Abstract
The advances in stem cell science have promoted research on their use in liver regenerative medicine. Beyond the demonstration of their ability to display metabolic functions in vitro, candidate cells should demonstrate achievable in situ differentiation and ability to participate to liver repopulation. In this work, we studied the in vivo behavior of adult liver mesenchymal stem/progenitor cells (ADHLSCs) after transplantation into immunodeficient mice. The kinetics of engraftment and in situ hepatogenic differentiation were analyzed. Response of transplanted ADHLSCs to regenerative stimulus was also evaluated. Nondifferentiated ADHLSCs were intrasplenically transplanted into SCID mice. Efficiency of transplantation was evaluated at the level of engraftment and in situ differentiation using immunohistochemistry, in situ hybridization, and RT-PCR. After bromodeoxyuridine (BrdU) implantation, proliferation of transplanted ADHLSCs in response to 20% hepatectomy was assessed using immunohistochemistry. We demonstrated that ADHLSC engraftment in the SCID mouse liver was low but remained stable up to 60 days posttransplantation, when albumin (ALB) immunopositive ADHLSCs were still detected and organized as clusters. Coexpression of ornithine transcarbamylase (OTC) demonstrated ADHLSC in situ differentiation mostly near the hepatic portal vein. After 20% hepatectomy on 1 month transplanted mice, the percentage of BrdU and human ALB immunopositive ADHLSCs increased from 3 to 28 days post-BrdU implantation to reach 31.3 ± 5.4% of the total analyzed human cells. In the current study, we demonstrate that transplanted ADHLSCs are able to differentiate in the non preconditioned SCID mouse liver mainly in the periportal area. In response to partial hepatectomy, integrated ADHLSCs proliferate and participate to recipient mouse liver regeneration.
Introduction
Liver is the ideal target organ for cell-based therapies. Because of its unique blood supply and architecture, the infused cell suspension is easily transferred and distributed within the hepatic parenchyma. Infused cells can engraft and repopulate the target organ if the conditions are optimal. Liver cell-based therapies have been developed as alternatives to conventional transplantation to restore missing liver function. Besides in vitro data, studies on animal models should prove that candidate cells display an ability to migrate into recipient liver, to engraft, and to function in situ. Adult hepatocytes, the smallest and pivotal functional entities of the liver, are fully active metabolic systems. Transplantation of isolated hepatocytes in animal models of genetic metabolic disorders such as the fumarylacetoacetate hydrolase knockout (FAH-/-) mouse, a model of tyrosinaemia type 1, and the Gunn rat, a model of Crigler–Najjar syndrome, has convincingly shown their ability to provide metabolic support (2,4,12). Similarly in human, hepatocytes isolated from donor livers have been used to treat miscellaneous inborn errors of metabolism (6,8,21,24,25,27,32,33,36,37). As in animal models, transplantation of hepatocytes in humans showed their ability to engraft within the recipient livers and to supply metabolic functions. However, their routine use remained hampered by several limiting factors such as significant donor shortage as well as their low resistance to isolation, cryopreservation, and in vitro culture processes (9,27,35). These factors are influencing both the engraftment quality of transplanted cells and the durability of the posttransplantation metabolic effect.
Adult liver, a quiescent organ, has the particularity to regenerate itself in response to several injuries. Regeneration is mainly due to hepatocyte proliferation. When hepatocyte proliferation is altered, stem/progenitor compartment can be activated. Increasing knowledge about liver regeneration capacity and its reservoir of stem/progenitor cells has brought valuable information to understand pathways involved in such process (1,7,28). In addition, their ability to proliferate and to differentiate in vitro as well as their resistance to cryopreservation rendered the stem/progenitor cells as serious candidates for liver cell therapy development. Adult stem/progenitor cells have been isolated and characterized from various tissues like bone marrow (19,20), adipose tissue (31), cord blood (10), umbilical cord (5), peripheral blood (18), and skin (22). A great interest has been given to these cells because of their involvement in tissue regeneration and repair. Despite a limited potential of differentiation into various tissues, as compared to embryonic stem cells, their multipotency raises the question of ectopic tissue formation after transplantation. We previously isolated from collagenase digested healthy human liver, a cell population with hepato-mesenchymal phenotype [adult human liver stem cell (ADHLSC)] and potency to proliferate and to differentiate into hepatocyte-like cells (26). After intrasplenic transplantation into mice, ADHLSCs are able to migrate and to engraft within the recipient liver parenchyma.
In the current study, we particularly studied the kinetics of ADHLSC engraftment as well as their in situ hepatogenic differentiation potency after transplantation into severe combined immunodeficient (SCID) mice. We also evaluated the interaction of engrafted ADHLSCs with host liver environment by analyzing their behavior after a regenerative stimulus induced by 20% partial hepatectomy.
Materials and Methods
The animal procedures were conducted according to the guidelines for humane care for laboratory animals established by the Université Catholique de Louvain, in accordance with European Union Regulation and protocols approved by the local ethic committee.
ADHLSC Isolation
ADHLSCs were isolated from the livers of three healthy cadaveric donors at 9 (male), 16 (female), and 37 (female) years old. After primary culture, emerging ADHLSCs become predominant after the second passage according to our previous documented observations (26). The purity of the ADHLSC population was confirmed by the morphology, hepato-mesenchymal phenotype [albumin (ALB)+, α-smooth muscle actin (α-SMA)+], and absence of epithelial markers such as cytokeratins (CK) 7, 8, 18, and 19 as well as cluster of differentiation 133 (CD133) using immunocytochemistry, RT-PCR, and flow cytometry (26). The hepatogenic differentiation potential is also evaluated as described elsewhere (17,26). The differentiated cells are analyzed at the morphological, genetic, and functional levels. As for instance, the differentiated ADHLSCs should exhibit significant cytochrome P450, family 3, subfamily A, polypeptide 4 (CYP3A4) activity, de novo production of glucose, and urea synthesis. Such characterization is necessary for their release and use in transplantation studies.
Mice Transplantation
All animal experiments were performed in compliance with Belgian law for animal protection and approved by the local ethical review board.
Nondifferentiated ADHLSCs cultivated on collagen type 1-coated flasks (BD Biosciences, Erembodegem-Aalst, Belgium and Greiner-Bio, Wemmel, Belgium, respectively) are detached using Trypsin-EDTA (Invitrogen, Gent, Belgium) solution for 5 min at 37°C. All cells used in this study are from the third passage. After centrifugation and washing, cells were suspended at 1 million cells/100 μl of PBS, pH 7.4 (Lonza, Verviers, Belgium). One million nondifferentiated ADHLSCs (viability > 90% using the trypan blue exclusion assay; Sigma, Diegem, Belgium) were injected into the spleen of 6-week-old SCID mice (Charles River, Lille, France) (n = 6–8 female mice).
To investigate migration of cells from the splenic site of injection toward the liver parenchyma, mice (n = 2) were sacrificed 10 min after transplantation, and their organs were immediately fixed in Bouin's solution (Prosan, Gent, Belgium) to preserve cell structure and morphology. We also investigated long-term engraftment and in vivo hepatic differentiation of transplanted human ADHLSCs. The SCID mice were sacrificed 7 (n = 8), 30 (n = 6), and 60 (n = 6) days posttransplantation, and their livers were fixed in paraformaldehyde 3.5% (VWR International, Leuven, Belgium) and embedded in paraffin for immunohistochemistry and in situ hybridization analyses. A small liver sample (5 × 5 × 5 mm) of transplanted mice at 7 and 60 days posttransplantation was incubated in Tripure isolation Reagent (Roche, Vilvoorde, Belgium) for total RNA extraction and RT-PCR analysis.
For hepatectomy studies, after general anesthesia of mice and small upper midline incision, the median lobe of liver was tied with silk suture (Covidien, Dublin, Ireland) and then resected with no general apparent postoperation complication and morbidity. The removed liver lobe was harvested for further immunohistochemical and PCR analyses. Its weight was determined and compared to the total mice liver.
Cell Proliferation Analysis by BrdU Uptake
Cell proliferation was evaluated by incorporation of BrdU (bromodeoxyuridine; Alzet, Charles River) into DNA during mitosis. One month after ADHLSC transplantation, mice (n = 6 for each group) were implanted intra peritoneum with an osmotic pump (Alzet, Charles River) continuously delivering BrdU at 20 μg/h for 3, 14, and 28 days. Half of these mice were subjected to 20% hepatectomy and evaluated for its stimulus effect on proliferation of implanted ADHLSCs. Harvested livers were fixed in paraformaldehyde 3.5% and embedded in paraffin for double immunodetection of BrdU and human hepatocyte markers.
Immunohistochemistry Analysis
Five-micrometer liver sections were deparaffinized and rehydrated in graded alcohol series. Endogenous peroxidase activity was blocked by incubation for 15 min in a 3% hydrogen peroxide–methanol solution (Sigma). Antigen retrieval was performed by incubating the sections in citric acid monohydrate solution (pH 6.0; Dako, Heverlee, Belgium) at 97°C for 90 min. Nonspecific immunostaining was prevented by 1 h incubation in PBS buffer containing 1% normal goat serum (Sigma) at room temperature. Slices were incubated overnight with polyclonal anti-human Alb (Calbiochem, Nottingham, UK) (1/2500), monoclonal anti-human vimentin (Progen, Heidelberg, Germany) (1/100), polyclonal anti-human ornithine transcarbamylase (OTC; Sigma) (1/200), mouse anti-human α-SMA (Dako) and monoclonal anti-BrdU (Sigma) (1/100) in 1% normal goat serum at 4°C. Staining detection was visualized by Envision Dako anti-mouse or anti-rabbit (Dako) using diaminobenzidine (Sigma) as chromogenic substrate. The nuclei were counterstained using Mayer's hematoxylin (Sigma) for 10 min and mounted for analysis.
Human vimentin immunostaining was particularly performed using Animal Research Kit (Dako) that is based on avidin-biotin and peroxidase methodologies to minimize cross-reactivity of secondary anti-mouse antibody with endogenous immunoglobulin that may be present in the liver section. For each marker, three different sections have been analyzed.
For double immunostaining, nuclear markers [BrdU or Alu sequences (described below)] were stained prior to cytoplasmic markers (human ALB or human OTC), respectively, using diaminobenzidine and 3-amino 9-ethylcarbazole (Dako) as chromogenic substrates. Slices were mounted using Dako Faramount Aqueous Mounting Medium (Dako) and examined using an HP50 inverted microscope coupled to a DFC camera (Leica, Heerbrugg, Switzerland). Digital images were acquired using Leica IM50 Image Manager Software.
Detection of Human Alu Sequences by In Situ Hybridization
Three-micrometer liver sections were deparaffinized and rehydrated. After antigen retrieval steps, slices were washed in distilled water for 10 min at room temperature and immersed in increasing graded ethanol for 1 min. DNA was denatured at 95°C for 5 min, and hybridization process was performed with human Alu-DNA Probe (Biogenex, Fremont, CA, USA) at 37°C overnight. Hybridized slices were washed in standard saline citrate (pH 7.0; Dako) at room temperature for 10 min, 56°C for 10 min, and room temperature for 15 min. Nonspecific binding was prevented by incubation in PBS containing 3% bovine serum albumin (Sigma) for 1 min followed by a two-step avidin and biotin blocking (Vector Laboratories Ltd., Brussels, Belgium) for 15 min at room temperature. To detect hybridized Alu sequences, slices were incubated with biotinylated goat anti-fluorescent antibody (1/100) (Vector Laboratories Ltd.) for 1 h, and the signal was revealed using streptavidin/horseradish peroxidase (Dako) with diaminobenzidine as the chromogenic substrate.
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from the liver samples using Tripure isolation Reagent (Roche). cDNA was generated using the Thermoscript™ RT kit (Invitrogen). The PCR amplification was performed for 28 cycles at 95°C for 1 min, 58°C for 1 min, and 72°C for 2 min using Elongase® Enzyme Mix (Invitrogen) and specific primers for human ALB (forward primer 5′-CCTTGGTGTTGATTGCCTTTGCTC-3′ and reverse primer 5′-CATCACATCAACCTCTGGTCTC ACC-3′) or human GAPDH (forward primer 5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and reverse primer 5′-AGCCTTCTCCATGGTGGTGAAGAC-3′). PCR products were subjected to 1.5% agarose gel electrophoresis (Life Technologies, Gent, Belgium) and visualized human ALB at 308 pb and human GAPDH at 307 pb after ethidium bromide (Sigma) staining.
Statistical Analyses
Results are expressed as mean ± standard error of the mean (SEM). Statistical differences were determined by Student's t test for two groups' comparison or by one-way ANOVA followed by the Newman–Keul's post hoc test for multiple comparisons between more than two groups. Differences were considered significant when p < 0.05 (*p < 0.05, **p < 0.01, ***p < 0.001).
Results
In Vivo Migration and Liver Parenchyma Engraftment of ADHLSCs
Before transplantation, ADHLSCs were well characterized in vitro for both their hepato-mesenchymal phenotype and hepatogenic differentiation potential. The ADHLSCs we used for transplantation studies are ALB+, α-SMA+, CK18-, and CK19- and exhibit features of advanced functional hepatogenic differentiation as described elsewhere (17,26). We firstly followed the in situ engraftment and differentiation of transplanted ADHLSCs in the non-preconditioned (i.e., untreated prior to transplant) liver SCID mice. Ten minutes postintrasplenic transplantation, hematoxylin-eosin staining of spleen slices revealed the presence of individual viable cells as well as organized clusters (Fig. 1A, B). In parallel, migrating individual ADHLSCs were already observed in the recipient liver mainly in the vascular structures (Fig. 1C, D). Few cells were entering the mouse liver parenchyma. Immunohistochemistry analysis using antibodies specific for human vimentin and α-SMA confirmed the human origin, the phenotype and revealed the organization of the transplanted cells both in the spleen and the liver (Fig. 1E, F).

Splenic and hepatic localization of ADHLSCs 10 min posttransplantation in two severe combined immunodeficient (SCID) mice. Hematoxylin and eosin staining revealed clustered or isolated adult human liver stem cells (ADHLSCs; black arrows) in the spleen (A, B), the hepatic portal vein (C), and the liver parenchyma (D) 10 min posttransplantation. Immunoreactivity for vimentin (E) and α-smooth muscle actin (α-SMA) (F) respectively confirmed the human origin of infused ADHLSCs both in spleen and after migration into mice liver parenchyma. Original magnification: 200× (A) and 400× (B–F).
Thereafter, in situ engraftment and differentiation of infused ADHLSCs was followed after 7, 30, and 60 days posttransplantation (Fig. 2). Using immunohistochemistry, we checked the presence of human ALB-positive cells in recipient mouse livers and found that transplanted human cells are detected up to 60 days posttransplantation. At the morphology level, immunodetected transplanted human cells adopted a hepatocyte-like shape within the recipient SCID mouse parenchyma. These transplanted ADHLSCs were seen as scattered individual cells or organized clusters. Comparison of the three transplanted animal groups revealed that 7 days posttransplantation, ADHLSCs were mostly scattered and localized near vascular structures (Fig. 2A). After 30 and 60 days posttransplantation, scattered cells were observed predominantly inside the hepatic parenchyma (Fig. 2B, C). Few clusters of three to four cells were observed after 7 days posttransplantation (Fig. 2D). The size of cell clusters was significantly decreased after 30 days posttransplantation (Fig. 2E). In the third group of transplanted mice, the number of scattered cells and clusters (Fig. 2C, F) increased progressively to reach the values detected after 7 days posttransplantation. The specificity of the immunostaining was demonstrated by using nontransplanted mice (Fig. 2G) and human liver sections (Fig. 2H).

Human albumin immunoreactivity of ADHLSCs in the liver of SCID mice 7, 30, and 60 days posttransplantation. Human albumin immunostaining revealed scattered or clusters of ADHLSCs (brown) in the SCID mice liver at 7 (A, D), 30 (B, E), and 60 (C, F) days posttransplantation. Nontransplanted mice (G) and human (H) livers were used as negative and positive controls respectively. Original magnification: 400×. To analyze ADHLSC kinetic implantation, human albumin positively stained cells were counted and the total number estimated after 7, 30, and 60 days is represented as scattered cells (I), clusters (J), or the ratio of human albumin positive/total host hepatocytes (K). The number of engrafted ADHLSCs significantly decreased 1 month posttransplantation and was enhanced after 2 months. *p < 0.05, **p < 0.01, ***p < 0.001.
Human ALB-positive ADHLSCs and mouse liver cells were counted in each 5-μm liver slice and in all hepatic lobes and the determined number of cells was normalized per mm2 of surface. Comparison of the three transplanted mouse groups revealed that a significant increase of human ALB-positive scattered cells and clusters was seen after 60 days posttransplantation (Fig. 2I, J). A significant augmentation of the cluster's size was also noticed (Fig. 2F).
Quantification analysis also confirmed a low engraftment level at day 7 posttransplantation (0.53 ± 0.06%, n = 8) (Fig. 2K). At day 30, the number of human ALB-positive cells significantly decreased to 0.23 ± 0.42% (n = 6) (Fig. 2K). Sixty days posttransplantation, the number of scattered human cells reincreased to reach an engraftment level of 1 ± 0.33% (n = 6) (Fig. 2K). This increase in human cell number was confirmed after evaluation of human ALB mRNA expression, as we only detected the presence of a PCR product in liver samples isolated from mouse group sacrificed 60 days posttransplantation (Fig. 3). When analyzing the presence of human Alu sequences using in situ hybridization, we demonstrated the presence of both scattered and clustered human Alu sequence-positive cells (data not shown) that were coimmunostained for human ALB (Fig. 4A, B). Furthermore, we did not observe any human cells positive for Alu sequences but not for ALB at the different time points analyzed. Specificity of the staining was demonstrated by using liver slices of nontransplanted mice as well as human (Fig. 4C, D).
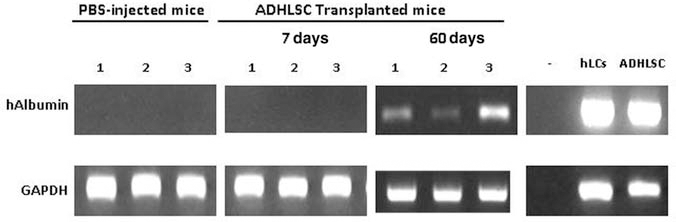
Human albumin mRNA is detected in the liver of transplanted SCID mice after 60 days posttransplantation. Conventional RT-PCR has been performed on transplanted liver mice mRNA extracts, and analysis demonstrated the presence of the human albumin transcript in three of six transplanted mice sacrificed 60 days posttransplantation. No human albumin detection is observed in the group transplanted for 7 days as well as nontransplanted mice. Freshly isolated human liver cells (hLCs) and ADHLSCs were used as positive controls.

Double immunostaining of human Alu sequences and human albumin. To demonstrate specificity of human marker positivity, we performed double immunostaining with human Alu sequences (brown nucleus) and albumin (brown cytoplasm). One month posttransplantation, coimmunostaining for human albumin and Alu sequences (black arrows) (A, B) was obtained for ADHLSCs integrated into the mouse liver parenchyma. Nontransplanted mice (C) and human liver (D) were analyzed as negative and positive controls. Original magnification: 400×.
In Situ Differentiation of Engrafted ADHLSCs
Microscopic evaluation of immunostained liver slices revealed that human ALB-positive cells engrafted in the mice liver parenchyma and adopted hepatocyte-like morphology. To test whether this correlated to in situ hepatogenic differentiation, we investigated on serial slices the expression of mesenchymal and hepatocyte markers. As shown in Figure 5, we did not detect human vimentin-positive immunostaining on human ALB-positive cells either after 7 days (Fig. 5A, B), 30 days (Fig. 5C, D), and 60 days posttransplantation (Fig. 5E, F). In parallel, we also checked on serial slices if OTC, one of the six enzymes of the urea cycle, was coexpressed in human ALB-positive cells. Our data revealed that 7 days posttransplantation, few ALB-positive cells were immunostained for OTC (Fig. 6A, B). After 30 and 60 days posttransplantation, we demonstrated that the number of human ALB-positive cells that are coimmunostained for human OTC had increased (Fig. 6C–F). Interestingly, OTC-positive detected ADHLSCs were mostly located near the hepatic portal vein (Fig. 6B, D, F), a specific zone for urea metabolism.

Transplanted ADHLSCs lose their mesenchymal phenotype after transplantation. Immunostaining for human albumin (brown) (A, C, E) and human vimentin (brown) (B, D, F) on serial liver sections revealed that most of the human albumin-positive cells lost vimentin expression at 7 (A, B), 30 (C, D), and 60 (E, F) days posttransplantation. Human vimentin staining was performed in parallel on nontransplanted mice (G) and human (H) livers as controls. Original magnification: 400×.

In situ differentiation of transplanted ADHLSCs in the mice liver parenchyma. Immunostaining of human ornithine transcarbamylase (OTC; B, D, F) was studied in transplanted mouse livers in order to investigate the in situ differentiation of transplanted ADHLSCs. Human albumin immunoreactivity (A, C, E) was in parallel evaluated on serial liver sections. Human cells coexpressing human OTC and albumin were rarely detected at 7 days posttransplantation (A, B). Their number increases after 30 (C, D) and 60 (E, F) days posttransplantation. Nontransplanted mice (G) and human (H) livers stained with human OTC were performed in parallel as negative and positive controls. Original magnification: 400×.
These data revealed that ADHLSC engraftment in the non preconditioned SCID mouse liver remained stable up to 60 days posttransplantation. Engrafted cells were shown to integrate into the recipient parenchyma and acquired morphological and phenotypic features indicating their in situ hepatogenic differentiation.
Engrafted ADHLSCs Proliferation After a Regenerative Stimulus
We thereafter investigated whether transplanted ADHLSCs are able to respond to hepatic regenerative stimulus such as hepatectomy by participating to recipient liver regeneration. To test our assumption, 1-month-old transplanted mice were subcutaneously implanted with an osmotic pump that continuously releases BrdU for 3, 14, and 28 days. If nuclear division occurs, BrdU molecules will integrate into cell DNA by replacing thymidine and immunostaining with anti-BrdU will be used to reveal newly proliferating cells. The human origin of cells will be in parallel confirmed using antibodies specifically directed against human ALB and OTC (Fig. 7).

Proliferation of engrafted ADHLSCs and participation in mouse liver regeneration. Thirty-day transplanted mice were subjected to bromodeoxyuridine (BrdU) uptake for 3 (A, D), 14 (B, E), and 28 (C, F) days (n = 6/group). Half of these mice were subjected to 20% hepatectomy (D, E, F) to evaluate if transplanted ADHLSCs proliferate and/or participate in recipient liver regeneration. Double immunostaining for human albumin (red cytoplasm) and BrdU (brown nucleus) analysis revealed that no human albumin-positive cells expressing BrdU was detected at 3 days (A) in control mice, whereas an increase of double immunostaining (black arrows) was detected at 14 (B) and 28 (C) days. In hepatectomized mice, host hepatocytes increased proliferation to reestablish the loss of hepatic mass. Very few human albumin cells were proliferated at 3 days (D), whereas an increase (arrows) was demonstrated at 14 (E) and 28 days (F). Human liver (G) and nontransplanted mouse (implanted with BrdU for 28 days) liver (H) and intestine (I) were analyzed for both markers in parallel as controls. We demonstrated that the human liver tissue was stained for albumin but not for BrdU. On the contrary, liver and intestine of nontransplanted mice was positive for BrdU but not for albumin. Original magnification: 400×. Percentage of BrdU-positive cells among engrafted ADHLSCs [human albumin (hAlb)-positive cells] or host hepatocytes (human albumin negative cells) were presented as a histogram (J) in control and hepatectomized mice following 3, 14, and 28 days BrdU implantation to illustrate kinetic regeneration response. Statistical analysis revealed significant proliferation of ADHLSCs following partial hepatectomy after 14–28 days post-BrdU implantation. *p < 0.05, **p < 0.01, ***p < 0.001.
Three days post-BrdU implantation, counted mouse hepatocytes that are positively stained for BrdU represented 4.3 ± 1.8% of scanned microscopic fields (Fig. 7J). In this group, no human ALB-positive cells were immunostained with anti-BrdU (Fig. 7A, J). After 14 and 28 days post-BrdU implantation, the number of mice hepatocytes expressing BrdU increased up to 12.3 ± 3.5% and 16.2 ± 5.1%, respectively (Fig. 7B, C, J). The number of human ALB-positive cells was also enhanced in both series (Fig. 7B, C, J). In the 28 days post-BrdU implantation group, a number of ALB immunopositive cells were also immunostained for BrdU (Fig. 7C).
Following 20% hepatectomy, the proliferation of mouse hepatocytes increased to restore the loss of hepatic mass. In the transplanted mice with BrdU implant, only a few proliferating cells positively stained for human ALB at day 3 postimplantation (Fig. 7D).
In this group, recipient proliferating hepatocytes represented approximately 17.1 ± 3.2% of the total analyzed cells (Fig. 7D, J). Quantification of immunopositive cells revealed that human cell proliferation at day 3 was lower than that of mice hepatocytes (2.6 ± 2.3% vs. 17.1 ± 3.2%) (Fig. 7J). Host hepatocytes continued to proliferate to restore the whole liver mass. The number of BrdU-positive mouse hepatocytes increased to reach 23.6 ± 4.1% and 40.2 ± 8.2% after 14 and 28 days, respectively (Fig. 7E, F, J). The percentage of BrdU and human ALB immunopositive cells (vs. the total analyzed human cells) increased in parallel up to 14 and 28 days (20.5 ± 6% and 31.3 ± 5.4%, respectively) (Fig. 7E, F, J), with no statistical difference for human or mice cells at 14 and 28 days (ns; p > 0.05) (Fig. 7J).
The specificity of the immunostaining was confirmed by the absence of BrdU staining in human liver (Fig. 7G) and its presence in nontransplanted mice (implanted with BrdU for 28 days) liver (Fig. 7H) and intestine (Fig. 7I). In contrast, the human ALB positivity revealed on the human liver slice (Fig. 7G) was not detected in the liver and the intestine of nontransplanted mice (Fig. 7H, I).
Similarly, cells proliferating and immunopositive for human OTC were not detected at 3 days post-BrdU implantation. Their number increased at 14 and 28 days post-BrdU implantation in hepatectomized mice (data not shown).
Discussion
In the current study, we demonstrate that transplanted ADHLSCs are able to differentiate in non-preconditioned mouse recipient liver by displaying hepatocyte markers up to 8 weeks posttransplantation. We also show that engrafted ADHLSCs proliferate in situ in response to mild hepatic regenerative stimulus.
ADHLSCs, originally isolated from collagenase-digested adult human livers, are of hepato-mesenchymal phenotype and exhibit liver metabolic functions after in vitro hepatogenic differentiation (26). Furthermore, ADHLSCs present significant self-renewal potential that remains maintained after cryopreservation. Such properties support the evaluation of their prospective use for liver cell therapy development. Hence, the current study aims at investigating the in vivo behavior of transplanted ADHLSCs in SCID mouse liver environment. Nondifferentiated ADHLSC suspension was infused in the spleen of SCID mice and hepatic engraftment was followed up to 8 weeks posttransplantation. Our data confirm the ability of infused undifferentiated ADHLSCs to migrate into the mouse liver. Indeed, ADHLSCs quickly reach recipient SCID mouse liver and are detected after only 10-min posttransplantation. Cells were mainly observed near the portal tract as demonstrated using both HE staining and immunopositivity for mesenchymal markers (vimentin and α-SMA). This is in accordance with previous studies demonstrating a rapid transit of intrasplenically transplanted cells towards liver plates (14,30). Isolated hepatocytes, initially used for liver cell therapy studies, reach distal portal spaces and sinusoids 20–60 min after intrasplenic transplantation.
Seven days posttransplantation, infused ADHLSCs already engraft within the SCID mouse hepatic parenchyma as revealed using anti-human ALB antibody staining. Quantification of immunopositive human cells (mainly scattered) reveals that the percentage of engrafted cells is very low (<1%). This corroborates with previous documented studies showing that a large proportion of transplanted cells is cleared early after transplantation (13). ADHLSCs used for our transplantation studies have more than 95% viability and are more resistant compared to hepatocytes. This suggests that marginal viability of the cell suspension is not in accordance with the observed cell loss and that cell entry to recipient liver parenchyma, at a given time, is highly regulated. Such observations also indicate that SCID mouse immune clearance mechanisms involved in the control of early cell engraftment is still significantly efficient. Adult MSCs have been shown to be susceptible to lysis by activated natural killer (NK) cells, which are still present in SCID mice (11).
The number of engrafted human ALB-positive cells, scattered and organized in clusters, decreases 30 days posttransplantation, indicating that late clearance mechanism occurs. The organization of ADHLSCs within recipient liver is in line with what has been documented for different hepatic cell types as for instance liver foetal progenitor cells (15). One also could stipulate that, when engrafted into inappropriate zones of the liver parenchyma, transplanted cells may not survive because of incomplete differentiation and integration. At 8 weeks posttransplantation, the number of engrafted ADHLSCs increases as demonstrated using in situ hybridization for human Alu sequences and RT-PCR assays. Immunohistochemical quantification of survived human cells reveals a low but stable engraftment level of ADHLSCs. This is in line with previous experiments performed in several xenogeneic models using differentiated hepatic cells like hepatocytes or foetal epithelial cells (29). Immunodetection of ADHLSCs at 8 weeks posttransplantation supports a delayed engraftment of ADHLSCs probably preserved in the peri-portal area or a proliferation of the engrafted cells between 4 and 8 weeks posttransplantation. Recently, it has been proposed that both the integration into hepatic plates and the nature of the cells may be critical factors for the engrafted cells to survive and expand (16). However, it has been demonstrated that a further increase in transplanted cell number does not allow higher repopulation (15).
Besides engraftment, we demonstrate using coimmunostaining that transplanted ADHLSCs differentiate in situ. Engrafted human ALB-positive cells lose their initial mesenchymal markers (vimentin) as early as from 7 days posttransplantation. In addition, human ALB-positive cells acquire mature hepatocyte markers such as OTC from day 30 posttransplantation. Hepatocytic differentiation was also documented at 60 days posttransplantation when enriched Thy1+ bone marrow cells were transplanted directly into rat livers treated with monocrotalin and 2-acetylaminofluorene following 2/3 partial hepatectomy (PH) (3). Here we demonstrate that ADHLSCs are able to differentiate in a liver environment with no regenerative stimulus applied, a feature probably supported by their initial hepatic origin.
Furthermore, costained ADHLSCs are exclusively concentrated in hepatic portal area, the specific zone of urea metabolism (34). No OTC staining is observed in the perivenous area, where the urea cycle is not functional, suggesting that engrafted human cells adapt to their zonal environment. The absence of engrafted ADHLSCs in other liver zones suggests that 8 weeks follow-up may be not enough time for the cells to reach very advanced differentiation features, which accelerates their local exclusion.
In the last part of the study, we evaluate the ability of engrafted differentiated ADHLSCs to respond to a regenerative stimulus and to participate to mouse liver regeneration. SCID mice were transplanted with nondifferentiated ADHLSCs and implanted with BrdU pumps 30 days later. Mice were then analyzed for different periods of time to analyze the proliferative status of engrafted human cells. In nonhepatectomized mice, no ADHLSC proliferation was observed after 3 days post-BrdU implantation, whereas only few human ALB-positive cells were immunodetected 28 days post-BrdU implantation. Such cells were mainly located at the periportal area.
After 20% hepatectomy, few proliferating cells were positively stained for human ALB 3 days post-BrdU implantation. The number of ADHLSC proliferating cells and immunopositive for ALB increases from 2 to 4 weeks post-BrdU implantation to reach equivalent level than mouse hepatocytes. The same observation is made for ADHLSCs immunopositive for OTC suggesting that proliferation in response to hepatectomy involves the newly differentiated human cells. If in situ proliferation has also been observed for early human fetal hepatoblasts without conditioning the donor (23), it is in our knowledge the first report of proliferation of in situ differentiated mesenchymal stem/progenitor within the liver. This reveals the ability of differentiated ADHLSCs to adapt to murine liver environment and to respond to mild hepatectomy without activation of EMT.
In conclusion, we demonstrate here that, in this xenogeneic model, transplanted nondifferentiated ADHLSCs are able to engraft and to differentiate in the non-preconditioned SCID mouse liver mainly in the periportal area. Engrafted ADHLSCs are also able to proliferate and to participate to mouse liver regeneration after partial hepatectomy. The analysis of the behavior of such cells in successfully restoring deficient hepatic functions and diseased livers remains mandatory before their potential development for liver cell-based therapies.
Footnotes
Acknowledgments
D.N.K. is a Télévie recipient. M.N. is a researcher of IREC. We also thank Prof. Isabelle Leclercq for her support and useful advices. This work was financially supported by Région Wallonne-DGTRE (Grant Waleo-HEPATERA) and FNRS-Télévie, Belgium. The authors declare no conflicts of interest.
