Abstract
The aim of this study was to evaluate the importance of nonalbumin-predominant proteinuria on kidney function (KF) after islet transplantation (ITx). Twenty-four-hour proteinuria and albuminuria were available in 27 recipients. KF was assessed by serum creatinine and estimated glomerular filtration rate (eGFR) was calculated by Modification of Diet in Renal Disease formula. Correlations between eGFR and albuminuria (r = −0.422, p < 0.001) were higher than with proteinuria (r = −0.223, p < 0.001; p = 0.006 for comparison between correlations). Nineteen (70%) subjects had proteinuria ≥ 300 mg/24 h during the follow-up. Subjects were divided into three groups according to urinary protein excretion patterns: no proteinuria (n = 8), nonalbumin-predominant (n = 8), and albumin-predominant (n = 11) proteinuria. Proteinuria ≥ 500 mg/24 h was observed only among patients with albumin-predominant proteinuria (64%; p = 0.002) and these patients had the lowest eGFR means post-ITx (no proteinuria: 84.2 ± 16.4 vs. nonalbumin: 69.1 ± 13.8 vs. albumin-predominant proteinuria: 65.5 ± 16.6 ml/min/1.73 m2, p = 0.044 for first vs. last group). In conclusion, high frequency of proteinuria was observed after ITx. However, it seems to be milder and have less impact on KF when albumin is not the major source of proteinuria. Prospective evaluation of proteinuria, including tubular function markers, should be performed to elucidate the mechanisms of kidney damage in this population.
Introduction
Immunosuppressive therapy is associated with renal dysfunction in naive (1) as well as in kidneys grafts (2), with tubular toxicity being implicated in the majority of cases (11). Urinary leakage of proteins other than albumin can indicate tubular instead of glomerular damage (14) and this nonalbumin pattern of proteinuria has been recently described as a risk factor for graft loss and mortality after kidney transplantation (7). The relative contribution of albumin to the total excreted urinary protein could be an important tool to define the specific mechanisms of kidney damage associated with chronic immunosuppression.
The clinical course of kidney function after islet transplantation (ITx) is still under debate (6, 8, 12, 15). In addition, it is not clear whether the decrease in glomerular filtration rate (GFR) observed in some ITx cohorts is due to the progression of a pretransplant subclinical diabetic nephropathy (DN) or due to the effect of chronic immunosuppressive therapy or a combination of both. In our experience, we have observed proteinuria in the absence of abnormal levels of albuminuria in a subset of ITx recipients, which is possibly indicating immunosuppressive therapy-related tubular damage. The aim of this study was to investigate the frequency and importance of nonalbumin-predominant proteinuria on kidney function in ITx recipients.
Materials and Methods
Subjects
A retrospective cohort study was conducted on 36 subjects with type 1 DM and hypoglycemia unawareness that have been followed at a single center after ITx alone (48 ± 22 months) between 2000 and 2007. Subjects with baseline renal dysfunction (serum creatinine >1.6 mg/dl or albuminuria >300 mg/24 h) were not considered eligible for ITx. One subject was excluded from the analysis due to early withdrawal of immunosuppressive therapy (102 days after transplant) and subsequent graft failure after a severe adverse event (aspiration pneumonia). For the purpose of this study, only patients with at least three concomitant measurements of albuminuria and proteinuria were included (n = 27) in the analysis. The patients excluded (n = 8) had similar age and diabetes duration when compared to the included ones (data not shown).
Islet Transplant Procedures
The pancreatic islet isolation, infusion, and immediate post-ITx management were performed as previously described (5). The induction was performed with either five-dose course of daclizumab (1 mg/kg biweekly; Zenapax®, Roche, Nutley, NJ, USA, n = 22) or alemtuzumab (20 mg IV, two doses before the transplant; Campath®, Genzyme, Cambridge, MA, USA, n = 5). The maintenance immunosuppressive regimen was based on tacrolimus (Prograf®, Astellas-Pharma US, Inc., Deerfield, IL, USA; target trough level 4–6 ng/ml) and sirolimus (Rapamune®, Wyeth Pharmaceuticals, Inc., Madison, NJ, USA; target trough level 10–15 ng/ml for 3 months, 8–12 ng/ml thereafter). A subset of subjects (n = 12) were converted from tacrolimus or sirolimus to mycophenolate mofetil (MMF; CellCept®, Roche, Nutley, NJ, USA) or mycophenolate sodium (MS; Myfortic®, Novartis, East Hanover, NJ, USA), targeting maximum tolerable dosage (maximum of 2000 and 1440 mg, respectively) due to side effects (n = 7, nephrotoxicity in two cases) (4, 13) or per protocol (n = 5).
All protocol procedures were approved by the University of Miami health research ethics board (IRB) and appropriate informed consent was obtained from each subject.
Laboratory Analysis
Kidney function was evaluated by serum creatinine (Jaffé method, Roche Diagnostics, Roche Cobas-Mira, inter- and intra-assay variation: 1.4% and 2.1%), 24-h urinary albumin excretion (UAE; immunoturbidimetry, Beckman-Synchron/CX9, Ramsey, MN, USA), and 24-h urinary protein excretion (pyrogallol red, Diagnostic Chemicals Limited, Charlottetown, Canada). The subjects were classified as normoalbuminurics (UAER <30 mg/24 h), microalbuminurics (30–299 mg/24 h), or macroalbuminurics (≥300 mg/24 h) based on two out of three pre-ITx measurements and the same criterion was employed for kidney status definition during follow-up. Subjects were considered to have abnormal proteinuria when 24-h urinary protein excretion was ≥300 mg/24 h, according to National Kidney Foundation (NKF) guidelines for chronic kidney disease (CKD) (10). Three groups, divided according to urinary protein and albumin excretion patterns, were analyzed: no proteinuria (proteinuria <300 mg/24 h at all the post-ITx time points; n = 8), nonalbumin-predominant proteinuria (proteinuria >300 mg/24 h on at least one time post-ITx and albuminuria <30 mg/24 h at all post-ITx time points; n = 8), and albumin-predominant proteinuria (proteinuria ≥300 mg/24 h and albuminuria ≥30 mg/24 h in at least one time point post-ITx; n = 11). The estimated GFR (eGFR) was calculated by the Modification of Diet in Renal Disease (MDRD) formula (9):186* [serum creatinine–1.154 *age−0.203*(0.742 if female)*(1.210 if afro-American)] and renal failure was considered when eGFR reached stage 3 of CKD (moderate dysfunction: <60 ml/min/1.73 m2) or lower, based on NKF guidelines (10).
Glycemic profiles were evaluated by fasting plasma glucose (hexokinase method) and A1c [high performance liquid chromatography (HPLC), Variant II Hemoglobin Testing System, BioRad, Richmond, CA, inter- and intra-assay variation: 1.7% and <2.0%, normal values 4.2–6.1%]. Fasting lipids (total cholesterol, HDL cholesterol, and triglycerides) were measure by the enzymatic method and LDL cholesterol was determined by the Friedewald equation (3).
Statistical Analysis
Statistical analysis and graphics were done using Excel® for Windows® and SSPS® 15.0 software. Results of continuous variables were expressed as means ± SD except for albuminuria and triglycerides [median (interquartile range)] and categorical as number of cases (%). Variables with nonnormal distribution were log-transformed. Correlations were performed by Pearson test and differences between two correlations were compared by Fisher r-to-z transformation. One-way ANOVA (with post hoc Turkey test), chi square, or log-rank tests were used to compare variables among groups. Values of p < 0.05 (two-tailed) were considered to be statistically significant.
Results
Patient age at the time of first transplant was 43.8 ± 9.0 years with a diabetes duration of 28.2 ± 12.6 years. All subjects were white and 10 (37%) were males. BP means were 117 ± 12/71 ± 8 mmHg and A1c was 7.1 ± 1.0%. Eight subjects (30%) had hypertension and 13 (48%) had dyslipidemia. Chronic diabetes complications at baseline included microalbuminuria in 15% (n = 4), diabetic retinopathy in 63% (n = 17; nine proliferative), peripheral neuropathy in 22% (n = 6), autonomic neuropathy in 11% (n = 3), and no subject had cardiovascular disease.
Inverse correlations were found between eGFR and albuminuria (r = −0.422, p < 0.001) and eGFR and proteinuria (r = −0.223, p < 0.001). When the correlations were compared, albuminuria showed a stronger association with each given eGFR value than proteinuria (p = 0.006 for comparison between correlations) (Fig. 1).

Correlations between estimated (e)Glomerular filtration rate and albuminuria (left) and eGlomerular filtration rate and proteinuria (right). p = 0.006 for comparison between correlations.
In order to further evaluate whether proteinuria in the absence of abnormal albuminuria had an impact on kidney function of ITx recipients, subjects were analyzed according to urinary protein and albumin excretion patterns. Nineteen subjects (70%) had at least one proteinuria value >300 mg/24 h, while eight (30%) had normal urinary protein levels during the entire follow-up. From the group with proteinuria >300 mg/24 h, eight subjects (42%) had normal albuminuria and 11 (58%) had micro-and/or macroalbuminuria at some point during the follow-up. Only one subject belonging to the normal proteinuria group (<300 mg/dl) had urinary albumin levels in the microalbuminuric range after transplant.
The clinical and laboratory characteristics of subjects according to renal protein leakage are presented in Table 1. Interestingly, subjects developing albumin-predominant proteinuria had higher A1c at baseline (no proteinuria: 6.5 ± 0.9 vs. nonalbumin predominant proteinuria: 7.0 ± 0.7 vs. albumin-predominant proteinuria: 7.6 ± 1.0%, p = 0.042), but the islet graft survival did not differ among groups (estimated time to graft dysfunction: 12.6 ± 3.3 vs. 19.0 ± 4.5 vs. 15.7 ± 4.6 months, p = 0.951; estimated time to graft failure: 41.8 ± 8.9 vs. 64.7 ± 8.4 vs. 63.1 ±6.1 months, p = 0.218). The prevalence of microvascular complications was higher in subjects with albumin-predominant proteinuria (microalbuminuria: 0% vs. 0% vs. 36%, p = 0.023; diabetic retinopathy: 38% vs. 50% vs. 91%, p = 0.016). Study follow-up was similar among groups and more patients showing albumin-predominant proteinuria required modification of the maintenance immunosuppressive protocol when compared to the other two groups (Table 1).
Baseline Clinical and Laboratory Characteristics According to Kidney Protein Excretion Pattern

No proteinuria versus albuminuria group.
p for trend.
Reason to switch—no protein group: both subjects switched from tacrolimus as per protocol; nonalbumin-predominant group: the subject was switched from sirolimus due to migraine; and albumin-predominant proteinuria group: 8 subjects were switched from tacrolimus (3 as per protocol; 1 due to eczema; 1 depression; 1 neurotoxicity, and 2 nephrotoxicity) and 1 from sirolimus (mouth ulcer).
No proteinuria and nonalbuminuria versus albuminuria group.
Proteinuria values >500 mg/24 h were found only among patients with albumin-predominant proteinuria (64%; p = 0.002). Moreover, a tendency for higher frequency of low eGFR (<60 ml/min/1.73 m2) was observed as subjects progress from no proteinuria to nonalbumin- and albumin-predominant proteinuria groups (0% vs. 24% vs. 36%, p = 0.069) (Fig. 2). Similarly, subjects with albumin-predominant proteinuria had the lowest eGFR values at the end of the follow-up (84.2 ± 16.4 vs. 69.1 ± 13.8 vs. 65.5 ± 16.6 ml/min/1.73 m2, p = 0.044 for differences between no proteinuria and albumin-predominant proteinuria groups) (Fig. 3).
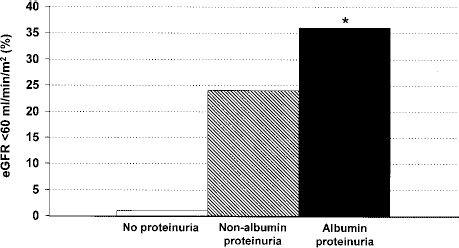
Development of low estimated glomerular filtration rate (<60 ml/min/1.73 m2) during the follow-up according to the urinary protein and albumin leakage pattern. No albuminuria (white bar), nonalbumin-predominant proteinuria (gray bar), and albumin-predominant proteinuria (black bar). *p for trend = 0.069. eGFR, estimated glomerular filtration rate.

Estimated glomerular filtration rate (eGRF) preislet transplant (dark gray) and in the most recent follow-up (light gray) according to the urinary protein and albumin leakage pattern.
Discussion
In this sample of ITx recipients, a high frequency (70%) of abnormal proteinuria (>300 mg/24 h) was observed during the post-ITx follow-up. Notably, proteinuria levels were higher and had a stronger impact on eGFR when a concomitant increase in albuminuria was present.
Posttransplant proteinuria has been reported in kidney (7) and heart graft recipients (1), but the nonalbumin fraction was only studied in a kidney transplant cohort (7). Similar to that reported in kidney recipients (7), maximum proteinuria values were lower in subjects with nonalbumin-predominant proteinuria than in those with concomitant increase in albuminuria. ITx recipients with concomitant high proteinuria and abnormal albuminuria had the lowest eGFR at the end of follow-up in our study, and no association between nonalbumin-predominant proteinuria and lower eGFR was observed. These results are different from those reported in kidney recipients (7).
This could be a result of differences between studies. Our series is composed of subjects exposed to the diabetes milieu for a long time pre-ITx, while only the minority of kidney graft recipients in the reported study had diabetes (~7%) (7). Even though the majority of subjects in this sample were normoalbuminuric at baseline, the presence of diabetic retinopathy suggests that they are at risk for developing DN during the course of the disease. Therefore, our results reflect the effect of immunosuppressive drugs on naive kidneys at risk for DN.
Accordingly, patients with nonalbumin-predominant proteinuria seem to be the intermediary state of the post-ITx nephropathy spectrum. These patients fall between subjects without proteinuria (and normal eGFR) and those with concomitant proteinuria and albuminuria (and higher prevalence of microvascular complications and A1c since baseline, which resulted in a lower eGFR). The last group (albumin-predominant proteinuria) probably represents a mixture of DN and immunosuppressive-induced nephrotoxicity, while the nonalbumin-predominant proteinuria group might express only the effect of immunosuppressive toxicity.
The individual role of each immunosuppressive agent (tacrolimus vs. sirolimus) to determine the protein excretion pattern could not be assessed in the present study, due to the small number of patients requiring protocol modifications (n = 12, only two switched from sirolimus). However, the majority of immunosuppressive protocol modifications were preformed in those patients with albumin-predominant proteinuria. In only two cases the alteration was attributed to nephrotoxicity, whereas in the other cases it was due to protocol requirements (n = 3) or to other side effects (n = 4) (4, 13) not related to the kidney function. Possible explanations for this observation could be a simple by chance association, or that albumin values influenced the decision to switch the therapy even in patients with other side effects and/or that patients with nonrenal toxicity could have as well an increment in albuminuria levels.
The limitations of this study are the retrospective analysis and the relative small sample size, which precluded a more comprehensive evaluation of tubular function markers in all subjects or finding statistical differences among the nonalbumin-predominant proteinura group and the other two groups. The availability of kidney biopsies would have enhanced our results, as it is the gold standard used to define the pathogenesis of kidney dysfunction. However, a systematic evaluation of the kidney histology in all ITx recipients would be difficult and not ethically justified, because the majority of the subjects have normal kidney function trough all post-ITx follow-up (8).
In conclusion, a high frequency of proteinuria was observed after ITx. However, it seems to be milder and have less impact on KF when albumin is not the major source of proteinuria. Therefore, albuminuria continues to be the best marker for kidney damage in patients with T1DM, including ITx recipients. The presence of microvascular disease pre-ITx, represented either by microalbuminuria or diabetic retinopathy, as well as the metabolic control, may indicate those recipients prone to develop albumin-predominant proteinuria and, consequently, lower GFR. However, as our results are based on observation of a small sample of ITx recipients, our findings should be replicated in other cohorts in order to confirm the concept. Moreover, systematic evaluation of proteinuria, including tubular function markers, should be performed prospectively in ongoing ITx trials, aiming at better characterization of the impact and mechanisms of immunosuppressive-mediated kidney damage in ITx recipients.
Footnotes
Acknowledgments
This study was supported by National Institutes of Health/National Center for Research Resources (U42 RR016603, M01RR16587); Juvenile Diabetes Research Foundation International (#4-2000-946, 4-2004-361); National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases (5 R01 DK55347, 5 R01 DK056953); State of Florida, and the Diabetes Research Institute Foundation (Hollywood, FL). C.B.L. was the recipient of a scholarship from Conselho Nacional de Desenvolvimento Cientifico e Tecnologico (CNPq).
