Abstract
Efficient repopulation by transplanted hepatocytes in the severely injured liver is essential for their clinical application in the treatment of acute hepatic failure. We studied here whether and how the transplanted hepatocytes are able to efficiently repopulate the toxin-induced acute injured liver. Male dipeptidyl peptidase IV-deficient F344 rats were randomized to receive retrorsine plus D-galactosamine (R+D-gal) treatment or D-galactosamine-alone (D-gal) to induce acute hepatic injury, and retrorsine-alone. In these models, retrorsine was used to inhibit the proliferation of endogenous hepatocytes while D-galactosamine induced acute hepatocyte damage. Wild-type hepatocytes (1 × 107/ml) were transplanted intraportally 24 h after D-galactosamine or saline injection. The kinetics of proliferation and repopulation of transplanted cells and the kinetics of cytokine response, hepatic stellate cell (HSC) activation, and matrix metalloproteinase (MMP2) expression were analyzed. We observed that early entry of transplanted hepatocytes into the hepatic plates and massive repopulation of the liver by transplanted hepatocytes occurred in acute hepatic injury induced by R+D-gal treatment but not by D-gal-alone or retrorsine-alone. The expressions of transforming growth factor-α and hepatocyte growth factor genes in the R+D-gal injured liver were significantly upregulated and prolonged up to 4 weeks after hepatocyte transplantation. The expression kinetics were parallel with the efficient proliferation and repopulation of transplanted hepatocytes. HSC was activated rapidly, markedly, and prolongedly up to 4 weeks after hepatocyte transplantation, when the expression of HGF gene and repopulation of transplanted hepatocytes were reduced afterward. Furthermore, the expression kinetics of MMP2 and its specific distribution in the host areas surrounding the expanding clusters of transplanted hepatocytes are consistent with those of activated HSC. Impaired hepatocyte regeneration after acute severe hepatic injury may initiate serial compensatory repair mechanisms that facilitate the extensive repopulation by transplanted hepatocytes that enter early the hepatic plates.
Introduction
Acute hepatic failure is a devastating disease with high mortality and a formidable challenge for clinicians. Liver transplantation continues to represent the primary therapy for patients with acute hepatic failure (1,6). However, only a limited number of eligible candidates can benefit from this option, given the shortage of donor organs, a high cost, and significant morbidity and mortality. Hepatocyte transplantation has been proposed as an attractive alternative since a few decades ago (3,5,19,24,25,28,37,38). The concept is that this procedure is simpler, less invasive, and less expensive than liver transplantation, and can alleviate donor organ shortage. However, application of this approach to the clinic is hampered, with only 38 acute hepatic failure patients allotransplanted with human hepatocytes reported worldwide up to 2007 (10,22). Most studies describing the potential of hepatocyte transplantation in acute hepatic failure models have focused on the improved survival (3,5,13,19,24,25,28,37,38). However, infusion of hepatocyte extract, culture supernatants, and bone marrow cells has also been shown to improve survival in acute hepatic failure in later studies (3,19,28). Most important, the repopulation efficiency by transplanted hepatocytes in the acutely injured liver has not been well studied, (5,41), thus raising the question about the true contribution of transplanted hepatocytes.
For hepatocyte transplantation to become a reliable alternative to liver transplantation, transplanted hepatocytes must proliferate efficiently in the host liver. This requires both the loss of host hepatocyte mass and a proliferative advantage by the transplanted hepatocytes (21,32,33,36,43). In humans, acute hepatic failure is characterized by various degrees of hepatocyte damage and impaired regeneration of the remaining hepatocytes (8,16). Although hepatic damage may trigger liver regeneration, the impaired regenerative response of the remaining hepatocytes results in a high mortality. However, the risky situation in acute hepatic failure may theoretically provide two major favorable factors, proliferation stimulus and selective growth advantage, for the transplanted cells. It should be reasonable to expect that transplanted hepatocytes would proliferate in the acutely injured liver, similar to that in genetic-based metabolic liver disease (21,33,36). Therefore, this study aimed to determine whether and how transplanted hepatocytes are able to integrate and repopulate efficiently the toxin-induced acute severely injured liver.
Materials and Methods
Animals
Dipeptidyl peptidase IV-deficient F344 rats were kindly provided by Professor Sanjeev Gupta from the Albert Einstein College of Medicine. Male DPPIV− rats were used as recipient animals. Normal male DPPIV+ F344 rats (age 8–10 weeks, 200–250 g) were purchased from the National Laboratory Animal Center in Taiwan and used as donor animals. These animals were in-house bred and maintained on standard laboratory chow and daily 12-h light/dark cycles. All of the animals received humane care in accordance with the guidelines of the National Science Council of Taiwan (NSC, 1997). All animal experiments were approved by the Institutional Laboratory Animal Care and Use Committee of the National Taiwan University.
Hepatocyte Isolation and Transplantation
In situ liver perfusion, collagenase digestion, and differential centrifugation were used to purify hepatocytes from normal male DPPIV+ F344 rats as previously described (42). The viability and purity of each preparation were assessed by evaluating trypan blue exclusion in a hemacytometer. Isolated hepatocytes were resuspended in PBS (pH 7.4) without serum to 1 × 107 cells/ml. The preparations typically contained >85% viable hepatocytes, which were transplanted within 1 h of isolation.
Hepatic Histology and Determination of Liver Repopulation
Fresh liver sections were fixed in formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin to demonstrate the hepatic histology. To identify transplanted hepatocytes in the recipient liver, DPPIV expression was determined by enzyme histochemistry in liver cryosections as previously described (35).
To analyze the liver repopulation, three to four sections from multiple liver lobes per rat (n = 6–8) were stained for DPPIV activity. Microphotographs were obtained from consecutively adjacent areas to include the whole section under 100x magnification using a digital camera. The area occupied by the transplanted hepatocytes was quantitated with J-Image software (National Cancer Institute, Bethesda, MD).
Histochemistry and Immunohistochemistry
All histochemical and immunohistochemical stainings were performed according to previously described protocols. The antibody to CD26 recognized the DPPIV antigen on hepatocytes. Double immunofluorescence staining for DPPIV (goat anti-CD26: R&D, Minneapolis, MN, USA) and Ki-67 (BD Biosciences, San Jose, CA, USA) (32), desmin (DAKO, CA, USA) (34), or MMP2 (Lab Vision, CA, USA) (20,32) were detected using the method described by Oertel et al. and Paku et al. Appropriate secondary antibodies used in various experiments included Alexa Fluor-488 donkey anti-goat IgG (Molecular Probes, OR, USA) and Alexa Fluor-594 donkey anti-mouse IgG (Molecular Probes).
Apoptosis of DPPIV+ versus the surrounding DPPIV-parenchyma was detected in situ by end-labeling the fragmented DNA of apoptotic cells using a commercially available kit (Roche, Minneapolis, MN, USA) and simultaneously stained with goat anti-CD26 (R&D) (32). The secondary antibody used was Alexa Fluor-594 donkey-anti-goat IgG (Molecular Probes).
Determinations of Proliferation Index (PI) and Apoptosis Index (AI)
PI and AI were determined in both DPPIV+ and DPPIV-areas in ten microphotographs randomly selected from microscopic fields per rat. The numbers of Ki-67-stained nuclei were counted under 100x magnification while the numbers of positively TUNEL-stained nuclei were determined under 200x magnification. Both indexes were expressed as percentages.
Serological Assay
Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, and total bilirubin were measured in a clinical laboratory using a standard automated assay.
Quantitative Real-Time PCR Assay
Total RNA isolated with Trizol reagent (Invitrogen, Carlsbad, CA, USA) from frozen tissue was reversed-transcribed according to the manufacturer's instructions. cDNA (200 ng) was subjected to quantitative real-time PCR using commercially available TaqMan probes for TNF-α, IL-6, NF-κB, TGF-α, HGF, and MMP2 (Applied Biosystems, Foster, CA, USA) and analyzed with the ABI prism 7700 sequence detection system (TaqMan, Applied Biosystems). Quantitative values were obtained from the threshold PCR cycle number at which an increase in signal associated with the exponential growth of the PCR product started to be detected. The relative mRNA levels in each sample were normalized to its glyceraldehyde-3-phosphate dehydrogenase (GAPDH) content.
Experimental Design
The retrorsine (Sigma, St. Louis, MO, USA) and d-galactosamine (Sigma) working solutions were prepared as described previously (11,42) and used immediately after preparation. Male DPPIV rats received two treatments of retrorsine (30 mg/kg, IP) or equal volumes of normal saline (IP) 2 weeks apart, at 6 and 8 weeks of age. Acute hepatic injury was induced by d-galactosamine treatment (0.7 g/kg, IP) 2 weeks after the second retrorsine or vehicle treatment. Isolated hepatocytes (1 × 107/ml) from wild-type F344 rats were transplanted intraportally 24 h after D-galactosamine or saline injection. Briefly, these groups were: retrorsine+D-galactosamine+ hepatocyte transplantation (R+D-gal+C group); D-galactosamine+hepatocyte transplantation (D-gal+C group); and retrorsine+hepatocyte transplantation (R+C group).
To analyze the engraftment and proliferation of the transplanted hepatocytes, the surviving rats were sacrificed and their livers were harvested on days 1 and 4, and on 1, 2, 4, 8, and 12 weeks after hepatocyte transplantation (R+D-gal+C group, n = 8 rats at each time point; D-gal+C group, n = 4-5 rats at each time point; R+C group, n = 3–5 rats at each time point). The rat body and liver weights were recorded. One piece of liver tissue from the right lobe was fixed in 4% formaldehyde and paraffin-embedded for histology. The other pieces from each liver lobe were snap-frozen in liquid nitrogen or embedded into optimum cutting temperature (OCT) compound and stored at −80°C.
Statistical Analysis
Data were presented as the mean ± SEM. The significance of differences was analyzed by t-test or analysis of variance (ANOVA) with the LSD method as appropriate using SPSS 9.0 (Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Retrorsine-plus-D-Galactosamine Caused Acute Severe Hepatic Injury with Delayed Recovery
The combination of retrorsine and D-galactosamine had not been previously used in animal studies to induce acute hepatic injury (7,29). Therefore, pilot studies on 75 rats were conducted to compare the courses of liver injury induced by D-gal-alone (n = 35) and R+D-gal (n = 40) with sublethal dose of D-galactosamine. Within 24 h of D-gal treatment, both protocols produced similar degree of severe liver injury (R+D-gal vs. D-gal-alone: ALT 1555.1 ± 315.0 IU/L vs. 1516.5 ± 213.7 IU/L; AST 2855.6 ± 523.5 IU/L vs. 3851.6 ± 572.8 IU/L; albumin 3.0 ± 0.1 g/dl vs. 2.9 ± 0.1 g/dl; p = N.S.; T-bilirubin 0.08 ± 0.01 mg/dl vs. 0.12 ± 0.02; t-test, p < 0.05) and of characteristic histological changes, including hepatocellular necrosis and fatty change, inflammatory cell infiltration, and expansion of nonparenchymal epithelial cells. However, the recovery from the acute hepatic injury was delayed to 4 weeks in the R+D-gal group compared to before 2 weeks in the D-gal-alone group.
Early Translocation of Transplanted Hepatocytes Into the Acute Injured Liver Induced by Retrorsine-plus-D-Galactosamine
Although panlobular necrosis and inflammatory cell infiltration became more prominent 24 h after hepatocyte transplantation in both R+D-gal and D-gal-alone treated models, 75.5 ± 3.6% of transplanted hepatocytes had infiltrated the necrotic and inflammatory hepatic parenchyma within 24 h in the R+D-gal-treated model. In contrast, 72.0 ± 12.3% of transplanted hepatocytes were still located in the portal vein radicals in the D-gal-alone injured liver at 24 h (t-test, p < 0.01) (Fig. 1).
Early translocation of transplanted hepatocytes into the acute injured liver induced by retrorsine-plus-D-galactosamine. (A) Transplanted hepatocytes had infiltrated into the necrotic and inflammatory hepatic parenchyma within 24 h in R+D-gal+C animals. In the D-gal+C animals, transplanted hepatocytes were still located in the portal vein radicals. DPPIV histochemistry and hematoxylin counterstain (original magnification 100x). (B) The fraction of transplanted hepatocytes in the hepatic parenchyma within 24 h was significantly higher in R+D-gal+C animals than that in D-gal+C animals. *p < 0.01, t-test. Each bar represents the mean ± SEM of 3–5 rats. Scale bars: 50 μm.
Transplanted Hepatocytes Extensively Repopulated the Acutely Injured Liver Induced by Retrorsine-plus-D-Galactosamine
At 1 week, transplanted hepatocytes were readily present in clusters of 2-10 cells throughout the R+D-gal-treated liver, occupying 2.9 ± 0.3% of the total liver area. In contrast, transplanted hepatocytes were rarely visualized, mostly singly, in the D-gal-alone or retrorsine-alone-treated liver, occupying 0.7 ± 0.1% and 0.1 ± 0.02% of the liver area, respectively (ANOVA with LSD, p < 0.001, vs. R+D-gal+C group).
At 2 weeks, DPPIV+ cell clusters had enlarged in size to 10–30 cells/cluster and comprised 9.8 ± 0.6% of the total area. By 4 weeks, islands of DPPIV+ hepatocytes had expanded extensively, reaching 100–300 cells/cluster, in round or oval shapes, with some clusters being already confluent. They comprised 54.6 ± 7.1% of the liver mass (Fig. 2). To confirm the dramatic expansion by transplanted hepatocytes from 2 to 4 weeks, an additional eight rats were analyzed at 3 weeks. The DPPIV+ clusters occupied 20.4 ± 1.8% of the liver mass. However, the extent of repopulation did not increase further after 4 weeks. The DPPIV+ clusters repopulated 42.4 ± 3.6% of the liver mass at 8 weeks (4 vs. 8 weeks, t-test, p = 0.1) and 38.1 ± 4.8% at 12 weeks (4 vs. 12 weeks, t-test, p = 0.03).
Transplanted hepatocytes efficiently repopulated the acutely injured liver induced by retrorsine-plus-D-galactosamine treatment, but not by D-galactosamine-alone or retrorsine-alone. (A) Shown are representative liver histologies after hepatocyte transplantation in R+D-gal+C, D-gal+C, and R+C animals at the indicated time points. DPPIV histochemistry and hematoxylin counterstain (original magnification 100x; R+D-gal+C, at 4 and 12 weeks, 40x). (B) Kinetics of liver repopulation by transplanted hepatocytes in R+D-gal+C (n = 8), D-gal+C (n = 4–5), and R+C animals (n = 3–5). Repopulation percentage by transplanted hepatocytes in R+D-gal+C animals reached a peak of 54.6 ± 7.1% at 4 weeks, and declined gradually after 8 weeks. Scale bars: 50 μm.
In contrast, in D-gal-alone-treated rats, the proliferation of transplanted hepatocytes was observed to a very limited extent, comprising 1.0 ± 0.4%, 1.3 ± 0.3%, 1.9 ± 0.2%, and 1.4 ± 0.1% of the liver areas at 2, 4, 8, and 12 weeks, respectively. In retrorsine-alone-treated rats, the extent of repopulation by transplanted hepatocytes was 0.4 ± 0.1%, 0.8 ± 0.3%, 4.1 ± 0.5%, and 11.7 ± 3.6%, respectively.
Temporal Expression of Cytokine and Growth Factor Genes in the Acute Injured Liver After Hepatocyte Ttransplantation
As shown in Figure 3A, TNF-α mRNA expression was significantly increased in R+D-gal+C livers compared to D-gal+C and R+C livers at 1, 4, and 7 days after hepatocyte transplantation (p = 0.001, p = 0.01, and p = 0.02, respectively, ANOVA with LSD). Likewise, IL-6 expression was significantly higher in the R+D-gal+C livers compared to the D-gal+C and R+C livers at day 1 (p = 0.003, ANOVA with LSD) (Fig. 3B).
Expression kinetics of TNF-α, IL-6, NF-κB, HGF, and TGF-α mRNA in the acutely injured liver after hepatocyte transplantation was analyzed using quantitative real-time PCR. Overall, the expression profiles were significantly early upregulation of (A) TNF-α (1, 4, 7 days) and (B) IL-6 (1 day) genes followed by prolonged significant upregulation of (C) NF-κB (1, 2, 4, 8 weeks), (D) HGF (1, 2, 4 weeks), and (E) TGF-α (1, 2, 4, 8 weeks) genes in the R+D-gal+C animals, compared with the D-gal+C animals. *p < 0.05, ANOVA with LSD. Each bar represents the mean ± SEM of 3-5 rats. All assays were performed in duplicate.
The expression of NF-κB mRNA was similarly decreased in all three groups at days 1 and 4 after hepatocyte transplantation but did significantly increase in the R+D-gal+C livers compared to the D-gal+C and R+C livers at 1, 2, and 4 weeks (p = 0.01, p = 0.03, respectively, ANOVA with LSD; p = 0.002, R+D-gal+C vs. D-gal+C, at 4 weeks, ANOVA with LSD) (Fig. 3C).
The expression of HGF mRNA increased in the R+D-gal+C livers from day 1 to 4 weeks after hepatocyte transplantation, and the expression level was significantly higher compared to the D-gal+C and R+C livers at 1, 2, and 4 weeks (p = 0.2, p = 0.06, p = 0.04, p = 0.02, and p = 0.03, respectively, ANOVA with LSD) (Fig. 3D). TGF-α mRNA expression was similar in all three groups at 1 and 4 days after hepatocytes transplantation but was significantly elevated in the R+D-gal+C livers compared to the D-gal+C livers from 1 to 8 weeks (p = 0.03, p = 0.03, p = 0.003, and p = 0.02, respectively, ANOVA with LSD) (Fig. 3E).
Overall, the expression kinetics of cytokine and growth factor genes in the R+D-gal+C livers after hepatocyte transplantation were the early activation of TNF-α and IL-6 genes at days 1 and 4, followed by prolonged significant upregulation of NF-κB, TGF-α, and HGF genes up to 4 weeks, which were parallel with the extensive proliferation of transplanted hepatocytes.
Different Response Between Transplanted and Host Hepatocytes to the Regenerating Signals in Retrorsine-plus-D-Galactosamine-Treated Rats
To determine the response of transplanted and host hepatocytes to the prolonged upregulation of growth factor genes in R+D-gal-treated rats, we used double immunofluorescence staining for DPPIV and Ki-67 or TUNEL assay. The transplanted hepatocytes exhibited the typical bile canalicular expressions of DPPIV and were already positive for Ki-67 staining (PI = 23.3 ± 0.6%) at day 4 (Fig. 4A). The host hepatocytes were also positive for Ki-67 (PI = 5.1 ± 0.2%, t-test, p < 0.001 vs. transplanted hepatocytes). The proliferative activity of the transplanted hepatocytes was significantly higher compared to the surrounding host hepatocytes up to week 4 (20.8 ± 3.5% vs. 2.3 ± 0.9%, 23.4 ± 1.8% vs. 1.1 ± 0.1%, and 1.3 ± 0.1% vs. 0.7 ± 0.1%, t-test, p = 0.007, p < 0.001, and p = 0.025, respectively), but not significantly different from 8 to 12 weeks (1.1 ± 0.3% vs. 0.6 ± 0.1%, 0.9 ± 0.2% vs. 0.5 ± 0.2%, t-test, p = 0.15 and 0.24, respectively).
(A) Proliferative and apoptotic activities of transplanted hepatocytes and surrounding host hepatocytes in the R+D-gal+C rats were analyzed using dual-label immunohistochemistry for DPPIV (green) and Ki-67 (red) (left column), and DPPIV (red) and TUNEL assay (green) (right column). Cell nuclei were stained with DAPI (blue) (original magnification, 200x). (B) The proliferative activity of transplanted hepatocytes was significantly higher than that of the surrounding host hepatocytes from 4 days to 4 weeks. (A, right column) Apoptotic host hepatocytes were observed throughout the liver parenchyma and at the periphery of DPPIV+ cell clusters (see arrows) (original magnification, 200x). (C) The apoptotic percentage of host hepatocytes was significantly higher than that of transplanted hepatocytes at 4 days only. *p < 0.05, t-test. Each bar represents the mean ± SEM of 3–5 rats. Scale bars: 50 μm.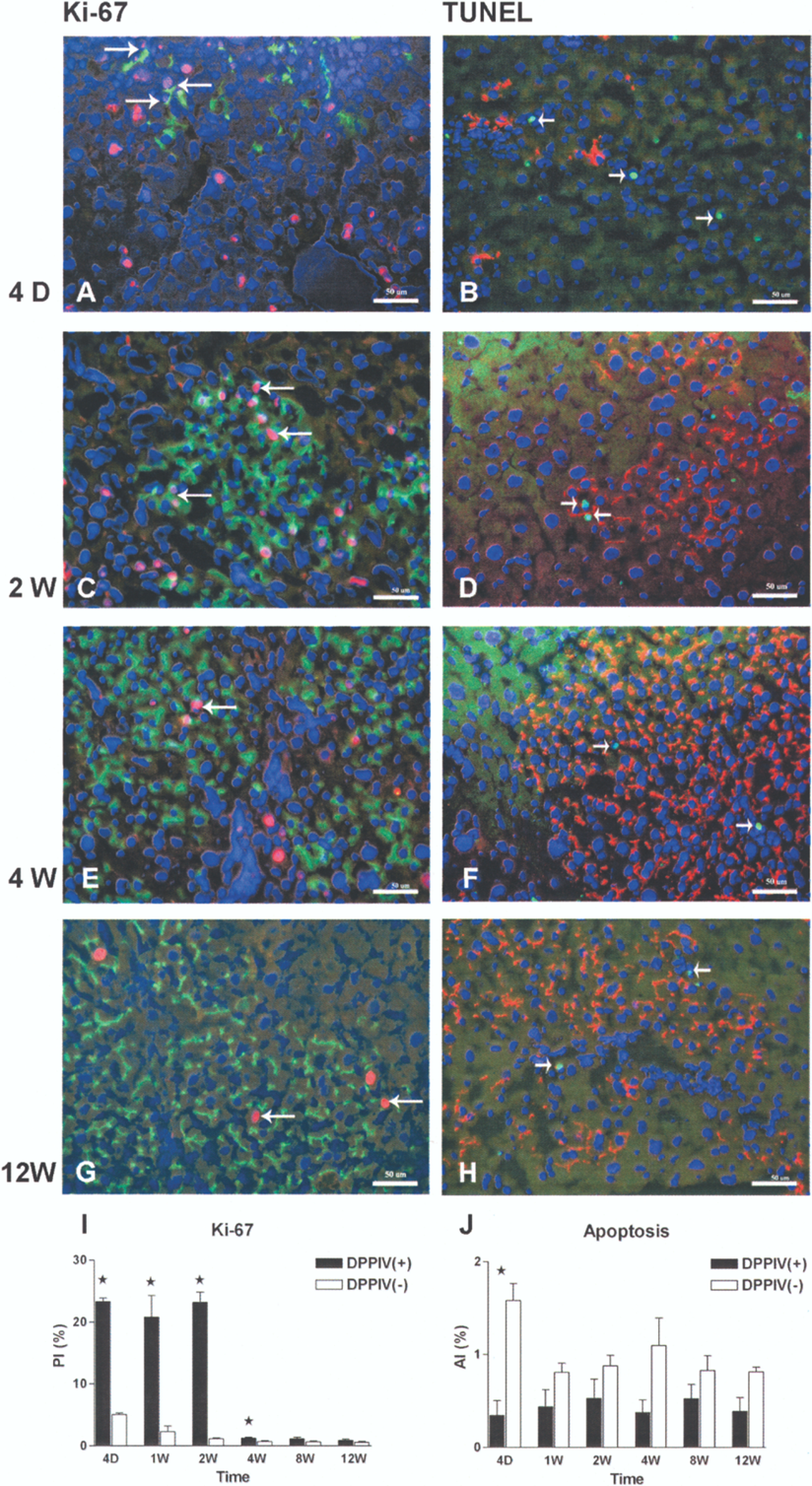
Apoptosis of the transplanted hepatocytes was rarely observed when first identified on day 4 and at all time points thereafter (AI = 0.4 ± 0.2%, 0.4 ± 0.2%, 0.5 ± 0.2%, 0.4 ± 0.1%, 0.5 ± 0.2%, 0.4 ± 0.1%, respectively) (Fig. 4A and B), and was randomly distributed within the transplanted cell clusters. In contrast, apoptosis of the host hepatocytes was most clearly seen on day 4 and was frequently observed throughout the liver parenchyma and at the periphery of proliferating clusters of transplanted hepatocytes from week 1 and thereafter by TUNEL assay (1.6 ± 0.2%, 0.8 ± 0.1%, 0.9 ± 0.1%, 1.1 ± 0.3%, 0.8 ± 0.2%, and 0.8 ± 0.1%, t-test, p < 0.001, p = 0.12, p = 0.19, p = 0.07, p = 0.2, and p = 0.05 vs. transplanted hepatocytes, respectively).
Activation of Hepatic Stellate Cells and Modulation of Eextracellular Matrix in the Regenerating Liver After R+D-gal+C Treatment
Activation of HSC is associated with increased desmin expression (23). To determine the potential role of HSC activation in the proliferation of transplanted hepatocytes in acute hepatic injury, we used double immunofluorescence staining for DPPIV and desmin. A marked increase in the number of desmin-positive HSC was observed from day 1 to week 2, followed by a decline after week 4 (Fig. 5A). The desmin-positive HSC was most prominent in the host liver areas surrounding the transplanted hepatocyte clusters.
HSC activation and liver repopulation in R+D-gal+C-treated rats. (A) Desmin-positive HSC increased rapidly and markedly up to 2 weeks after hepatocyte transplantation, and mostly distributed in areas surrounding the transplanted DPPIV+ hepatocytes (original magnification: 4D and 2W, 200x; 4W, 100x). (B, C) The expression kinetics of MMP2 gene and activity are consistent with the HSC activation. Increased MMP2 activity was also distributed mostly in areas surrounding expanding clusters of transplanted DPPIV+ hepatocytes (original magnification 100x). MMP2 gene expression was early and significantly upregulated in the R+D-gal+C liver. *p < 0.05, ANOVA with LSD. Each bar represents the mean ± SEM of 3–5 rats. All assays were performed in duplicate. Scale bars: 50 μm.
MMP2 expression played a significant role in the integration of transplanted hepatocytes (20,32). To explore if MMP2 expression was an important mechanism for the extensive repopulation by transplanted hepatocytes, we used quantitative real-time PCR and double immunofluorescence staining to determine the expression of MMP2 (17,20). As shown in Figure 5B and C, MMP2 mRNA expression was significantly elevated in the R+D-gal+C livers compared to the D-gal+C and/or R+C livers from day 1 to week 2 (p = 0.002, p = 0.03, p = 0.04, and p = 0.05, respectively, ANOVA with LSD). In parallel with gene expression, the expression activity of MMP2 was markedly increased from day 1 to week 2, followed by a decline after week 4, and was most prominent in the host liver areas surrounding the transplanted hepatocyte clusters.
Discussion
This study clearly demonstrates that the massive proliferation and repopulation of the liver by transplanted hepatocytes can occur in acute severe hepatic injury induced by R+D-gal treatment within weeks, but not by D-gal-alone or retrorsine-alone. This indicates that both impaired host hepatocyte proliferation and hepatocellular damage are required to efficiently repopulate the liver with transplanted cells. Although massive hepatocyte necrosis and impaired proliferative activity of the remaining hepatocytes represent the most important predictors of poor outcome in acute hepatic failure patients, our data suggest that they may yet provide a suitable microenvironment for transplanted hepatocytes to proliferate and expand. Our results therefore provide experimental evidence supporting the reported clinical experiences of successful treatment of acute liver failure patients with hepatocyte transplantation (10).
In this study, we observed prolonged and significant upregulation of NF-κB, TGF-α, and HGF genes from day 1 to 4 weeks in the R+D-gal+C-treated livers. These growth factors play critical roles in promoting cellular replication during liver regeneration (39). Therefore, their prolonged upregulation may indicate an attempted compensatory response to the impaired proliferative capacity of host hepatocytes after hepatic injury. The significantly higher proliferative activity of transplanted hepatocytes compared to that of host hepatocytes and parallel kinetics of these gene expressions and repopulation of transplanted cells through the same time period suggest that transplanted hepatocytes take over these regeneration signals and proliferate. This finding is clinically important. HGF and TGF-a levels were strongly elevated in patients with acute hepatic failure (2,40). Transplantation of normal hepatocytes might take this advantage to proliferate, and thus have a great therapeutic potential for this disease context.
We observed rapid infiltration of transplanted hepatocytes into the necrotic and inflammatory hepatic parenchyma within 24 h in the R+D-gal-treated liver, but not in the D-gal-alone or R-alone-treated liver. Previous studies indicated that Kupffer cells contribute to clear the transplanted cells in the portal radicles and that hepatocytes need to be primed by cytokines before they can proliferate (14,27). Therefore, the early entry of transplanted cells into the hepatic plates in the R+D-galtreated liver prevented them from early clearance by Kupffer cells and facilitated their priming by the significantly high expression of TNF-α and IL-6. Subsequently, they can fully respond to the prolonged upregulation of HGF and TGF-α and proliferate efficiently and extensively. These findings support the previous reports that superior cell integration accelerates the kinetics of liver repopulation by donor cells (12,26). In addition, the rapid entry into the hepatic plates also implies the early resolution of portal hypertension caused by transplanted hepatocytes emboli. The delayed resolution of portal hypertension due to prolonged portal obstruction by transplanted cell emboli in an acutely injured liver has previously been shown to increase mortality (42).
Activation of HSC is crucial in liver repair after acute hepatic injury and plays a role in the early engraftment of transplanted hepatocytes (4,23). Loss of activated HSC is a hallmark of successful repair (30). In our study, the number of desmin-positive HSC was increased rapidly and markedly up to 2 weeks and declined after 4 weeks in the regenerating liver after R+D-gal+C treatment. This response kinetics is prolonged as compared to that after D-gal-alone-induced, CCl4-induced, and partial hepatectomy-induced liver injury, where HSC activation subsides in 7 days (23,30). The prolonged HSC activation may result from the impaired regeneration of host hepatocytes. However, while the HSC activation declined after 4 weeks, the expression of HGF gene and repopulation of transplanted hepatocytes were reduced. These data suggest that prolonged activation of HSC in acute hepatic injury is beneficial to the proliferation of transplanted cells.
Another potential mechanism used by the activated HSC to regulate the repopulation of transplanted hepatocytes is through modulation of extracellular matrix (18, 31). In this study, the expression kinetics of MMP2 gene and MMP2 activity are consistent with the kinetics of HSC activation. Furthermore, the MMP2 distribution in the regenerating liver, mostly in the host areas surrounding the expanding clusters of transplanted hepatocytes, was similar that of activated HSC and was consistent to previous studies (20,32). These findings suggest that the host extracellular matrix is actively remodeled by MMP2 to create spaces for the transplanted cells.
We acknowledge that the condition induced by R+D-gal regimen is experimental and thus striking in promoting liver replacement by transplanted cells. However, many features in R+D-gal-induced acute hepatic injury, such as diffuse hepatocellular damage, long-lasting impaired proliferative capacity of remaining hepatocytes, and persistently elevated cytokine and growth factor levels, are similar to those in human acute hepatic failure. Furthermore, although the classical retrorsine + partial hepatectomy regimen allowed comparable repopulation kinetics by transplanted hepatocytes to R+D-gal system, the former requires invasive surgery and the remaining host liver is normal (21). Therefore, the data presented in this study has significant clinical implications. The R+D-gal system is a less-invasive and useful model to study cell transplantation therapy in acute massive hepatocyte necrosis.
In conclusion, impaired hepatocyte regeneration after acute severe hepatic injury may initiate serial compensatory repair mechanisms which facilitate the extensive repopulation by transplanted hepatocytes that enter early the hepatic plates. Hepatocyte transplantation has a great potential in the treatment of patients with acute hepatic failure.
Footnotes
Acknowledgments
This study was supported by grants from the National Science Council, R.O.C (NSC93-2752-B-002-010-PAE; NSC94-2752-B-002-009-PAE; NSC95-2752-B-002-009-PAE; NSC96-2752-B-002-009-PAE), and from the Buddhist Tzu-Chi General Hospital, Taipei Branch (TCRDTPE-95-22).
