Abstract
Islet transplantation has recently emerged as an effective therapy and potential cure for type 1 diabetes mellitus. Recent reports show that the two-layer method (TLM), which employs oxygenated perfluorochemical (PFC) and University of Wisconsin (UW) solution, is superior to simple cold storage in UW for pancreas preservation in islet transplantation. Moreover, we recently reported that islet yield was significantly higher in the ET-Kyoto solution with ulinastatin (MK)/PFC preservation solution compared with the UW/PFC preservation solution in the porcine model and that the advantages of MK solution are trypsin inhibition and less collagenase inhibition. In this study, we compared ulinastatin with another trypsin inhibitor, Pefabloc, in preservation solution for islet isolation. Islet yield before purification was higher in the MK/PFC group compared with the ET-Kyoto with Pefabloc (PK)/PFC group. The stimulation index was higher for the MK/PFC group than for the PK/PFC group. These data suggest that ET-Kyoto with ulinastatin was the better combination for pancreas preservation than ET-Kyoto with Pefabloc. Based on these data, we now use ET-Kyoto solution with ulinastatin for clinical islet transplantation.
Introduction
Since the report of the Edmonton protocol (37), islet transplantation has advanced significantly and more than 600 type 1 diabetes patients in more than 50 institutions have undergone islet transplantation to cure their disease. The cadaveric pancreas is injured due to brain death, hypotension, and vasopressor therapy, and subsequently from warm ischemia after donor cross-clamping and cold ischemic storage. There is a clear relationship between these injuries and the reduced success of subsequent islet isolation (4,12). In Japan, pancreatic islets are isolated from non-heart-beating donors (NHBDs) for clinical islet transplantation because donations from heart-beating brain-dead donors are only two to five cases per year and most of their pancreata are used for pancreas organ transplantation. We therefore need to develop an efficient isolation technique for NHBD pancreata.
We have recently demonstrated that islet isolation and transplantation with NHBDs using the modified Ricordi method (Kyoto islet isolation method) effectively cures type 1 diabetes (23). The transplantation rate (transplantation number/isolation number) is more than 80%, higher than recently published data using brain-dead heart-beating donors (14,21,31). The isolation method includes an in situ cooling system for pancreas procurement (19), ductal injection (28), the modified two-layer method (MK/PFC) (27,30), and iodixanol-based purification (14). We previously showed that MK/PFC preservation significantly improved islet yields, compared with UW/PFC preservation (30). MK solution includes a trypsin inhibitor (ulinastatin), which is one of the advantages of this solution. Indeed, pancreas preservation using MK solution was superior to preservation with ET-Kyoto solution without the trypsin inhibitor in a rat model (30).
In this study, we compared ulinastatin with another trypsin inhibitor, Pefabloc, in preservation solution for islet isolation.
Materials and Methods
Preservation Solution
We used ET-Kyoto solution (5,32) with ulinastatin (Miraclid®, Mochida Pharmaceutical, Tokyo, Japan; ET-Kyoto + ulinastatin = “MK solution”) or Pefabloc (Roche Applied Science, Germany; ET-Kyoto + Pefa bloc = “PK solution”). The components of the solutions are shown in Table 1.
Composition of Each Preservation Solution

Measurement of Trypsin Inhibition Ability of Solutions
In order to assess the trypsin inhibition of MK solution, PK solution, and ET-Kyoto solution without trypsin inhibitors (control), 3 ml of 0.3 mM N-benzoyl-L-arginine ethylester reagent (BAEE; Sigma, Tokyo, Japan) were incubated for 5 min at 25°C and then 5 μl of 1 mg/ml trypsin and 45 μl of each solution were added. Trypsin activity was measured by absorption spectrophotometry (λ253 nm) using BAEE for the trypsin substrate, according to a previous report (17). Absorbance was measured every minute for 6 min. A BAEE unit was defined as a change in optical density of 0.001/min.
Porcine Islet Isolation
Porcine pancreata were obtained at a local slaughterhouse. The operation was started about 10 min after the cessation of heart beating. After removing the pancreas, we immediately inserted a cannula into the main pancreatic duct, infused each preservation solution for ductal protection, and put the pancreas into a two-layer preservation container that had one of the preservation solutions (preservation solution/PFC). Operation time was defined as the time elapsed between the start of operation and removal of the pancreas. Warm ischemic time (WIT) was defined as the time elapsed between cessation of heart beating and placement of the pancreas into the preservation solution. Cold ischemic time (CIT) was defined as the time elapsed between placement of the pancreas into the preservation solution and the start of islet isolation.
Islet isolation was conducted in accordance with the method described in the Edmonton protocol (15,16,33,34,37). In brief, after decontamination of the pancreas, the ducts were perfused in a controlled fashion with a cold enzyme blend of Liberase HI (1.4 mg/ml; Roche Molecular Biochemicals, Indianapolis, IN). The distended pancreas was then cut into nine pieces, placed in a sterilized Ricordi chamber, and shaken gently. While the pancreas was being digested by recirculating the enzyme solution through the Ricordi chamber at 37°C, we monitored the extent of digestion with dithizone staining by taking small samples from the system. Once digestion was completed, RPMI-1640 medium (Gibco, Carlsbad, CA) was introduced into the system, and the system was cooled to stop further digestive activity. The digested tissue was collected and washed with fresh medium to remove the enzyme. The phase I period was defined as the time between placement of the pancreas in the Ricordi chamber and the start of collecting the digested pancreas. The phase II period was defined as the time between the start and end of collection.
Islets were purified with a continuous density gradient with Iodixanol-Kyoto solution in an apheresis system (COBE 2991 cell processor, Gambro Laboratories, Denver, CO). Because Iodixanol has a lower viscosity than Ficoll, it needs less force during centrifugation, which causes less damage to islets. For the solution, low-density (density: 1.077) and high-density (density: 1.100–1.125) Iodixanol-Kyoto solutions were produced by changing the volumetric ratio of Iodixanol and Kyoto solution.
Islet Evaluation
The crude number of islets in each diameter class was determined by counting islets after dithizone staining (3 mg/ml, final concentration) (Sigma Chemical Co., St. Louis, MO) using an optical graticule. The crude number of islets was then converted to the standard number of islet equivalents (IE; diameter standardizing to 150 μm) (35). Gross morphology was qualitatively assessed by two independent investigators scoring the islets for shape (flat vs. spherical), border (irregular vs. well-rounded), integrity (fragmented vs. solid/compact), uniformity of staining (not uniform vs. perfectly uniform), and diameter (least desirable: all cells < 100 μm/most desirable: more than 10% of the cells >200 μm) (16,35). Each parameter was graded from 0 to 2 with 0 equaling the worst and 2 the best score, so that the worst islet preparations were given a cumulative score of 0 and the best a score of 10. Spherical, well-rounded, solid/compact, uniformly stained, and large islets were characterized as the best islets.
Islet viability after purification was assessed using acridine orange (10 μmol/L) and propidium iodide (15 μmol/L) (AO/PI) staining to visualize living and dead islet cells simultaneously (3,16,35). Fifty islets were inspected and their individual viability was determined visually, followed by calculation of their average viability (16).
In Vitro Assessment of Islet Function
Islet function was assessed by monitoring the insulin secretory response of the purified islets during glucose stimulation according to a procedure described by Shapiro and colleagues (37). Briefly, 1200 IE were incubated with either 2.8 or 25 mM glucose in RPMI-1640 for 2 h at 37°C and 5% CO2. The supernatants were collected and insulin levels were determined using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (ALPCO Insulin ELISA kit; ALPCO Diagnostics, Windham, NH). The stimulation index was calculated by determining the ratio of insulin released from islets in high glucose concentration to the insulin released in a low concentration. The data were normalized to total protein from the cell lysate. All assessments were made in triplicate and the data (mean ± SE) were expressed as a percentage of the control values in each experiment to eliminate variables caused by differences among donor pancreata.
Recently, Goto et al. showed that the measurement of the ADP/ATP ratio correlated with transplantation outcome (8). The ADP/ATP ratio was measured to evaluate the energy status of cultured islets, using the Apo Glow™ kit (Cambrex Bio Science Nottingham Ltd., Nottingham, UK). In brief, 80 IEs were washed in PBS and then mixed with 100 μl of nucleotide-releasing reagent for 10 min at room temperature. Thereafter, 20 μl of nucleotide-monitoring reagent was added to the solution. The ATP levels were measured using a luminometer (FB 12 Luminometer, Berthold Detection Systems GmbH, Pforzheim, Germany) and expressed as the number of relative light units (RLU). After 10 min, the ADP in the solution was converted to ATP by adding 20 μl ADP converting reagent and then measured as the number of RLU. Subsequently, the ADP/ATP ratio of the islets was calculated.
In Vivo Assessment of Islet Function
Mice with severe combined immunodeficiency disease (SCID; CLEA Japan, Inc., Meguro, Tokyo) were used for the experiments. The recipients were rendered diabetic by a single injection of streptozotocin (STZ) at a dose of 220 mg/kg. Hyperglycemia was defined as a glucose level of >350 mg/dl detected twice consecutively after STZ injection. The 2000 IE pig islets obtained from each group were transplanted into the renal subcapsular space of the left kidney of diabetic SCID mice. During the 30-day posttransplantation period, the nonfasting blood glucose levels were monitored three times per week. Normoglycemia was defined when two consecutive blood glucose level measurements showed less than 200 mg/dl. No statistical differences in either pretransplantation blood glucose levels or pretransplantation body weight were observed among the four groups of mice. Mouse studies were approved by the Institutional Animal Research Committees of Kyoto University, Nagoya University, and Fujita Health University.
Statistical Analysis
Values for the data represent the mean ± SE. Two or three groups were compared by Student's t-test with Bonferroni correction.
Results
Inhibition of Trypsin Activity
Previous reports show that trypsin inhibition with TLM preservation improves islet yields (17,30). We examined whether MK and PK solutions inhibited trypsin activity. Both solutions inhibited trypsin activity (Fig. 1) compared with ET-Kyoto solution (control) (p < 0.01), suggesting that these solutions could be useful in reducing trypsin activity during pancreas preservation.

Impact of MK or PK solution on trypsin activity. BAEE was incubated for 5 min at 25°C and then trypsin along with MK solution (n = 7), PK solution (n = 7), or ET-Kyoto (control; n = 7) was added. Trypsin activity was measured by absorption spectrophotometry (λ253 nm) using BAEE reagent. Absorbance was measured every minute for 6 min. A BAEE unit was defined as a change in optical density of 0.001/min. MK and PK solutions inhibited trypsin activity significantly more than ET-Kyoto (p < 0.01). Data are expressed as the mean ± SE.
Porcine Islet Isolation Characteristics
The characteristics of porcine islet isolation protocols are shown in Table 2. There were no significant differences in pancreas size, operation time, WIT, or CIT between the two groups. Phase I and phase II periods were also similar for the two groups.
Pig Islet Isolation Characteristics

Data are expressed as mean ± SE.
Islet yield before purification was significantly higher in the MK/PFC group (n = 5) than the PK/PFC group (n = 3) (MK/PFC; 9676 ± 635 IE/g, PK/PFC; 6999 ± 844 IE/g, p < 0.05) (Fig. 2A). The islet yield after purification for the MK/PFC group was higher than the PK/PFC group (MK/PFC; 6608 ± 927 IE/g, PK/PFC; 4964 ± 1153 IE/g) but not significantly so (Fig. 2B). Other porcine islet characteristics are shown in Table 3. The stimulation index was higher for the MK/PFC group than for the PK/PFC group (p < 0.05). There were no other significant differences in characteristics between the two groups.
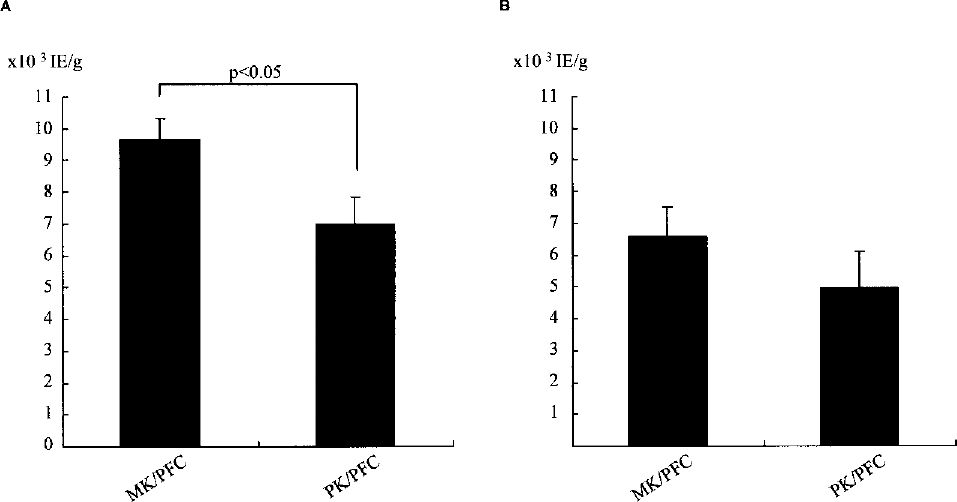
Islet yield before purification (A) and after purification (B). The MK/PFC group had significantly better islet yield before purification than the PK/PFC group (p < 0.05). Data are expressed as the mean ± SE.
Pig Islet Characteristics

Data are expressed as mean ± SE.
Stimulation index was higher for the MK/PFC group than for the PK/PFC group (p < 0.05).
Assessment of Islet Function In Vitro and In Vivo
Recently, Goto et al. showed that the measurement of the ADP/ATP ratio correlated with transplantation outcome (8). To assess the islet graft function of each group in vitro, the ADP/ATP ratio was measured. There was no significant difference in ADP/ATP ratio between the groups (data not shown).
To assess the islet graft function of each group in vivo, 2000 IE of each group were then transplanted below the kidney capsule of STZ-induced diabetic SCID mice. There was no significant difference between the groups with respect to the attainability of posttransplantation normoglycemia (data not shown). Morphologic studies showed the presence of islets under the kidney capsule of all SCID mice 30 days after transplantation. The islet grafts of each group in the normoglycemic mice showed intense insulin staining (data not shown).
Taken together, these data suggest that MK/PFC preservation is superior to PK/PFC preservation.
Discussion
Islet allotransplantation can achieve insulin independence in patients with type 1 diabetes (37). Since the Edmonton protocol was announced, islet transplantations from brain-dead donors (9,10) as well as from non-heart-beating donors (13,14,21) and living donors (15) have been performed. These advances were based on advanced pancreas transport systems (10,16,17,30), revised immunosuppressant protocols (24,25), improved islet isolation methods (17), and enhanced islet engraftment (26). Although experiments of β-cell regeneration from stem cells have proceeded (20,22,29), there is still no reliable method for producing β-cells. Until a new method to generate β-cells is developed, improving the efficacy of islet transplantation seems the most realistic and prudent method to cure diabetes.
Donor pancreata are usually preserved with University of Wisconsin (UW) solution. Recent reports have shown that the two-layer method (TLM), which employs oxygenated perfluorochemical (PFC) and UW solution, is superior to simple cold storage in UW to preserve not only the whole pancreas but also individual islets for transplantation (16,17). However, UW solution has several disadvantages: it is chemically unstable, it must be cold stored until use, and its short shelf life makes it expensive. It is also highly viscous, which may complicate the initial organ flush (39). Recently, our university developed a new preservation solution, ET-Kyoto solution, and its effectiveness in cold lung storage has been demonstrated in clinical lung transplantation (5,32). It also is effective for skin flap storage and its clinical application is beginning in this field (40). Although high potassium in UW solution causes insulin release from pancreatic β-cells (7), ET-Kyoto solution has a high sodium/low potassium composition. Moreover, UW solution inhibits the activity of Liberase, an enzyme blend for pancreatic digestion (6,36), but ET-Kyoto solution with ulinastatin inhibits Liberase less (30).
Trypsin from pancreatic acinar cells destroys islets. Previous studies have shown that trypsin inhibition by Pefabloc during human pancreas digestion improves islet yield and reduces the fraction of embedded islets (11,17), suggesting that trypsin may degrade the ductules and thus reduce the delivery of collagenase solution to the immediate neighborhood of the islets. Previously, we demonstrated that modifying TLM preservation, by including ulinastatin, eliminated trypsin activity during pancreas preservation, and ET-Kyoto/PFC preservation without ulinastatin resulted in lower islet yields (30). In this study, we showed that MK solution was synthetically superior to PK solutions. It may be due to differences in inhibitory effects of cytokines. Ulinastatin has been shown to inhibit not only trypsin activity but also the release of neutrophil elastase. It also downregulates transcription of tumor necrosis factor mRNA, the activation of endothelial cells, and the expression of ICAM-1 induced by endotoxin in vitro (1,2,18). It has been shown that administration of ulinastatin decreased the ischemia-reperfusion injury (38) and attenuated the elevation of inflammatory cytokines and C-reactive protein, a marker of inflammation (41) in transplanted small intestine.
In conclusion, we show that ET-Kyoto with ulinastatin is a better combination for pancreas preservation than ET-Kyoto with Pefabloc. Based on these data, we now use the ET-Kyoto solution with ulinastatin for clinical islet transplantation from NHBD pancreata. MK/PFC preservation makes it feasible to use NHBDs for efficient islet transplantation into type 1 diabetes patients.
Footnotes
Acknowledgments
The authors wish to thank Dr. Yusuke Nakai (Kyoto University), Dr. Bashoo Naziruddin, Dr. Nicholas Onaca, Mr. Andrew Jackson, and Ms. Yoshiko Tamura (Baylor Research Institute) for technical advice, Dr. Carson Harrod for his careful reading and editing of this manuscript, and Ms. Nobuyo Hatanaka (The University of Tokyo) and Maki Watanabe (Fujita Health University) for assistance. This work was supported in part by the Juvenile Diabetes Research Foundation International (JDRFI); the Ministry of Education, Science and Culture, the Ministry of Health, Labour and Welfare; and Baylor All Saints Health Foundation.
