Abstract
Oval cells and hepatocytes rarely proliferate simultaneously. This study aimed to determine the impacts of hepatocyte transplantation on the response and fate of oval cells that are activated to proliferate in acute severe hepatic injury. Retrorsine + D-galactosamine (R+D-gal) treatment was used to induce acute hepatic injury and to elicit extensive activation of oval cells in male dipeptidyl peptidase IV-deficient F344 rats. These rats were then randomized to receive wild-type hepatocyte transplantation or vehicle intraportally. The kinetics of oval cell response and their differentiation fate were analyzed. Results showed that oval cells were activated early and differentiated into hepatocytes in R+D-gal-treated rats without hepatocyte transplantation. With hepatocyte transplantation, the oval cells were recruited later and continued to proliferate in parallel with the massive proliferation of transplanted hepatocytes. They formed ductules and differentiated into biliary cells. When hepatocytes were transplanted at the day when oval cells were at their peak response, the numerous activated oval cells ceased to differentiate into hepatocytes and remained in ductular form. The ductular oval cells were capable of differentiating into hepatocytes again when the donor hepatocytes were inhibited to proliferate. We conclude that hepatocyte transplantation changes the mechanism of liver reconstitution and affects the differentiation fate of host oval cells in acute severe hepatic injury.
Introduction
Acute hepatic failure is a devastating disease with high mortality and a formidable challenge for clinicians. Liver transplantation continues to represent the primary therapy for patients with acute hepatic failure (1,3). However, because of the shortage of donor organs, a high cost, and significant morbidity and mortality, only a limited number of eligible candidates can benefit from this option. Mature hepatocytes have an enormous proliferative potential (20). Transplanted hepatocytes are able to proliferate extensively in selected animal models with inherent genetic defects or preconditioned with retrorsine + PH or CCL4 (6,12,15,22,27). Therefore, hepatocyte transplantation has been proposed as an alternative strategy for the treatment of acute hepatic failure (8).
In response to liver injury, the mature hepatocytes can rapidly proliferate to repair liver damage (6,20). When hepatocyte proliferation is inhibited or overwhelming liver injury occurs, hepatic injury leads to the activation of oval cells, which are a group of small proliferating cells with an oval-shaped nucleus and a high nuclear to cytoplasm ratio that appear in the periportal areas (7,31,32). After activation, oval cells are able to proliferate and differentiate to replenish the hepatic mass. Although high activation of oval cells is a sign of disease severity and indicative of poor outcome, their activation and differentiation into hepatocytes are vital for survival after acute hepatic injury (14,18).
We have recently demonstrated that massive repopulation by transplanted hepatocytes can also occur in acute severe hepatic injury (39). As a general rule, hepatocytes and oval cells rarely proliferate simultaneously (6,7,31,32)). As such, when oval cells are activated in acute severely injured liver, the question arises whether the extensive proliferation of transplanted hepatocytes in acute hepatic injury will exclude the activation of host oval cells, or if donor hepatocytes will cooperate with the activated oval cells for reconstitution. The answer is yet to be determined. Therefore, this study aimed to determine the impact of donor hepatocytes on the response of oval cells and the fate of their differentiation in acute hepatic injury.
Materials and Methods
Animals
Dipeptidyl peptidase IV-deficient (DPPIV-) F344 rats were kindly provided by Professor Sanjeev Gupta from the Albert Einstein College of Medicine. Male DPPIV- rats were used as recipient animals. Normal male DPPIV+ F344 rats (aged 8–10 weeks, 200–250 g) were purchased from National Laboratory Animal Center, Taiwan and used as donor animals. These animals were in-house bred and maintained on standard laboratory chow and daily 12-h light/dark cycles. All of the animals received humane care in compliance with the guidelines of the National Science Council of Taiwan. All animal experiments were approved by the Institutional Laboratory Animal Care and Use Committee of the National Taiwan University, College of Medicine and College of Public Health.
Retrorsine Administration and Induction of Acute Hepatic Injury
The retrorsine (Sigma, St. Louis, MO) and d-galactosamine (Sigma, St. Louis, MO) working solutions were prepared as described previously and used immediately after preparation (10,38). The 6-week-old male DPPIV- rats (100–130 g) received two treatments of retrorsine (30 mg/kg, IP) 2 weeks apart (n = 150). d-Galactosamine (0.7 g/kg, IP) was administered 2 weeks after the second treatment. More than 90% of treated rats survived the experimental procedures.
Hepatocyte Isolation and Transplantation
In situ liver perfusion, collagenase digestion, and differential centrifugation were used to purify the hepatocytes from normal male DPPIV+ F344 rats as previously described (38). The viability and purity of each preparation were assessed by evaluating trypan blue exclusion in a hemacytometer. Isolated hepatocytes were resuspended in PBS (pH 7.4) without serum to 1 × 107 cells/ml. Preparations typically contained >85% viable hepatocytes. The hepatocytes were transplanted within 1 h of isolation.
A bolus of 1 × 107 fresh hepatocytes in 1 ml of PBS was infused through the portal cannula within 1–2 min, performed between 24 and 26 h after D-galactosamine administration (38). Patency of the portal cannula was confirmed by blood sampling before cell infusion.
Histochemistry and Immunohistochemistry
All histochemical and immunohistochemical stainings were performed according to previously described protocols. To detect oval cells, cryosections were stained with OV6 mouse monoclonal antibody (R&D, Minneapolis, MN, USA) (24). Fluorescence detection was carried out with Alexa Fluor 594 donkey-anti-mouse IgG (Molecular Probes, OR, USA). The antibody to CD26 recognized the DPPIV antigen on hepatocytes. Double immunofluorescence staining for DPPIV (R&D) and OV6 or laminin (DAKO, CA, USA), or OV6 and laminin were detected using the method described by Paku et al. (24). Appropriate secondary antibodies used in various experiments included Alexa Fluor 488 donkey anti-goat IgG (Molecular Probes) and Alexa Fluor 594 donkey anti-mouse IgG (Molecular Probes).
To identify the transplanted hepatocytes in the recipient liver, DPPIV expression was determined by enzyme histochemistry in liver cryosections as previously described (26). OV6 (R&D) and CK19 (Novocastra, Newcastle, UK) were detected by ABC peroxidase method using diaminobenzidine (DAB) as chromogen (Elite kit; Vector Laboratories, CA, USA) (23). Gamma-glutamyltranspeptidase (GGT) was detected by the method of Rutenberg et al. (29) and glucose-6-phosphatase (G6Pase) by the method of Dabeva et al. (4).
Determination of Oval Cell Response
The oval cell response was classified by microscopic examination of liver sections stained with OV6 antibody. The prevalence of the oval cell response was assessed separately by two investigators in at least 50 portal fields per rat and scored from 1 to 5 using the method of Braun et al. (2). A mean value of the two scores was assigned to the liver.
Experimental Design
Male DPPIV-deficient rats received two treatments of retrorsine (30 mg/kg, IP) 2 weeks apart, at 6 and 8 weeks of age. Acute hepatic injury was induced by D-galactosamine treatment (0.7 gm/kg, IP) 2 weeks after the second retrorsine treatment. The rats were then randomized to receive wild-type hepatocyte transplantation (1 × 107/ml) or vehicle intraportally 24 h or 4 days after D-galactosamine treatment. Some rats received additional two doses of retrorsine at the second and fourth weeks after hepatocyte transplantation. Briefly, these groups were: (A) retrorsine + D-galactosamine treatment with no hepatocyte transplantation (R+D-gal group), (B) retrorsine + D-galactosamine + hepatocyte transplantation at 24 h (R+D-gal+C group), (C) retrorsine + d-galactosamine + hepatocyte transplantation at the fourth day (R+D-gal+C4d group), and (D) retrorsine + D-galactosamine + hepatocyte transplantation at 24 h + retrorsine at the second and fourth week (R+D-gal+C+R group).
To analyze the activation of oval cells and the proliferation of donor hepatocytes, surviving rats were euthanized and their livers were harvested at days 1 and 4, weeks 1, 2, 4, and 8 after hepatocyte or vehicle transplantation, and indicated times (R+D-gal group, n = 6–8 rats at each time point; R+D-gal+C group, n = 8 rats at each time point; R+D-gal+C4d group, n = 3 at weeks 1, 2, and 4; R+D-gal+C+R group, n = 3 at weeks 2, 4, and 6). Rat body and liver weights were recorded. One piece of liver tissue from the right lobe was fixed in 4% formaldehyde and paraffin embedded for histology. The other pieces from each liver lobe were snap-frozen in liquid nitrogen or embedded into optimum cutting temperature (OCT) compound and stored at −80°C.
Statistical Analysis
Data were analyzed by the Mann-Whitney rank sum test or Kruskal-Wallis test with the Bonferroni correction as appropriate using SPSS 9.0 (Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Retrorsine + d-Galactosamine Treatment Induced Oval Cell Response in Rats
D-Galactosamine treatment was able to induce oval cell response (4,16,32). However, R+D-gal treatment had not been previously used as a protocol to induce oval cell reaction. To verify and characterize, we conducted pilot studies on 75 rats that were sacrificed at several time points (3 h to 14 days) to compare the oval cell reaction induced by D-gal alone (n = 35) and R+D-gal (n = 40) with sublethal dose of D-galactosamine (0.7 gm/kg). More than 90% of treated rats in each group survived. Oval cells in the D-gal-alone injured livers in our hands were morphologically and phenotypically similar to those reported in previous studies (Fig. 1A) (4,16). In the R+D-gal-treated rats, H&E-stained liver sections showed an increased number of oval cells, spreading from the periportal region (Fig. 1B). These cells were further confirmed by labeling with the OV6 antibody, the most frequently used antibody to identify oval cells (Fig. 1C and D) (24). Hepatocyte megalocytosis was clearly visible by 3 days after R+D-gal treatment (Fig. 1B). Clusters of small hepatocyte-like cells were rarely observed in the livers of R+D-gal-treated rats.
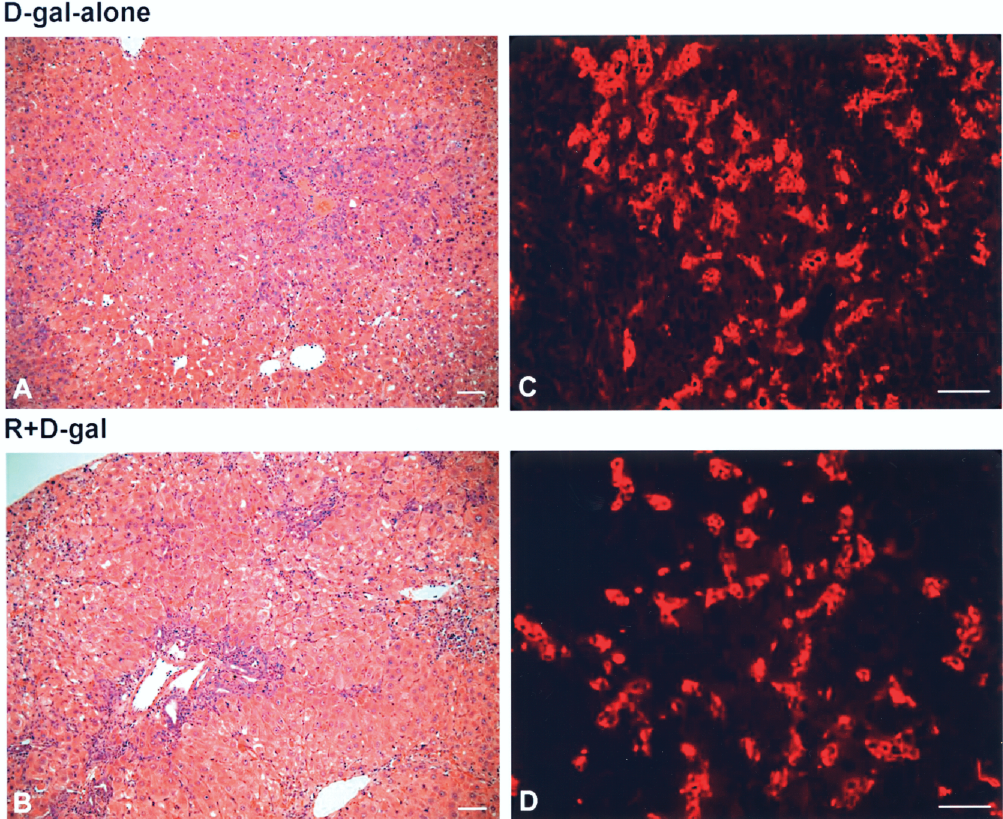
Activation of oval cells in rats treated with D-galactosamine (D-gal) alone (A, C) or retrorsine + D-galactosamine (R+D-gal) (B, D). H&E staining of liver sections on day 3 after either treatment showed clusters of small and dark blue cells in the periportal areas representing activated oval cells (A, B). Immunofluorescence for OV6 (red) further confirmed the activation of oval cells after either treatment (C, D). Original magnification: A, B, 100x; C, D, 200x. Scale bars: 50 μm.
Hepatocyte Transplantation Changed the Kinetics of Oval Cell Response in R+D-gal-Treated Rats
In the R+D-gal-treated rats without hepatocyte transplantation, the oval cell response emerged at day 1 and peaked at days 4–5, when there was extensive oval cell presence through the hepatic parenchyma. The oval cell response declined after 1 week. With hepatocyte transplantation, the number of oval cells at day 1 (2 days after R+D-gal treatment) was similar to that without hepatocyte transplantation. However, the number of oval cells markedly decreased as compared to the peak oval cell response at day 4–5 without hepatocyte transplantation. In contrast to the declined oval cell reaction after 1 week without hepatocyte transplantation, the oval cells continued to expand at day 7, reached a peak response at week 4 after hepatocyte transplantation, and declined after week 8 (Fig. 2). Because the oval cells often formed duct-like structures originating from the periportal region and spreading into the liver acinus in both the R+D-gal-treated and R+D-gal+C-treated rats, the oval cell response was classified and scored by microscopic examination of liver sections stained with OV6 antibody (n = 6–8 rats at each time point) (Fig. 3A).

Kinetics of oval cell response in retrorsine + d-galactosamine-treated rats without (R+D-gal, left column) or with hepatocyte transplantation (R+D-gal+C, middle and right columns) were analyzed using immunofluorescence for OV6 (red) and double immunofluorescence staining for DPPIV (green) and OV6 (red). In R+D-gal-treated rats, the oval cell response peaked at days 4–5, when there was extensive oval cell presence through the hepatic parenchyma, and declined after 1 week. In R+D-gal+C-treated rats, oval cells increased from week 1 and reach a maximal response at week 4. The proliferation of oval cells was in parallel with the massive proliferation of donor hepatocytes. Original magnification: 100x. Scale bars: 50 μm.

(A) Oval cell response in R+D-gal-treated and R+D-gal+C-treated rats. The oval cell response was classified by microscopic examination of liver sections stained with OV6 antibody. The prevalence of the oval cell response was scored on scales of 1 to 5: 1 = oval cells are absent or rare; 5 = extensive oval cell presence throughout all liver zone; 2 to 4 = intermediate response. *Significant difference (Kruskal-Wallis test with Bonferroni correction, p < 0.05) versus control group (D0). **Significant difference (Mann-Whitney rank sum test, p < 0.05) between groups at each time point. Each bar represents the median with minimal and maximal scores of 6–8 rats. (B) The kinetics of oval cell response was in parallel with the repopulation percentage of transplanted hepatocytes in R+D-gal+C-treated rats. Each bar represents the median with minimal and maximal scores (oval cell score) or mean ± SEM (repopulation percentage) of 8 rats.
To assess the relative distribution of transplanted hepatocytes and oval cells, we used double immunofluorescence staining for DPPIV and OV6. At weeks 1 and 2, the oval cells were distributed through the injured hepatic parenchyma and located mostly at the periportal area. At weeks 4 and 8, the numerous oval cells often formed ductular structures and commonly surrounded the donor-derived hepatocyte clusters (Fig. 2). Furthermore, the repopulation percentage by transplanted hepatocytes was quantitated from 3–4 liver sections stained for DPPIV activity with J-Image software (National Cancer Institute, Bethesda, MD) (n = 8 rats at each time point). The kinetics of oval cell response was clearly parallel with the massive proliferation of transplanted hepatocytes throughout the same time period (Fig. 3B).
Hepatocyte Transplantation Affected the Oval Cell Differentiation Fate in R + D-gal-Treated Rats
To determine whether the different kinetics of oval cell response with/without hepatocyte transplantation in R+D-gal-treated rats was associated with oval cell differentiation fate, we used the histochemical staining for GGT. GGT activity was limited to the bile duct cells in the portal triad in normal livers. However, GGT was also expressed in oval cells and differentiating hepatocytes but not in mature hepatocytes (4,11,16). As shown in Figure 4, two distinct patterns of oval cell differentiation were readily demonstrated by GGT staining. In R+D-gal-treated rats without hepatocyte transplantation, the numerous oval cells at day 5 were stained positive for GGT. These GGT-positive oval cells gave rise to GGT-positive hepatocyte clusters in the periportal areas at day 7. The new GGT-positive hepatocyte clusters continued to expand towards the central veins from week 2 to week 8 and lost the GGT expression at week 12.

Fate of oval cell differentiation in retrorsine + d-galactosamine-treated rats without (R+D-gal, left two columns) or with hepatocyte transplantation (R+D-gal+C, right three columns) were analyzed using immunohistochemical staining for OV6 and histochenmical stainings for gamma-glutamyl-transpeptidase (GGT) and DPPIV in serial liver sections. Two distinct patterns of oval cell differentiation were demonstrated. In R+D-gal-treated rats, the numerous oval cells at day 5 were stained positive for GGT. These GGT-positive oval cells differentiated into GGT-positive hepatocyte clusters in the periportal areas at 1 week. The GGT-positive hepatocyte clusters continued to expand towards the central veins from week 2 to week 8. In R+D-gal+C-treated rats, the GGT activity was confined to the expanding oval cell ductules and the bile duct cells in the portal triad after week 2. These GGT-positive oval cells remained in ductular structures and did not differentiate into GGT-positive hepatocyte clusters at week 8 and afterward. Original magnification: 100x. Scale bars: 50 μm.
In contrast, after hepatocyte transplantation, the GGT activity was confined to the expanding oval cell ductules and the bile duct cells in the portal triad after week 2. These GGT-positive oval cells remained in ductular structures and did not give rise to GGT-positive hepatocyte clusters at week 8 and afterward (Fig. 4).
To further characterize the oval cell fate after hepatocyte transplantation, we used imunohistochemical stainings for DPPIV, OV6, and CK19 in serial sections, and double immunofluorescence stainings for DPPIV and OV6 or laminin, or OV6 and laminin. CK19 was an established bile duct specific marker (25,26), and laminin was a well-established marker of the basement membrane, sharply circumscribed the bile ducts (23,24). It is generally accepted that the oval cells are surrounded by laminin. When the oval cells expressed CK19, they differentiated into biliary cells (25). If they lost the surrounding laminin, they differentiated into hepatocytes (23,24). As shown in Fig. 5, the numerous oval cell ductules at week 4 were stained positive for CK19 and were always surrounded by laminin. The precise colocalization of OV6 and CK19, laminin, or GGT in oval cell ductules indicated that they would differentiate into biliary cells. In addition, these oval cell ductules were elongated extensions of the bile ducts and maintained close contact with the randomly distributed DPPIV+ hepatocyte clusters (Fig. 5D–I). Confocal microscopy was used to confirm these findings on representative sections (Fig. 5J–L).
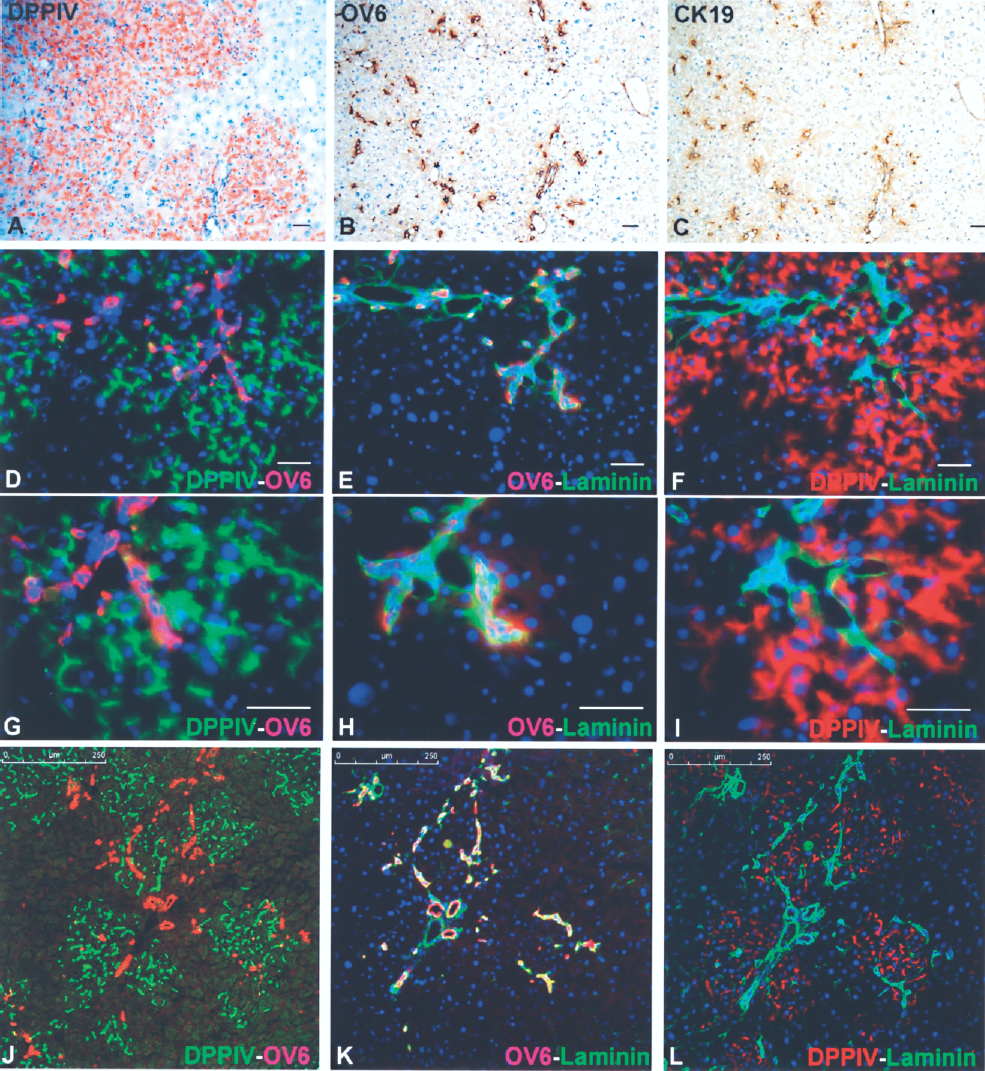
Oval cells differentiate as bile ductules in retrorsine + D-galactosamine-treated rats with hepatocyte transplantation. (A–C) Serial liver sections that were stained histochemically for DPPIV, and immunohistochemically for OV6 and CK19. At the peak of oval cell response (week 4), the numerous oval cell ductules were stained positive for bile duct-specific CK19. Original magnification: 100x. (D–L) Double immunofluorescence stainings for DPPIV (green) and OV6 (red) (left), OV6 (red) and laminin (green) (middle), and DPPIV (red) and laminin (green) (right) in serial liver sections showed that the oval cell ductules were always surrounded by laminin. These oval cell ductules were elongated extensions of the bile ducts and maintained close contact with the DPPIV+ hepatocyte clusters. DAPI was used for nuclear staining (blue). All of the data are shown as merged images. Original magnification in A–F, 100x; in G–I, 200x. Scale bars: A–I, 50 μm; J–L, 250 μm.
To demonstrate that the oval cell-derived GGT-positive hepatocytes in R+D-gal-treated rats or donor-derived hepatocytes in R+D-gal+C-treated rats expressed the functions characteristic of mature hepatocytes, we used histochemical stainings for G6Pase, GGT, and DPPIV in serial sections. G6Pase is expressed uniquely in hepatocytes (4,13), while DPPIV is expressed at the apical surface of the mature hepatocytes (26). The oval cell-derived GGT-positive hepatocytes showed high expression of G6Pase (Fig. 6). Similarly, donor-derived hepatocytes also expressed G6Pase. All of the donor-derived hepatocytes displayed the canalicular expression pattern of DPPIV typical of metabolically active hepatocytes, but did not express GGT. Notably, the donor-derived hepatocyte clusters were not cholestatic in H&E stain (data not shown).
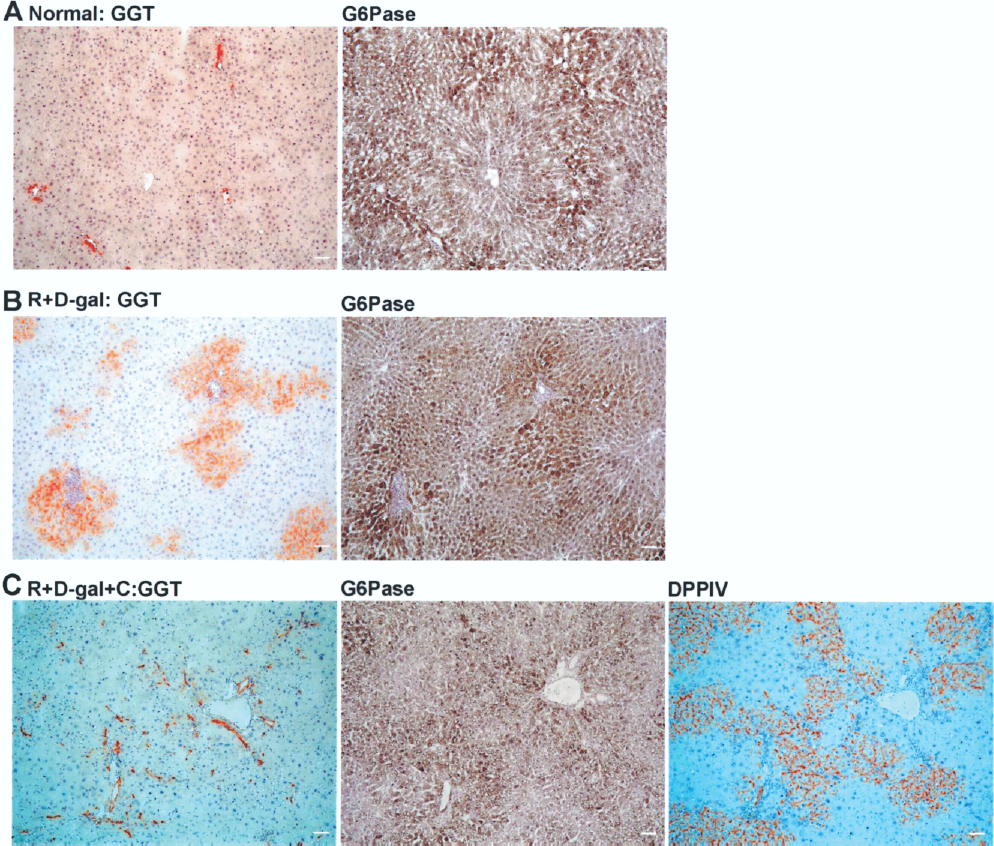
Oval cells differentiate into hepatocytes in retrorsine + d-galactosamine (R+D-gal)-treated rats without hepatocyte transplantation. Each row displays serial liver sections that were stained histochemically for GGT, glucose-6-phosphotase (G6Pase, a protein expressed uniquely in hepatocytes), and DPPIV. (A) In normal rats, only mature bile duct cells were intensely stained for GGT (left). Hepatocytes were positive for G6Pase, with a higher expression in the periportal area (right). (B) In R+D-gal-treated rats, oval cell-derived GGT-positive hepatocytes (left) showed high expression of G6Pase as the surrounding mature hepatocytes (right). (C) In R+D-gal+C-treated rats, DPPIV+ hepatocyte clusters showed varied expression levels of G6Pase, depending on their location in the liver lobules. GGT staining was restricted to bile ducts in the portal area and numerous oval cell ductules. Original magnification: 100x. Scale bars: 50 μm.
Hepatocyte Transplantation Blocked the Oval Cell Differentiation Into Hepatocytes in R+D-gal-Treated Rats
To directly address that hepatocyte transplantation changes the differentiation fate of oval cells, we transplanted hepatocytes at the fourth day after R+D-gal treatment when the host oval cells were at their peak response (Fig. 7A). GGT- and OV6-stained tissues from these rats demonstrated that oval cells were still numerous at 7 days and ceased to differentiate into GGT-positive hepatocytes after hepatocyte transplantation (Fig. 7B). Subsequently, the differentiation pattern of oval cells was similar to that in standard R+D-gal+C protocol. The expanded oval cells remained in ductular form and were commonly in contact with donor-derived hepatocyte clusters.
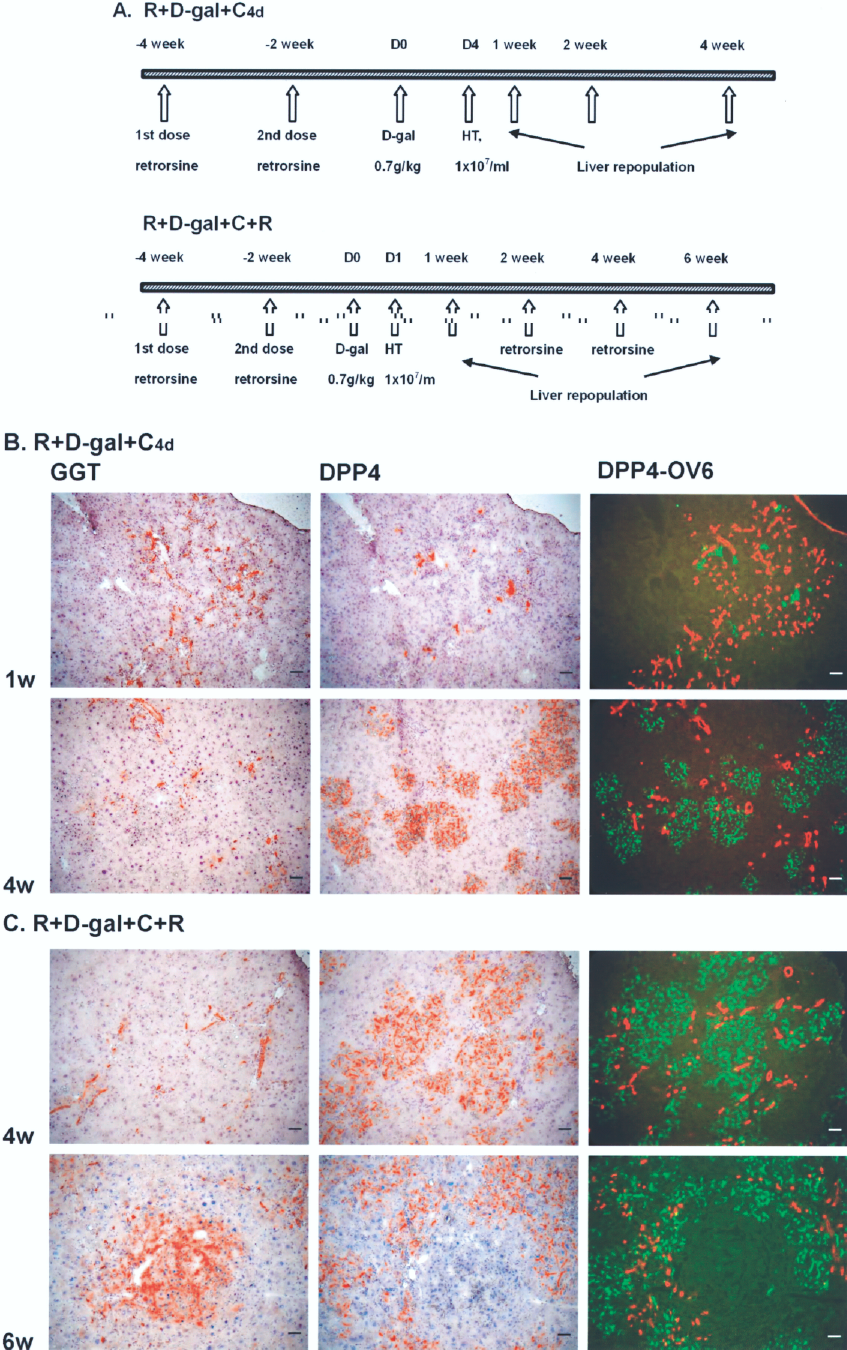
Hepatocyte transplantation blocks the oval cell differentiation into hepatocytes in retrorsine + d-galactosamine treated rats. (A) R+D-gal+C4d group: retrorsine + D-galactosamine + hepatocyte transplantation at the fourth day when the host oval cells were at their peak response; R+D-gal+C+R group: retrorsine + D-galactosamine + hepatocyte transplantation at 24 h + retrorsine at second and fourth week. (B) In R+D-gal+C4d-treated rats, GGT- and OV6-stained tissues demonstrated that oval cells were still numerous at 1 week and ceased to differentiate into GGT-positive hepatocytes. The expanded oval cells remained in ductular form and were commonly in contact with donor-derived hepatocyte clusters (n = 3 rats at each time point). (C) In R+D-gal+C+R-treated rats, the host and transplanted hepatocytes were inhibited to proliferate by retrorsine and became hypertrophic. The oval cells continued to expand in ductular form at 4 weeks and gave rise to GGT-positive hepatocyte clusters at 6 weeks (n = 3 rats at each time point). Original magnification: 100x. Scale bars: 50 μm.
Next, we inhibited the proliferation of transplanted hepatocytes by treating standard R+D-gal+C-conditioned rats with additional two doses of retrorsine at the second and fourth weeks after hepatocyte transplantation, when transplanted hepatocytes and host oval cells underwent extensive proliferation (Fig. 7A). The host and transplanted hepatocytes were incapable of proliferation by retrorsine treatment and became hypertrophic. The oval cells continued to expand in ductular form at 4 weeks. These ductular oval cells gave rise to GGT-positive hepatocyte clusters in the periportal areas at 6 weeks (Fig. 7C).
Discussion
This study clearly shows that hepatocyte transplantation changes the kinetics of oval cell response and affects their differentiation fate in acute severe hepatic injury. Transplanted hepatocytes have priority over host oval cells in replenishing the lost hepatocyte mass. However, oval cells retain flexibility and are ready to give rise to hepatocytes or to provide biliary cells, as the need arises. To our knowledge, this is the first study to illustrate the interaction between donor hepatocytes and host oval cells in reconstituting the acute severely injured liver.
It is generally accepted that oval cells are activated by hepatic parenchymal damage and differentiate preferentially into hepatocytes. They rarely proliferate with hepatocytes simultaneously (6,7,31,32)). However, our study shows that oval cells proliferate in parallel with the massive proliferation of transplanted hepatocytes. The numerous oval cells form ductules while spreading into parenchyma. They express CK-19 and GGT and are surrounded by laminin, indicating they differentiate into biliary cells. They maintain close contact with the randomly distributed donor-derived hepatocyte clusters. Regarding the fact that functional mature hepatocytes require proper bile duct architecture, otherwise they will be cholestatic (5), the lack of cholestasis in the donor-derived hepatocyte clusters indicates that the oval cell ductules are functional, bile-conducting ducts. Based on the findings here, we hypothesize that the functional need of the donor-derived hepatocyte clusters is the mechanism for inducing oval cell response and its differentiation into biliary lineage in the R+D-gal-treated rats after hepatocyte transplantation.
Although oval cells proliferate robustly in R+D-gal-treated livers, their differentiation into hepatocytes is blocked after hepatocyte transplantation. They are capable of giving rise to hepatocytes again when the donor hepatocytes are inhibited to proliferate. These findings have significant implications for liver regeneration and cell transplantation therapy of liver diseases. Oval cells have been considered as a promising cell type for cell transplantation therapy (21,32–34,36). However, oval cells have only limited ability to repopulate normal or diseased livers. To efficiently repopulate the liver with oval cell transplantation, substantial modifications that block the proliferation of endogenous hepatocytes is required (37). These studies, together with our findings, indicate that there exists a cell lineage hierarchy and host priority in regenerating lost or damaged hepatocytes, which is host hepatocytes, followed by donor hepatocytes, and the last, host and/or donor oval cells. Therefore, a successful cell transplantation therapy for acute and/or chronic hepatic injury should take into account the remaining cell type for regenerating the host liver and select the most appropriate cell type with prior hierarchy for transplantation.
Transdifferentiation of rat hepatocytes into biliary cells can occur in a situation when the bile duct is ligated and the host biliary cells and/or oval cells are damaged and inhibited to proliferate by methylene diamiline (9, 19). However, this biliary transdifferentiation of hepatocytes is unlikely to account for the numerous oval cell ductular reactions in the R+D-gal-treated rats after hepatocyte transplantation for the following reasons. First, oval cells are activated to proliferate by R+D-gal treatment. Second, host hepatocyte proliferation is impaired by retrorsine. They are unable to divide and then became reprogrammed. Finally, if the donor hepatocytes do transdifferentiate into biliary cells, there should have seen a substantial number of DPPIV+ bile ductules (19). Together, these data suggest that the direct recruitment of host oval cells and their differentiation into biliary cells may be the simplest way to provide new bile duct systems for the randomly distributed donor-derived hepatocyte clusters.
Does the observed oval cell response in the R+D-gal-treated rats after hepatocyte transplantation arise by some oval cell contaminants in the transplanted cells? This is not likely in this study. For one, R+D-gal treatment induces host oval cell activation. Also, the transplanted hepatocytes have been isolated from normal healthy livers, which normally do not harbor oval cells. Lastly, based on the discussion about cell lineage hierarchy and host priority above, if a small number of oval cells do contaminate the transplanted cells during the isolation procedure, they should have no proliferative advantage over the host oval cells enough for the dramatic oval cell proliferation.
We acknowledge that our findings are based on the drastically experimental conditions induced by R+D-gal and R+D-gal+C+R regimen. However, we have shown in our previous study that R+D-gal-induced acute hepatic injury has many features similar to those in human acute hepatic failure (39). In addition, the oval cell and ductular reactions induced by R+D-gal regimen often occur in various human liver diseases such as chronic viral hepatitis and nonalcoholic steatohepatitis in which the hepatocytes are inhibited to replicate by chronic diseases or develop premature senescence with impaired ability to regenerate (17,28,30,35)). Although the classical d-galactosamine (D-gal-alone) or 2-acetylaminofluorene + partial hepatectomy (2-AAF+PH) regimen induce comparable oval cell response to R+D-gal system, the liver regenerates rapidly after D-gal-alone injury majorly by remaining mature hepatocytes and transplanted hepatocytes do not proliferate in the D-gal-alone system (4,39). Oval cells in the 2-AAF+PH model are capable of giving rise to hepatocytes in a time course that is 2-AAF dose dependent (24); however, the percentage of repopulation by oval cell-derived hepatocytes has not been determined and the 2-AAF+PH system has never been used for hepatocyte transplantation study. We speculate that the administration of 2-AAF after PH may block the proliferation of transplanted hepatocytes and thus limit the further application of 2-AAF+PH system in cell transplantation study. While the classical retrorsine + partial hepatectomy (R+PH) regimen allows comparable repopulation kinetics by transplanted hepatocytes to R+D-gal system (15,39), R+PH induces the emergence of small hepatocyte-like progenitor cells (SHPCs) instead of oval cells and hepatocyte transplantation blocks the proliferation and expansion of SHPCs (10). Therefore, the data presented in this study have significant clinical implications. The R+D-gal system is a useful model to study the mechanisms for the interactive cross-talk between hepatocytes and oval cells.
In conclusion, the liver has multiple mechanisms of repair. Our study demonstrates that hepatocyte transplantation changes the mechanism of liver reconstitution from oval cell-mediated to donor hepatocyte-mediated in R+D-gal-induced acute severe hepatic injury, indicating that hepatocytes are the most efficient cell type of repair. However, the oval cells can proliferate with the transplanted hepatocytes simultaneously and differentiate into biliary cells. Nonetheless, the fate of expanded oval cells is not invariable. They are capable of differentiating into hepatocytes again when the transplanted hepatocytes are incapable of proliferation. Our study provides strong evidence that the differentiation fate of oval cells is affected by transplanted hepatocytes. Although the molecular mechanisms governing the complex interaction between hepatocytes and oval cells are far from deciphered, the present study provides new insight in the cell transplantation therapy of acute hepatic failure patients and regenerative biology of the liver.
Footnotes
Acknowledgments
This study was supported by grants from the National Science Council of the Republic of China (NSC95-2752-B-002-009-PAE; NSC95-2745-B-002-001; NSC96-2752-B-002-009-PAE; NSC96-2321-B-002-022; NSC97-2321-B-002-021-MY2), from the Buddhist Tzu-Chi General Hospital (TCRD-TPE-96-30), and from the Buddhist Tzu-Chi University (TCMRC-C95007-02).
