, 15). Moreover, we previously reported that MK preservation, including ulinastatin, eliminated trypsin activity during pancreas preservation, and that Kyoto/PFC preservation without ulinastatin resulted in lower islet yields in our rat and porcine models (25). In this study, islet yields before purification in the HNC and MK groups, both of which include a trypsin inhibitor, were similar, suggesting that the effects on pancreas preservation may be similar between the HNC and MK solutions. The differences of islet yield after purification are due to the effect of these solutions on collagenase inhibition.
ATP is utilized to drive a sodium pump, maintain cell integrity, and repair warm ischemic injury (9, 11, 35, 37, 38). ATP levels in islets after culture reflected islet viability (10), and a recent paper showed that the ATP/DNA ratio is a better indicator of islet cell viability than the ADP/ATP ratio (36). In this study, there was no difference in ATP levels between the two groups. Moreover, an in vivo study found that the attainability of posttransplant normoglycemia was similar between the two groups. These data suggest that the quality of islets was similar between the two groups.
In conclusion, MK solution is superior to HNC solution in human islet isolation due to the higher percentage of islets that can be isolated from the donor pancreas. MK solution has been used in clinical islet isolation in Japan (18) and should be the solution of choice for clinical isolations.
Footnotes
Acknowledgments
The authors wish to thank Dr. Carson Harrod for editing the manuscript and Ms. Yoshiko Tamura for technical support. This work was supported in part by the Juvenile Diabetes Research Foundation International (JDRFI), Otsuka Pharmaceutical Factory, Inc., and the All Saints Health Foundation.
